IB Biology HL: Proteins
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
Proteins
Large, complex molecules consisting of amino acid residues joined by peptide bonds. account for 50% of the dry weight of most cells.
Structural
Type of protein used for support (collagen, keratin).
Storage
Type of protein used for storing amino acids (casein, plant seeds).
Transport
Type of protein used for moving other substances (hemoglobin).
Hormonal
Type of protein used for the coordination of an organism's activities (insulin).
Receptor
Type of protein used for the response of cell to chemical stimuli.
Contractile
Type of protein used for movement (actin, myosin).
Defensive
Type of protein used for protection against disease (antibodies).
Enzymatic
Type of protein used to accelerate chemical reactions (enzymes).
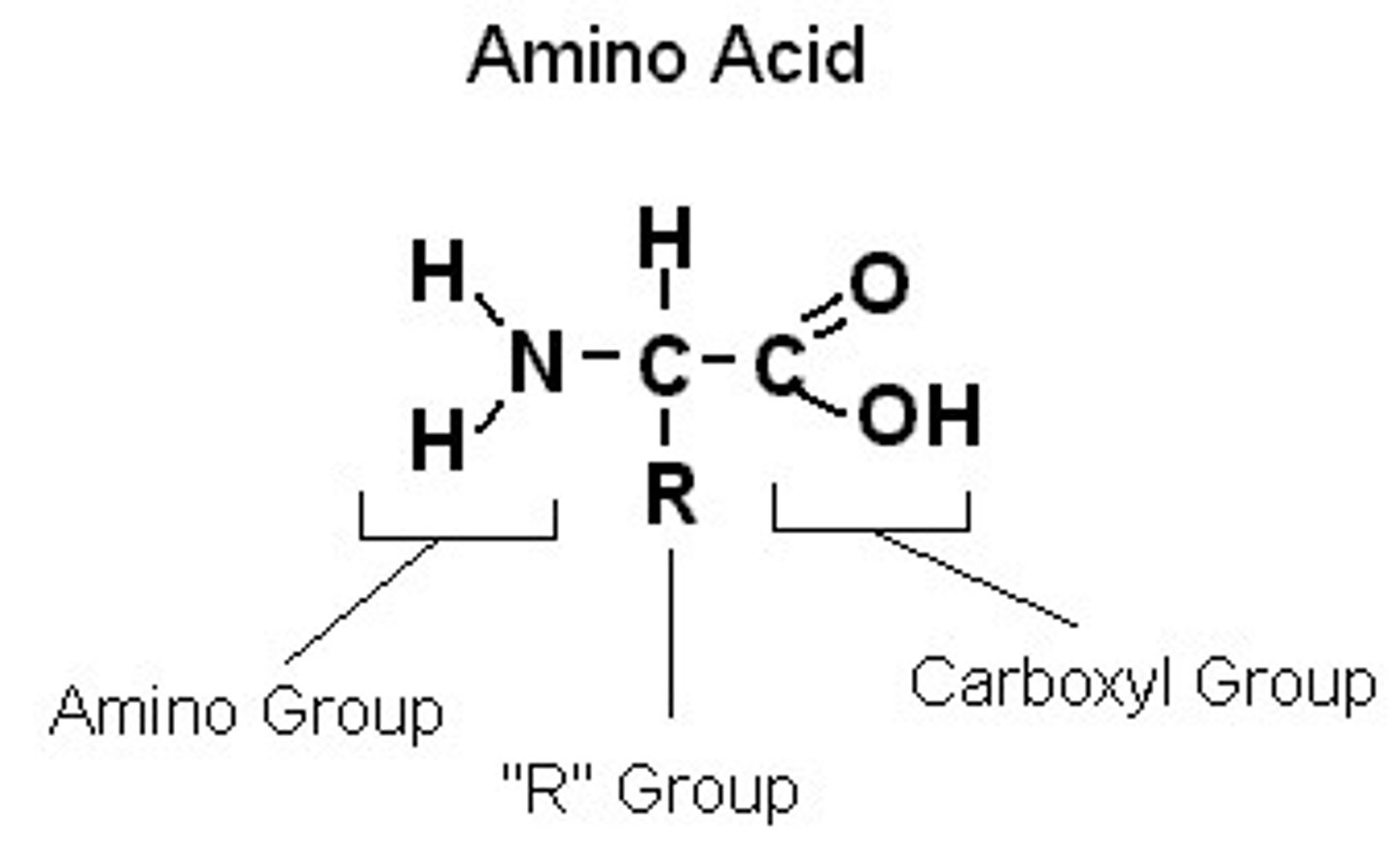
Amino Acids
Building blocks of proteins.
Essential Amino acids
Amino acids required in the human diet and cannot be synthesised from other metabolites
R-group
A functional group that defines a particular amino acid and gives it special properties.
Polypeptides
Polymers of amino acids. Joined together by peptide bonds.
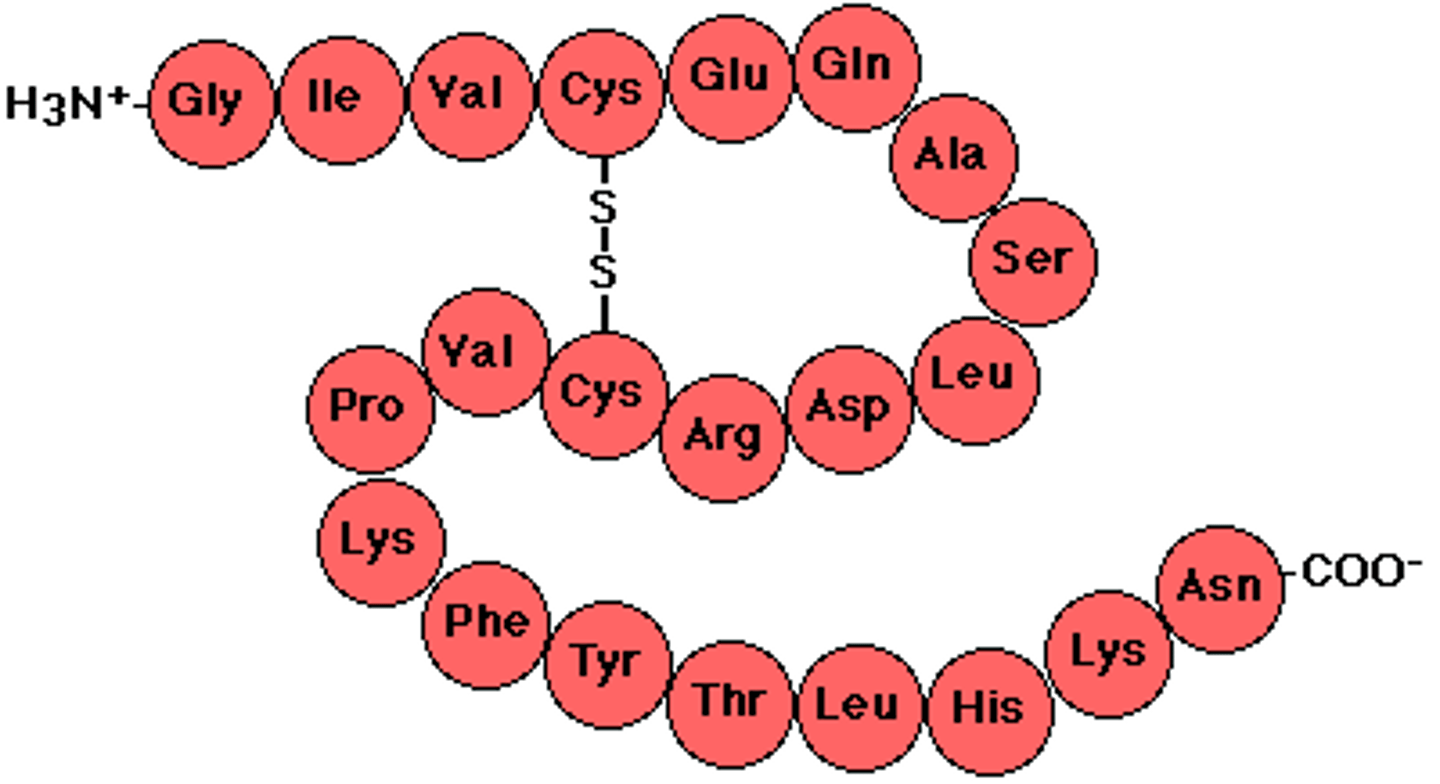
Explain how the amino acid sequence detremines the 3D conformation of apolypeptide?
