free radical substitution of alkanes
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
what is a free radical/radical
any species with an unpaired electron
shown as a dot
highly reactive species - react with alkanes
reactions involving free radicals are called
free radical substitution
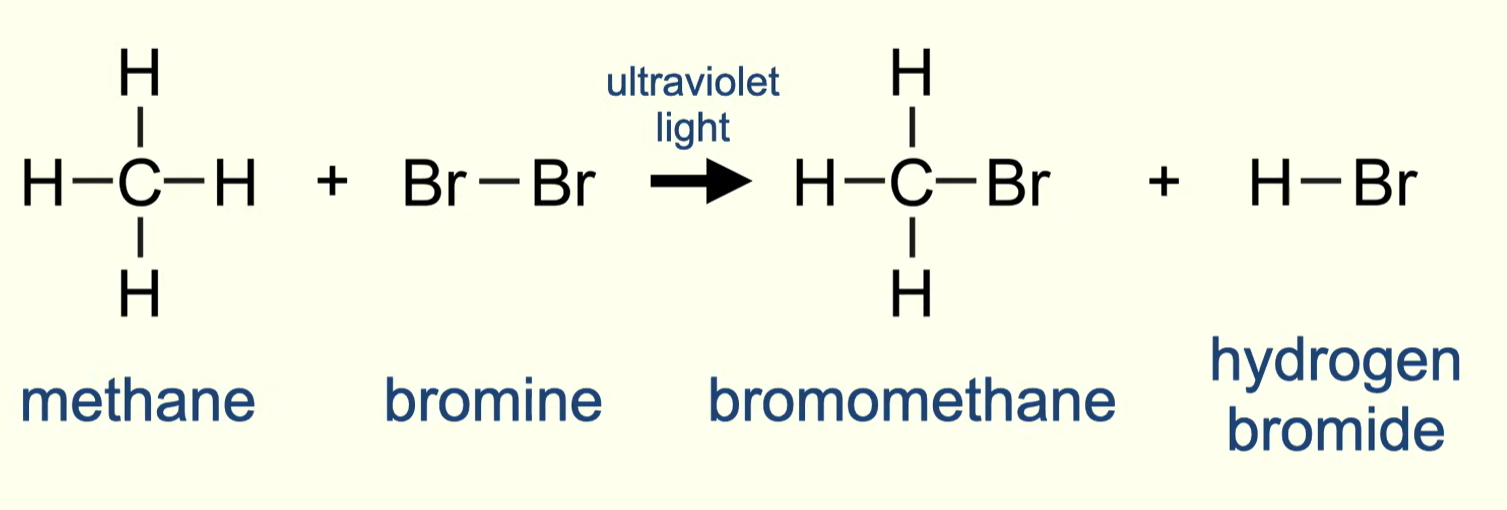
stages for reaction between any alkane and chlorine/bromine
3 stages:
initiation
propagation
termination
describe the initiation between any alkane and chlorine/bromine
shine ultraviolet light onto reaction mix
energy causes single covalent bond between two chlorine/bromine atoms to break
