VSEPR chem 142
1/24
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
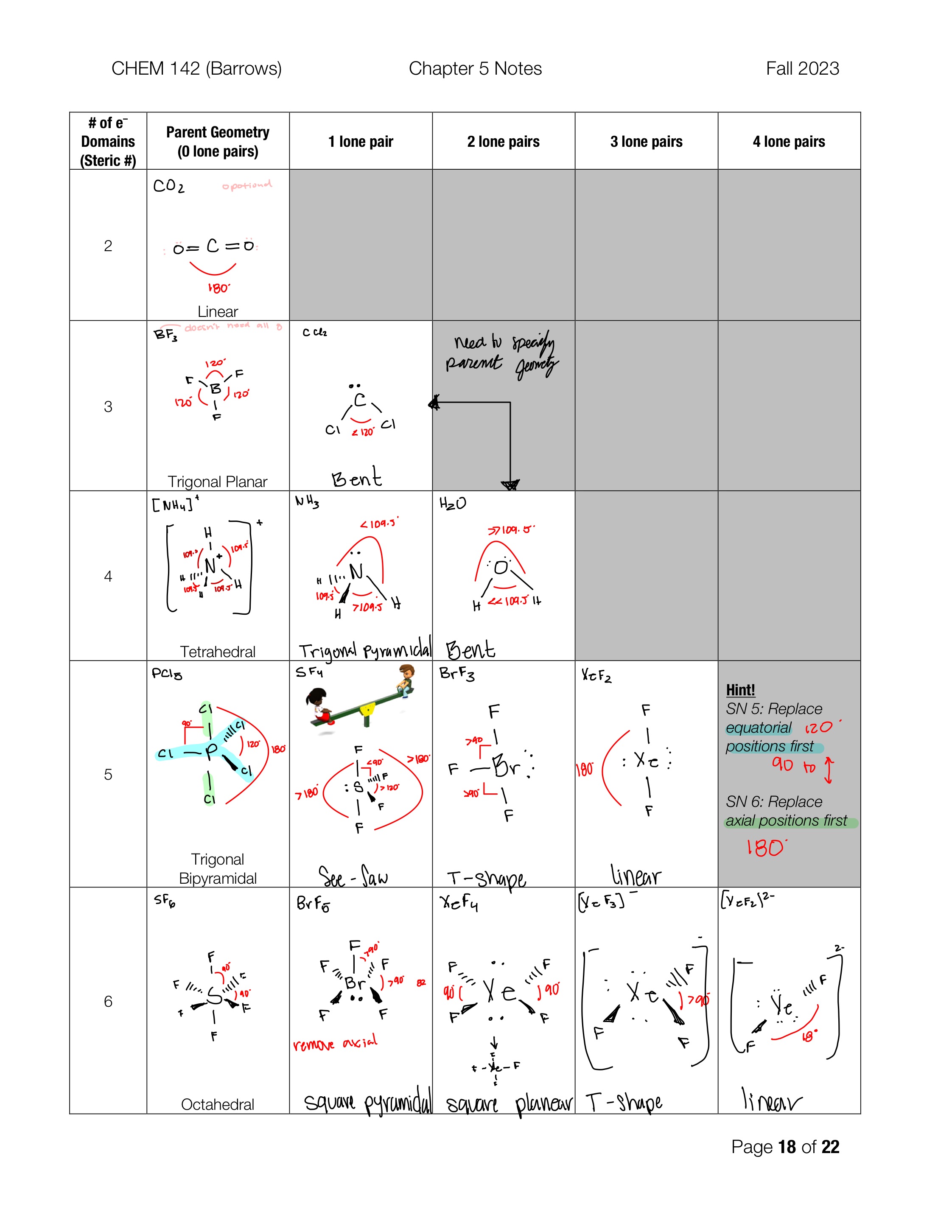
Linear
Electron Domain: 2
Trigonal planar
Electron Domain: 3
Bent
Molecular geometry: 3 electron domains, 1 lone pair
Tetrahedral
Electron Domain: 4
Trigonale pyramidal
Molecular geometry: 4 electron domains, 1 lone pair
Bent
Molecular geometry: 4 electron domains, 2 lone pair
Trigonal bipyramidal
Electron Domine: 5
See-Saw
Molecular geometry: 5 electron domains, 1 lone pair
T-shape
Molecular geometry: 5 electron domains, 2 lone pair
Linear
Molecular geometry: 5 electron domains, 3 lone pair
octahedral
Electron Domain: 6
Square pyramidal
Molecular geometry: 6 electron domains, 1 lone pair
square planer
Molecular geometry: 6 electron domains, 2 lone pair
T-shape
Molecular geometry: 6 electron domains, 3 lone pair
linear
Molecular geometry: 6 electron domains, 4 lone pair
equatorial postion
the plane around the x axis of the atom
Axial position
the plane around the y axis of the atom
SN5
Remove equatorial positions first and replace with lone Paris
SN6
Remove axial positions first and replace with lone pairs
All
ya
Linear Bond angle
180
ideal trigonal planar bond angle
120
Ideal tetrahedral bond angle
109.5
Ideal trigonal bipyramidal bond angles
120 and 90
ideal octahedral bond angle
90