pmcol final pt 2
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
90 Terms
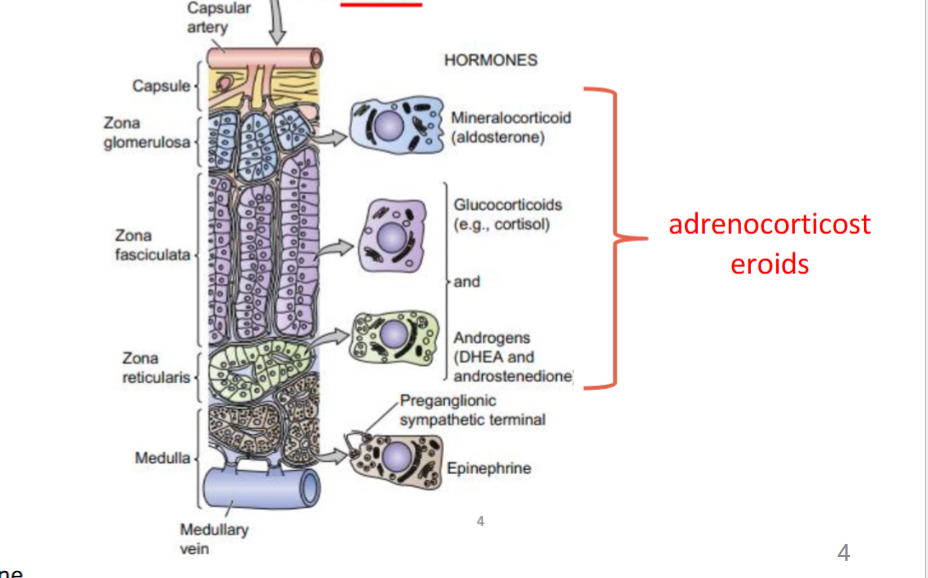
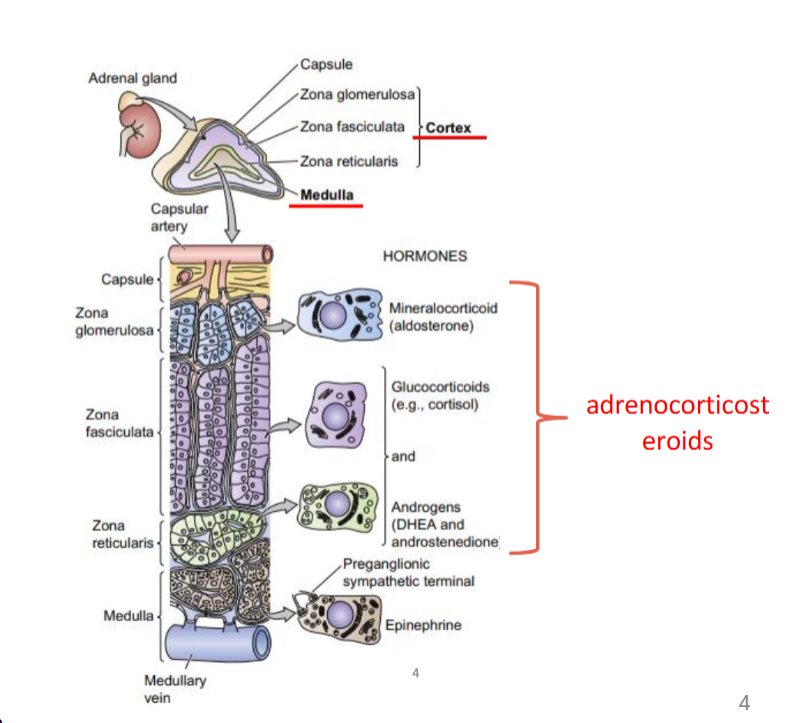
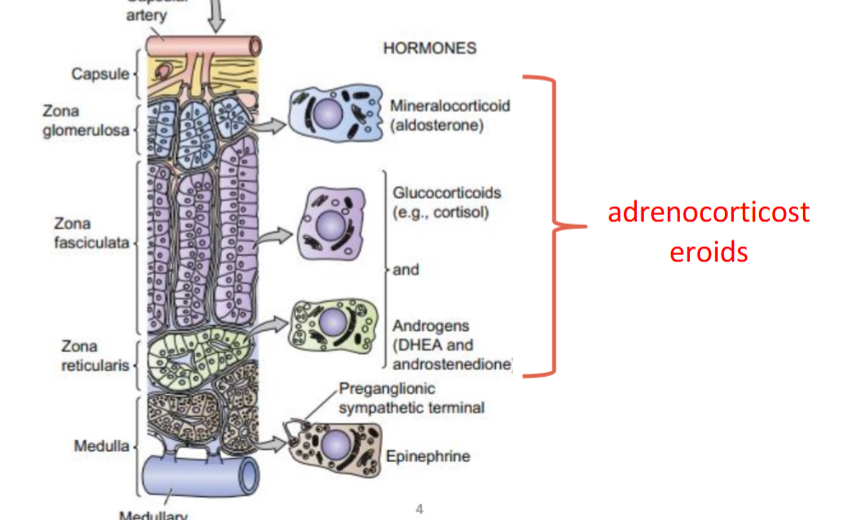
adrenocorticosteroids
steroids secreted by adrenal glands in cortex
three categories of adrenocorticosteroids + examples
glucosteroids: cortisol
mineralocorticoids: aldoesterone
adrenal androgens: DHEA, androstenodione (testosterone and estrogen)
what is the major source of estrogen post menopause
androsteonodione
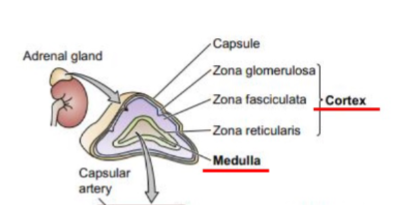
layers of adrenal cortex
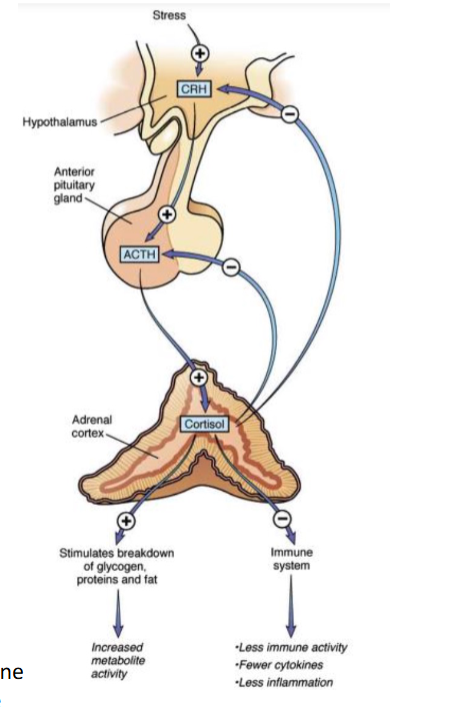
how are adrenocorticosteroids regulated
stress amplifies CRH release from hypothalamus
CRH stimulate anterior pituitary to release ACTH
ACTH act on adrenal cortex to stimulate cortisol
negative feed back of CRH and ACTH when done
zona glomerulosa
mineralocorticoid release
zona fasciculata
glucocorticoids and androgens release
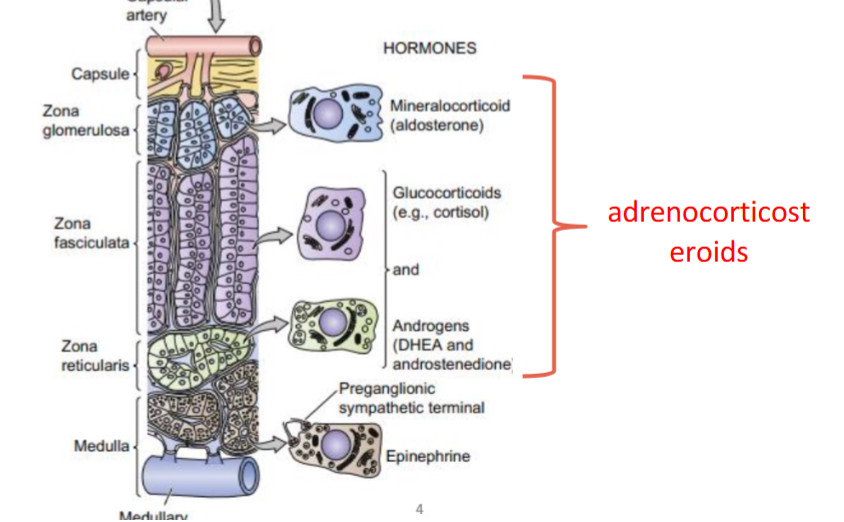
zona reticularis
glucocorticoids and androgens release
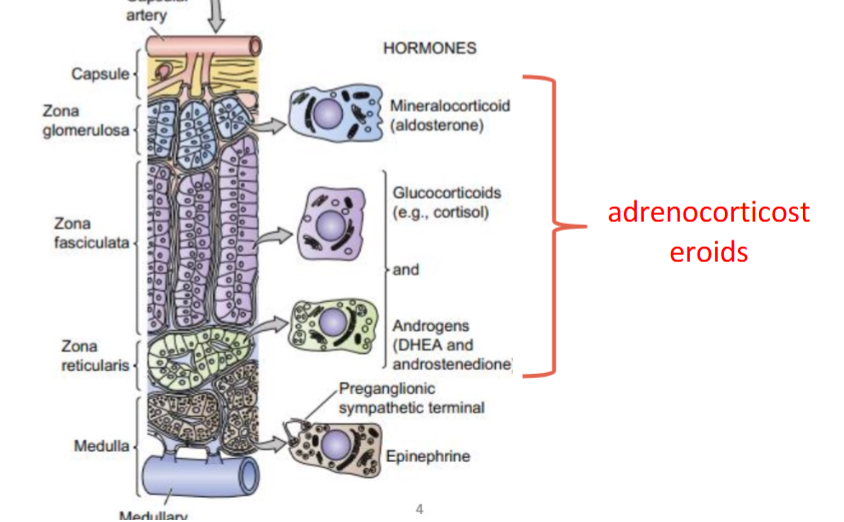
where does cholesterol come from
LDL
how are aderenocorticosteroids made
cholesterol turns to prenenolone in mitochondira (rate limiting step) under influence of ACTH
prenenolone diffuse back to cytosol and remainder of processing steps occur in SER
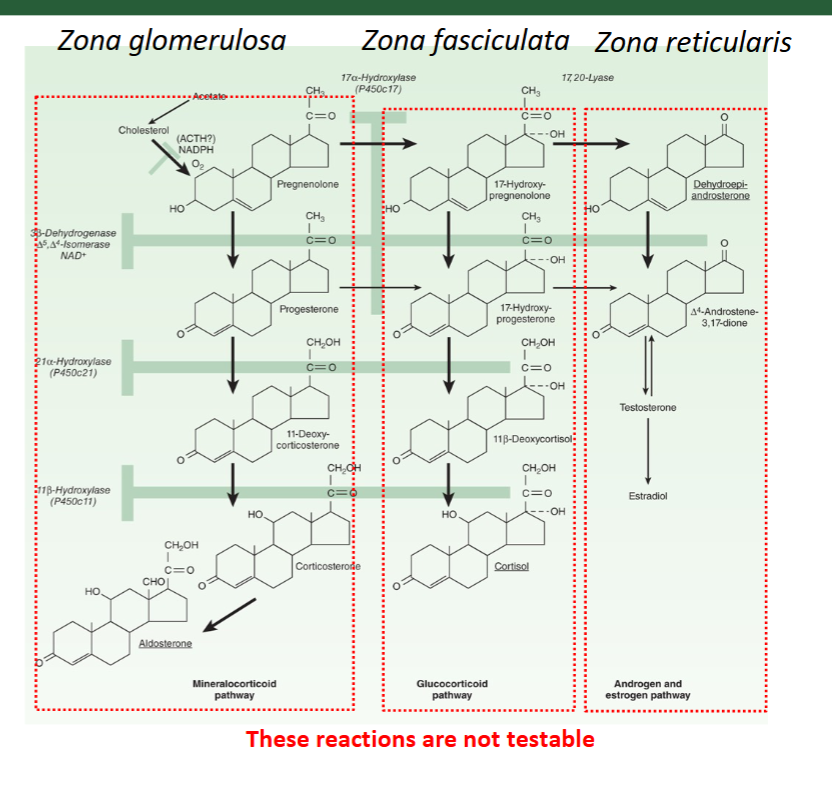
17 alpha hydroxylase is _____ expressed in the Zona glomerulosa
not; that is why prenenolone gets directed to mineralocorticoid path
metabolic effects of glucocorticoids on carbs
