chem exam 2
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
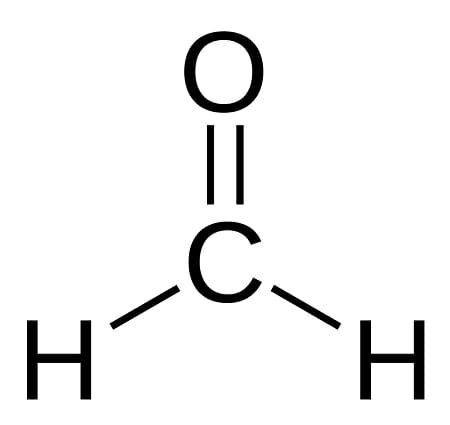
formaldehyde (methanal)
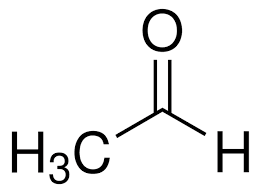
acetaldehyde (ethanal)
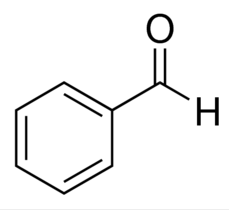
benzaldehyde (benxenecarbaldehyde)
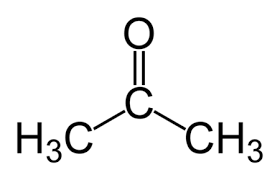
acetone
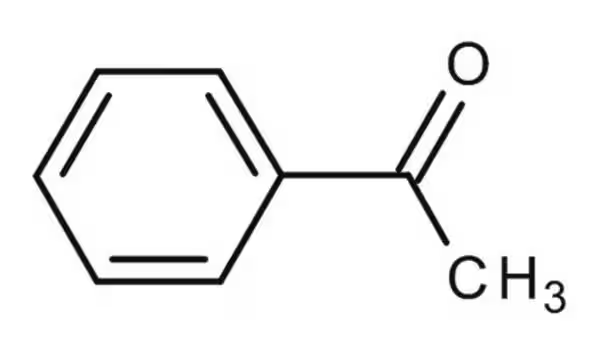
acetophenone
increasing boiling point and strength of intermolecular forces
hydrocarbons → aldehydes/ketones → alcohols
formaldehyde use
preservation of biological specimens
acetone in health
byproduct of fatty acid metabolism, produced in excess in diabetes when the disease is not properly controlled
aldehydes are oxidized to
carboxylic acids
oxidation
replacing one C-H bond with a C-O bond on the carbon bearing OH group
ketone oxidation
not possible due to the lack of hydrogen on the carbon next to oxygen
hydrogenation
addition reaction where a group is added to both atoms on a double bond
acetial
has two OR groups bonded to the same carbon

hemiacetal
has one OH and one OR group bonded to the same carbon

hemiacetal formation
the addition of an alcohol to an alydehyde or ketone, catalyzed by H2SO4
acetal formation
when reacting with excess alcohol, the OH is removed and replaced with the alcohol group