Bonds between amino acids’ R-groups hold the final conformation
Hydrophilic amino acids end up on the edges
the sequence of amino acids influence folding
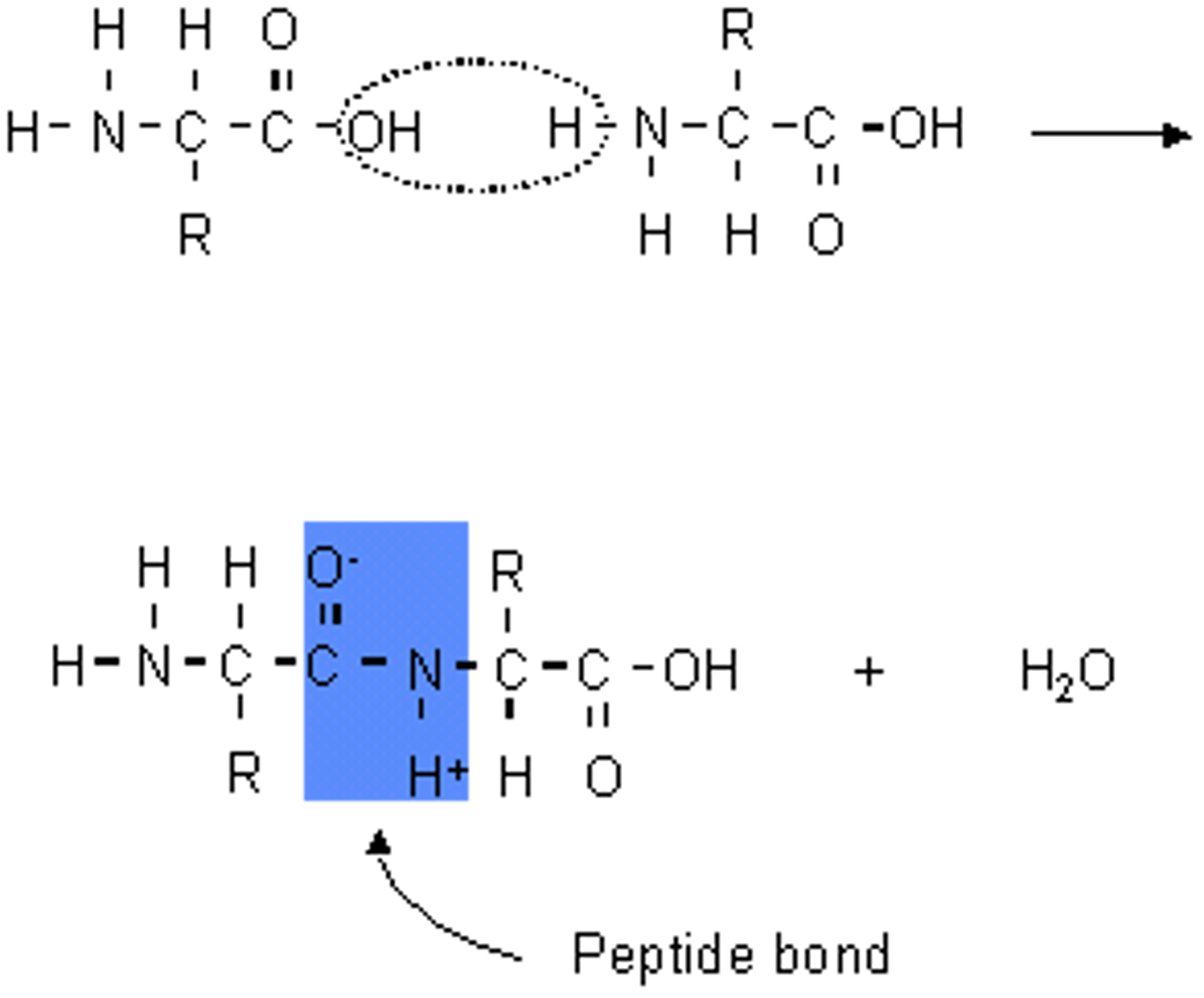
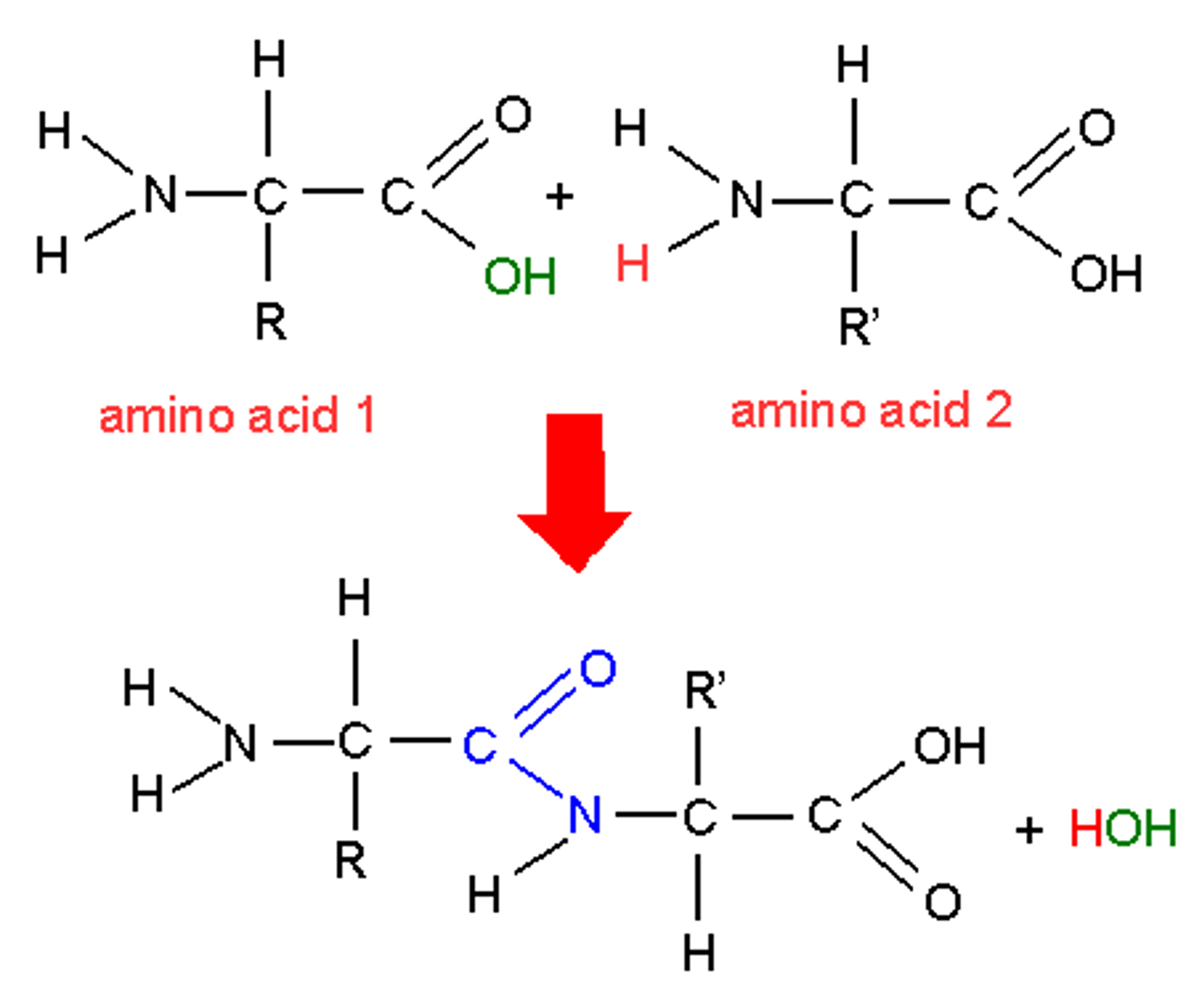
Peptide Bond
The chemical bond that forms between the carboxyl group of one amino acid and the amino group of another amino acid.
