Chemistry - chemical changes
1/115
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
116 Terms
What is the reactivity series?
A list of elements ordered by their reactivity
The reactivity series
Potassium
sodium
calcium
magnesium
aluminium
carbon
zinc
iron
lead
hydrogen
copper
silver
gold
Which elements in the reactivity series react with oxygen?
everyone except gold
Reaction with water (K - Ca)
fizz, give off hydrogen gas and leave behind an alkaline solution of metal hydroxide
Reaction with water (Mg - Fe)
very slow reaction
Reaction with water (Sn and Pb)
slight reaction with steam
Reaction with water (Cu - Au)
no reaction even with steam
Reaction with dilute acid (K - Li)
explodes
Reaction with dilute acid (Ca - Fe)
fizzing, giving off hydrogen gas and forming a salt
Reaction with dilute acid (Sn and Pb)
reacts slowly with warm acid
Reaction with dilute acid (Cu - Au)
no reaction
What are displacement reactions?
A reaction in which one substance replaces another. The more reactive element "pushes out" a less reactive element from a compound.
What is oxidation in terms of oxygen?
gain of oxygen.
What is reduction in terms of oxygen?
loss of oxygen
What is oxidation in terms of electrons?
loss of electrons
What is reduction in electrons?
gain of electrons
What are redox reactions?
chemical reactions that transfer electrons between reactants
Example of redox reactions
Zn + CuSO₄→ZnSO₄ + Cu
Zn is oxidised, and copper is reduced
What are ionic equations?
equations that only show the atom and ions that change and take part in a chemical reaction
What are spectator ions?
The ions that don't change in the reaction. they don't change form and appear on both sides of the equation
Ionic equation for neutralisation
H⁺(aq) + OH⁻(aq) --> H2O(l)
How are the metals above carbon extracted?
by electrolysis because of their high reactivity
How are the metals below carbon to copper extracted?
reduction with carbon in a blast furnace to cause a replacement reaction, removing the oxygen
How are metals like silver or gold extracted?
They are found as pure metals because they don't form ores
What are acids?
- Substances that release hydrogen ions in a solution
- They have a Ph less than 7
What makes solutions acidic?
A solution is acidic when it has a high concentration of hydrogen ions
Examples of acids
- hydrochloric acid (HCl)
- Nitric acid (HNO₃)
- Sulfuric acid (H₂SO₄)
What are buffers?
Chemicals that resist changes to pH (keep pH constant)
What is the pH scale?
a scale that measures how acidic or alkaline a solution is.
What is ph?
a scale used to measure the concentration of H⁺ ions
What does a low pH mean?
The solution is acidic
What does a high pH mean?
The solution is alkaline
What does a pH of 7 mean
The solution is neutral
What does a high concentration of H+ ions mean?
A solution is highly acidic and has a low pH
What does a low concentration of H+ ions mean?
It indicates a weak acid, a highly diluted acid, or an alkali
How does the pH scale measure acids?
- by the concentration of H+ ions
- the higher the concentration, the lower the pH
What does pH stand for?
potential of hydrogen
What does a decrease mean on the pH scale?
- Every decrease of 1 on the scale is an increase in H+ by a factor of 10
- e.g, 0.1 mol/dm³ of H⁺ ions = pH 1
- 0.01 mol/dm³ of H⁺ ions = pH 2
- 0.001 mol/dm³ of H⁺ ions = pH 3
What is the universal solvent?
H₂O
What is the pH of a substance with a concentration of 0.1 mol/dm³ of H⁺ ions?
pH 1
What is the pH of a substance with a concentration of 0.01 mol/dm³ of H⁺ ions?
pH 2
What is the pH of a substance with a concentration of 10⁻⁵ mol/dm³ of H⁺ ions?
pH 5
What is the pH of a substance with a concentration of 10⁻⁹ mol/dm³ of H⁺ ions?
pH 9
What is a base?
- any substance that neutralises an acid to form only salt and water
- They are typically metal oxides, metal hydroxides, or metal carbonates.
- they have a pH greater than 7
How do bases neutralise acids?
They are hydrogen ion acceptors. When they react, they accept H+ions.
What is an alkali?
- a soluble base that dissolves in water to produce hydroxide ions
- they have a pH greater than 7
What is the difference between bases and alkalis?
A base is any substance that neutralises acids to form water and salt, while an alkali is specifically a base that is soluble in water
Examples of bases
- Sodium hydroxide (NaOH)
- Lime water Ca(OH)2
- Ammonia NH3
What makes solutions alkaline?
A higher concentration of OH⁻ ions than H⁺ ions
What are alkalis used for?
cleaning, industrial manufacturing, and neutralising acids
Everyday examples of alkalis
drain cleaner, oven cleaner, and bleach use strong alkalis to break down grease and fats.
