fsct326: exam 2
1/100
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
101 Terms
CDC 2016 List of Notifiable Foodborne Diseases, Bacterial
• Botulism (All forms including foodborne, infant)
• Brucellosis (Undulant Fever)
• Campylobacteriosis
• Cholera
• Listeriosis
• Q-Fever (Coxiella burnetii)
• Salmonellosis
• Shiga toxin-producing E. coli (STEC) disease, acute/chronic
• Shigellosis
• Vibriosis
• Yersiniosis (Not CDC, but is mandatory in Texas)
foodborne disease outbreak
typically occurring with 2 or more persons consume shared food item (contaminated) within localized timeline and/or geographic area
shared food vehicle → not alive carrier of disease
vector → living that carries disease to you
exception: C. botilinum disease → only 1 case required to define as outbreak
sporadic disease: typically single patients/cases, not affiliated with other persons or food items (not outbreak related)
outbreaks can range from very small and localized to very large (multi-state, -national, -continent)
Bacillus (Latin = “small staff“)
belongs to the family Bacillaceae
aerobic spore-forming rods (obligate aerobes), bearing psychrotolerant, mesophilic, and thermophilic spp.
some capable of anaerobic fermentation of glucose
proteolytic and non-proteolytics
spores do not distend (expand, disshapen) the sporangium (mother cell)
environmental reservoirs (ecological niche in which foodborne pathogens gains access to food-producing environment/processing) exist (soils, grasses, crop production, environments)
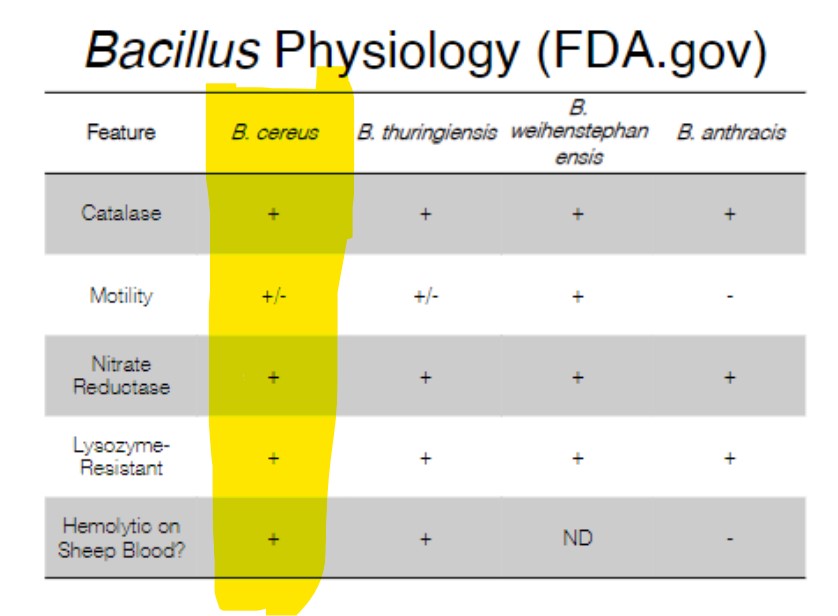
Bacillus cereus group: B. cereus, B. anthracis, B. thuringiensis, and B. weihenstephanensis → plasmid-borne pathogenesis elements
B. cereus and human disease
isolates capable of producing 1 or 2 key toxins
emetic toxin (intoxication)
intoxication → bacteria/toxin formed prior to consumption
emesis = throwing up, vomiting
enterotoxin/diarrheal toxin (toxico-infection)
toxico-infection → must consume bacterium, survives stomach, gets in lower intestinal tract, secretes enterotoxin and disrupts stomach balance)
entero- = gut in Latin
commonly associated with starchy foods and/or high proteins foods (milk, fish, meats)
contamination of animal or milk
spores survive pasteurization
some psychrotolerant strains identified- milk safety during refrigeration (growth at > 4°C)
B. cereus foodborne disease
characteristic | enterotoxin | emetic toxin |
# of cells needed for illness | 10^5 - 10^7 CFU total | 10^5 -10^9 CFU/g food |
toxin production? | small intestine | pre-formed in food |
incubation period | 8 -16 hr (typical) | 0.5 - 6 hr |
duration | 12- 24 hr (or longer) | 6 - 24 hr |
symptoms | abdominal pain, watery diarrhea, nausea | nausea, vomitting |
emetic toxin
a form of cereulide (amino-acid based)
enzymatically synthesized, plasmid-encoded
tolerant to large pH range (pH 2-11) and to heating/cooking (90 min at 121 °C)
longer than mandatory decontamination for some bio-waste in micro labs…
similar process tolerance as S. aureus enterotoxin
no impact of protease treatment
toxin production conditions
a_w, pH, O2 content, temperature, nutrients, [NaCl] → growth of microbes, respiration → toxin production
other Bacillus spp.