stimulate gluconeogenesis in liver → inhibit glucose uptake in muscle and adipose → increases plasma glucose
metabolic effects of glucocorticoids on proteins
breakdown muscle proteins to amino acids (used as substrates for gluconeogenesis → increase plasma glucose
metabolic effects of glucocorticoids on lipids
breaks down fats (lipolysis) to free fatty acids + glycerol (substrates for gluconeogenesis) → inc. plasma glucose
genomic path of glucocorticoids
induce lipocortin-1 (inhibit phospholipase A2)
inhibit arachidonic path → prostaglandin and leukotrine decrease
transpress inflammatory transcription factors + inhibit cytokine production + COX 2
inhibitory transcription factors (2)
NFkB + AP-1
cytokines that are inhibited due to genomic anti-inflammatory path
IL-2. IL-6, TNF-alpja
non genomic anti-inflammatory effects
cell membrane fluidity, inhibit MAPK and PI3K/ Akt path
how are the metabolic and anti-inflammatory effects of glucocorticoids granted
cortisol that circulates blood is bound to CBG and only free from can enter cells
signals glucocorticoid receptor to transcribe proteins involved in gluconeogenesis, lipolysis and inflammation suppression
what are three synthetic glucocorticoids
hydrocortisone
prednisone/prednisolone
dexamethasone
mechanism similar to endogenous cortisol
what are the 3 side effects of synthetic glucocorticoids
hyperglycemia/ early onset of diabetes
osteoporosis
risk of infections
where does aldosterone affect Na+ reabsorption
kidney, colon, sweat glands