Acid + base
→salt + water
Metal + Acid
→ salt + hydrogen
Acid + alkali
→salt + water
Acid + Carbonate
→salt + water + CO₂
metal oxide + acid
→salt + water
Neutralisation ionic equation
H⁺(aq) + OH⁻→H₂O
Acid + ammonia
→ ammonium salt
What is a salt?
an ionic compound made from any acid-base reaction
What does concentrated mean?
There is lots of solute in a particular volume
What does dilute mean?
There is little solute in a particular volume
What is a concentrated acid?
an aqueous solution containing a high proportion of acid molecules and very little water
What is the pH of a concentrated acid?
0-4
What is a dilute acid?
an aqueous solution containing a low proportion of acid molecules and lots water
What is the pH of a dilute acid?
4-6
Why don't acids form ionic bonds?
because the water molecules get in the way
What is a strong acid?
- an acid that fully dissociates in aqueous solution
- they produce a higher concentration of H⁺ ions so they have a lower pH (0-4)
Why are strong acids more reactive than weak acids at the same concentration?
They have a higher concentration of H⁺ ions, which means there are more reactive particles available, leading to faster, more vigorous reactions with substances like metals, bases, or carbonates compared to weak acids.
Examples of strong acids
- hydrochloric acid (HCl)
- sulfuric acid (H₂SO₄)
- nitirc acid (HNO)
Equation for full dissociation
HCL→H⁺ + Cl⁻
What is a weak acid?
- An acid that does not completely ionise/dissociate in water
-They have a higher pH than stronger acids because they release fewer H⁺ ions (4-6)
Examples of weak acids
- Ethanoic acid,CH₃COOH
- Citric acid,C₆H₈O₇
- Carbonic acid, H₂CO₃
Equation for dissociation of weak acids
CH₃COOH ⇌ H⁺ + CH₃COO⁻
Why is the dissociation of weak acids reversible?
they only partially ionise in water, creating a dynamic equilibrium between the molecules and their ions
What are strong bases?
compounds that dissociate completely into hydroxide ions in aqueous solution, resulting in a high pH. (11-14)
Examples of strong bases
- hydroxides of alkali metals (Group 1) and alkaline earth metals (Group 2)
- LiOH, NaOH, KOH, RbOH, CsOH, Ca(OH)₂, Sr(OH)₂, Ba(OH)₂
What is a weak base?
- a substance that only partially ionizes or dissociates in water, producing a low concentration of hydroxide ions
- they have a lower pH than strong acids (8-11)
Why does diluting acids increase the pH?
Because it reduces the concentration of H⁺ ions
What is electrolysis?
Electrolysis is the breakdown of a substance by passing an electric current through it.
What is a buffer?
- A solution which resists a pH change when small amounts of acid or alkali are added
- they either release H⁺ to lower the pH or take up H⁺ to raise the pH
What is an electrolyte?
An ionic compound that dissociates into ions when placed in water.