Forms dipeptide and water
What kind of reaction is breaking down polypeptides into amino acids ?
Hydrolysis - it requires water
There is an ______ variety of polypeptide chains
Infinite
Dipeptide
Two amino acids bonded together.
Carboxyl Group
Part of an animo acid that reacts with another amino acid's amine group.
N and C
Elements between which a peptide bond is formed.
Covalent
Type of bond a peptide bond is.
Ribosomes
Site of protein synthesis- facilitate the formation of peptide bonds
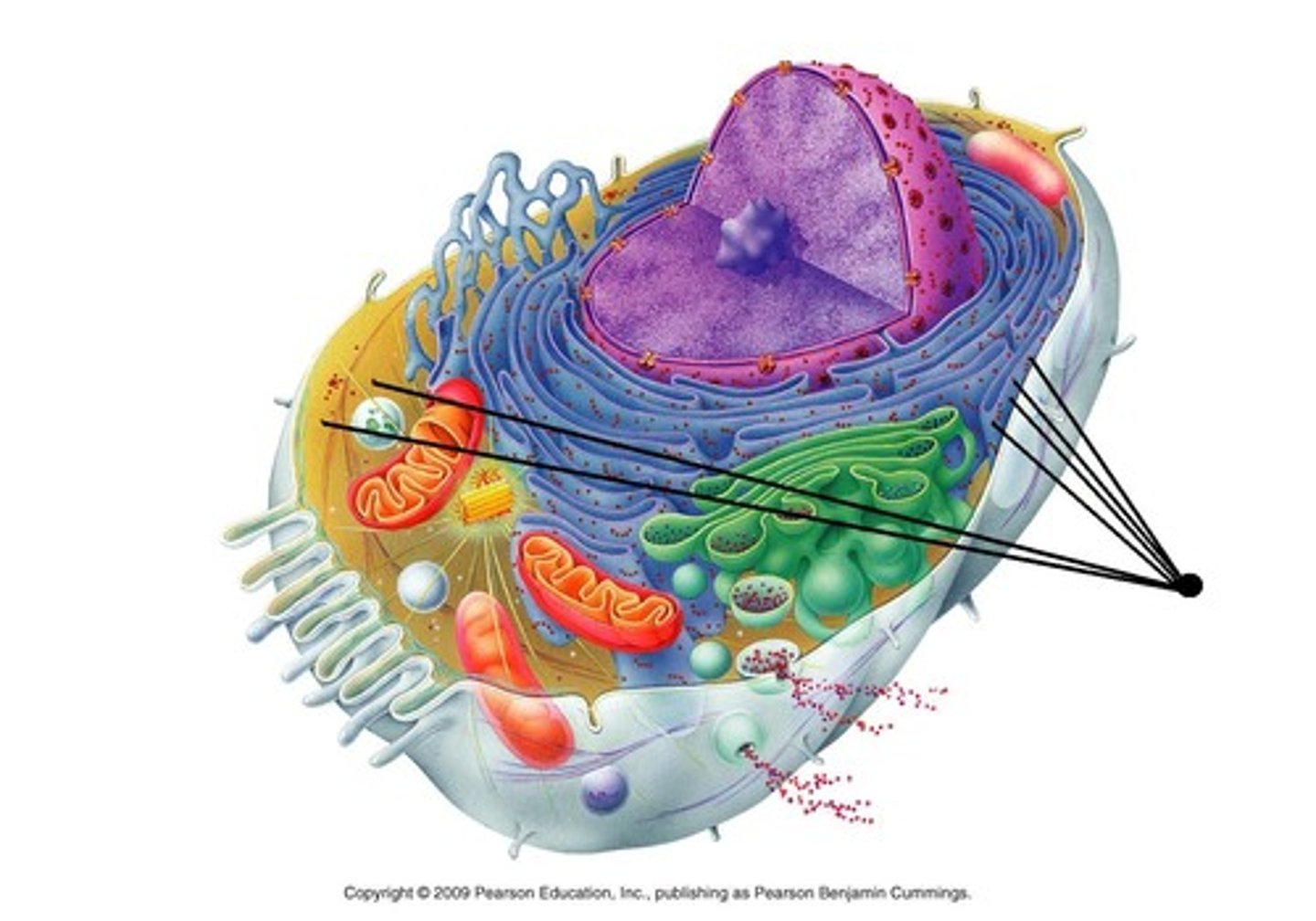
DNA
Provides information for construction of polypeptides.
1
Proteins are made up of at least ___ polypeptide.
Protein _______ determines function
shape
Primary Structure
Unique sequence of amino acids in a protein- determined by DNA
- Specific order will determine how the protein folds. Amino acids in a chain bonded to each other with peptide bonds.
- To avoid tangling and breaking parts of the chain are stabilised as they are made (Hydrogen bonds, ionic bonds, hydrophic interactions and disulfide bonds)

Sickle Cell Anemia
Genetic disorder resulting from a single change in the amino acid sequence of a cell.

