BIOC 4331 Lecture 2
1/33
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
Why is biochemistry considered a unifying theme of life?
Because living organisms are remarkably similar at the molecular level, despite enormous diversity.
What are non-covalent interactions, and how strong are they compared to covalent bonds?
Non-covalent interactions are weak interactions (≈ 4–75 kJ/mol) compared to covalent bonds (≈ 400 kJ/mol). (Individually weak, collectively powerful in macromolecules)
Why are non-covalent interactions biologically important?
Their strength is similar to thermal energy at room/body temperature, so they form and break constantly, enabling binding, dissociation, enzyme catalysis, and signaling.
What are the four types of non-covalent interactions and their approximate energies?
Ionic interactions: ~ 75 kJ/mol (strongest)
Hydrogen bonds: ~ 25 kJ/mol
Hydrophobic interactions: ~ 4 kJ/mol
van der Waals interactions: ~ 4 kJ/mol
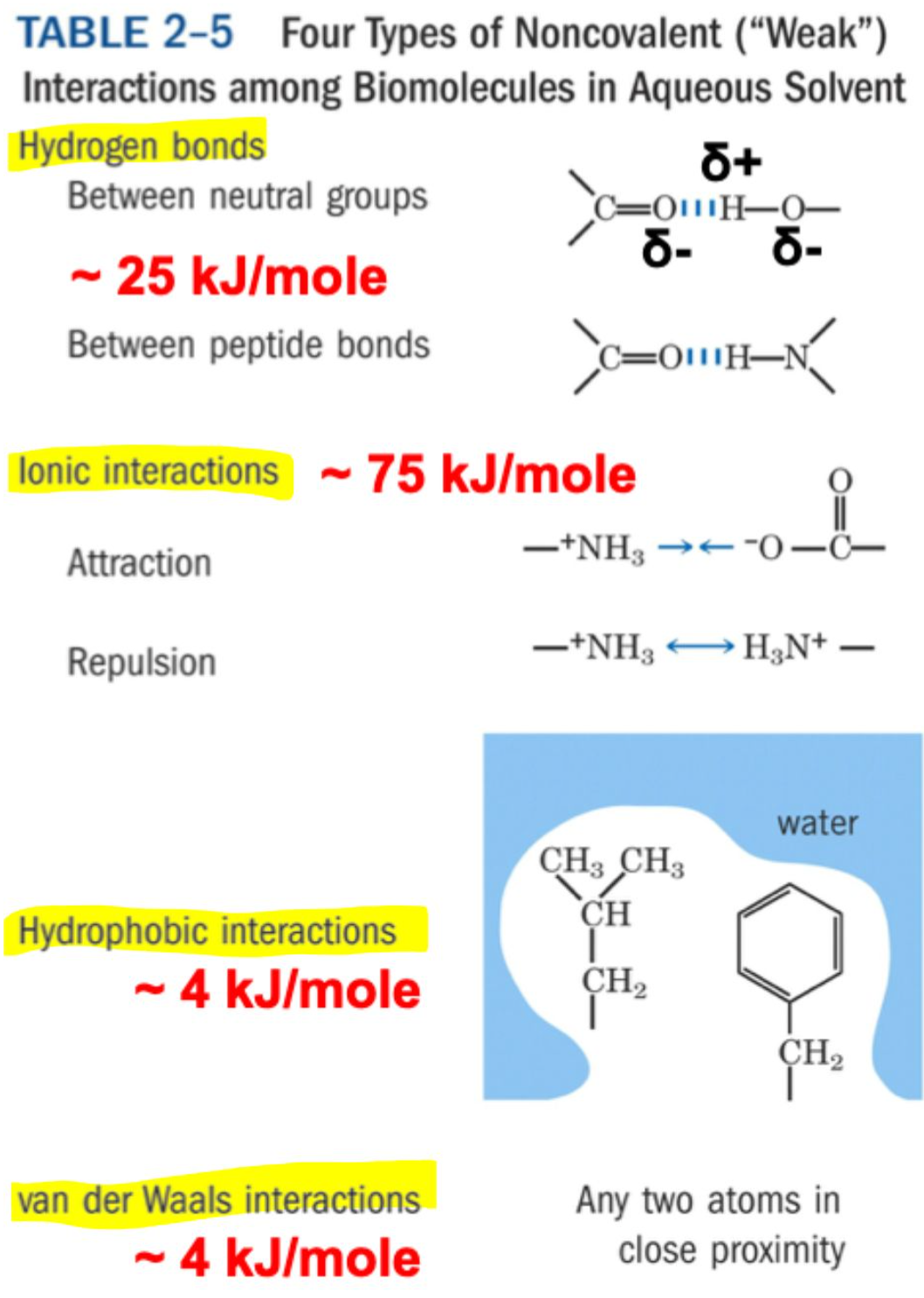
What causes van der Waals interactions?
Transient electric dipoles formed by random electron movement induce opposite dipoles in nearby atoms → weak attraction.
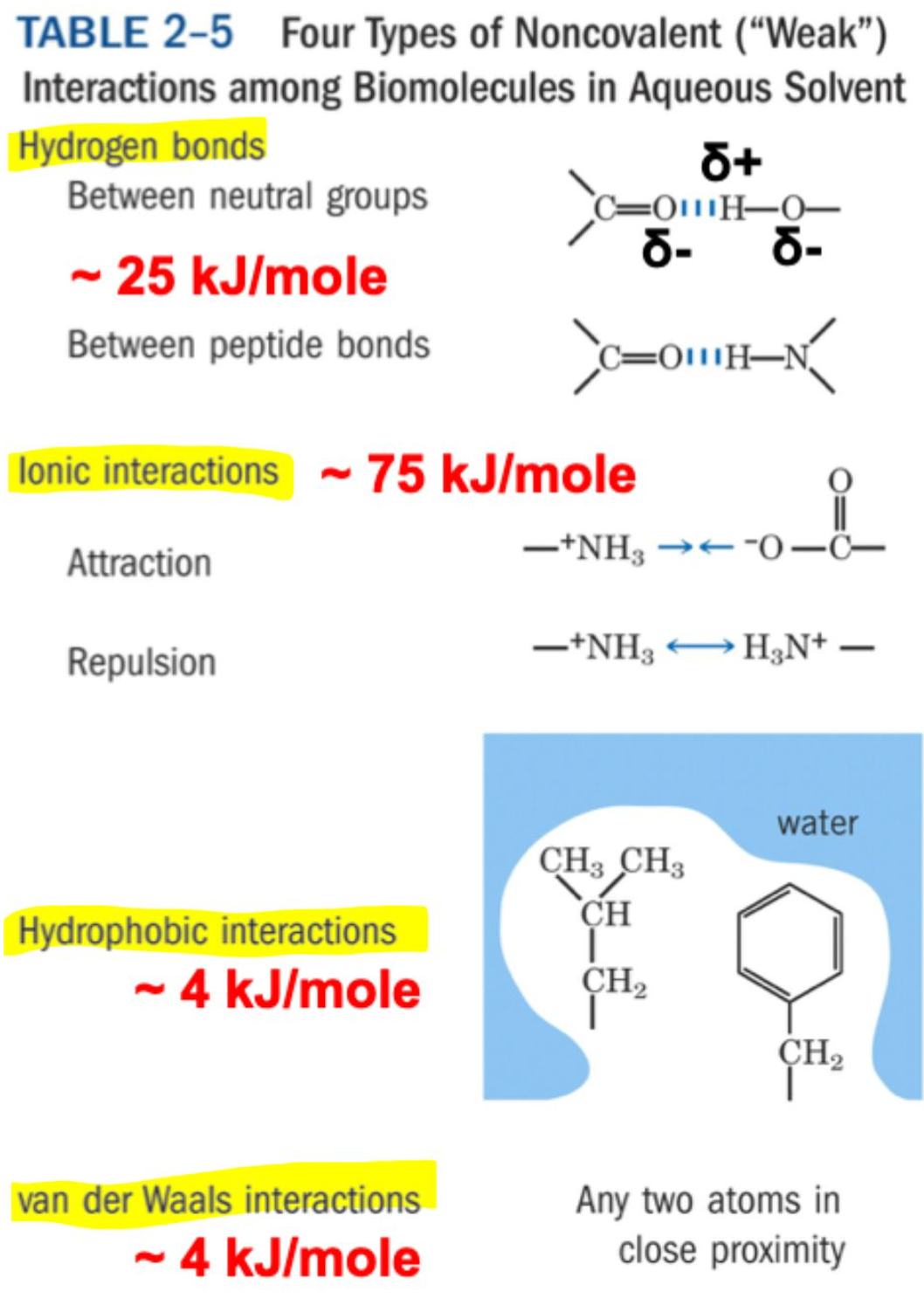
What limits how close atoms can get in van der Waals interactions?