dairy spoilage
B. weihenstephanensis
B. wiedmanii (2016 discovered)
other genera derived, reclassified from Bacillus
Paenibacillus (“Like Bacillus): P. polymyxa, P. macerans
Geobacillus: G. stearothermophilus - Thermophilic flat
sour spoilage LACF
Clostridium botilinum
family Clostridiaceae (Greek = spindle, small spindle) (type species = C. butyricum)
C. botulinum first described in connection to consumption of raw undercooked blood sausages (Botulus: Latiin- sausage)
anaerobic regularly-shaped spore-forming rod (spore distends sporangium)
psychrotrophic, mesophilic, and thermophilic species
C. botulinum possess both psychotropic and mesophilic strains
C. botulinum physiology
cells may be pleomorphic in shape
motile by flagella (peritrichous)
may ferment various acids and can produce gas from sugars
typically catalase negative
C. botulinum classifications
8 toxin types: A. B, C_1, C_2, D, E, F, G
human disease : A, B, E, and F
groups:
1 (toxin A and proteolytic strains) → associated with large outbreaks and meat/poultry dishes
2 (non-proteolytics, type E)
3 (toxin types C and D)
4 (toxin type G)
foodborne botulism
adult, infant, wound, and indeterminate form
flaccid paralysis → blocking of acetylcholine release from muscle/nerve junction
adult botulism → foodborne intoxication
food vehicles: home-canned vegetables/foods (low-acid), vacuum-packed smoked fish, under-cooked canned foods, honey (infants), baked potatoes wrapped in foil, warmed-over extended holding foods
intoxicating dose: 0.4 ng (10^-9 g) per kg of body weight
symptoms of disease
incubation 12-36 hr post-consumption
symptoms
nausea, abdominal pain, vomiting
descending paralysis
droopy eyelids
slurred speech, loss of motor control in face
can have respiratory failure, inability to breathe (leading cause of botulism-derived fatality)
abdominal pain follows, constipation (typically infant botulism)
disease therapies
providing antiserum to bind and remove toxin
enema to expel toxin from GI tract rapidly
stomach pump, or induced vomiting
ventilator to assist, maintain breathing
most critical to proper treatment- proper diagnosis!
botulism in the US
increasingly associated with home-preserved foods
home-canning of vegetables, meats
low-acid foods
often a result of insufficient heating in jars (spores survive and germinate post-cooling)
proper cooking in home requires use of pressure cooker
cooking lowers E_h supporting growth
botulism cases, US, 2009-2018
about 80 cases of disease in US, 5 fatalities
most cases arise from consumption of contaminated fish, seal meat, grains, and canned veggies
recent foodborne botulism outbreaks in US
2017: nacho cheese from convenience stores (CA): 10 cases
2015: potato salad (home-canned potatoes)- OH (29 cases confirmed, suspected) [Described as largest botulism outbreak in 40 years in US]
2014: pesto (canned)- OH/CA (2 cases of adult females with disease)
2012: home-fermented tofu- NY (2 cases)
botulinal neurotoxin
two components: light and heavy chains
chains must separate to exact final toxin activity
Zn-metalloprotease: blocks acetyl-choline release
thermally labile (sensitive):
cooking 80 °C, 10 min (or boiling)
best practices of home canning (US CDC)
use recommended pressure canner holding at least four 1 qt. jars upright
ensure pressure cooker gauge accuracy
clean lid gaskets and components via manufacturer recommendations
vent canner before pressuring and follow recommended cooling procedures
use current process time/temperature combinations for specific food items of interest, jar size, method of food packaging in jar (with brine, without, salted, unsalted)