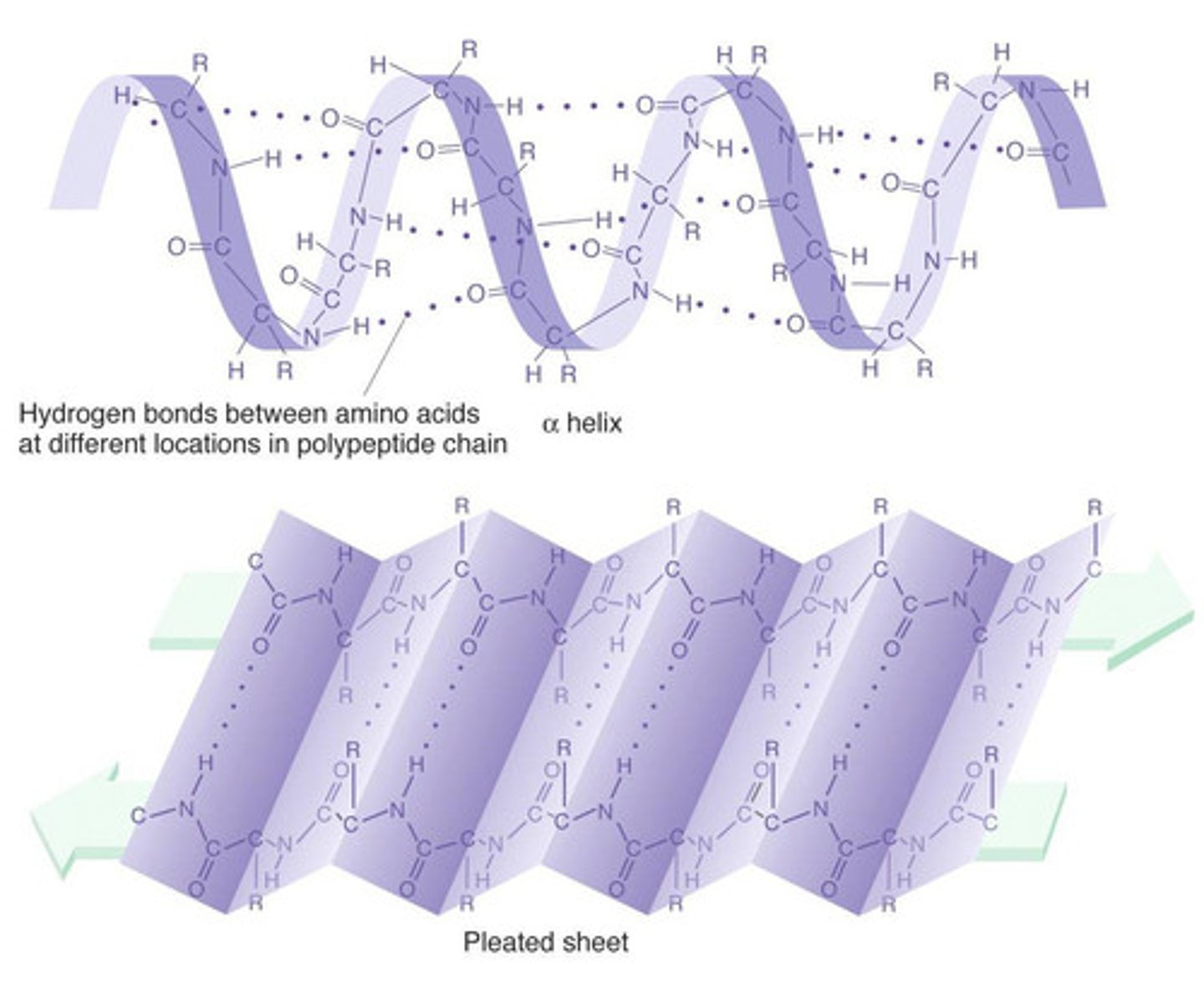
Secondary Structure
Alpha helix = hydrogen bonds formed between coiling chain
Beta pleated sheets= parallel hydrogen bonds between folded chain
Hydrogen bonds between C=O and N-H
Sidechain
The portion of amino acids not included in the backbone of a protein.
Alpha-Helix
A spiral shape constituting one form of the secondary structure of proteins, arising from a specific hydrogen-bonding structure.
Beta-Pleated Sheet
the polypeptide chain folds back and forth and the fold are held together by parallel hydrogen bonds.
In an alpha-helix, hydrogen bonds exist between every _____ amino acid.
4th
In a beta-pleated sheet, hydrogen bonds form between parts of the backbone in __________ regions.
parallel
Teritary Structure
folding of a polypeptide chain into a 3d conformation
hydrogen bonds between polar r-groups
hydrophobic interaction between non-polar R-groups
covalent bonds/ dissulfide bridges between R-groups of cysteine amino acids
Ionic bonds between R-groups with opposite charges
This gives protein specific shape - function
Hydrophobic Interactions
Clustering of hydrophobic R-groups away from water
Hydrogen Bonds
Very weak bonds;
Between R-groups
H- - -O
Ionic Bonds
Formed when one or more electrons are transferred from one atom to another.
Disulfide bridge
Covalent bond between two cysteine amino acids.
Quarternary Structure
has 2 or more polypeptide chains
has prosthetic groups
forms conjugated proteins
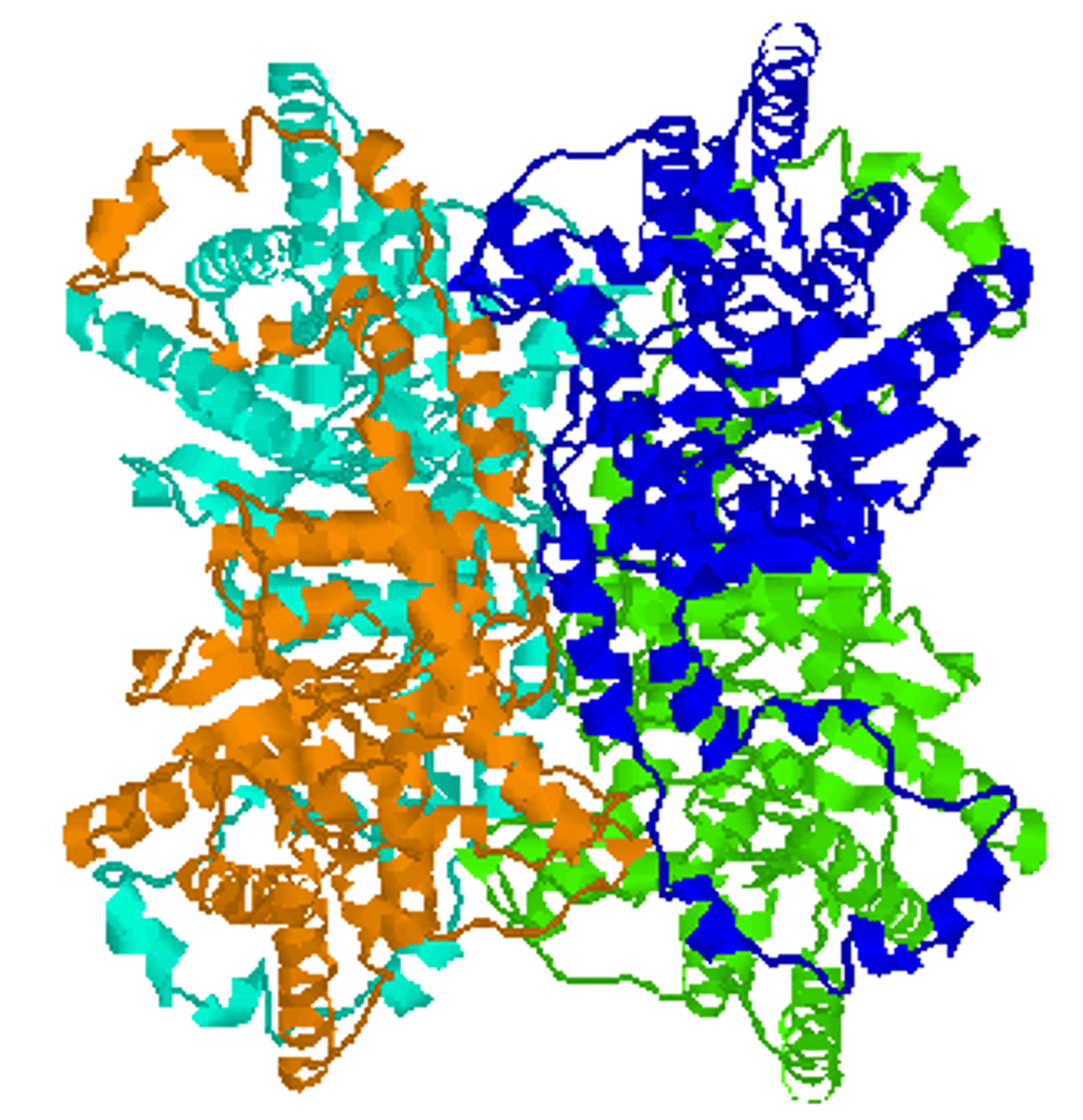
conjugated proteins
protein molecules combined with another kind of biomolecule (prosthetic group) in the final molecule e.g. Haemoglobin
Non-conjugated protein
A protein that is made up of only amino acids/ polypeptide chains
Fibrous Protein
only has a secondary structure;
generally insoluble;
long, narrow, coiled,
repetitive amino acid sequences
collagens, elastins, and keratins.