Repulsion of electron clouds → defines the van der Waals radius.
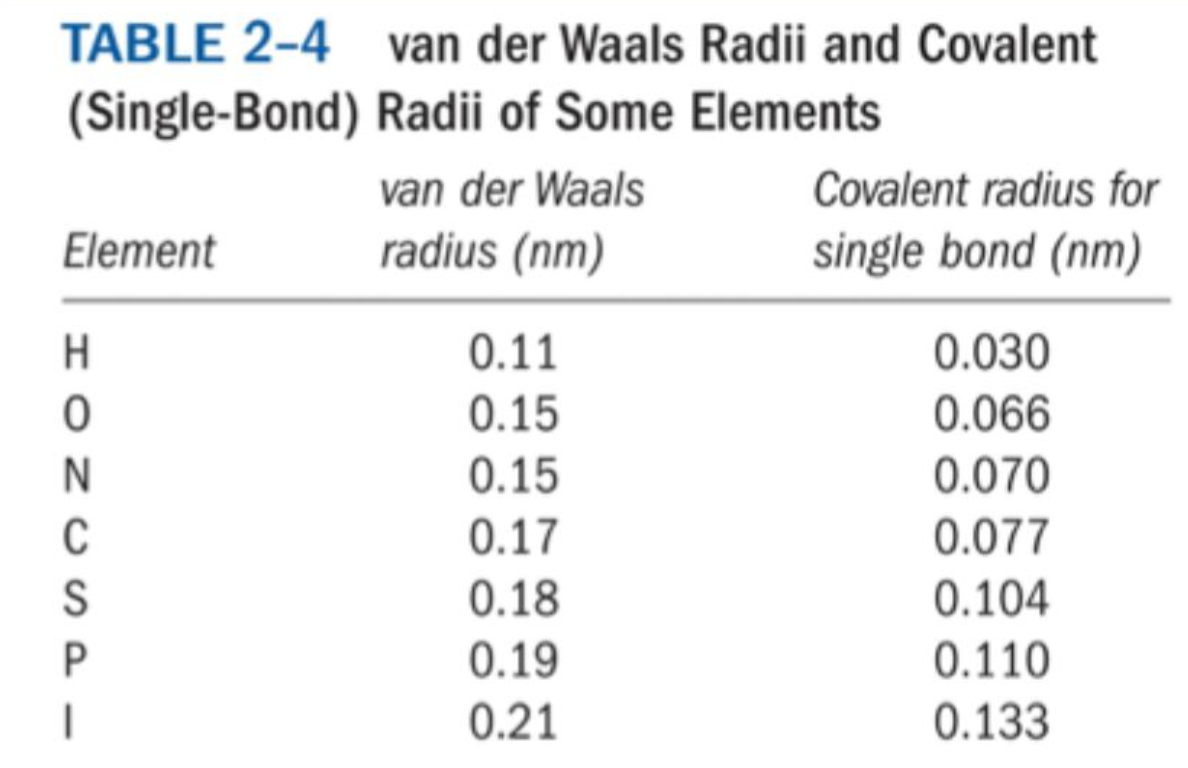
How do you calculate the optimal van der Waals distance between two atoms?
Add the van der Waals radii of the two atoms.
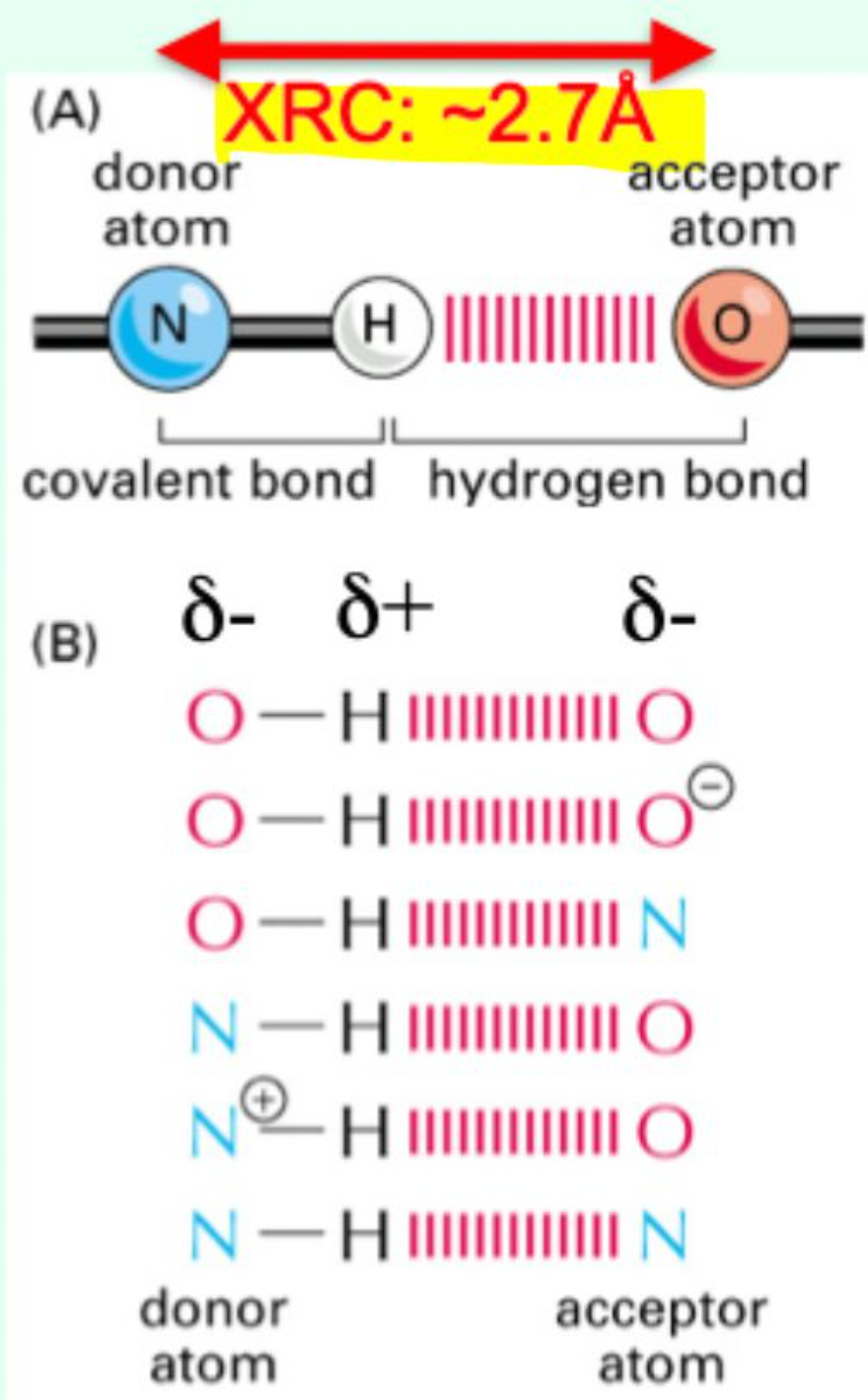
What defines a hydrogen bond?