What kind of compounds are used in electrolysis?
ionic compounds, either molten or dissolved in water (aqueous solution)
What are electrodes made from?
inert substances such as graphite or platinum
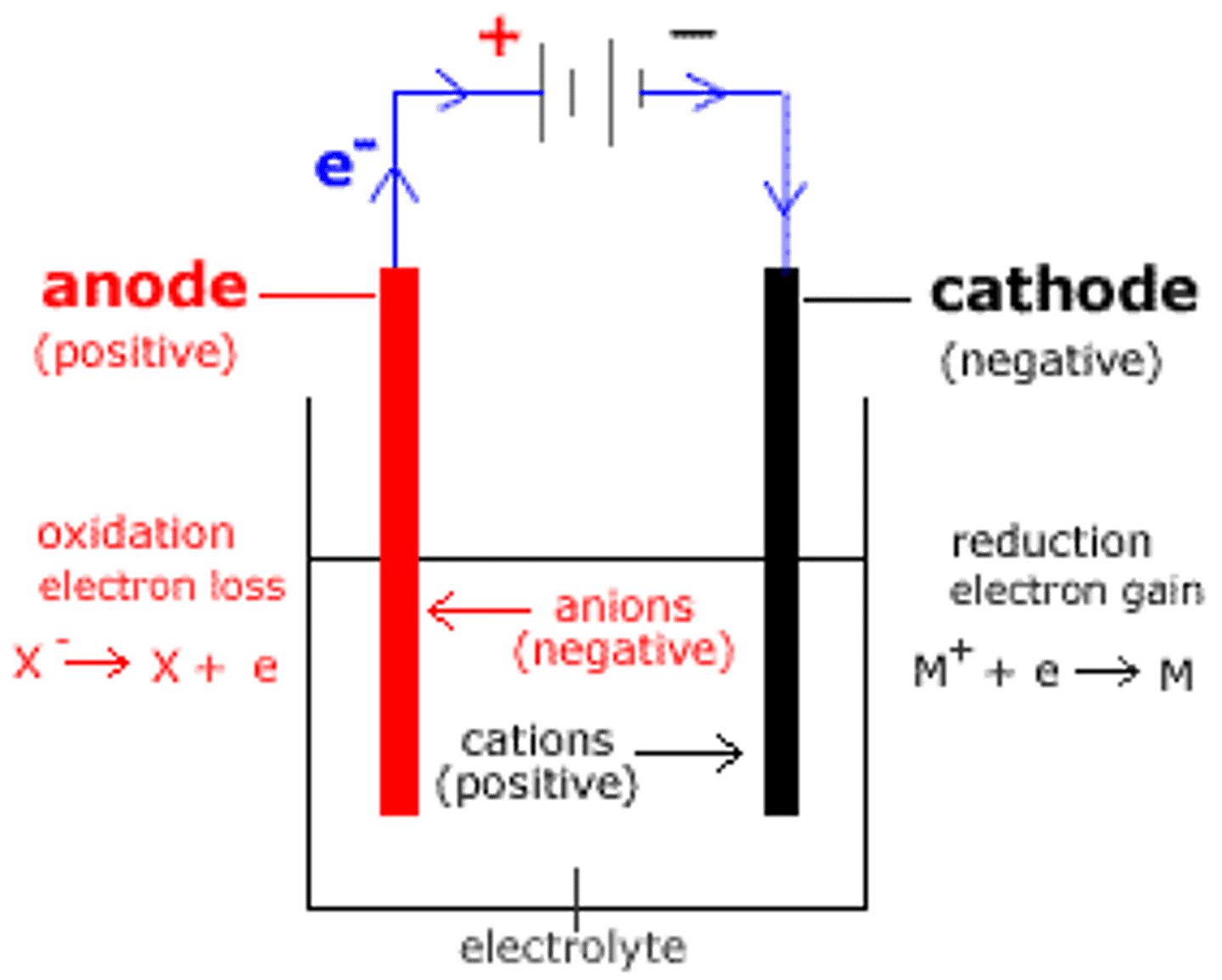
What is the cathode in electrolysis?
- The negatively charged electrode, where reduction (gain of electrons) occurs
- it is connected to the negative terminal of the power supply
What is the anode in electrolysis?
- the positively charged electrode where oxidation (loss) of electrons occurs
- it is connected to the positive terminal
What does PANIC stand for?
Positive
Anode
Negative
Is
Cathode
What are anions in electrolysis?
negatively charged ions in electrolysis that are oxidised by the positive electrode (anode)
What are cations in electrolysis?
positively charged ions that are reduced by the negative electrode (cathode) during electrolysis
How does electrolysis work?
- One electrode is connected to the positive terminal of the power supply. This is called the anode
- The other electrode is connected to the negative terminal and is called the cathode
When current flows:
- positive ions (cations) move to the negative electrode (cathode) and gain electrons (reduction)
- negative ions (anions) move to the positive electrode (anode) and lose electrons (oxidise)
- When the ions reach the electrodes, they lose their charge and become elements
Half equation of oxygen
4OH⁻ →O₂ + 2H₂O +4e⁻
Half equation for chlorine
2Cl(l)⁻→ Cl₂(g) + 2e⁻
Half equation for zinc
Zn²⁺(aq) + 2e⁻→ Zn(s)
Half equation for hydrogen
2H⁺ + 2e⁻ →H₂
Why are electrodes made from graphite?
- it is a good conductor of electricity
- it is inert (non-reactive)
- it is capable of withstanding extreme temperatures
- it is cost-effective
What are the rules of electrolysis in solution?
- if the metal ion is more reactive than hydrogen, hydrogen will form at the cathode
- if there is a halogen involved, the halogen will form; if not, then oxygen will form
What happens when there are two elements that can be formed at an electrode?
the more reactive metal will form
Why do electrodes need to be constantly replaced?
- Positive carbon anodes must be constantly replaced because they burn away
- The oxygen produced at the anode reacts with the hot carbon, forming carbon dioxide gas, which consumes the electrode
What is cryolite?
a mineral used as a solvent in the electrolytic extraction of aluminium from molten aluminium oxide
Why do electrodes need to be inert?
to conduct electrons without chemically reacting with the electrolyte or products
Why is cryolite used in the electrolysis of aluminium oxide?
- to lower its melting point from over to around. This significantly reduces energy consumption and costs
- acts as a solvent for the aluminium oxide
- increases electrical conductivity.