1.7 Water
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Why is water a polar molecule? (3)
- Water is a polar molecule due to the uneven distribution of charge within the molecule.
- The oxygen atom is more electronegative than the hydrogen atoms.
- This attracts the electrons more strongly, causing one end of the molecule to be more positive than the other.
What is the significance of water's high specific heat capacity? (2)
- A lot of energy is required to change its temperature, which minimises temperature fluctuations in aquatic environments.
- This allows organisms in rivers and lakes to survive in different seasons.
What is the significance of water's high latent heat of vaporisation? (2)
- A large amount of energy is needed for it to evaporate.
- This provides a significant cooling effect with little water loss, for example, through sweating.
What are cohesion, adhesion, and capillarity in water? (3)
- Cohesion is the sticking together of water molecules due to hydrogen bonding between adjacent molecules.
- Adhesion is when water molecules adhere to the sides of tube-like cells, such as in xylem vessels.
- Capillarity is the passive movement of water through narrow tubes.
Why is ice less dense than liquid water? (3)
- Water reaches its maximum density at 4 degrees Celsius; in ice, the molecules are spread out and fixed in place, making it less dense.
- Therefore, ice floats on top of water, creating an insulating layer.
- This prevents large bodies of water from freezing solid, increasing the chance of survival for aquatic organisms during winter.
How does the incompressibility of water support organisms? (2)
- It provides structural support in hydrostatic skeletons, for example, in earthworms.
- It also allows hydraulic movement, such as pressure changes in xylem or muscle cavities.
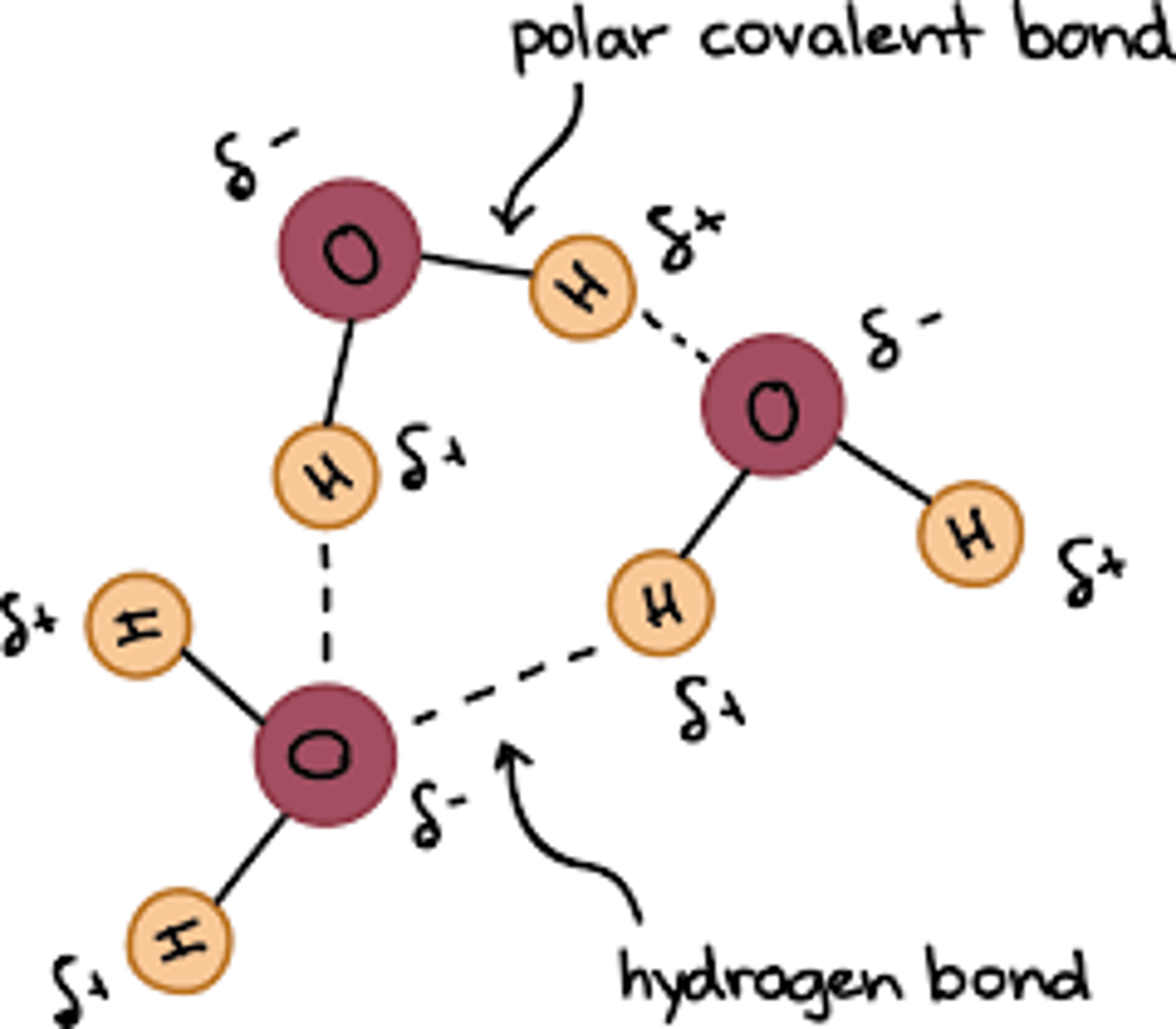
How does the dipole nature of water allow for hydrogen bonding? (3)
- Water molecules are polar and have a slight positive charge on hydrogen and a slight negative charge on oxygen.
- The oxygen nucleus is stronger than the hydrogen nuclei, so electrons are attracted much closer to the oxygen atom.
- Opposite charges between adjacent water molecules attract, forming hydrogen bonds.
How does the high specific heat capacity of water benefit aquatic organisms? (2)
- It prevents rapid temperature changes in their aquatic environment.
- This provides stable conditions for enzyme-controlled reactions to occur efficiently.