Forms between:
A hydrogen with a slight positive charge (δ⁺)
Bonded to a more electronegative donor atom
And an acceptor atom with a slight negative charge (δ⁻)
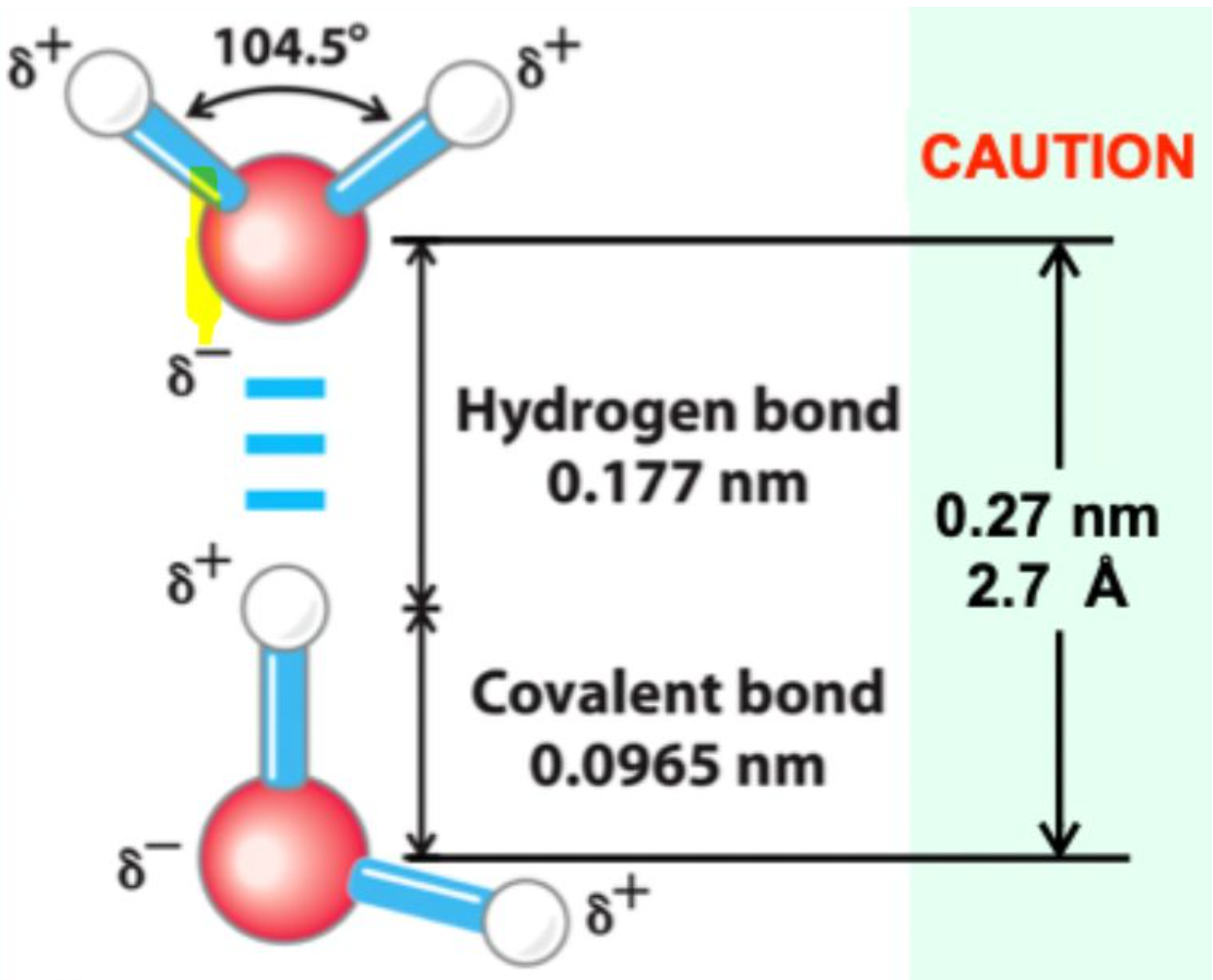
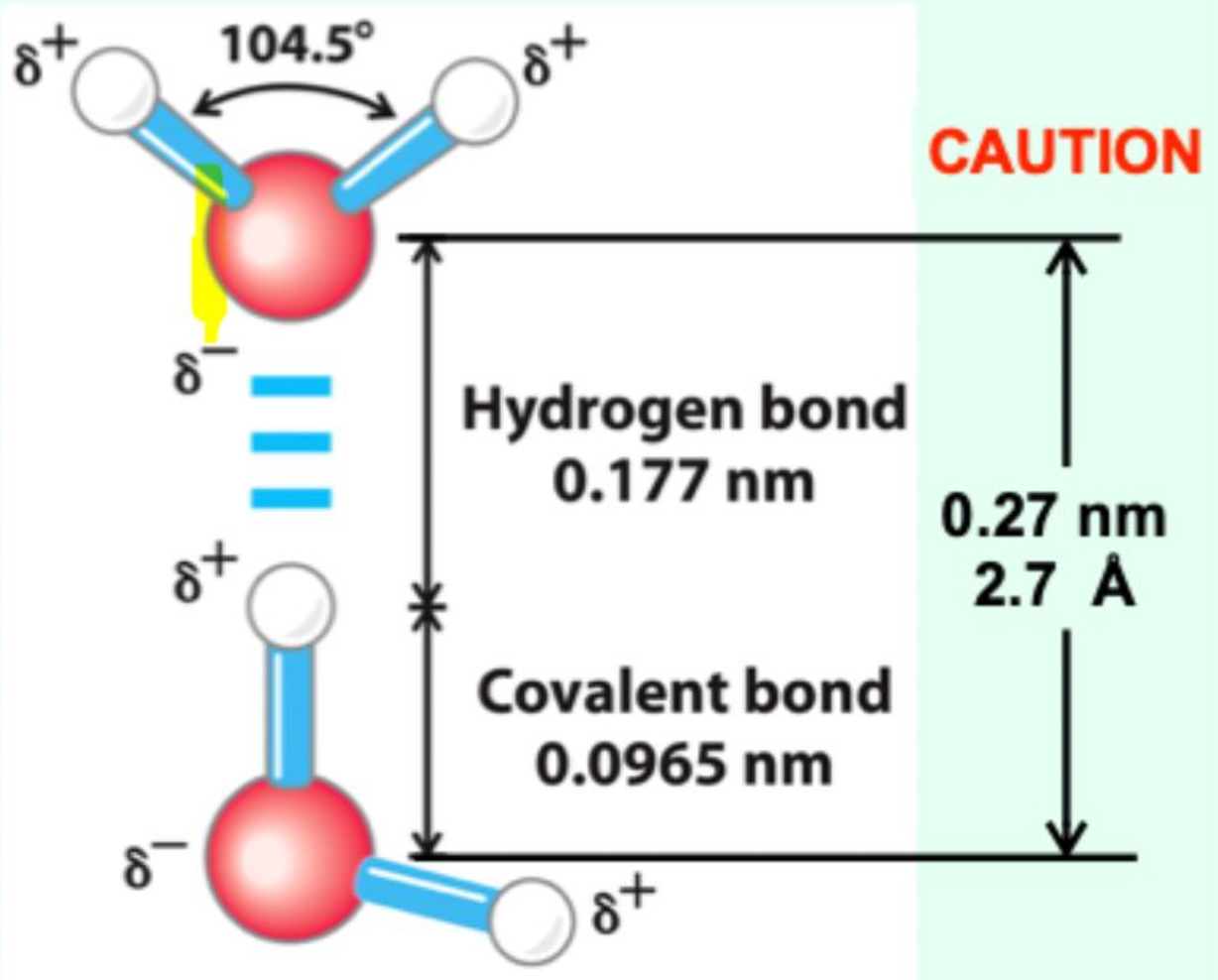
What is the typical hydrogen bond distance?