Clostridium perfringens
similar to other Clostridium species: rod-shaped, sporeformer, mesophilic pathogen
originally named C. welchii
some identify as facultative anaerobe due to plating capacity, but typically behaves as obligate anaerobe
non-motile by flagella
encapsulated organisms
bacterial capsules: excreted polysaccharides bonded to outer layers of cell wall, teichoic acids in Gram-positives
useful for attaching to solid surfaces, protecting against dehydration
capable of very rapid doubling times (10 min) under optimal conditions
optimal growth: 40-45 °C
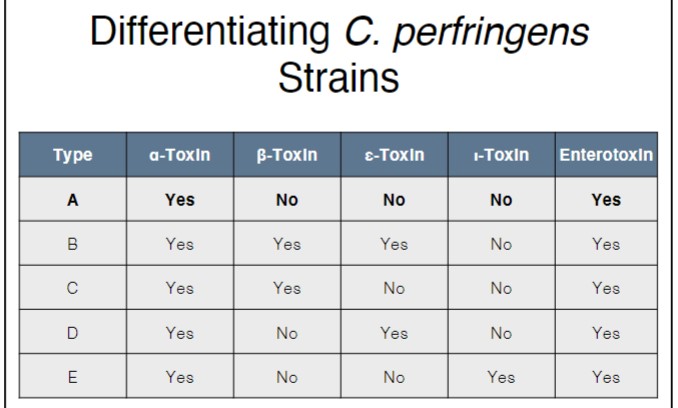
differentiating C. perfringens strains
foods associated with C. perfringens disease
common food vehicles are high protein-containing
stews, roasts, gravies, meat + gravy, seafood, poultry
13 essential amino acids cannot be synthesized (source effectively in high protein foods)
often foods undercooked or are properly cooked but cooled too slowly
vegetative cells destroyed; spores survive
re-heating shocks spores into germination and replication
preventing disease in the home
cooks foods thoroughly
use meat thermometer; check to ensure proper cooking tp recommended internal temperatures
consider size of meat dish in cooling plans
vegetative cells possess elevated heat tolerance
chill rapidly
use the refrigerator to chill leftovers- that’s why it was purchased; don’t use counter-top to “cool“
preventing C. perfrigens growth in food processing
very similar to consumer recommendations: proper cooking to destroy vegetative cells, rapid chilling to prevent spores from germinating/replicating
USDA-FSIS 1999: appendix B- stabilization
demands cooling of fully cooked roasts, meat, poultry products
increase in C. perfringens counts: no more than 1.0 log_10-cycle allowed during cooling
130 to 80 °F in 5 hr
addition of curing agents increases the amount of time required for cooling by preventing spores from germinating
processing hurdles to C. perfringens survival/growth
heat (thermal processing) and cold (refrigerated storage at/or below 6 °C)
reduction in pH by acidification; isolates don’t tend to grow below pH 5.0
reduction in a_w: 0.93 lower limit to growth
maintenance of elevated E_h (not always an option and there are tradeoffs)
foodborne perfringenosis
diarrheal disease (toxico-infection): commonly associated with type A toxin-producing C. perfringens (sometimes also type E)
acute (explosive) diarrheal with cramping (12-24 hr symptoms) → symptoms painful but short-lived
incubation period 8-12 hr
necrotic enteritis (pigbel; Darmbrand)
uncommon in US (observed in Papua New Guinea and surrounding regions today in non-industrialized countries
observed with C. perfringens producing the β-toxin (typically type C)
characterized by necrosis of jejunum and ileum; can be fatal if not diagnosed quickly and treated aggressively
also identified to occur in livestock animals via toxigenic C. perfringens
CDC estimates as 3rd leading cause of bacterial foodborne human disease!
characteristic of disease
typically requires moderate consumed dose (10^6 - 10^7 CFU/g); 8-16 hr incubation
fever is uncommon
passage through stomach in meaty protects during stomach passage
toxin released in small intestine as consequence of sporulation (accumulates in cell prior to mother cell lysis)
toxin causes damage to villus tips in intestinal cells
diarrheal symptoms continues to flush out excess toxin particles
Listeria monocytogenes
gram-positive cocco-bacillus facultatively anaerobic psychrotrophs
tumblee motility by flagella
over 15 species and sub-species (most identified within the last 5 years!)