Why do ocean temperatures change less than land temperatures? (3)
- Water has a high specific heat capacity, so it absorbs lots of energy without a large temperature change.
- This prevents large temperature fluctuations in oceans.
- This helps to keep enzyme activity and oxygen availability stable for marine life.
Why are globular proteins soluble in water? (2)
- Hydrophilic R-groups on a globular protein face outwards, interacting with water.
- Water forms hydrogen bonds with these polar R-groups, allowing the proteins to dissolve.
What is the basic structure of a water molecule? (3)
- A water molecule is a simple covalent molecule.
- It consists of one oxygen atom and two hydrogen atoms.
- It is a dipolar molecule, which means it has opposite charges and displays electrical polarity.
Why is water considered a good solvent? (2)
- Due to hydrogen bonding, polar water molecules can attract ions.
- This attraction allows water to disassociate ionic compounds, causing them to dissolve.
What is the role of water in transport within biological systems? (3)
- Water's solvent properties allow substances to be dissolved in it for easy transport.
- Important biological fluids like blood plasma, tissue fluids, and lymph are predominantly water.
- Digestive juices and tears also use water to transport substances like salts and enzymes.
How is water involved in the removal of metabolic wastes? (2)
- Metabolic wastes like ammonia and carbon dioxide are transported in a water solution.
- This allows for a greater concentration of the substance to be diffused through membranes for efficient removal from the body.
What is the difference between hydrophilic and hydrophobic substances? (2)
- Hydrophilic substances are polar and are attracted to water.
- Hydrophobic substances are non-polar and are repelled by water.
What is hydrolysis and why is water a good medium for chemical reactions? (3)
- Hydrolysis is a reaction in which water is used to break down large substances, such as proteins into amino acids.
- All chemical reactions in living organisms occur in an aqueous solution.
- This allows the molecules to move freely and react with each other.
How does water act as a medium for dispersal in plants? (2)
- Mosses and ferns rely on water to transfer the male gamete for fertilisation.
- In the squirting cucumber, a build-up in osmotic pressure helps to disperse the seed.
What is the role of water in hearing and balance? (3)
- The watery endolymph and perilymph fluids are involved in hearing and balance.