Is less sensitive to changes in heat and pH than globular proteins
STRUCTURAL

Globular Proteins
compact,circular
soluble
irregular amino acid sequences,
catalase, hemoglobin, insulin.
FUNCTIONAL / PHYSIOLOGICAL
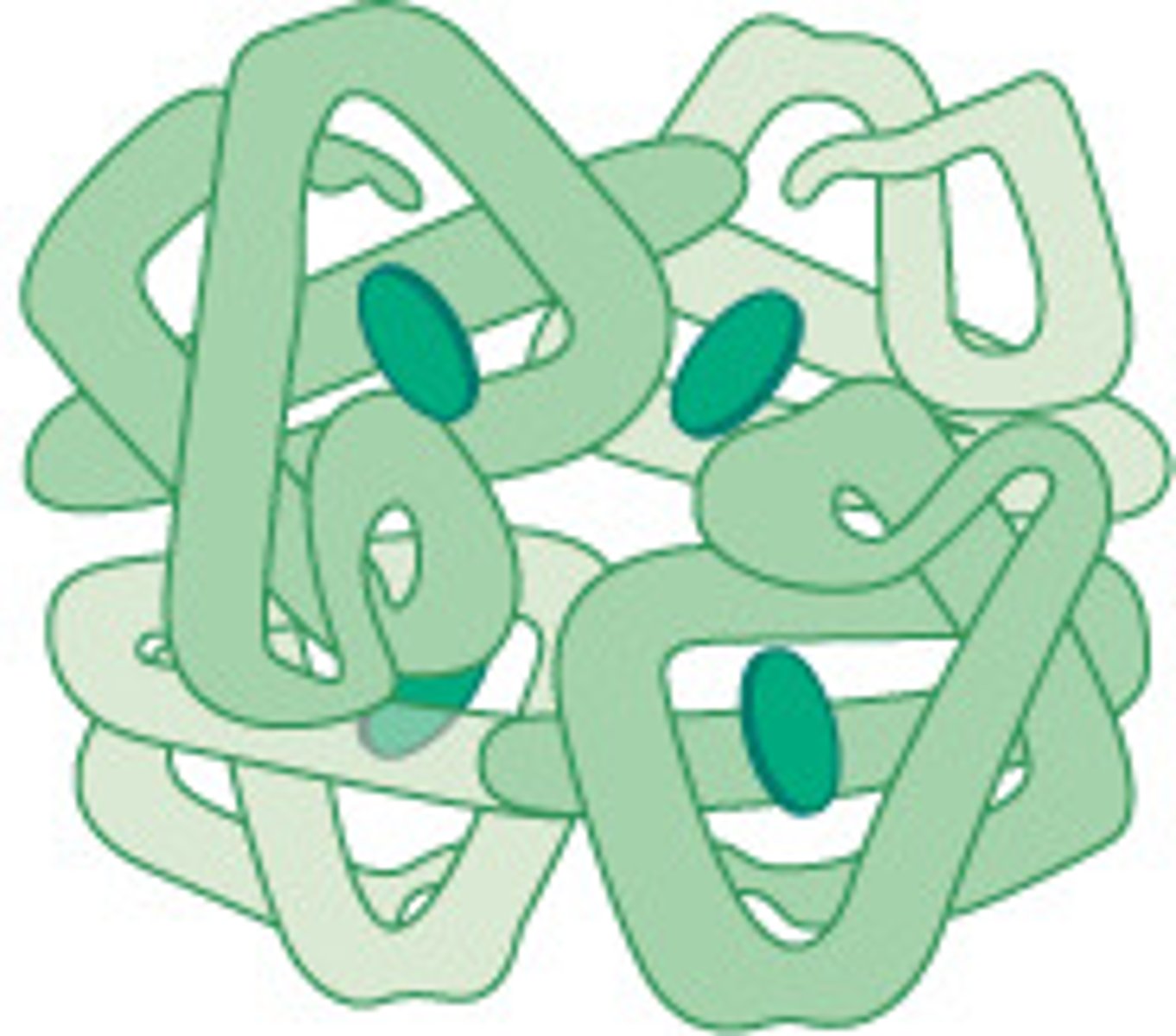
RNA
A single-stranded nucleic acid that passes along genetic messages. Moves to the ribosome to bond together amino acids.
Transcription
The organic process whereby the DNA sequence in a gene is copied into mRNA.
Translation
