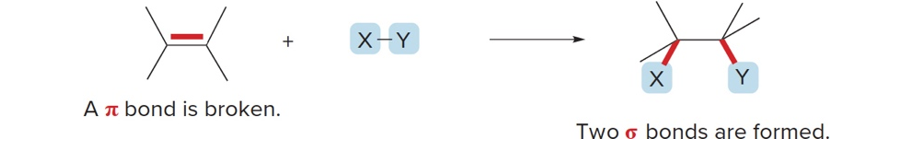1) Kinds of organic reactions
1/9
Earn XP
Description and Tags
page 1-2
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
What does hv (nu) stand for
light is needed in the reaction
What does the triangle (delta) stand for
heat is added to create products
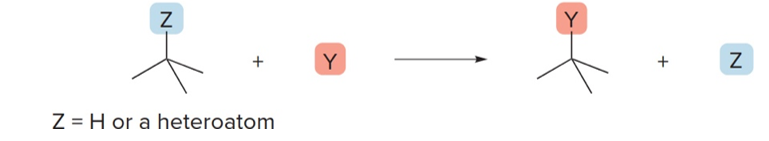
What is a substitution reaction?
An atom is replaced by another group of atoms
What does a substitution reaction involve?
Breaking and forming of sigma bonds
What does a substitution rxn usually swap out for?
swaps a less EN atom for a more EN atom
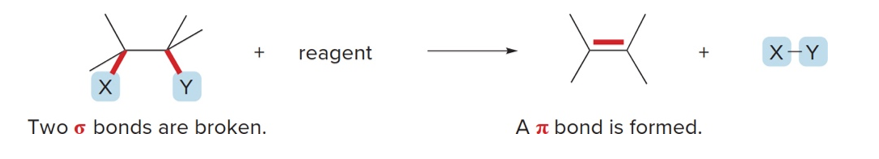
What is an elimination reaction?
The starting material is “lost” and a pi bond forms
What does an elimination reaction involve?
Breaking 2 sigma bonds, creating a new pi bond
1
2
What is an addition reaction?
Atoms are added to starting material
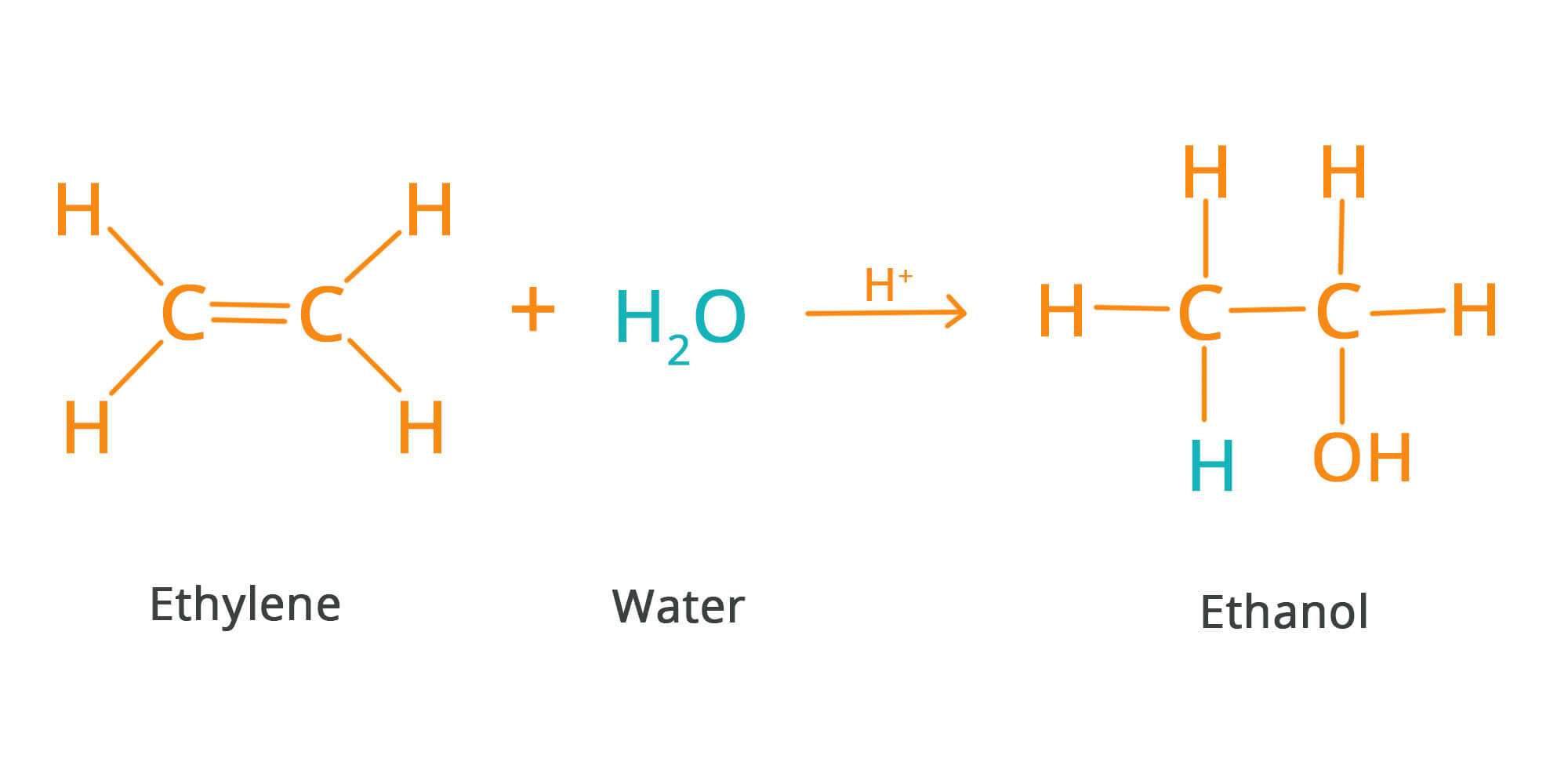
What does an addition reaction require?
The breaking of 1 pi bond, forming 2 sigma bonds