C1- Atomic structure and C2-Structure & bonding
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
what is an element?
a substance made of only one type of atom
what is a mixture?
A mixture is a substance made up of two or more different types of atoms or molecules that are not chemically bonded together.
what is a compound?
substance formed when two or more different types of atoms are chemically bonded together
what is an atom?
smallest unit of matter
what is the charge and mass of a proton?
mass= 1 charge= +1
what is the atomic number of an atom?
number of protons in an atom
what is the mass number of an atom?
number of protons + number of neutrons
how do you work out neutrons in an element?
protons - electrons
why are atoms neutrally charged?
as there are the same number of protons and electrons their charges cancel out
how many electrons can go in each shell?
2,8,8,2
how did Dalton describe atoms
as solid spheres
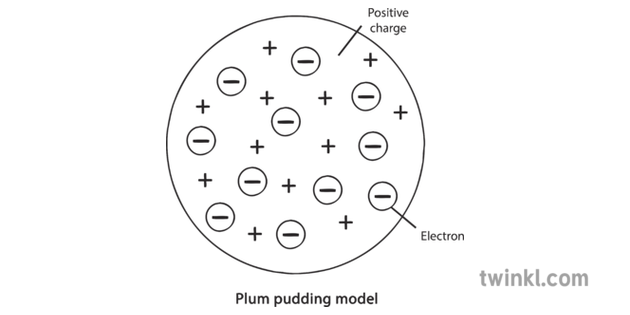
describe the plum pudding model of the atom
a cloud of positive charge with negative electrons embedded throughout it
what did the gold foil experiment prove?
the atoms have dense nuclei with a positive charge
what did Chadwick discover?
the neutron
what did Bohr’s experiment show?
that electrons are in specific shells
approximately how large are atoms?
radius is about 0.1nm
what are Isotopes?
atoms of the same element with the same number of protons but a different number of neutronsh
how is the modern periodic table arranged?
by their atomic number and in groups according to chemical properties
before the discovery of protons, electrons and neutrons how did scientists organise the elements?
by their atomic weight
why did Mendeleev leave gaps in his periodic table?
for elements that had not been discovered yet
what is group 1 called?
noble gases
what is group 7 called?
halogens
what is group 1 called
alkali metals
why are the group 0 elements unreactive?
they have full outer shells so do not need to lose or gain electrons
how does the boiling point of group 0 elements change down the group?
increases down the group
what happens to the electrons in a covalent bond?
they are shared
what two types of substances have covalent bonds?
giant covalent substances and small molecules
how many bonds does each carbon have in each diamond?
4
explain why diamond and silicon dioxide have high melting points?
giant structures, many strong covalent between the atoms, requires a lot of energy to break
explain why most covalent substances do not conduct electricity?
there are no electrons or ions that are free to move and carry charge
explain why graphite can act as a lubricant?
weak forces between layers which are free to slide over each other
what type of substances are methane and water?
small molecules
describe the structure of small molecules?
strong covalent bonds between atoms, weak intermolecular forces holding the molecules together
explain why small molecules have low melting points?
require little energy to break
what is a polymer?
many small molecules, monomers, joined together in a chain to form a large molecule
what are the three types of bond?
covalent, ionic, metallic
what happens to the electrons in an ionic bond?
they are transferred
what type of metals will form ionic bonds
non-metal and metal
what is the charge on elements from group 1 and 2?
group 1: 1+ group 2: 2+
what is the charge on elements from group 6 and 7?
group 6: 2- group 7: 1-
describe the structure and ionic bonding in an ionic compound?
giant ionic lattice held together by strong electrostatic forces of attraction which require a lot of energy to break
describe the structure and bonding in a metal?
layers of positively charged metal ions with delocalised electrons
why are metals good conductors?
delocalised electrons are free to move through the structure and carry charge
what is an alloy?
a mixture of a metal with another element
why are alloys harder than pure metals?
the layers are disrupted and so not able to slide making the alloy harder than a pure metal
why do larger molecules have higher melting points than smaller ones?
intermolecular force strengthens with increased molecular size
what is graphene and what is it used for?
one layer of graphite- electronics and composite materials
what are nanotubes and what are they used for?
electronics, nanotechnology and materials