genus shares similarities to Clostridium, Brochothrix, and other gram-positive genera
only L. monocytogenes (Lm) and L. ivanovii are identified pathogens (L. ivanovii is animal pathogen)
described as being environmental ubiquitous
Lm physiology
psychrotrophic growth (1-45 °C)
t_d (hr) = 43 @ 4 °C, 1.1 hr @ 37 °C)
can grow at low pH (4.3 - 4.4) but most rapid at neutral pH
growth at > 0.93 a_w
not very strong competitor in mixed microbial ecologies
ferments D-xylose, L-rhamnose, D-mannitol
Listeriosis disease syndrome
foodborne infectious infectious, intracellular pathogen
syndomes
adult onset: symptoms of meningitis, septicemia with fatality of 1/5 to ¼ (immuno-compromised adults at highest risk)
pregnant mother: flu-like symptoms with possible long-term shedding, other complications
late-onset neonatal: exposure during vaginal birth, post-delivery nosocomial from shedding adults
early-onset neonatal: exposure to fetus during gestation, potentially resulting in stillbirth, spontaneous abortion, or systemic infection post-natal
feverish gastroenteritis: those with no other predispositions; often requires high consumption for infection to occur with incubation periods 12-72 hr
foodborne listeriosis: generalities
higher fatality rate versus other viral, bacterial pathogens (maybe 3rd highest amongst foodborne pathogens)
observed in multiple types of fresh and processed foodstuffs
pathogen (Lm) can perisist in numerous of food process systems
can form biofilms, resist sanitizer attack, and tolerate acid, moderate heat exposure
incubation period can be very long! (up to 70 days)
infectious dose may be low (as few as 1 CFU/gram of product in some outbreaks)
intracellular pathogen (invades, moves within human GI cells escaping immune response)
pathogenic Lm differentiated into 13 serotypes: ½a, ½b, and 4b are most commonly identified in human disease
often associated with ready-to-eat (RTE) food products: cheeses, processed meats, produced foods (whole and sliced)
environmental transmission to foods
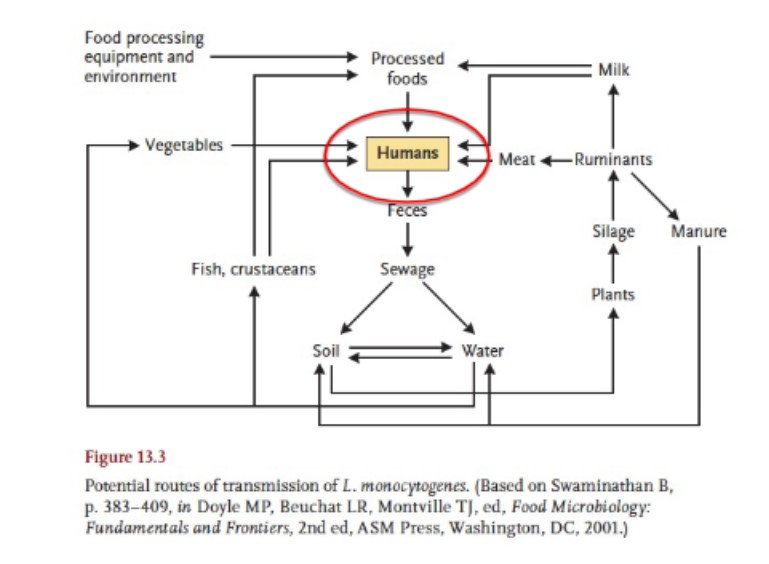
foods of concern: raw milk and dairy
raw milk: cross-contamination in milk collection equipment, holding tank, on farm/cow teats
outbreaks of disease associated with raw milk-made cheeses occurring all over US
1985: Los Angeles, CA → 142 cases (93 associated with pregnant women, fetuses)
2001: Winston-Salem, NC → 12 cases (10 pregnant women, 5 stillborns, 3 premature, 2 post-delivery infected)
2015: Blue Bell…
RTE meat, poultry, seafood
pathogen may infiltrate post-lethality environment, causing contamination risk on processed product
some products are more supportive of growth than others
fermented sausages, seafood less supportive at low pH