where does aldosterone affect K+ excretion
kidney, colon
where does aldosterone affect H+ excretion
kidney
how does mineralocorticoids work/ signal
aldoesterone binds albumin to some extent in blood only free form can enter cells
signals via mineralocorticoid receptor —> ENac + Na-K ATPase → Na reabsorption to blood/ K+ excretion
H+ ATPase increased expression
although cortisol has affinity for MR, why is it ineffective in kidneys
because kidney expresses 11 beta-hydroxysteroid dehydrogenase type 2 that converts cortisol to cortisone that has no MR affinity
what are the 2 synthetic mineralocorticoids
fludrocortisone and desoxycorticosterone
mechanism similar to endogenous aldoesterone
what are the side effects of synthetic minteralocoticoids
hypolakemia, hypertension, metabolic alkalosis
addison disease + treatment
too little adrenocortical insufficiency → use hydrocortisone + fludrocortisone
Cushing syndrome + treatment
adrenal cortex produce excessive levels of cortisol
surgical removal/ radiotion for tumor, ketoconazole + metyrapne
ketoconazole
broad effect on all hormones ; non selective
used for Cushing
metyrapone
blocks final step in cortisol synthesis; selective treatment for Cushing
primary aldosteronism + treatment
adrenal cortex produce excessive aldoesterone leads to hypertension
spironolactone + eplerenone
metastasis
abnormal cell growth with potential to spread to other parts of the body
cancer fundamentally
these cells are heritable
G1 Phase
checkpoint to ensure cell is ready for DNA synthesis
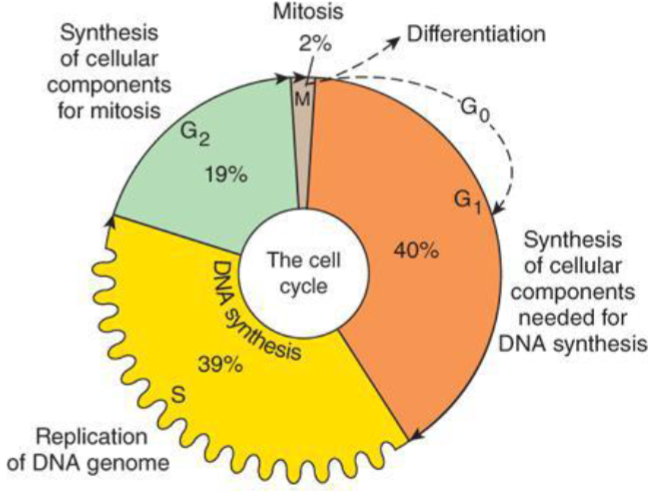