isomers
different compounds with the same molecular formula
constitutional isomers
differ in the way atoms are connected to each other
stereoisomers
differ in the 3-dimensional arrangement of atoms that always have the same functional group
cis-isomer
two methyl groups on the same side of the double bond
trans-isomer
two methyl groups on the opposite side of the double bond
chiral
a molecule or object that is not superimposable on a mirror image (cannot align all parts of an object with its mirror image)
achrial
a molecule or object that is superimposable on its mirror image (can align all parts of an object with its mirror image)
enatiomers
mirror images that are not superimposable
chirality center
a carbon atom bonded to four different GROUPS
plane polarized light
light that consists of waves that oscillate in a single plane, arises from ordinary light though a polarizer
polarimeter
an instrument that allows plane polarized light to travel through a sample tube containing an organic compound
achiral compound optical activity
optically inactive, does not change direction of plane polarized light
chiral compound optical activity
optically active, rotates plane polarized light
detrorotary
rotation of light is clockwise, labeled as the (+) enantiomer
levorotary
rotation of light is counter clockwise, labeled as the (-) enantiomer
diastereoisomers
stereoisomers but not mirror images of eachother
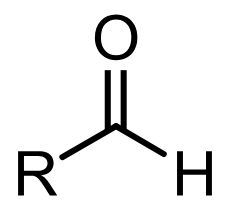
aldehyde
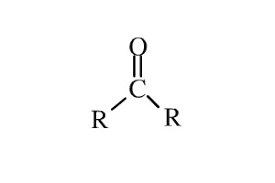
ketone
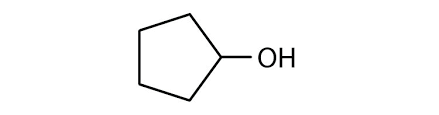
alcohol
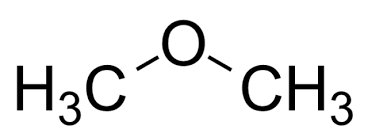
ether
alkyl halide
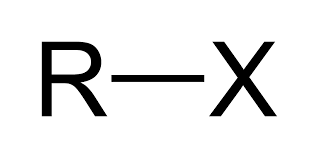
thiol
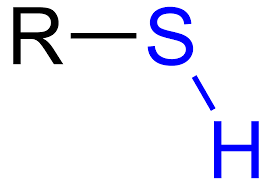
dehydration reaction
loss of H2O from a starting material
elimination reaction
a reaction where elements of the starting material are lost and a new multiple bond is formed
zaitsev rule
the major product in elimination is the alkene with more alkyl groups (CH3 groups) bonded to it
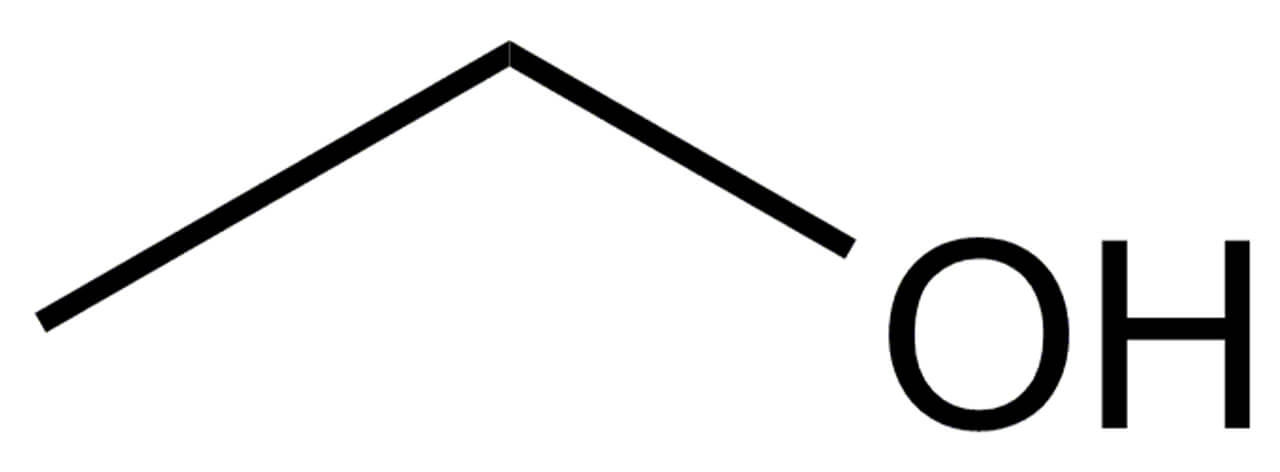
ethanol
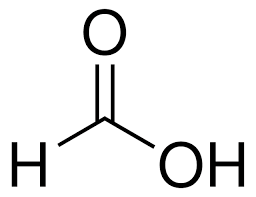
formic acid
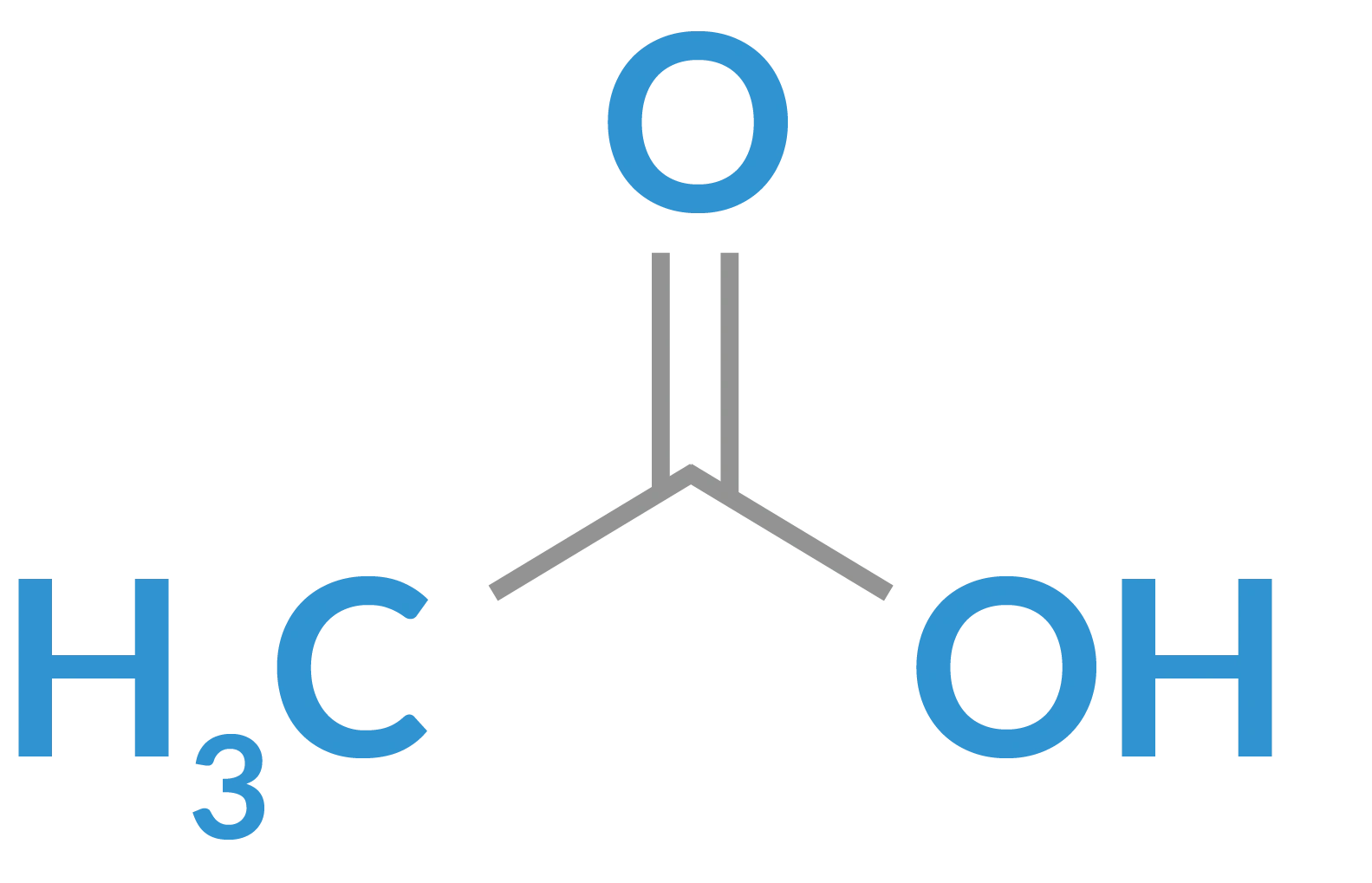
acetic acid
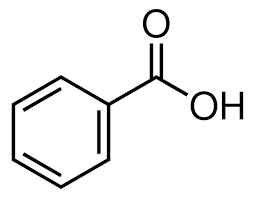
benzoic acid
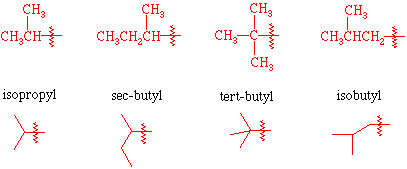
isopropyl
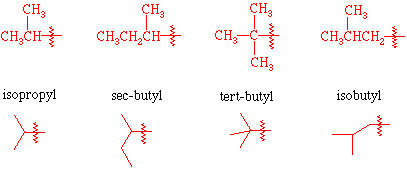
tert butyl
tertiary carbon attached to branched alkyl group
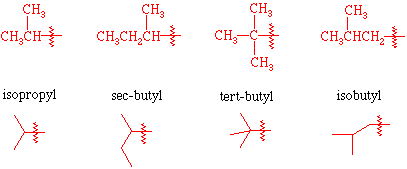
sec-butyl
secondary carbon attached to branched alkyl group