one electron now goes to each chlorine/bromine atom - these are now chlorine/bromine radicals. this is called homolytic fission
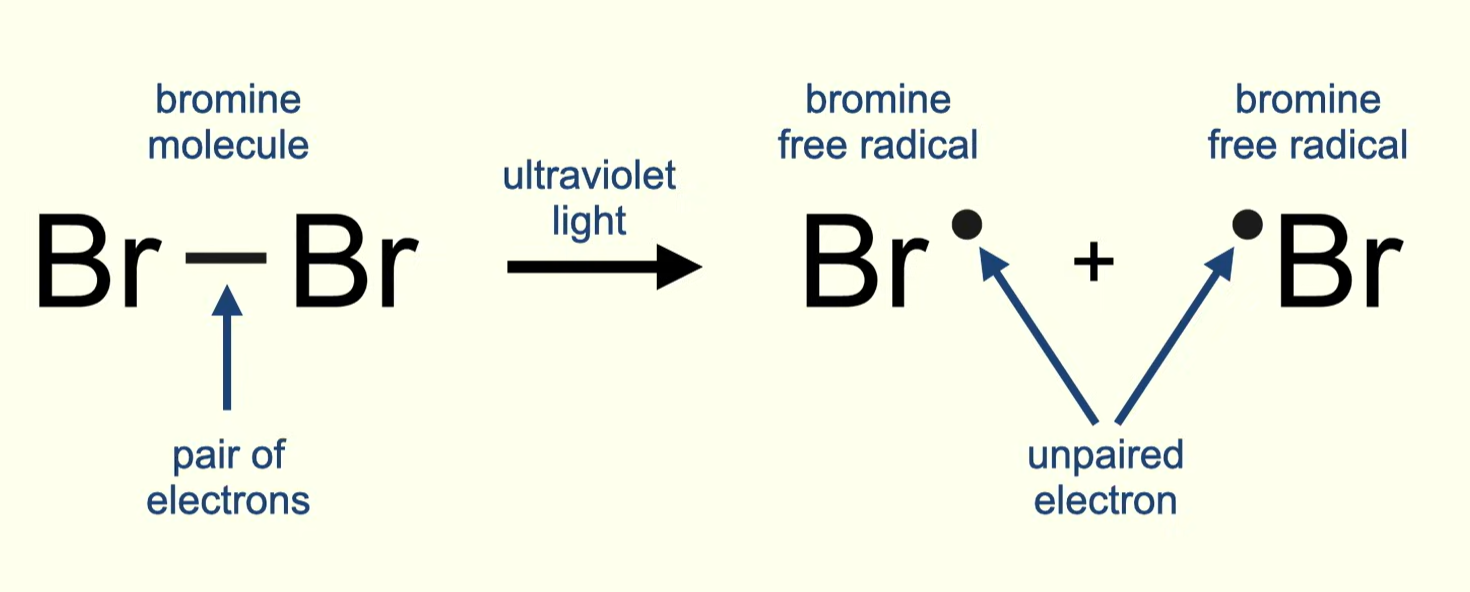
what is homolytic fission
in an initiation reaction, one electron now goes to each chlorine/bromine atom - these are now chlorine/bromine radicals
describe the STEP 1 propagation between any alkane and chlorine/bromine
a chlorine/bromine radical reacts with an alkane molecule
the radical takes a hydrogen atom plus one electron from the alkane
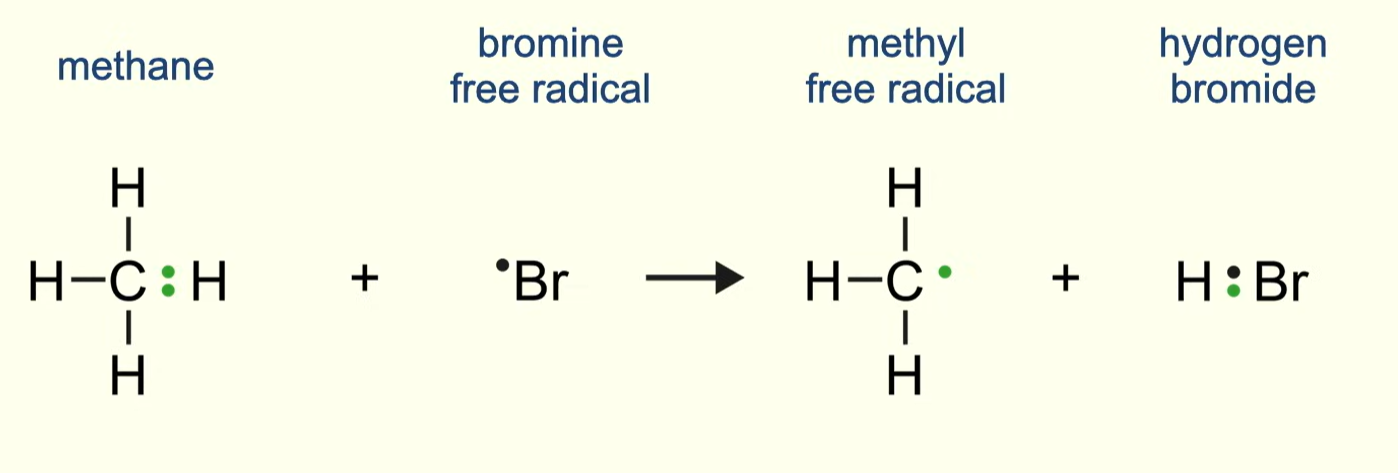
describe the STEP 2 propagation between any alkane and chlorine/bromine