S Phase
DNA synthesis (many of cancer drugs show efficacy)
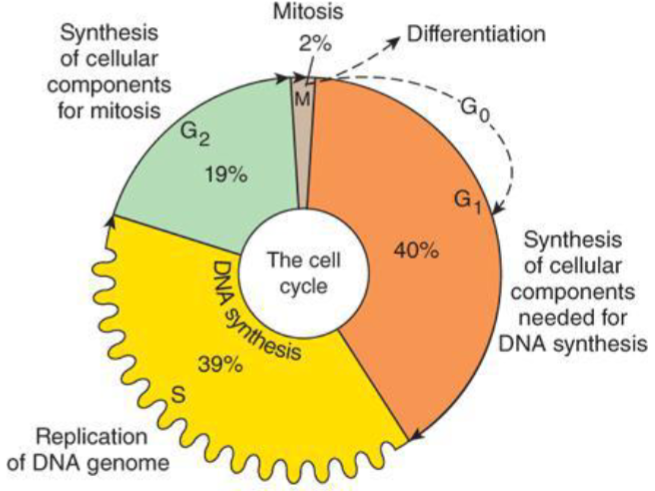
G2 phase
Checkpoint to ensure cell ready for mitosis
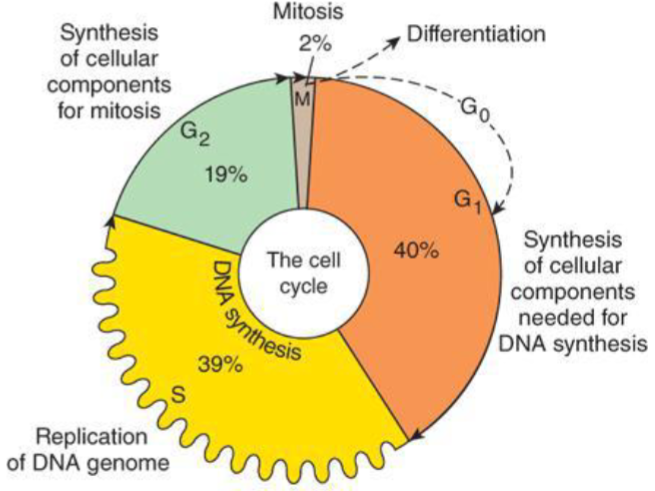
M phase
mitotic phase where cell divides (another important target for cacner drugs)
G0 phase
quiescent/ dormacy phase
what are the 2 groups that genes importnat for regulating cell cycle
tumor suppressor genes
protooncogenes
tumor suppressor genes
repress cell cycle or promote apoptosis + inhibit cell division following irreversible DNA damage
BRCA
DNA repair proteins (part of tumor suppressor genes)
p53
tumor suppressor protein that marks damage and slows s phase down (is mutated in 50% of cancer)
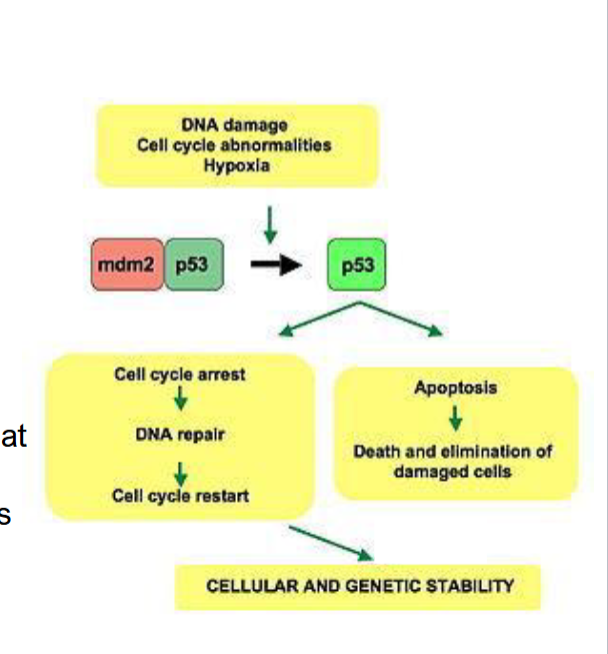
oncogenes
mutated forms of normal genes that cause normal cells to grow out of control → cancer cells
proto-oncognees
genes that normally control how often a cell divides and the degree to which it differntiates or specializes
what happens when proto-oncogene turns to an oncogene
it becomes permanently turned on/ activated when its not supposed to be
how does proto-oncogene become an oncogene
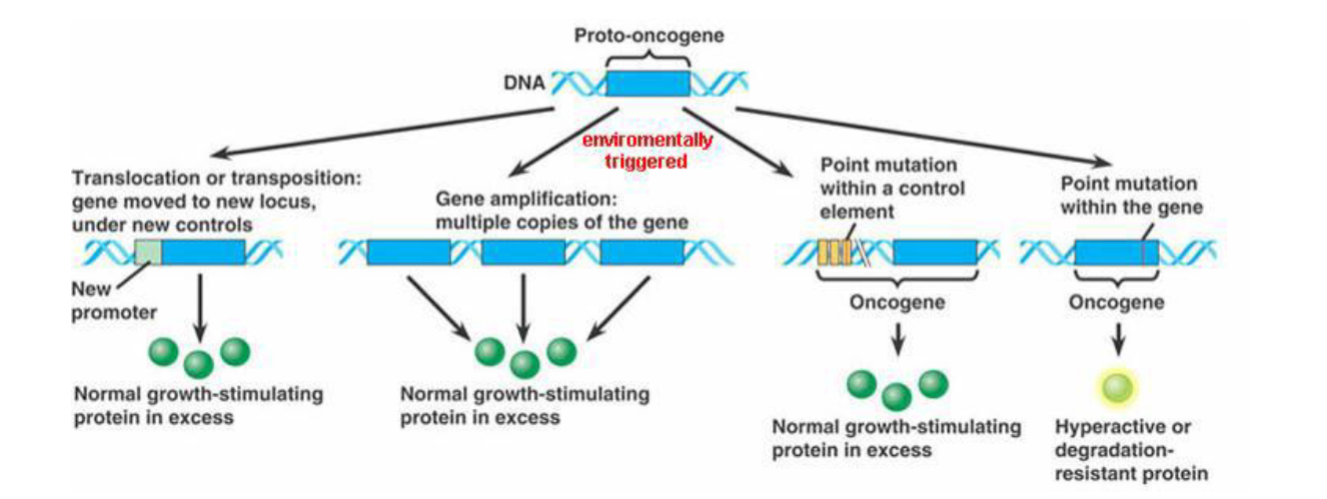
what are the major 4 classes of oncogenes
growth factors and their receptors
signal transducers (between cell receptors and nucleus)
transscription factors/ nuclear transducers
programmed cell death regulators
neoplasms
cells that exhibit combinations of mutations
Knudson’s multiple-hit hypothesis
environmental factors hit genes to become faulty