Movement of mRNA to ribosomes to bond together amino acids.
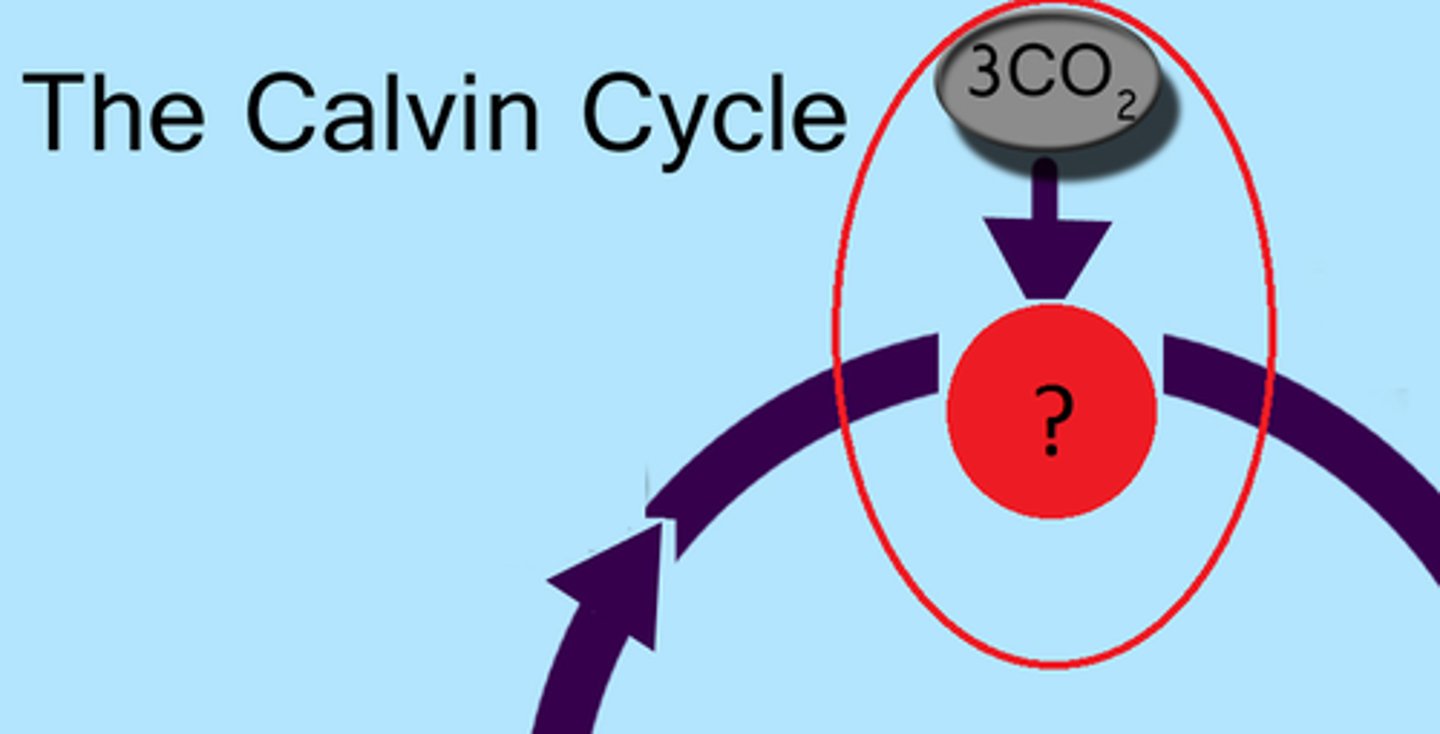
Rubisco
Enzyme with an active site
fixes carbon during photosyntheis
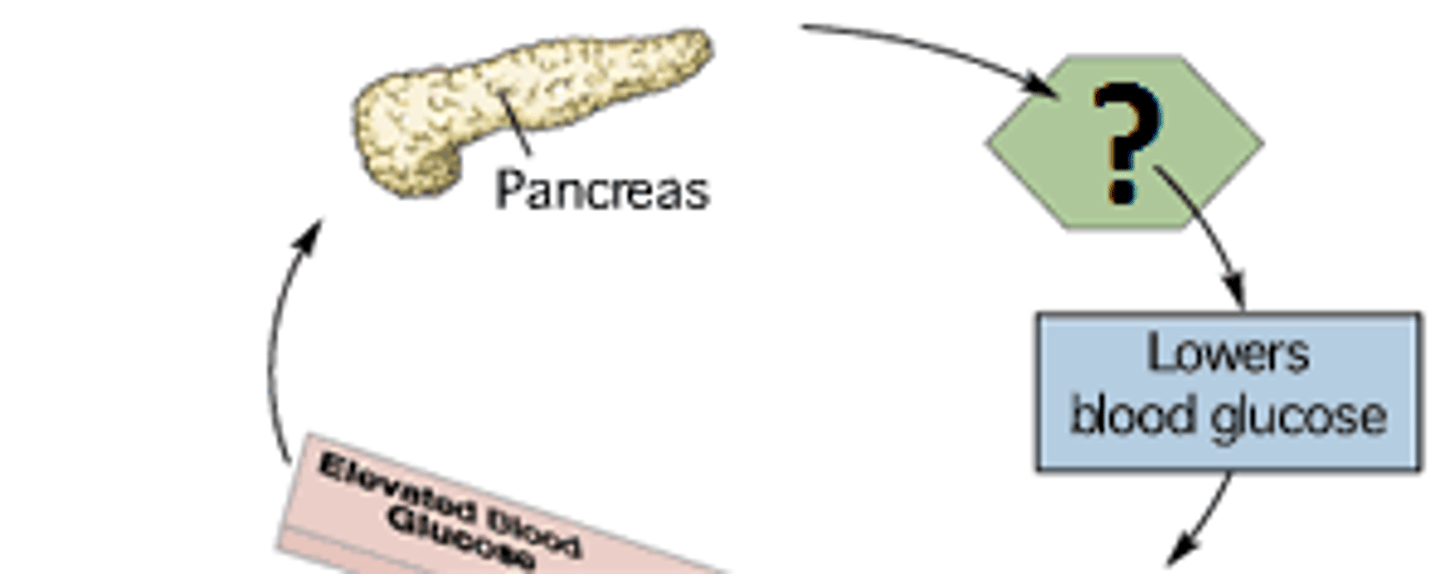
Insulin
Hormone
soluble in bloods
converts glucose into glycogen
Type 1 diabetics- pancreas does not produce enough insulin
Non-conjugated - 2 polypeptide chains
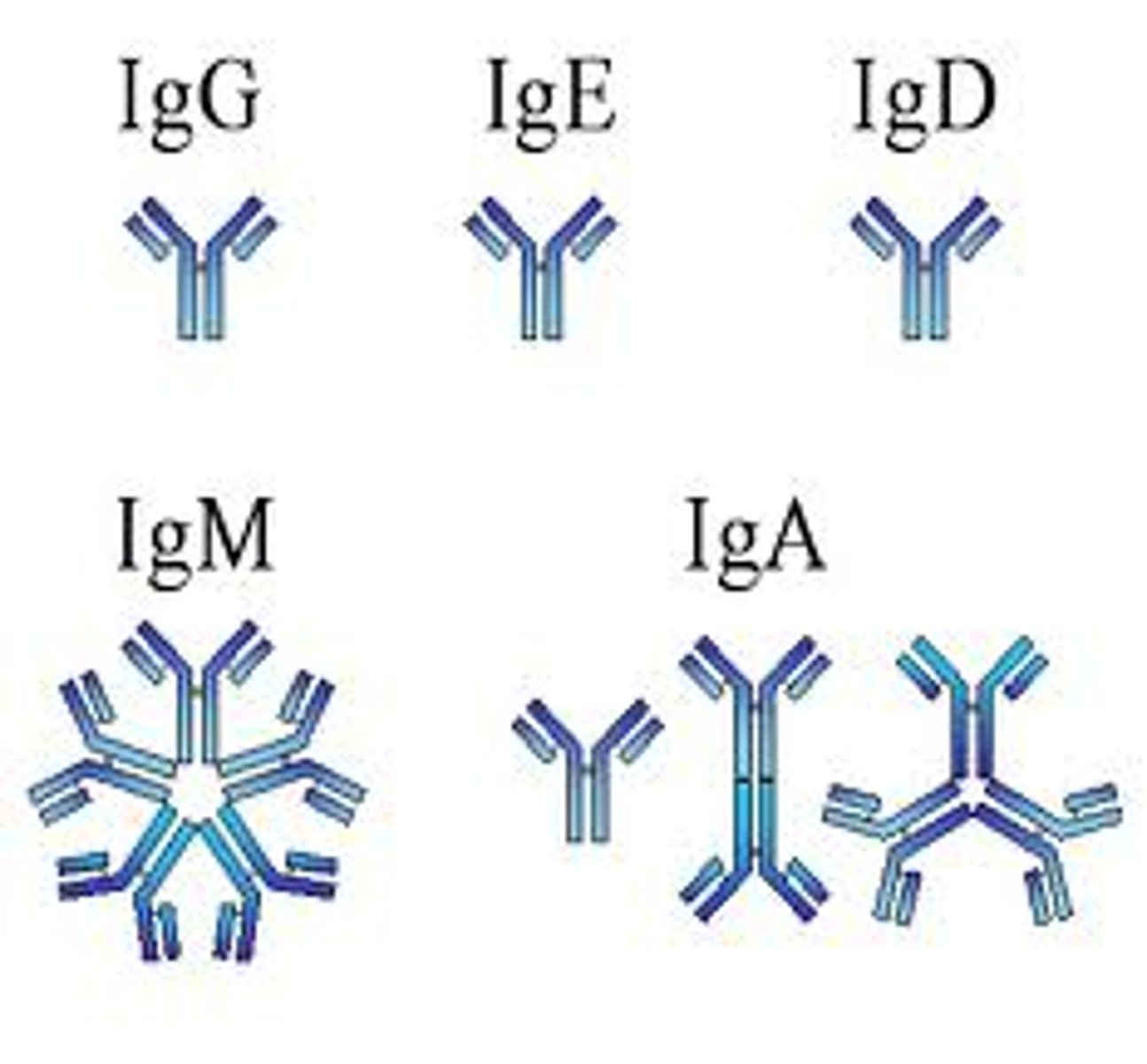
Immunoglobulins
Antibodies that bind to antigens and pathogens.
