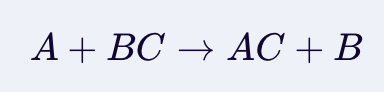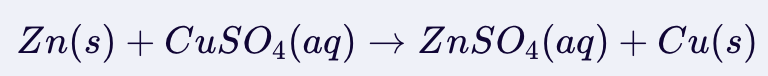Displacement - Forming Salts
1/4
Earn XP
Description and Tags
SOLUBILITY RULES, Double/Single DIsplacement, precipitation
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
What is a salt?
an ionic compound formed from the neutralisation reaction of an acid and a base, consisting of positively charged cations and negatively charged anions.
What neutralization
The reaction between a salt and base to form a salt and water.
Double Displacement Reaction
In a double displacement, the formation of a precipitate depends on the solubility of the resulting products.
Two aqueous solutions react and if they form an insoluble (in water) product, precipitate will form.
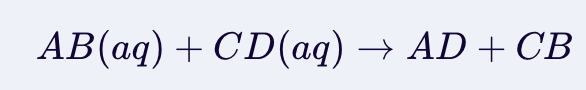
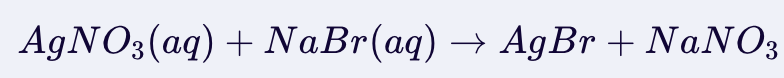
NAG-SAGS (solubility rules)
Nitrates, Acetates, Group 1, Sulfates, Ammonium, Group 17 (halides). = SOLUBLE
CH - Carbonate + Hydroxide = INSOLUBLE
Exceptions: INSOLUBLE
PMS - Lead sulfate/chloride/iodide/bromide, Mercury , Silver chloride/bromide
Castro Bear (Calcium, Strontium, Barium) for Sulfates.
Single Displacement
Depends on reactivity to determine if a reaction will occurd to form salt.