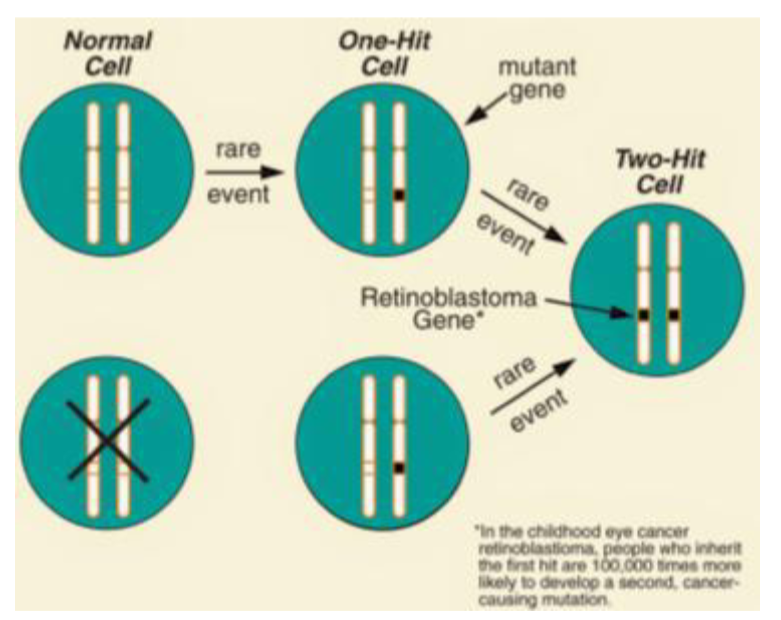
what happens at the time of 30 cell doublings + another 10 doublings
2 cm diameter tumor, can be diagnosed by unnoticed in many organs
20 cm in diameter → lethal
therefore neoplasm is silent for ¾ of its existence
cancer is often advacned by what
time of detection
fundamental issue of cancer regarding cell number
you cannot leave any cancer cells behind after treatment ends
drugs will not have 100% efficacy maybe 99.99% (could still have 10 million cells left over!) → why surgery is best
what are some basic treatments for cancer
1/3 are treated with local treatment (surgery + radiotherapy). some use anti-cancer drugs due to metastasis
fundamental issue of cancer therapy regarding cells
they are your own cells! → a little protection because your immune system wont touch them (immune to immune activity)
therapies have narrow theraputic window because they are meant to be cytotoxic
what are some other cells that cehmotheraputic drugs attack
liver, and GIT because they rapidly divide
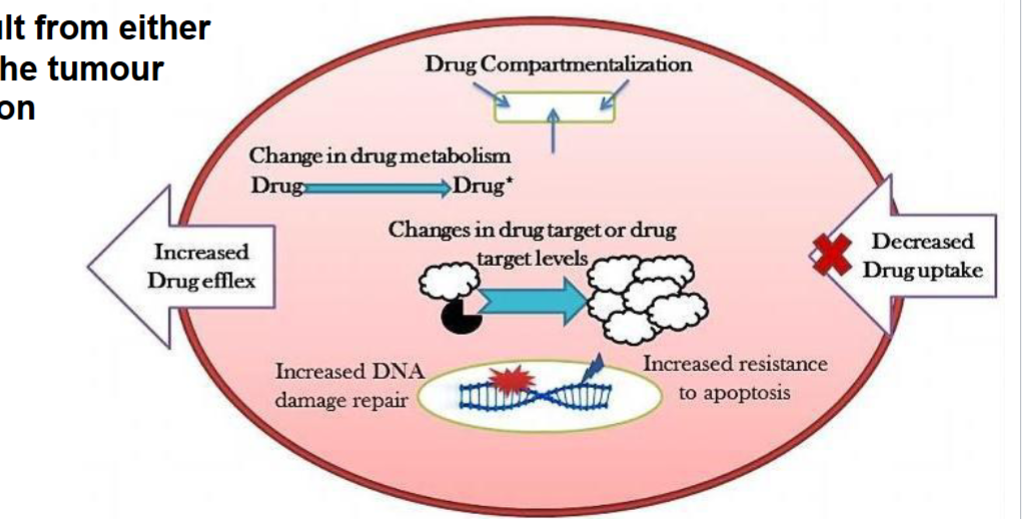
fundamental issue with cancer therapy regarding drug resistance (2 types)
primary drug resistance: present when drug is given (elevated TP proteins)
acquired drug resistance: result from adaptation from tumor cells or mutation (antibodies cannot bind)
if you kill the non-resistant cells you will give room to resistant cells to crazy proliferate
alkylating agents
nitrogen mustards + Cisplatin
cisplatin
aklylating agent that causes severe nausea and vomiting → can be nephrotoxic
