Management of Ocular Pain
1/73
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
74 Terms
What are the typical causes for acute ocular pain?
Conjunctival or Corneal FB
Conjucntival or corneal abrasion
Traumatic Iritis
Herpes Zoster
Post surgical PRK, PTK, superficial keratectomy among others
What are Nociceptors?
Specialized nerve endings in tissue that transmit pain signals to cortex.
What is the role of Substance P in pain signaling?
Substance P mediates transmission of pain from activated nociceptors
Enhances signaling at synapses involved in pain pathways
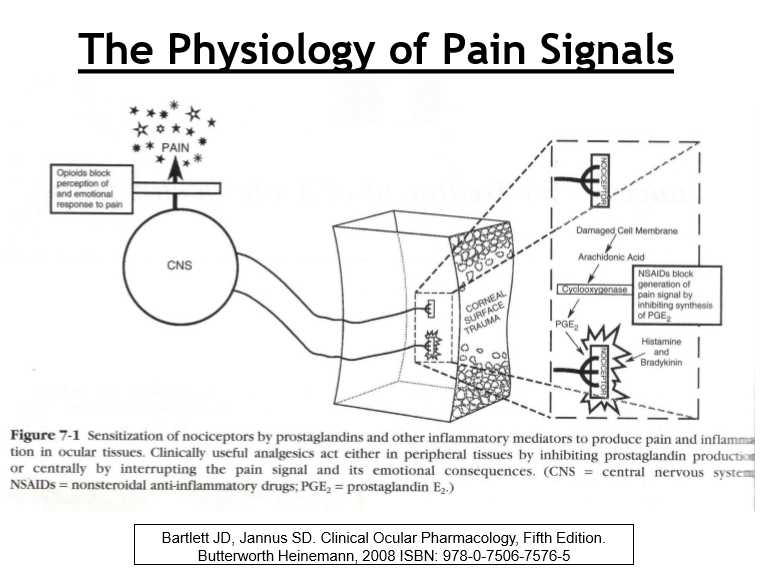
How do prostaglandins influence pain perception?
Arachidonic acid metabolites (prostaglandins) enhance/stimulate Substance P activity
Result: amplified pain signaling and increased sensitivity
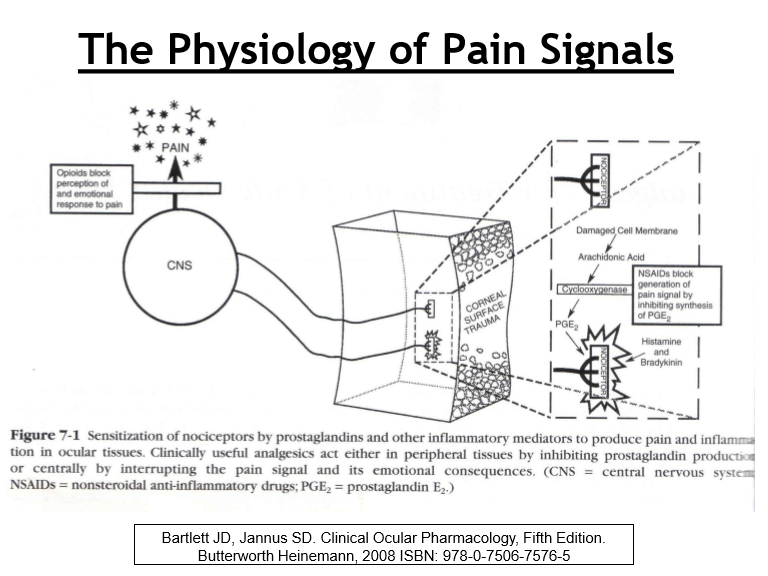
Where must pain signals be processed for pain to be consciously perceived?