≈ 2.7 Å (0.27 nm)
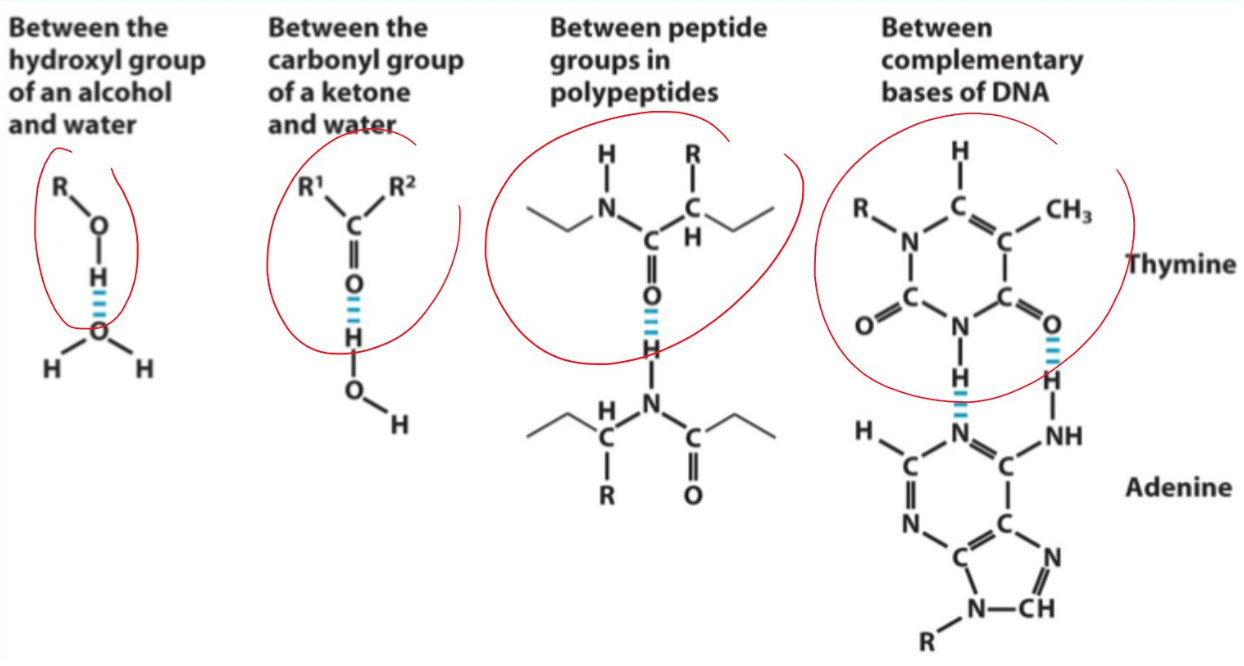
Name biologically important hydrogen bonds.
Alcohol ↔ water
Carbonyl ↔ water
Peptide backbone ↔ peptide backbone
Complementary DNA bases
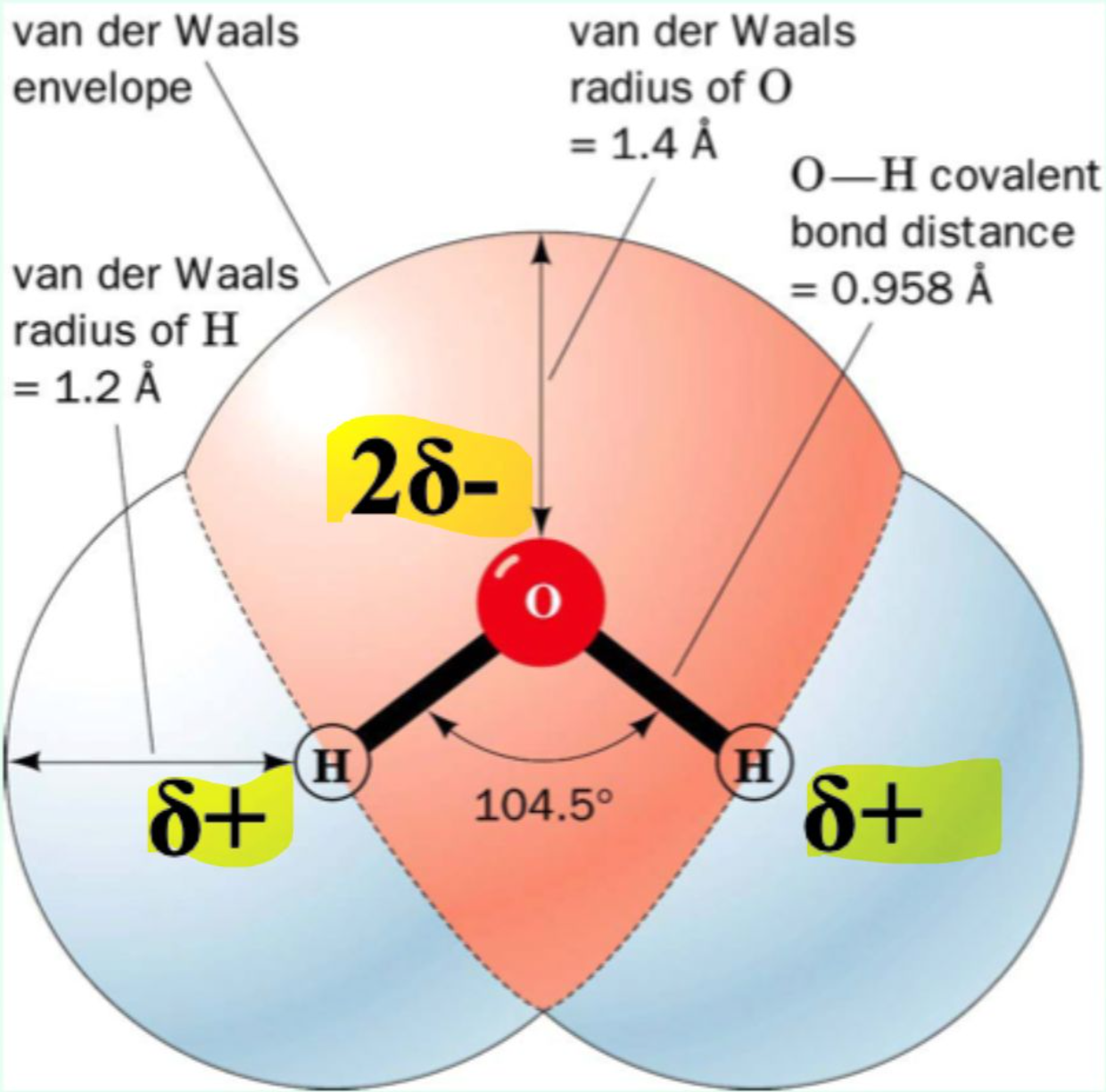
Why is water polar?
Oxygen is more electronegative, pulling electrons toward itself → δ⁻ on O, δ⁺ on H.