the alkane radical now reacts with the bromine/chlorine molecule producing a bromo/chloroalkane and a chlorine or bromine radical
propagation steps 1 and 2 form a chain reaction - the radical formed in step 2 can react with the alkane in step 1 - this continues until termination
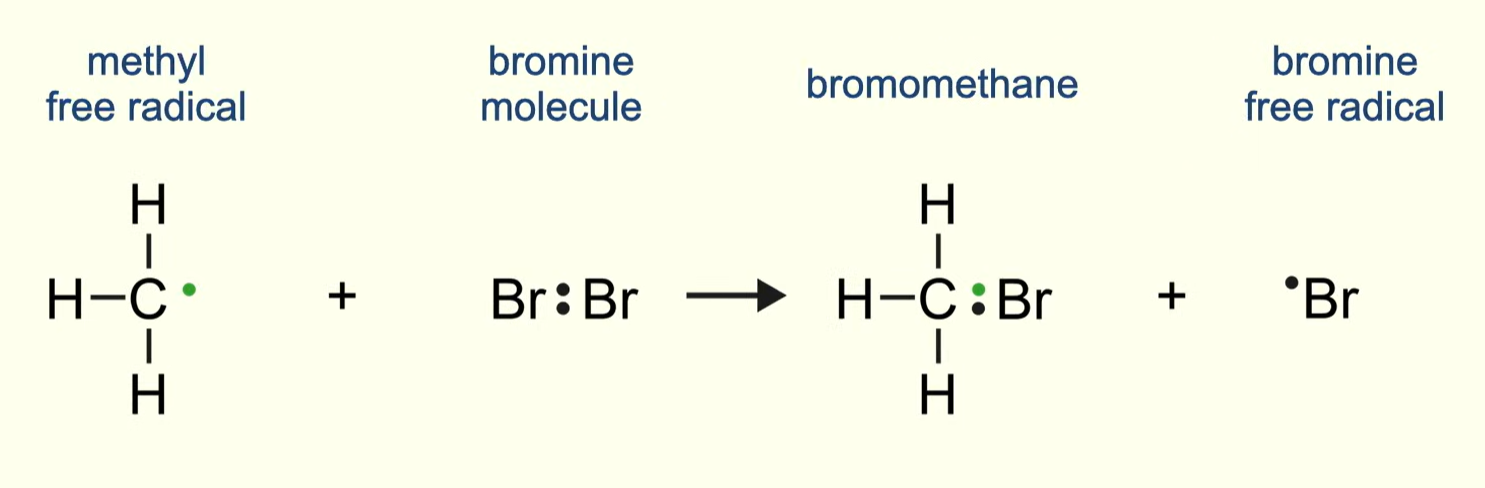
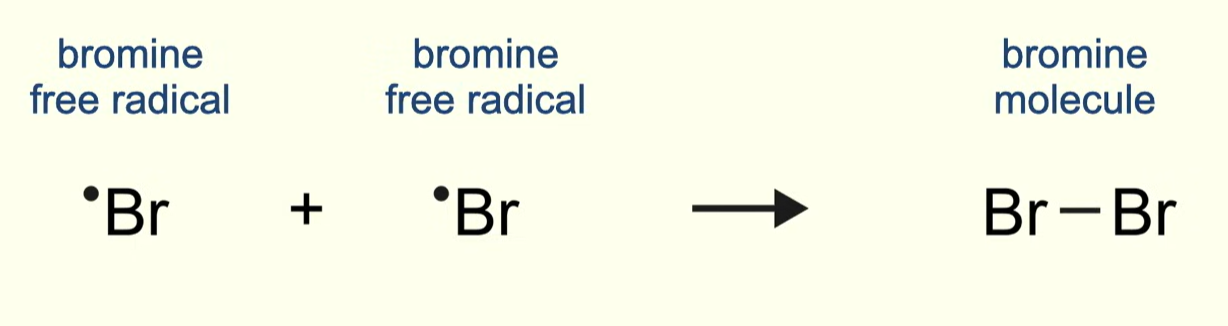
describe termination between any alkane and chlorine/bromine
two free radicals react together to form a molecule with no unpaired electrons
problem wit free radical substitution
can form a wide range of side products, including different isomers - need to separate products at end