revolutionalized treatment of germ cell tumors
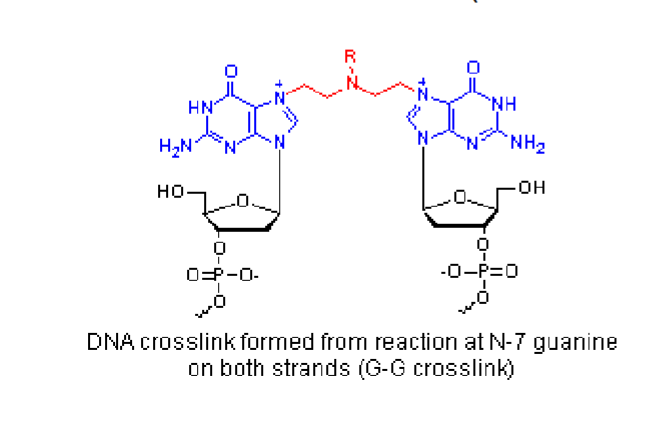
how do alkylating agents work
form higly reactive electrophilic intermediates and bind covalently to DNA (esp. N7 guanine)
bifuncitonal to cross link DNA strands
when are cancer cells most susceptible to alkylating agents
in late G1 and S phase due to abundance of DNA targets
anti-metabilite chemotheraputics
mimics natural metabolites → interfere with biosynthetic paths → inhibt nucleic acid synthesis
what are some anti-metabolite chemotheraputics 3
anti-folates
pyrimidine analogues
purine analogues
methotraxate
(MTX) analog for folic acid
converted to polyglutamates (pG-MTX)
together MTX and pG-MTX halt de novo nt biosynthesis
3 pyrimidine base analogues
cytosine arbinoside(nucleoside)
gemicitbine (nucleoside)
fluorouracil
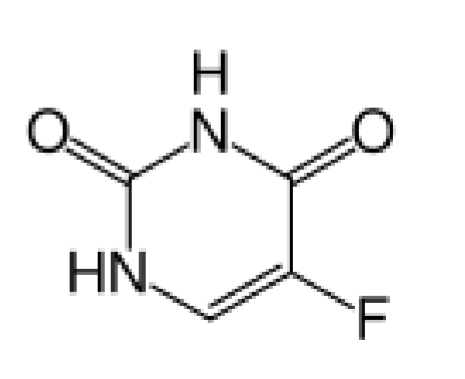
fluorouracil
nucleobase analogue
converted to F(UTP) and encorporated in RNA → slow translation
converted to dTMP which inhibits DNA synthesis
what are purine nucleobase analogues used for mainly
acute lymphoblastic leukemia
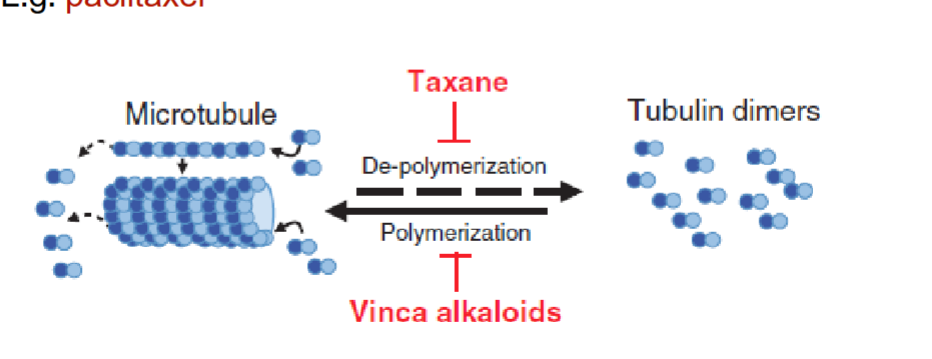
Vinca alkaloids
natural chemotheraputic derived from periwinkle plant
inhibits tubulin polymerization → distrupts assemplt of microtublules involed in mitiotic spindle M phase
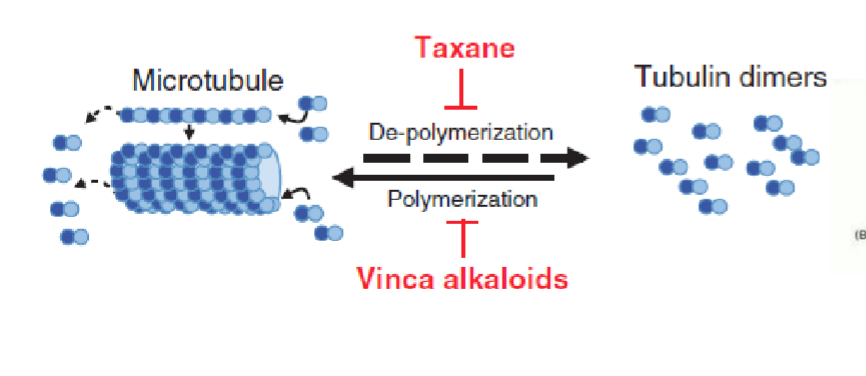
paclitaxel
(taxanes) natural chemo agent from Pacific yew tree
promotes high affinity microtubule assemnly → affects M phase
camptothecins
bind and stabilize the short lasting DNA -topisomerase I → cannot re-ligate = accumulate single stranded breaks