salted foods/meats, dried meats do not support growth of pathogen
opportunity exists to cross-contaminated post-lethality exposed product from food contact surfaces, adjacent surfaces, walls, floors, drains, and other niches
process facility sanitation systems are key yo Lm control on foods
isolates (strain) shown to have tolerance development to sanitizers (rotate sanitizers to inhibit this) → dubbed persistent Lm
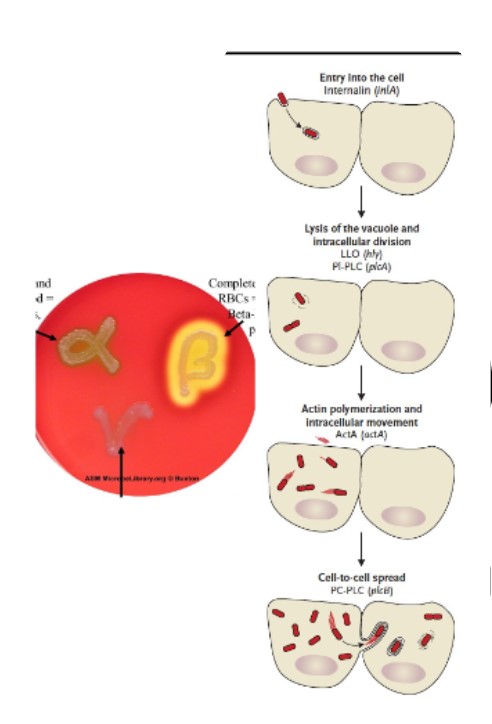
Lm pathogenesis
internalins: help pathogen to enter human GI epithalials (escape immune response, competitors, environmental stressors)
Listeriolysin O (LLO): aids escape of internalized (vacuolar) cells via destruction of vacuolar/phagosome membrane
hemolysin: Lm and β-hemolytic (escape phagosomes)
actin mobilization: assists movement through cell and translocation to adjacent GI cells
US regulations surrounding Lm
zero-tolerance for RTE foods
fully cooked meat, poultry, seafood
dairy products, produce
non-detectable in 1 or 2 identical 25 g food samples
USDA-FSIS: 9 CFR 430 (i.e., “The Listeria Rule“)
3 alternatives for processors to handle Lm in PLE foods with respect to processing and sampling
1) use of post-lethality treatment (1.0 log_10 reduction in Lm counts) and antimicrobial agent/process (< 2.0log_10 in growth over product shelf life)
2a, b) use of post-lethality treatment (2a) or microbial agent (2b)
3) sanitation-based prevention of contamination
provides additional processing requirements for manufacturers of deli products
Straphylococcus aureus (Greek: grape-like coccoid)
family Straphylococcaceae (S. aureus is type species)
facultatively anaerobic non-sporulating coccoid
cause foodborne intoxication: pre-formed toxin consumption
pathogen not required for disease
toxin not process sensitive
may also cause toxic shock syndrome (TSS), wound infections, and animal disease (mastitis)
S. aureus physiology, dissemination
catalase +, non-motile, environmentally ubiquitous
coagulase-variable; thermostable DNAse-variable
human reservoir; 1 in 2 to 1 in 3 carry at any time
skin, hair
nasal mucosa
food preparers often linked to pathogen contamination (duh…)
mesophilic facultative anaerobe (optimal growth: 35 °C)
exhibits growth to a_w > 0.85 (aerobic) and 0.9 (anaerobic); toxin production typically halts at a_w 0.9
describes as poor competitor: does not effectively sequester/control nutrients, water in mixed microbial ecology
the pathogen in foods and processing
meat processing equipment contamination sites
inadequate refrigeration or temperature abuse allows growth, toxin production
inadequate cooking, heating of foods
Staphylococcus environmental stress tolerance
most low a_w-tolerant bacterial pathogen!