Memorize the electronegativity order used in biology.
O > N > S > C > H
Why does water form so many hydrogen bonds?
Oxygen has two lone pairs, and hydrogen is small and polar → extensive H-bonding network.
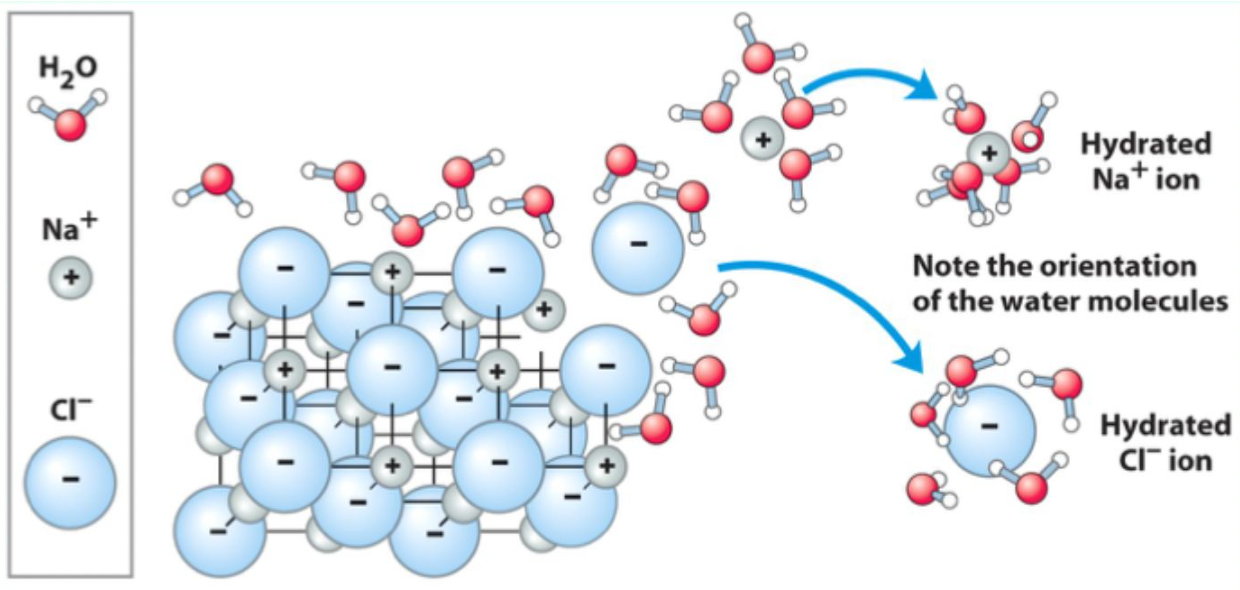
Why is water an excellent solvent?
Its polarity allows it to stabilize ions and polar molecules via hydration.
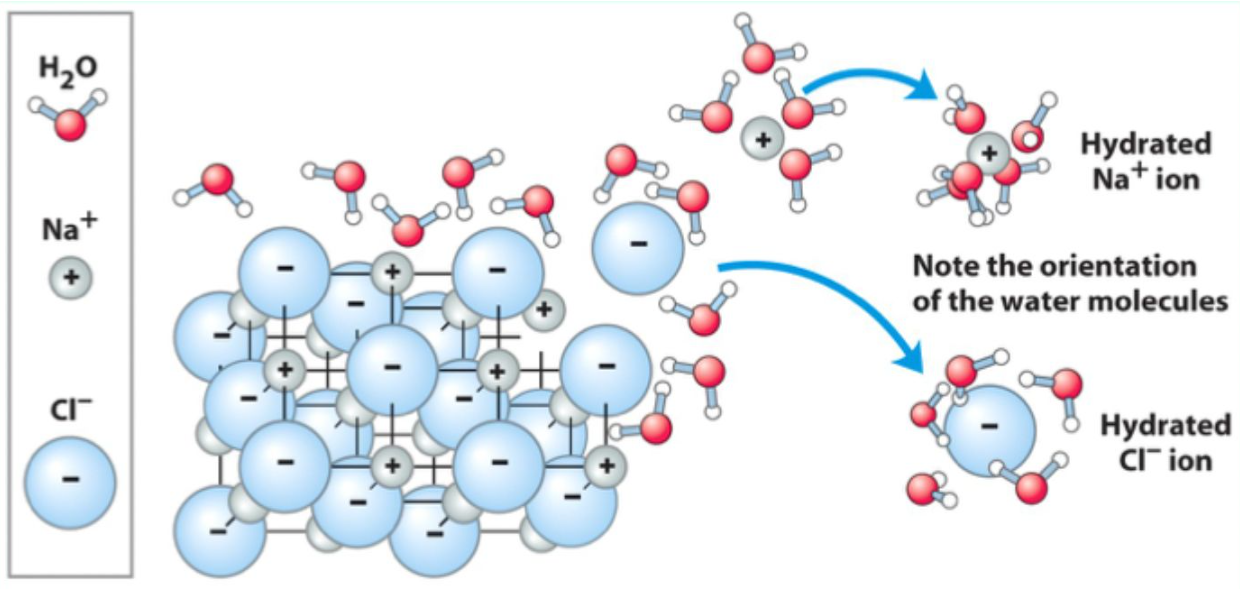
What is hydration?
Water molecules cluster around ions or polar groups, stabilizing them and allowing dissolution.
What unit are biomolecular molecular masses (m) commonly reported in?
Daltons (Da)
What is the definition of relative molecular mass (molecular weight), Mr?
The sum of the relative atomic masses of all the atoms in a molecule, compared with ¹⁄₁₂ of the mass of a carbon-12 atom.

What is the acid dissociation constant (Kₐ)?
Ka = [A-][H+]/[HA]
![<p>Ka = [A-][H+]/[HA]</p>](https://knowt-user-attachments.s3.amazonaws.com/ecafc328-7111-4827-bb20-c16e8f8d9e4b.png)
In aqueous solutions, what form does free H⁺ actually exist as?
Hydronium ion (H₃O⁺)
What is the pH of a 1 M strong acid?
pH = 0 (Dissociates completely)
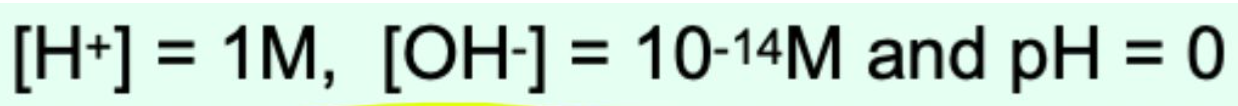
What is the pH of a 1 M strong base?
pH = 14 (Completely protonated)
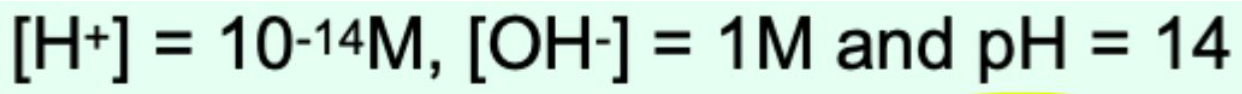
Do weak acids and bases fully dissociate or protonate in water?