S-phase (where DNA replication is happening)
antibiotics for cancer
doxorubin
bind DNA through intercalation = block DNA synthesis and cell replication
what are the 4 mechanisms of action for doxorubin
main focus
inhibit topisomerase
generate free radicals → stress → apoptosis
high binding affinity for DNA
bind cellular membrane to alter fluidity and ion TP
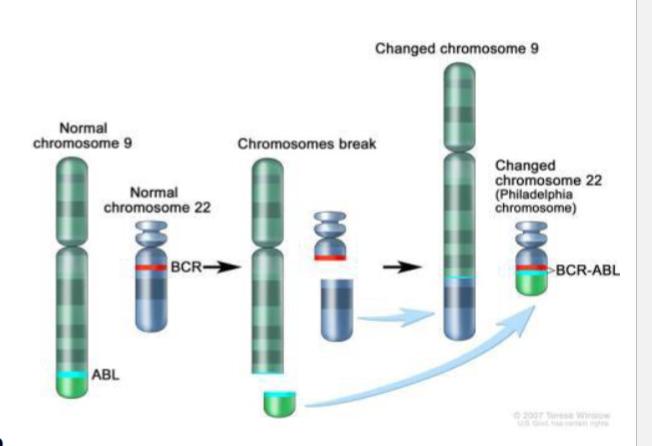
tyrosine kinase inhibitors
used to treat a type of leukemia containing Philadelphia chromosome
inhibits tyrosine kinase domain of Bcr-Abl oncoprotein
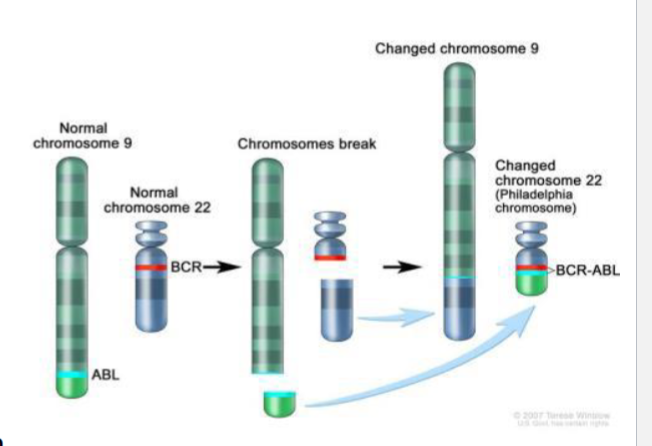
Philidelphia chromosome
specific genetic abonormality where choromosome 22 (BCR gene) and chormosome 9 (ABL 1 gene) combine
BCR-ABL leads to unregulated expression of protein tyrosine kinase activity → unreg cell division
cetuximab
a monoclonal antibody against extracellular domain of EGFR (inhibitor)
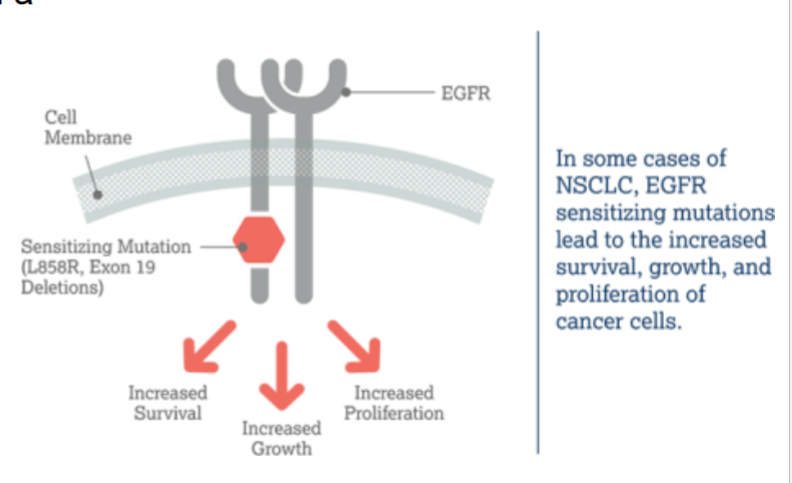
EGFR in cancer
over-expressed in a number of solid cancers and promotes cell growth, invasion and metastasis + angiogenesis (new blood vessels)
this dimerizes upon binding
tamoxifen
hormonal anti-cancer agent that is a selective estrogen receptor antagonist
blocks estrogen binding in estrogen sensitive cancer cells in breast tissue
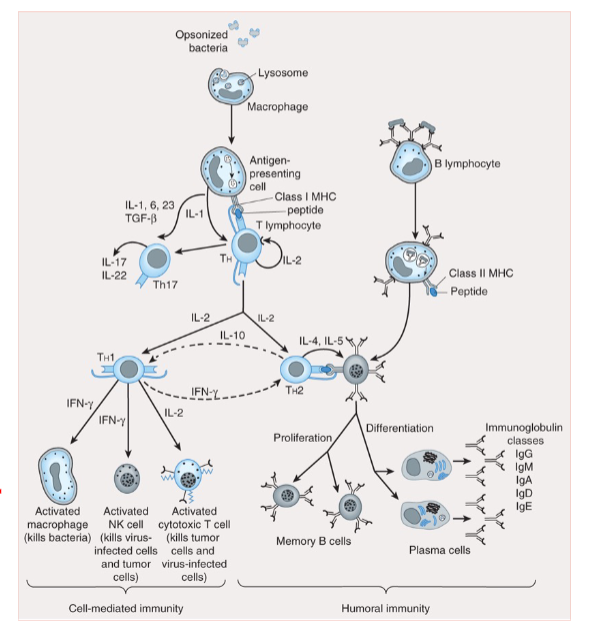
innate immune system
first line of defense against pathogens non-specific + always exists
skin/ mucous
enzymes (lysozyme, acid hydrolases)
immune cells: neutrophils, macrophages, natural killer cells, complement system