Allows specific immunity against many different diseases : very variable
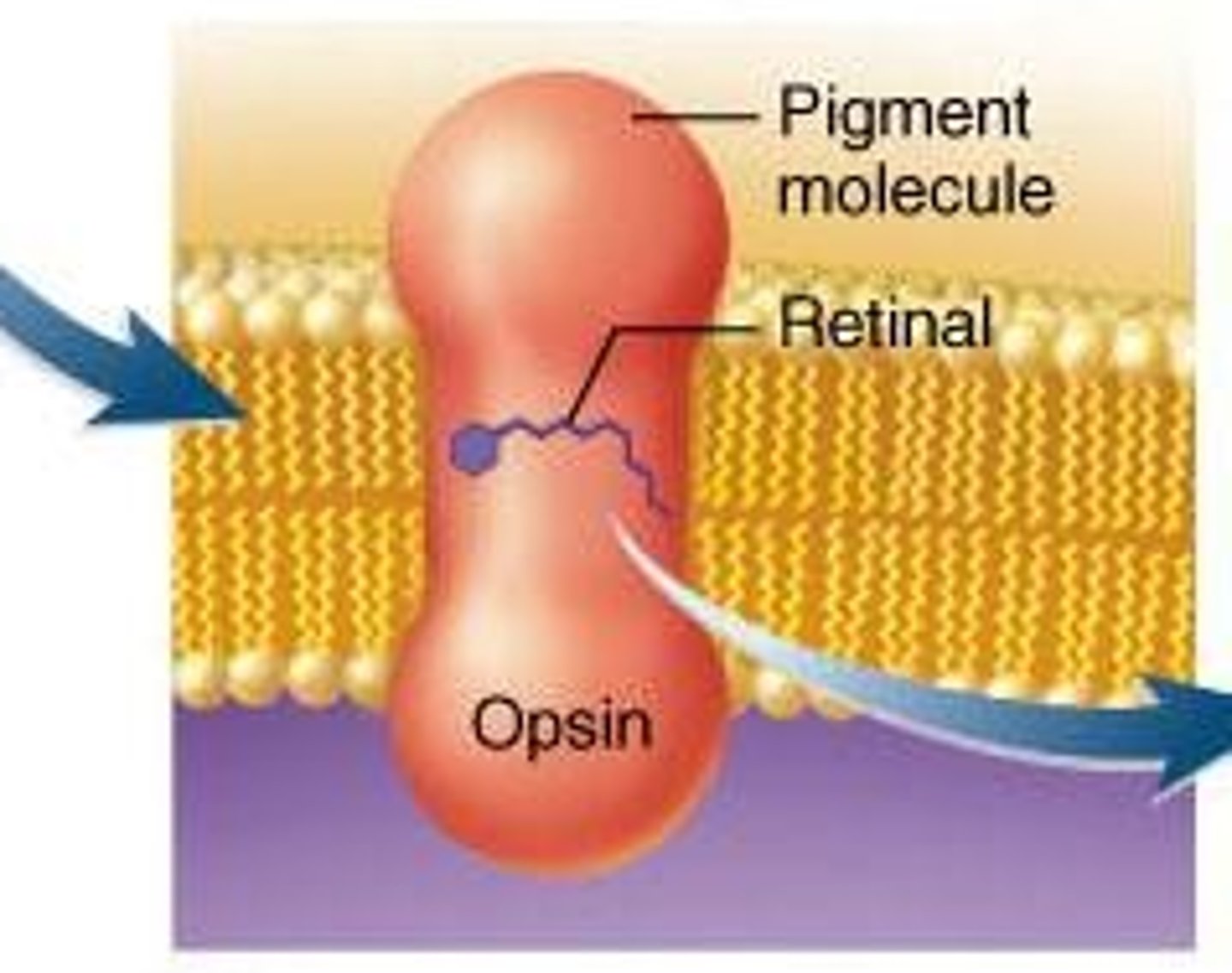
Rhodopsin
Pigment that makes the rod cells of retina light
sensitive. Rod cells send nerve impulses to the brain.
Collagen
Structural protein
3 polypeptides (non-conjugated)
rope-like conformation/ used
prevent tearing, in bones to prevent fractures, and in tendons to give tensile strength.
Fibrous
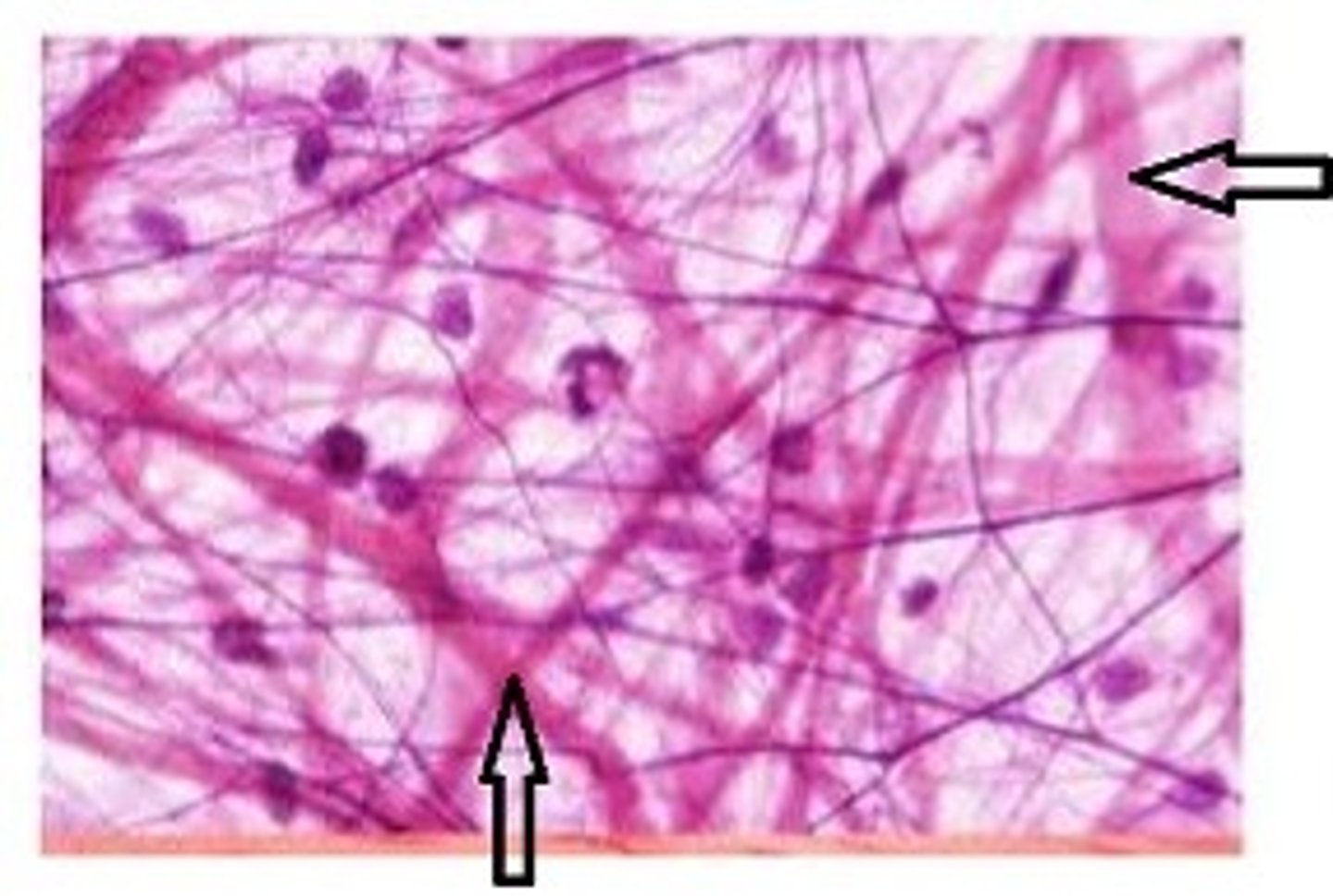
Why is Collagen a strong structural protein?