No
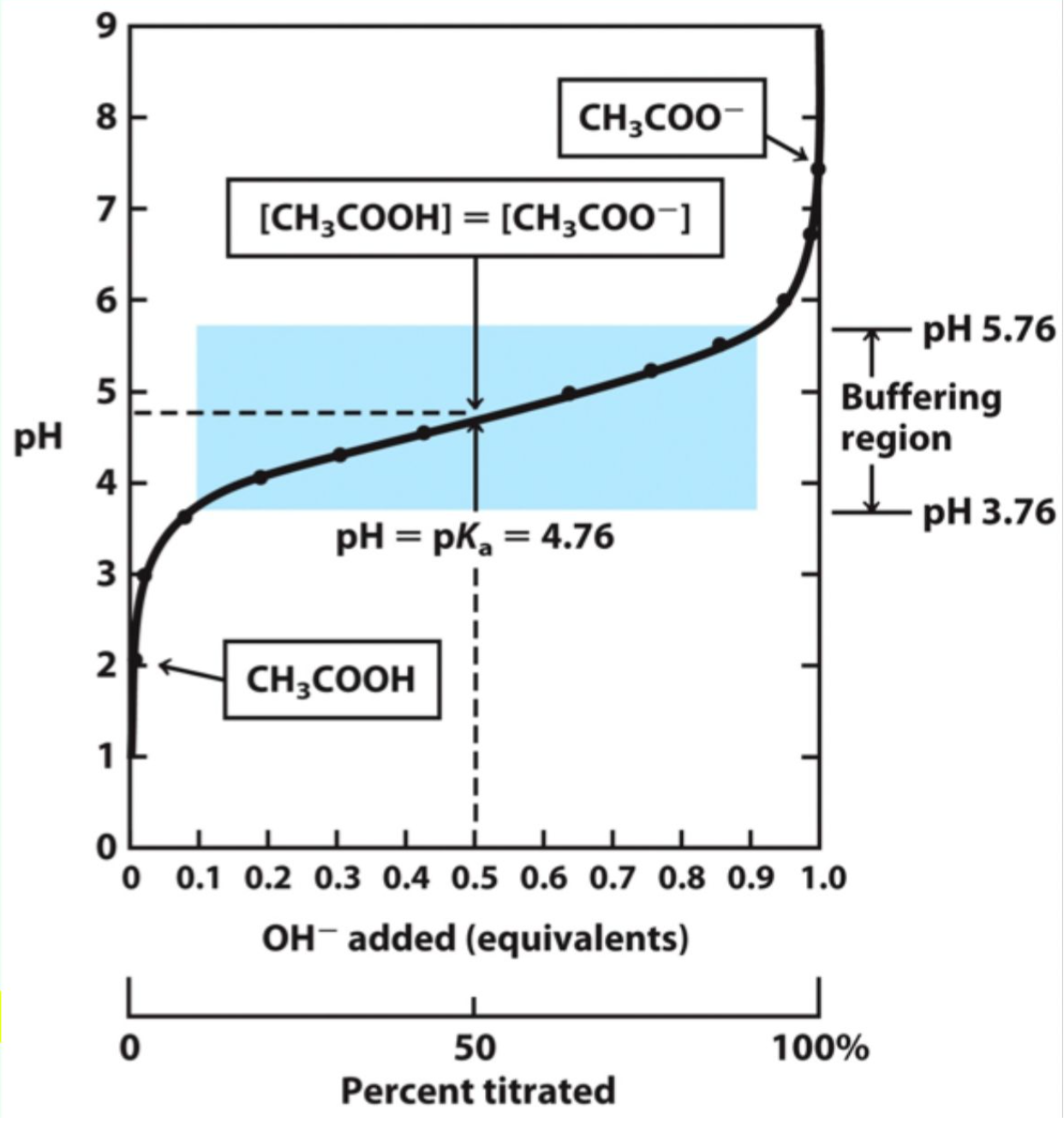
What does pKₐ represent?
pKa = −log(Ka)
What is special about pH = pKₐ?
The concentrations of the acid/base and its conjugate base/acid are equal.
What equation relates pH, pKₐ, and concentrations of acid/base?
pH = -log[H+] = pKa + log[A-]/[HA]
![<p>pH = -log[H+] = pKa + log[A-]/[HA]</p>](https://knowt-user-attachments.s3.amazonaws.com/b1ccce0f-e8e8-40ba-92d6-53a57fe42566.png)
What is the distance between two water molecules joined by a hydrogen bond?
2.7 Å (0.27 nm)
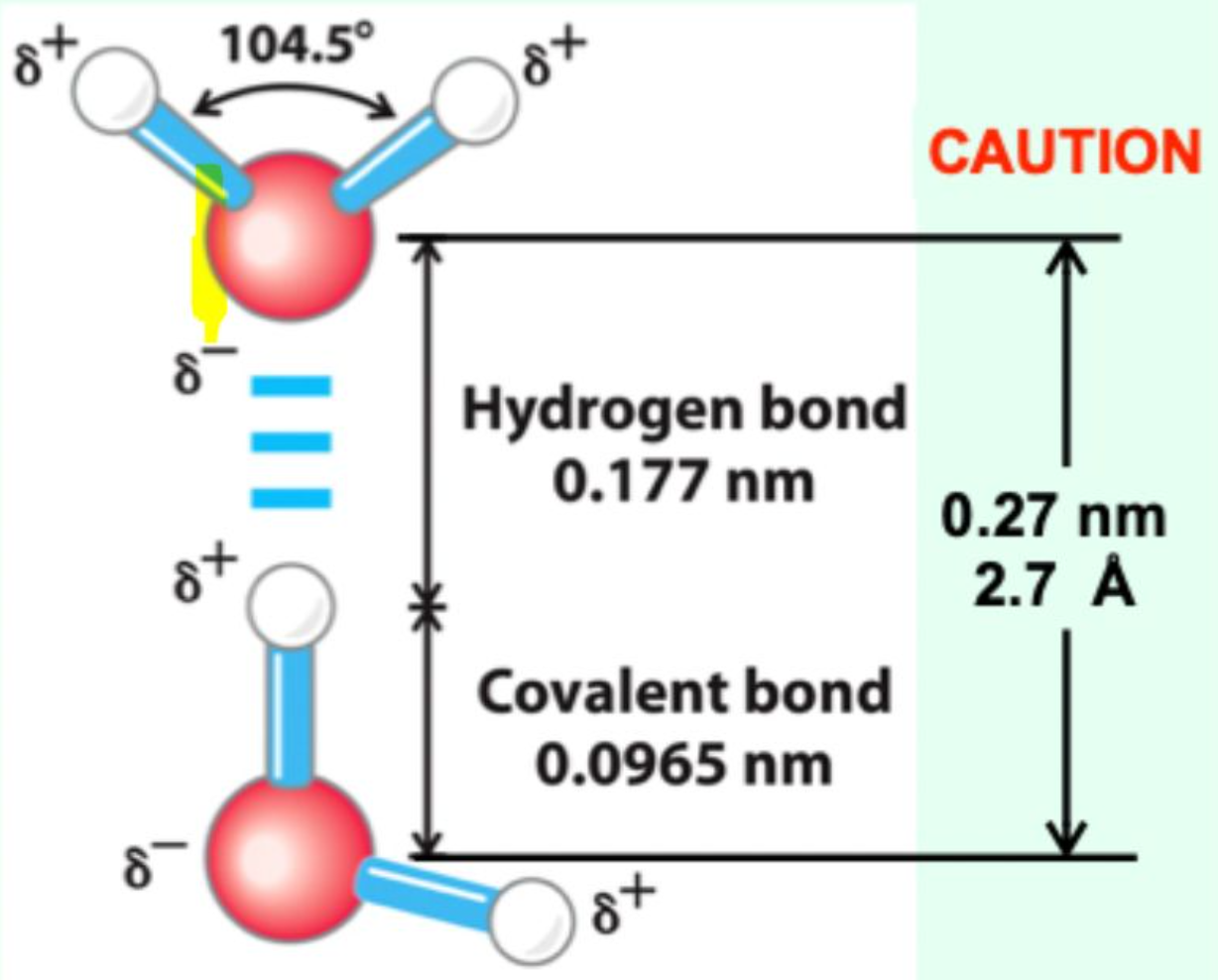
How does the hydrogen bond distance compare to a covalent O–H bond?