- Vibrations are carried by these fluids to the cilia, which detect motion and send impulses to the brain.
- An unequal distribution of this fluid can trigger a response in the brain concerning balance.
Why does water have surface tension? (3)
- Due to its electrical polarity, water molecules form hydrogen bonds with each other.
- These bonds create strong cohesive forces, pulling the molecules inwards.
- At the surface, this inward pull creates a skin-like effect.
How does surface tension support small organisms like pond skaters? (3)
- Water molecules are polar and form hydrogen bonds.
- Cohesive forces between water molecules pull them inward at the surface.
- This creates a surface tension strong enough to support small organisms.
How does water enable earthworms to change shape? (3)
- Water molecules are incompressible.
- This is because of strong cohesion due to hydrogen bonding.
- Hence, incompressibility aids in muscular contractions and allows changes in volume without compressing the fluid.
Why does the dipole nature of water lead to stable ocean temperatures? (2)
- The dipole nature of water molecules allows them to form numerous strong hydrogen bonds between them.
- This gives water a high specific heat capacity, meaning a large amount of energy is required to break these bonds and raise its temperature.
What is the latent heat of vaporization? (2)
- It is the energy required to overcome the hydrogen bonds between water molecules.
- This allows the molecules to separate and enter a gaseous state without a change in temperature.
How does sweating cool the body? (2)
- Water absorbs a large amount of energy from the body in order to evaporate.
- This evaporation removes heat, leaving a lower average kinetic energy level behind, which cools the surface.
What is specific heat capacity? (1)
It is the amount of heat needed to raise the temperature of a given mass of a substance.
Why is water's high specific heat capacity important for living organisms? (3)
- It ensures aquatic environments are thermally stable because temperature change is very slow.
- It buffers cells against sharp temperature changes.
This stability reduces the chances of enzyme denaturation.
Why is water a useful lubricant? (2)
- The viscosity of water makes it an effective lubricant.
- Lubrication helps to minimise damage from friction between surfaces.
What are three examples of water-based lubricants in the body? (3)
- Synovial fluid lubricates the movement between many vertebrate joints.
- Pleural fluid lubricates the movement of the lungs during breathing.
- Pericardial fluid lubricates the movement of the heart.
What is a hydrostatic skeleton? (3)
- A hydrostatic skeleton is a support system found in animals like earthworms.
- These animals are supported by the pressure of an aqueous medium within their bodies.
- Water is incompressible because its molecules lie close together due to large cohesive forces between them.
How are herbaceous plants supported by water? (2)
- Herbaceous plants are supported by the osmotic influx of water into their cells.
- This creates turgor pressure, which keeps the cells rigid.
What are two other ways water provides support in organisms? (2)
- In vertebrates, the shape of the eye is maintained by the aqueous and vitreous humours.
- Water provides support to the organisms which live within it.
Why is the stable temperature of oceans important for organisms like fish? (2)
- The body temperature of a fish, being a cold-blooded organism, fluctuates with the temperature of the surrounding water.
- A stable water temperature helps the fish maintain a constant and appropriate body temperature, which is vital for its enzymes to function correctly and for its metabolism.
How does the dipole nature of water assist transpiration? (3)
- The dipole nature of water allows for the formation of hydrogen bonds between water molecules.
- These hydrogen bonds create strong cohesive forces, causing water molecules to stick together and allowing them to be pulled up the xylem in a continuous column.
- The dipole nature also results in adhesive forces between the water molecules and the walls of the xylem, which helps to support the column of water against gravity.
How does a drought affect the quality of food for a herbivore? (2)
- During a drought, there is less water available in the soil for plants to take up through their roots.
- This reduces the transport of mineral ions from the soil into the plant, which lowers the nutritional quality of the plant tissue.
How does a drought affect the quantity of food for a herbivore? (3)
- The lack of water limits the rate of photosynthesis, as water is required for the light-dependent reaction.
- A reduced rate of the light-dependent reaction means less GALP and glucose are produced.
- This leads to a reduction in the overall growth and biomass of the plants, decreasing the quantity of food available.