forms a triple helix
triple helix contains both covalent + hydrogen bonds
many collagen finrils join to make collagen fibre
Spider Silk
fibrous
tensile strength
beta-pleated sheets
Becomes stronger when stretched resisting breakage.
Denaturation
high temperatures cause protein denaturing
deviation of pH from te optimum causes protein denaturing
Bonds within protein between R-groups break
3d conformation of protein changes
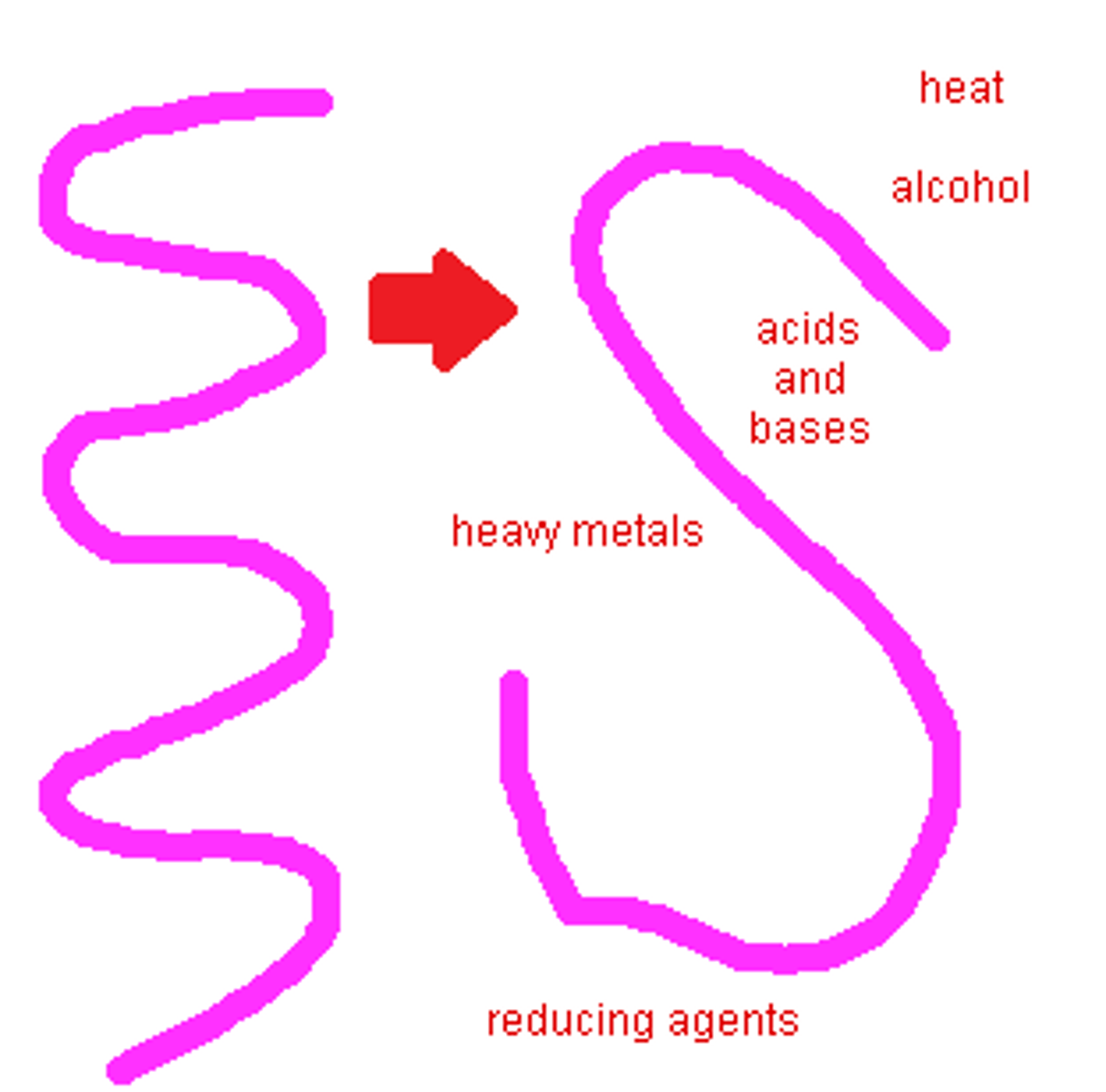
Why is denaturation of proteins fairly easy ?
The 3D structure of proteins are stabilised by bonds or interactions between R- groups of amino acids within the molecule - most of there are relatively weak and can be disrupted and broken by pH + temp
causes denaturation of proteins
heat- vibrations cause intermolecular bonds to break
pH, chemicals, salt concentrations
Charges of R groups of amino acids changing - break ionic bonds and form new ones
Thermophiles
Archaea that thrive in very hot environments, such as volcanic springs
Their proteins are stable at much higher temperatures (100 degrees)
What are the nutritional benefits of cooking protein rich foods before injesting them?
heat denaturation starts the process of digestion of proteins to amino acids
in the stomach, pH further denatures
amino acids readily absorbed in small intesine
can be used for synthesis fo human proteins
Proteomes
The complete complement of proteins that a cell or organism can make.
Genome
All the genetic information in an organism; all of an organism's chromosomes.
Explain why the genome of an organism is fixed but not the proteome?
all cells carry the same genome
not all cells make the same proteins
the range of proteins made in a cell can vary during its lifetime
Why does every organism have a unique proteome?
every organism has unique genome due to mutations
genes code for amino acid that code for proteins that code for
the range of proteins produced thus the proteome
When amino acids bond to form peptide bonds what kind of reaction is it ?
Condensation reaction - water removed
Polymerisation of amino acids
Amino acids can be combined by condensation polymerisation. The products of this reaction are a polypeptide and water.
Word equation for the polymerisation of amino acids
Amino acid 1 + Amino acid 2 = Dipeptide + water (H20)
Effect of pH on protein structure
The charges on R groups are changed- breaking the ionic bonds within the protein or causing New Ionic bonds to form
Effect of temperature on protein structure
- Can cause denaturation - vibrations within the molecule break the intermolecular bonds or interactions
why are some amino acids polar and some are non-polar ?
- Amino acids with hydrophobic R-groups are non-polar (like leucine)
- Amino acids with hydrophilic R-groups are polar (threonine)
Why are cellular enzymes mainly made up of proteins? (5 marks)
Enzyme function is detremined by shape of active site
shape of protein is determined by amino acid sequence- very variable
primary → secondary with a helix abd b pleated regions
secondary structure held by hydrogen bonds
secondary → tertiary (3d)
folding of primary = bonds between amino acids
hydrogen, ionic and disulfide