It is much longer (~2.7 Å vs ~1.0 Å), showing that hydrogen bonds are weaker and non-covalent.
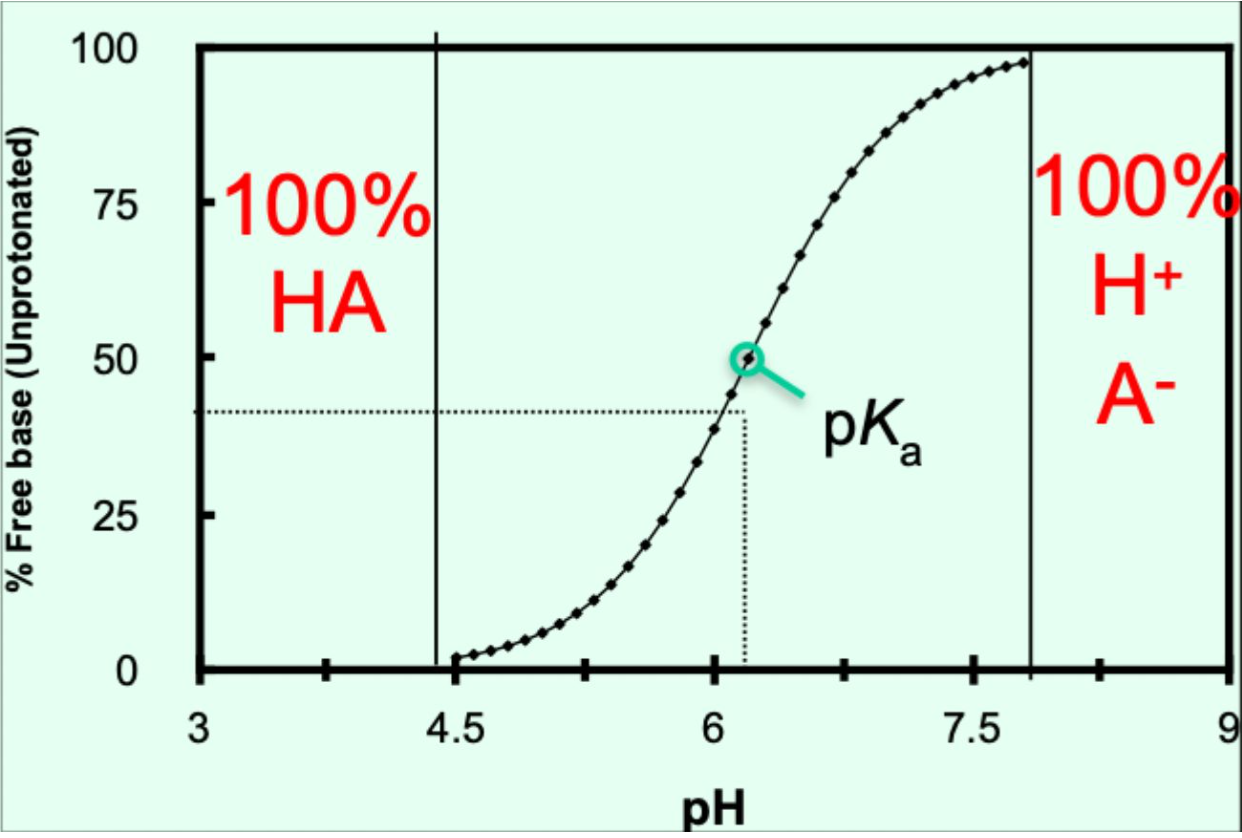
When does an acid have no charge (HA)?
When pH < pKₐ − 1.5
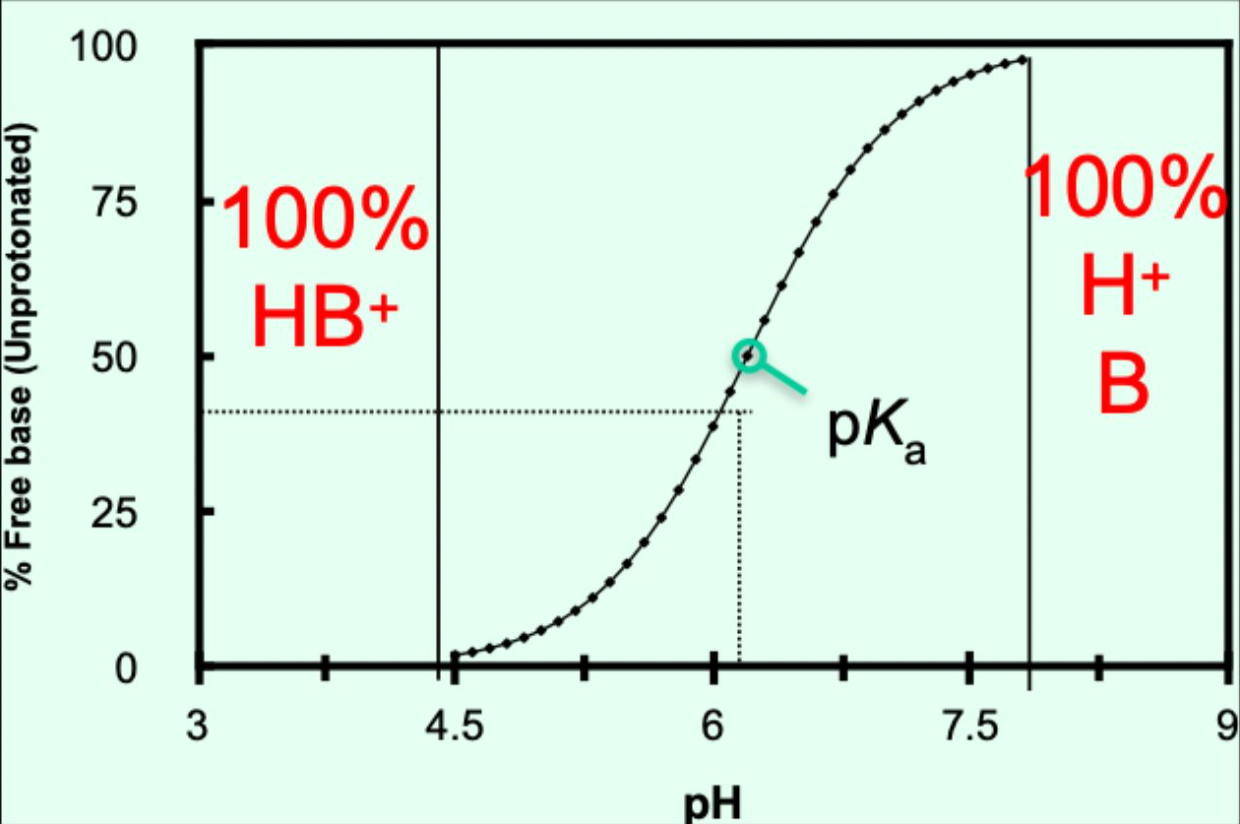
When does a base have no charge?
When pH > pKa + 1.5
What makes a solution a good buffer?
A weak acid/base and its conjugate within ~1 pH unit of its pKa.