accumulation of compatible solutes
reduction of cytoplasmic a_w to neat equal exterior environment → prevents water movement out of pathogen cell
may escape host immune systems via phagocytosis
ubiquity: being everywhere in production environment increases difficulty of control
cooperative in biofilm development, maintenance
foodborne Staphylococcal disease
characterized by short-lived, self-limiting disease with low fatality rate
CDC estimates ~ 241K cases per year in US with ~6 fatalities (highly under-estimated incidence)
infectious dose of enterotoxin: <1.0 CFU/g to achieve toxic doses of SE
incubation: 0.5-7 hr (dose consumed, sensitivity); duration is short (hrs - 1 day)
symptoms: nausea, heavy/violent vomiting (emesis), diarrhea (pathogen and/or toxin can be recovered from stools), dehydration
foods associated with Staphylococcal disease
meats, poultry/eggs, dishes requiring and/or human handling allowing for S. aureus contamination
confections, bakery items, meat/starchy salads (tuna, chicken, potato)
products with fully cooked ingredients still requiring human mixing/handling
some milk/dairy products (cheeses)
any foods where background microbes have been eliminated and food is kept under temperature abuse for extended period can be transmission vehicle for S. aureus
Staphylococcal enterotoxins
SEs are proteins (single chains) anti-genetically distinct from one another: A, B, C, D, E, G, H, J (now known over 20 distinct toxin types)
some cross-reaction with toxin form-specific antibodies
SEA is most common disease-causing form, but SEB is most severe form (symptoms)
SEA, B are super-antigens (bind to major histo-compatibility class II (MHC II) molecules expressed as antigen-presenting cells (e.g., macrophages)
classically used coagulase test to predict SE producing S. aureus strains (coagulase + staphylococci)
single peptides (25-28 kDa)
SEs are food process resistant
tolerant to protease attack (pepsin, trypsin) (pH dependent; may be degraded under conditions of severely low pH)
thermo-tolerant
can withstand autoclaving conditions, short term canning
microbe inactivated much more easily than toxin
heating tolerance best at low acidity or under sight alkaline condition
Enterococcus spp.
capable of growth at environmental temperatures ranging 10-45 °C, in up to 6.5% NaCl
may grow in conditions of pH 9.5-9.6, and can hydrolyze esculin in conditions of high bile salts concentration
will appear as cocci ~2 µm in diameter in pairs or chains (similar to Streptococci)
broken out into five distinct groupings based on biochemical capabilities
typical residents of GI tracts of both warm- and cold-blooded animals: can reside in human tract GI tract also
some utility as fecal residues indicator, but typically overlooked in US for this purpose
growth in these environments predicated on multiple systems of environmental tolerance, metabolism of various nutrients, and capacity to resist antimicrobials, reactive species, etc
Enterococcus diarrheal disease: humans
may possess low infectious dose, but estimates vary
typically acute, and self-limiting, but can become severe with possibly ½ chance of succumbing
symptoms can mirror those of staphylococcal food disease
E. faecium and sometimes E. faecalis
many isolates difficult to treat due to multi-drug resistance (e.g., vancomycin-resistant enterococci [VRE])
Enterococci in livestock species disease
E. durans: diarrheal disease reported in calves, piglets, foals
E. avium, E. faecalis, E. gallinarum: septicemia in bords
E. porcinus, E. villorum: piglet diarrhea/neonatal disease
GRAM-NEGATIVE bacterial foodborne pathogens
Brucella abortus
non-sporulating facultatively anaerobic coccobacilli; 11 species in genus
reference organism: B. melitensis
some species known animal pathogens (B. abortus, B. canis)
family Brucellaceae
zoonotic pathogen: can be spread between animals and humans
reservoirs: sheep, goats, bison, elk
human brucellosis (undulant fever)
uncommon in US; FDA/CDC estimates 120-840 cases/year (higher incidence in other countries with lower food safety demands, animal vaccination programs versus US)
infectious dose: ~500 cells (possibly fewer)
fatality rate: <2%
symptoms: chills, sweating, muscle weakness, headache, joint/muscle pain (acute)
chronic conditions: endocarditis, liver/spleen swelling, chronic fatigue, arthritis, recurring fever
animal farmers
meat industry employees (slaughter workers and QA laboratory)
vets
consumers of raw milk, fresh-made cheese
pregnant women
bovine brucellosis (bang’s disease)
primarily impacting cattle, bison, and some cervids (deer)
pathogens: B. abortus, B. melitensis, B. suis (swine)
typically no clinical symptoms appear: abortions or weakness in calves, fawns, piglets
pathogen eradication program present in US- significant cost-savings in production/meat processing
vaccination
surveillance
herd/animal quarantine
Campylobacter spp.