Pain is only felt if the signal is fully received and processed in the cerebral cortex
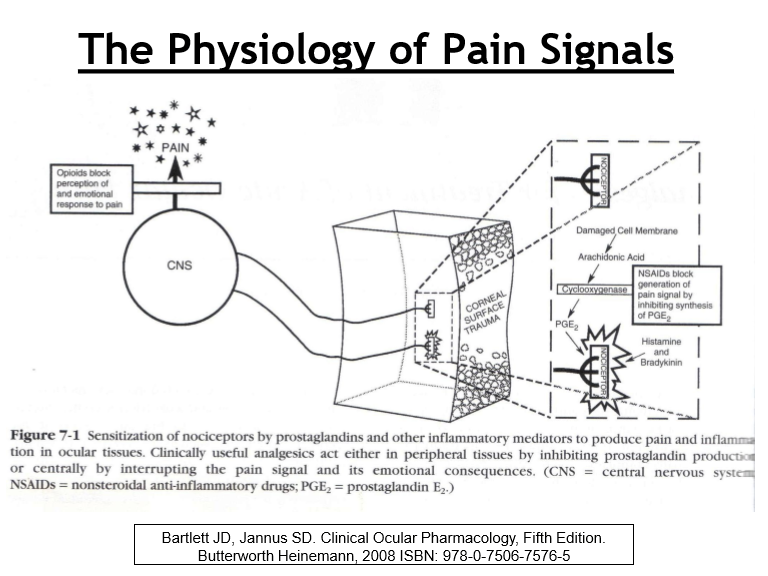
Where along the pain pathway can pain be pharmacologically interrupted?
Periphery: ↓ prostaglandins or substance P → ↓ nociceptor sensitization
Signal transmission: block nerve conduction → prevent signal reaching cortex
Central perception: alter pain processing in cerebral cortex
How do NSAIDs reduce pain at the peripheral level?
Inhibit COX → ↓ prostaglandin (PGE₂) synthesis
↓ sensitization of nociceptors to Substance P
Which drugs inhibit Substance P directly, and what is their limitation?
Capsaicin
Depletes Substance P → ↓ nociceptor activation
Dermatologic use only (not used intraocularly)
How do anesthetics reduce pain?
Block pain signal transmission along nerves
Prevent signal from reaching the cerebral cortex
How does acetaminophen reduce pain?
Acts centrally in cortex to alter pain perception
Minimal peripheral anti‑inflammatory effect
How do opioids reduce pain?
Alters central nervous system perception of pain via 3 receptor types:
Mu (μ): supraspinal analgesia, sedation, respiratory depression
Kappa (κ): spinal analgesia
Delta (δ): relatively unclear/less well‑defined effects
What are the advantages of using topical NSAIDs for ocular pain?
Direct application to site of injury/inflammation
Acts locally at nociceptor nerve endings
Limits systemic absorption and side effects
What are topical ocular NSAIDs used to treat?
Post Refractive Surgery
Post Catarct Surgery
Post FB removal
Pre/Post Betadine Treatment
Bullous Keratopathy & Acute Corneal Hydrops
How do NSAIDs work?
Cyclo-oxygenase inhibitor that prevent/decrease the formation of prostaglandins
Provides anagesia by reducing PGE2 to reduce pain
Anti-inflam by reducing PGD2 to decrease vasodilation and vascular permeability
Anti-platelet properties
Anti-pyretic properties
What are the NSAIDs and their dosages for acute ocular pain?
Ibuprofen 400-600mg PO QID (max: 2400mg/24hrs)
Ketoprofen 50mg PO q6-8hr
Naproxen sodium 250-500mg PO BID
Take with food to decrease GI effects
What are the SE of oral NSAIDs?
Hemorrhage
GI distress, heartburn, duodenal ulcer
Nephrotoxicity
What are the contraindications for oral NSAIDs?
Hyphema
Gastric/peptic ulcer
Renal insufficiency of CHF
Pregnant/nursing mothers
No aspirin for children d/t risk of Reye’s syndrome
What is Acetaminophe/tylenol’s therapeutic effect?
Analgesia
Antipyretic
Has no effect on inflammation or platelets
What are the SE of Acetaminophen?
No GI SE
No anticlotting/bleeding effects
No cross reactivity with NSAIDs
Safe for kids and during pregnancy
Liver toxicity if exceed recommended dosage or if in alcoholic or known liver disease
What are the contraindications for Acetaminophen?
Chronic alcoholics
What is the typical dosing of Acetaminophen?
325-1000mg PO q6 hour
Tylenol regular strength: 325mg
Tylenol extra strength: 500mg
In 2011, reduced maximal daily dosage from 4g/24hrs to 3g/24hr d/t high incidence of inducing liver disease
How is Acetaminophen and NSAIDs dosed?
Alternate drug every 3 hours

What are opiates/opioid analgesics, and what is the prototypical agent?
All derived from opium or opium‑like compounds
Prototypical opioid: morphine
What is the primary mechanism of action of opioid analgesics?
Alter central nervous system perception of pain
Act via opioid receptors in the brain and spinal cord
What are the three main opioid receptor types and their clinical effects?