adaptive immune system guys
antigen presenting cells (dendritic cells and macrophages)
T lymphocytes
B lymphocytes (antibodies)
adaptive immune system
responds to pathogens in an antigen specific manner and previous antigen via memory response
3 ways to modulate immune system to our advantage
suppress it when immune response is unintended/ exaggerated
stimulate it when immune response is dampened
manipulate it for targeted immune response
immunosuppresants
supress unwanted effects of:
solid organ/ bone marrow transplant rejection
autoimmune diseases
hypersensitivity/ allergy
immune rejection is ____ as GVHD
not the same (host immune attack donor) ; graft versus host disease (donor immune attack host)
immunomodulators/ stimulants
activate immune to eliminate tumors
cancer immunotherapy (only cancer cells)
calcineurin inhibitors
immunosuppresant → cyclosporin A + Tarcolimus
removes phosphate (autocrine ligand is IL2 and IL2 secretion)
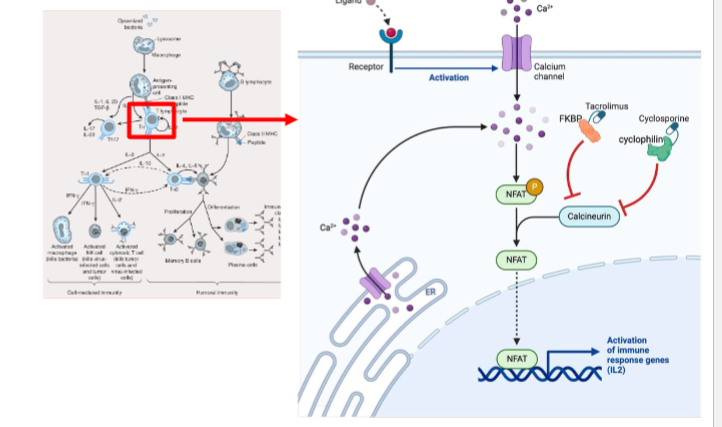
cyclosporin A
calcinuerin inhibitor that binds cyclophillin
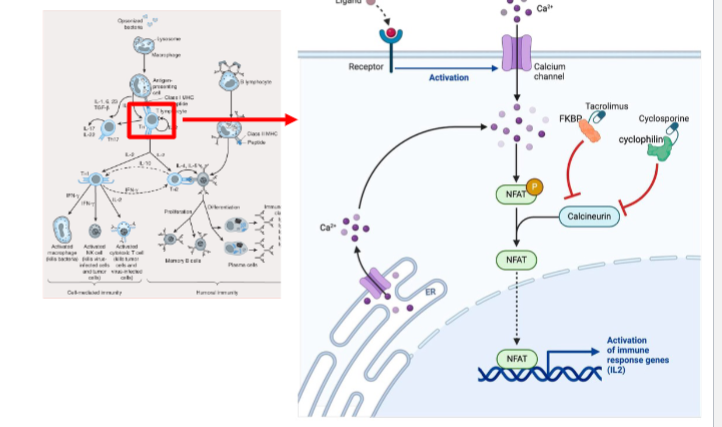
tarcolimus
calcineurin inhibitor that binds FKBP → inhibit calcineurin (phosphatase) → inhibit NFAT translocation to nucleus → inhibit IL-2 production
more potent than cyclosporin
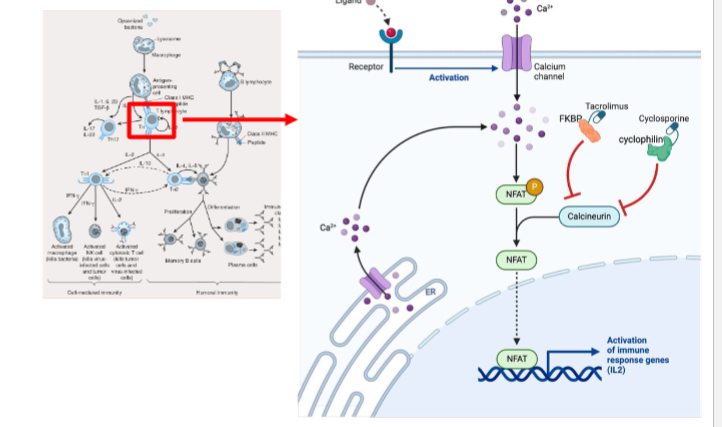
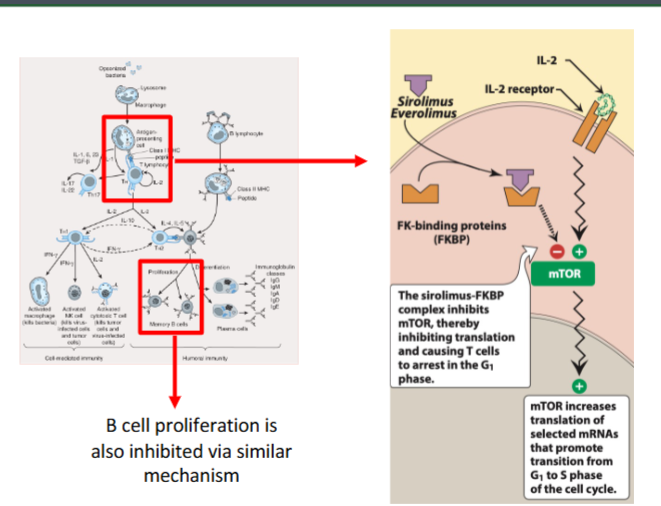
proliferation signal inhibitors
immunosuppresant → sirolimus + everolimus
binds to FKBP → inhibit mTOR → inhibit T cell proliferation (prevent cells from going