family Campylobacteraceae (greek: curved rod); type species: C. fetus (previously named Vibrio fetus)
over 30 species with genus
key human foodborne pathogenic species: C. jejuni subsp. jejuni, C. coli
some species known to cause disease/abortion in livestock: C. fetus, C. jejuni
spiral rods with one or more twists to body (0.2 -0.9 µm length)
organisms are microaerophiles (optimal: 3-5% O2, 10% CO2); high oxygen tension can be lethal
motile by flagella (mono- or amphi-trichous with one unsheathed flagellum at one, both poles)
optimal growth: 37-42 °C
nutritionally fastidious; unable to utilize/oxidize CHO (yield energy from amino breakdown) (does contain cytochrome oxidase [electron carrier])
Campylobacter jejuni
human foodborne disease (infectious)
thermo-tolerant Campylobacter (good growth: 37-42 °C; no proliferation <30 ° C)
some isolates shown to continue respiration at 4 ° C
does not generally catabolize CHO, but contains some genetics necessary for limited sugar (fucose) use in some strains
lactate. pyruvate, acetate, etc
glutamate, aspartate, serine, proline
colonization sites in animal, human GI tract with sufficient O2 availability
multiple respiratory components sustain growth in gut
Campylobacteriosis epidemiology
globally reported #1 cause of bacterial foodborne disease
CDC estimates between 1st to 3rd most common human foodborne disease
~1.5 million incidence cases/year estimated
reservoirs for pathogen identified in multiple meat animals, companion animals, and humans
outbreaks in animal-derived foods, produce, water, human-human cross-contacts
children mist likely impacted, along with adolescents and young adults
immune-compromised
HIV+
higher disease, outbreak frequency in warmer months/seasons
campylobacter in foods
principally associated with animal-derived foods not properly cooked, cross-contaminated
poultry, swine meat
non-pasteurized milks
fecal shedding leads to water, produce cross-contamination
full cooking of meat, poultry/pasteurization of milk destroys organism
human camoylicater enteritis
typically self-limiting/acute disease: up to 2-10 days of clinical symptoms
clinical symptoms resolve w/o medical treatment (self-limiting)
fever, diarrhea (heavy, frequent, contain undetected blood [occult]), abdominal pain, vomiting (violent, repeated)
infectious dose is low: <500 CFU (generally around 10^4)
incubation period: 2-5 days post-exposure
low frequency of chronic sequelae (post-acute disease) onset
bacteremia: bacteria in blood stream
inflammation: meningitis, pancreatitis, endocarditis
miscarriage of fetus, neonatal sepsis (C. fetus subap. fetus)
auto-immune disorders: Guillain-Barre Syndrome (GBS), reactive arthritis
pathogens in humans
factors aiding Campylobacter disease, human GI cell invasion
adhesions for GI cells with subsequent invasion
flagellar motility: critical for colonization of gut
GI Mucin (O2 tension; fucose secretion)- chemo-attractants
cytolethal distending toxin (Cdt)
leads to cell death eventually
damages host DNA
thermo-tolerance: greater survival- higher numbers consumed
LPS component: sialic acid (mirrors components of human gangliosides)
antibodies attack human nerve cell endings
autoimmune condition onset
control in food handling
use of sanitary/potable drinking water for animals
poultry/meat/food products: proper cooking
prompt chilling, full reheating leftovers
WASH YOUR HANDS!
frequently
working with food animals, pets, exotics
helicobacter pylori
previously Campylobacter pylori (pyloric; stomach)
spiral-shaped Gram-negative motile rod capable of motility and colonization or GI mucosum
widespread throughout world (50-66% infection/carriage rate); likely transmission/exposure through contaminated drinking water
fecal/oral; oral/oral routes of spread
causative of gastric ulcers; contributor to gastric cancers
may provide some protection against esophageal cancer
helicobacter pathogenesis
capable of survival in acidic conditions
motility: locate b/w tight cell junctions in stomach epithelium
urease secretion
chronic infection by organisms leads to inflammation, exacerbated by acid → ulcer
older adults; senior citizens mostly impacted
toxins
vacuolating toxin: disrupts GI tight cell junction
phospholipase
cytotoxic elements
vibrio spp.
family vibrionaceae: facultatively anaerobic irregularly shaped rods (curved rods), halophilic (require Na+ for survival and successful growth)
> 90 species in genus, 8 are foodborne pathogens
Infectious disease spp: V. cholerae, V.