Mu (μ): supraspinal analgesia, sedation, respiratory depression
Kappa (κ): spinal analgesia
Delta (δ): relatively unclear/less well‑defined effects
What are the therapeutic effects of opiates?
analgesia
Anesthesia
Cough suppression
Anti-diarrheal
What are the SE of opiates?
Constipation
Dry mouth
Nausea
Sedation, dizziness, weakness
Addition/dependence d/t euphoric effects
Respiratory depression, can be fatal
What are the contraindications for opiates?
Asthma, COPD
Beware of sleep apnea
Pregnancy
An unidentifiable cause of pain
What should be done or not done when taking opiates?
Take opiates with food to decrease GI effects
Avoid driving or other activities requiring alertness
Avoid alcohol, muscle relaxants
What determines a drug’s DEA schedule?
Abuse potential
Medical use in the U.S.
Risk of dependence (physical or psychological)
Schedule I = highest abuse, no accepted medical use → Schedule V = lowest abuse
What characterizes Schedule I controlled substances?
Very high abuse potential
No accepted medical use
Illegal to prescribe
What are common examples of Schedule I drugs?
Heroin
LSD
MDMA (ecstasy)
Peyote
Quaaludes
Marijuana (federally Schedule I despite state legalization)
What characterizes Schedule II controlled substances?
High abuse potential
Accepted medical use
Severe dependence liability
What are examples of Schedule II drugs?
Cocaine
Amphetamines (including ADHD medications)
Opioids: morphine, oxycodone, hydrocodone
What characterizes Schedule III controlled substances?
Moderate abuse and dependence potential
Accepted medical use
Less restrictive prescribing than Schedule II
What are examples of Schedule III drugs?
Ketamine
Anabolic steroids
Combination opioids (e.g., Tylenol #3 with codeine)
What characterizes Schedule IV controlled substances?
Lower abuse and dependence potential
Accepted medical use
Refills allowed
What are common Schedule IV drugs?
Analgesics: Tramadol
Anti-anxiety agents: Xanax, Klonopin, Valium, Ativan
Sleep aids: Ambien, Lunesta
What characterizes Schedule V controlled substances?
Lowest abuse potential
Accepted medical use
Limited quantities, lowest level of control
What are examples of Schedule V drugs?
Antitussives/antidiarrheals with codeine
FDA‑approved CBD‑containing medications
What are the ocular indications for opiates?
Severe, acute pain from:
Severe Corneal abrasion or FB
Post PRK or other corneal surface procedures
Herpes Zoster and post-herpetic neuralgia
Which opioid analgesics are within the scope of practice for Ohio optometrists?
Tramadol (recognized therapeutic amount)
Codeine ≤ 60 mg, only if combined with non‑narcotic ingredients
Hydrocodone ≤ 7.5 mg, only if combined with non‑narcotic ingredients
What formulation limits apply to codeine and hydrocodone prescribed by Ohio optometrists?
Must be combination products
Must contain other active non‑narcotic ingredients
Dose limits strictly enforced (codeine ≤ 60 mg; hydrocodone ≤ 7.5 mg)
What are the quantity limits on opioid prescriptions by Ohio optometrists?
Maximum: single 4‑day supply
Per episode of illness, injury, and/or treatment
No extended or repeat opioid prescribing
What is tramadol, and why is it relevant to optometric prescribing?
Synthetic opioid, analog of codeine
Trade name: ConZip®
Became a Schedule IV controlled substance in Oct 2014
Permitted for Ohio ODs, but requires a DEA number
What is the abuse potential and side‑effect profile of tramadol?
Low addiction potential
Similar side effects to codeine: nausea, dizziness, sedation
Still carries risk of dependence and CNS depression
What is the standard adult dosing for tramadol?
50-100 mg PO every 4-6 hours as needed for pain
Use lowest effective dose for shortest duration
What tramadol combination product is commonly used, and how is it dosed?
Tramadol 37.5 mg / Acetaminophen 325 mg
2 tablets PO every 4–6 hours
Max: 8 tablets in 24 hours
What drugs interact with Tramadol?
Anti-depressants
What is codeine, and how does it produce analgesia?
Prodrug of morphine
Central opioid analgesic effect
What is the DEA schedule and typical non‑analgesic use of codeine?
Schedule III controlled substance
Standalone codeine commonly used for cough suppression
What analgesic formulations of codeine are commonly used?
Combination products with acetaminophen:
15 mg codeine / 300 mg acetaminophen
30 mg codeine / 300 mg acetaminophen
60 mg codeine / 300 mg acetaminophen
What is the standard adult dosing for codeine used as an analgesic?