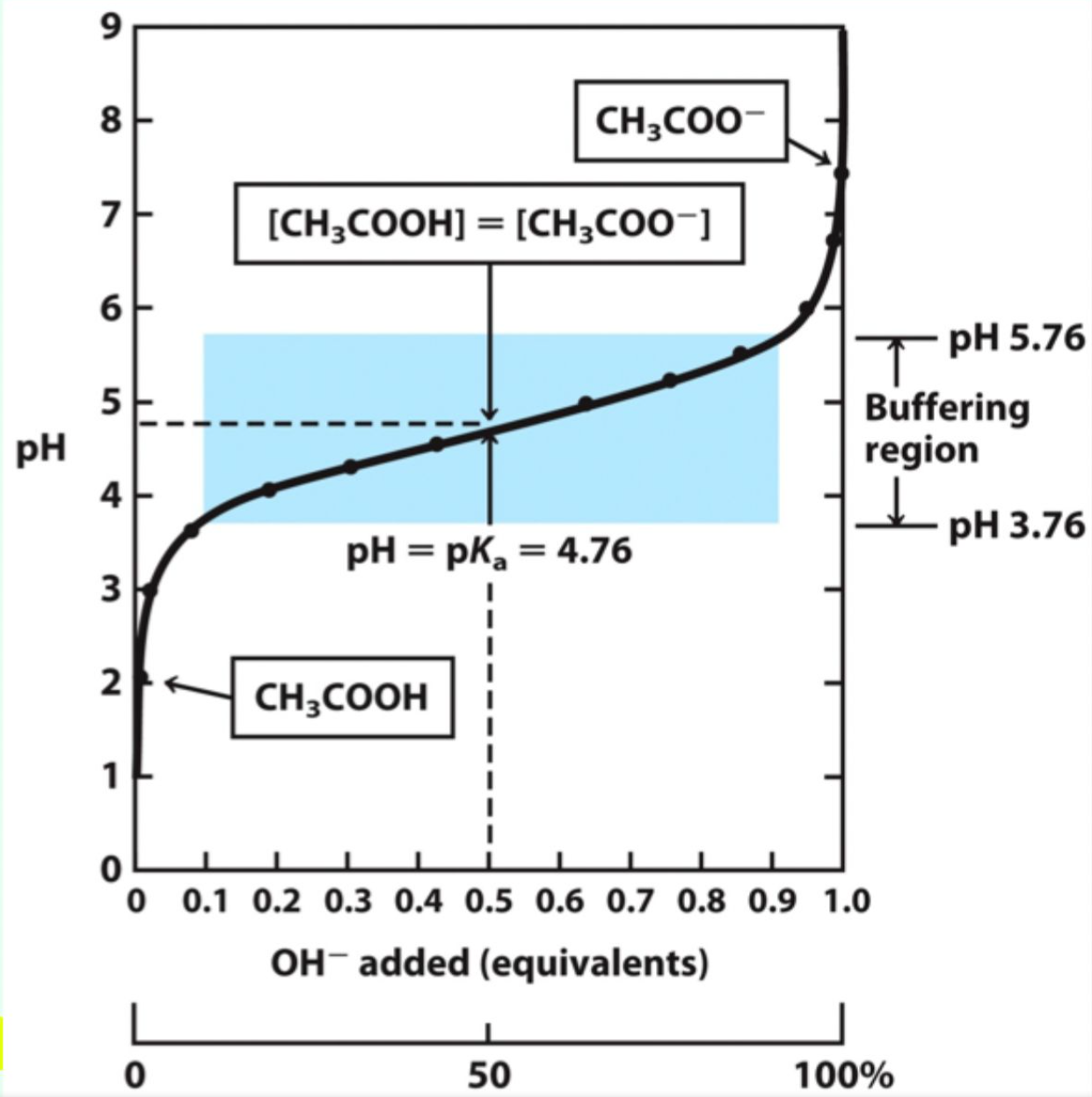
Why does a pH change very little near pKa?
Added H+ or OH- is absorbed by the conjugate base or acid.
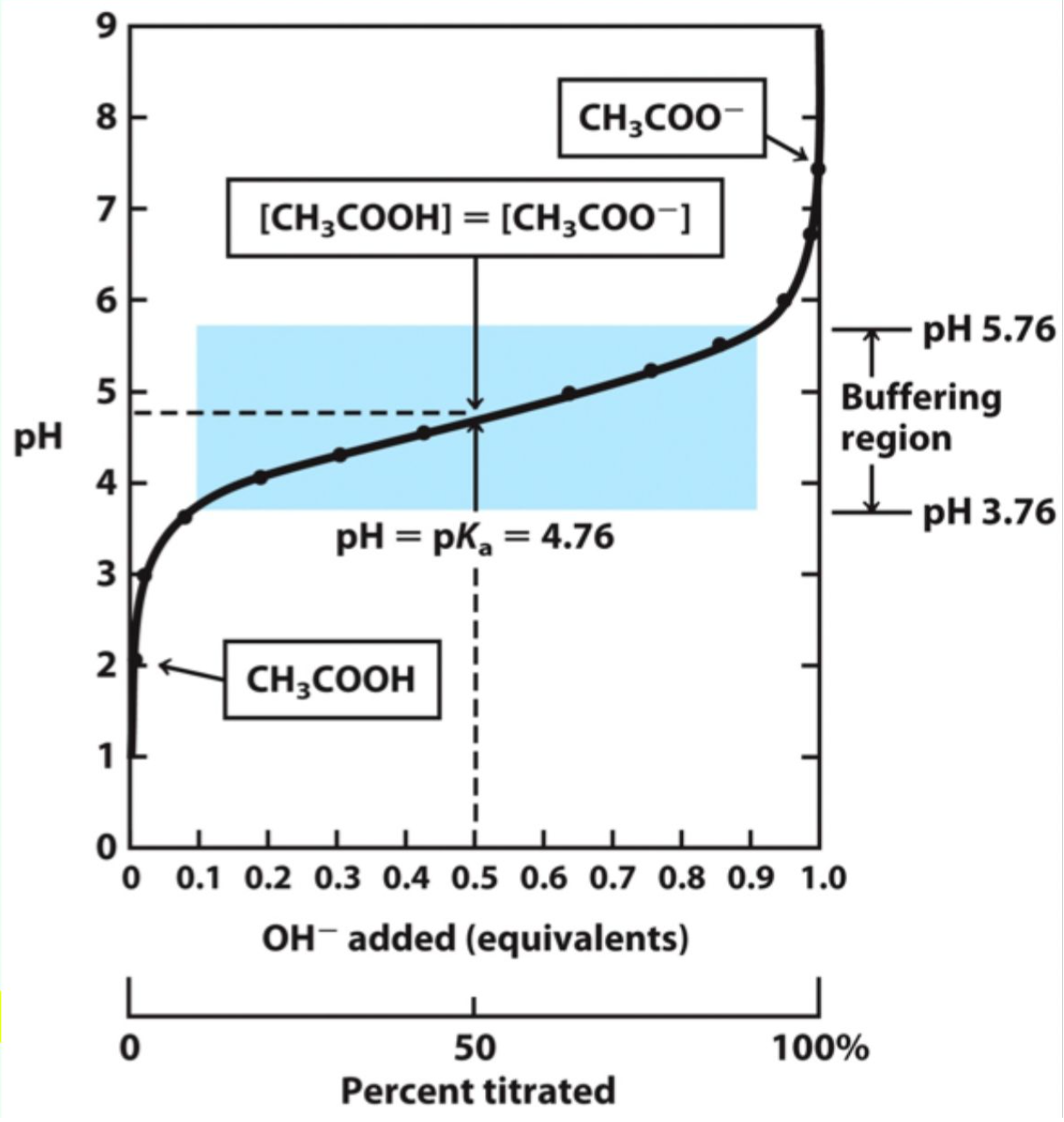
What happens when a small amounts of strong acid/base are added to a buffer?
Minimal or little change in pH.
What interactions stabilize biological macromolecules and depend on pH?
Ionic interactions
What are biological buffers composed of?
Weak acids and their conjugate bases