30-60 mg PO every 4-6 hours
Dose must be considered within combination product limits
What is hydrocodone and how is it classified by the DEA?
Semi‑synthetic opioid analgesic
Reclassified in October 2014 from Schedule III → Schedule II
Change due to widespread abuse and dependence risk
How does the potency of hydrocodone compare to codeine?
6 times more potent than codeine
Produces stronger analgesia at lower doses
What are the characteristic side‑effect differences of hydrocodone compared to codeine?
Less constipation
Less sedation
More euphoria
What is the typical adult dosing for hydrocodone?
5-7.5 mg PO every 4-6 hours as needed for pain
How is hydrocodone most commonly formulated?
Combination products with non‑narcotic analgesics
What hydrocodone-acetaminophen combination strengths are available?
2.5 mg hydrocodone / 325 mg acetaminophen
5 mg hydrocodone / 300–325 mg acetaminophen
7.5 mg hydrocodone / 300–325 mg acetaminophen
10 mg hydrocodone / 300–325 mg acetaminophen
Generic formulations only
What hydrocodone-ibuprofen combination strengths are available?
2.5 mg hydrocodone / 200 mg ibuprofen
5 mg hydrocodone / 200 mg ibuprofen
7.5 mg hydrocodone / 200 mg ibuprofen
10 mg hydrocodone / 200 mg ibuprofen
Generic formulations only
Why are Ohio optometrists prohibited from prescribing oxycodone?
Schedule II opioid
10-12× more potent than codeine
Produces significant euphoria
High risk of dependence and abuse
What are common trade names and formulations of oxycodone?
Standalone: OxyContin, Roxicodone, RoxyBond, Xtampza ER
Combination with acetaminophen:
Percocet
Endocet
Roxicet
What is hydromorphone and why is it excluded from OD prescribing?
Trade name: Dilaudid
Schedule II opioid
Very potent µ‑opioid agonist
High abuse potential and overdose risk
What is required to obtain a DEA number to prescribe controlled substances?
731$ nonrefundable fee
Covers a 3‑year registration period
Does a DEA number transfer between states?
No. A separate DEA application is required for each state in which you practice
Having a DEA number in another state does not exempt reapplication
What is the key mechanism of opioid addiction/overdose, and why is opioid use a major U.S. public‑health issue?
Addiction: driven by opioid‑induced euphoria
Overdose deaths: due to respiratory depression
Epidemic fueled by overprescribing and illicit use
U.S. = disproportionate consumer: ~5% of world population but ~80% of global opioid use
What are the key legal requirements for prescribing controlled substances in Ohio?
Must be written or e‑prescribed (cannot be called in)
Recommended to keep a log of controlled‑substance prescriptions with patient signatures
Patient education is mandatory (Ohio Revised Code):
Must inform patients of the addictive nature of opioids
Must provide an opioid/narcotics patient handout
What is OARRS and how does it help reduce substance abuse in Ohio?
Ohio Automated Rx Reporting System (OARRS)
Secure database that allows authorized providers to review a patient’s controlled‑substance prescription history
Helps detect doctor shopping, overprescribing, and misuse
All pharmacies must report Schedule II–V dispensations at least weekly
How is Opioid Overdose treated?
Naloxone (Zimhi)
An opioid antagonist used to reverse effects of opioid overdose
Injectable by paramedics and also as a nasal spray
What medications are used to treat opioid use disorder, and how do they work?
Methadone: long‑acting full opioid agonist used as morphine alternative
Buprenorphine: partial agonist with lower overdose risk
Naltrexone: opioid antagonist for long‑term relapse prevention
What are the key pharmacologic features and risks of methadone?
Synthetic full opioid agonist
Very long half‑life (~60 hrs; up to 150 hrs)
Takes ~10-12 days to reach steady state
High overdose risk → involved in ~30% of opioid‑related deaths
Why is buprenorphine considered safer than full opioid agonists?
Partial agonist at µ‑opioid receptors
Ceiling effect on respiratory depression
Lower risk of fatal overdose
What is the role of naltrexone in opioid use disorder treatment?
Opioid antagonist
Blocks opioid effects
Used for long‑term maintenance / relapse prevention
What are the main non‑opioid options for managing ocular pain and their indications?
Cycloplegia: uveitis, keratitis (reduces ciliary spasm)
Bandage contact lens: large corneal abrasions
Topical NSAIDs: corneal hydrops, bullous keratopathy
Oral NSAIDs: drug of choice for ocular and periorbital pain
Oral acetaminophen: hyphema present or as alternative to NSAIDs