parahaemolyticus, V. vulnificus, V. mimicus, V.
alginolyticus
foodborne disease most commonly associated with contaminated marine (salt-water) food animal meat
raw shellfish, sushi
cross-contaminated post-cooking
sensitive to cold storage; no growth at refrigeration/freezing conditions
can enter VNC state: shrink, coccoidal cells
foodborne disease trends
CDC estimate: ~80 K cases per year (~52K are foodborne)
overall increases in US vibriosis cases since 1996
CDC indicated higher disease rates in warmer months/waters (higher pathogen loads in harvest waters)
V. cholerae
foodborne GI disease and/or cholera (severe: cholera gravis)
can enter VNC states
cholera strains belong to serotypes O1 and O139 (encapsulated)
non-O1 and non-O139 V. cholerae can produce GI disease; not cholera-associated generally
reservoirs in marine and fresh waters (drinking water contaminated with fecal sewage common cause of disease)
cholera
estimated ~3 million cases globally per year, with 95K fatalities occurring
can take mild to heavy/severe forms; partially mediated by toxin expression capacity of isolate (cholera enterotoxin (CT))
CT induces export of ions and loss of transmembrane ion regulation ability- high ion content leads to flooding of water into colon
high infectious dose (~1 million cells), with incubation up to 3 days post consumption
mortality is high without treatment but can be successfully treated with antibiotics and rehydration therapy
severe diarrhea, pain, cramps, heart racing, muscle aches, rice water-stools (progressed)
small numbers of cases still occurring in US; most cases occur in under-developed countries, Africa
V. parahaemolyticus
optimal growth in 2% NaCl in medium; doubling time as little as -10 min at 37 ° C
produces thermostable direct hemolysin (TDH), AKA Kanagawa hemolysin (beta-hemolytic on Wagatsuma Agar)
infectious pathogen: 2% mortality common (elevated when patient (pt) is septic)
capable of very rapid replication numbers necessary for disease
infectious dose 10^5-10^7 cells; incubation period typically 24 hr
reservoirs: estuarine waters, seafood animals, sediments
symptoms typically 2-6 days (acute): diarrhea, abdominal pain, cramps. blood in stool, possible septicemia
V. vulnificus
highest fatality rate of pathogens discussed (35-60%), but very low incidence (~90 cases annually)
infections leads to septicemia
wound infections occur in addition to foodborne infections (20-25% fatality)
estuarine envirnoment reservoirs
similar to other vibrio in cold sensitivity, acid sensitivity
incubation period can be very long (up to 21 days); infectious dose estimated at 1,000 cells
typical symptoms of GI disease are not common in cases (diarrhea, vomiting)
highest disease incidence: US, Japan, Mexico, Taiwan, S. Korea
chronic hepatic disease predisposes to diseases(males more frequent pt)
syndromes:
primary septicemia
primary gastroenteritis
primary wound infection
Cronobacter sakazakii
formerly enterobacter sakazakii (family enterobacteriaceae) (genus change suggested in 2008)
rod-shaped mesophilic facultative anaerobe
encapsulated organism
enhanced biofilm participation
enhanced dehydration tolerance
non-pasteurization tolerant: contaminates post-process
human cronobacter disease
human infectious disease agent; CDC estimates 4-6 cases per year
processed powdered infant formula (PIF)
noenates (<2 months) at risk for disease, death
infection can cause bowel damage- septicemia
spread to brain- fatality (10-90%)
infectious dose estimated at 10 CFU
incubation is 2-5 days
symptoms: jaundice, persisting irritability, poor feedings, seizure, fluctuations in body temperature, etc
treatment often requires antibiotics, but some isolates are drug-resistant
FDA dictates zero-tolerance in cronobacter and salmonella in infant formulas
escheria coli and the EHEC
member of family enterobacteriaceae
gram-neg, oxidase-negative facultative anaerobic rod, motile by peritrichious flagella
mesophilic growth, with capacity to grow at pH 4.0-4.5 in some systems
acid tolerance response system (rpoS)
glutamate- and arginine-dependent systems
pathogenic and non-pathogenic E. coli
other pathogenic Escheria spp.: E. albertii, E. fergusonii
E. coli pathgroups
disease syndromes differ between patho-groups, as well as high-risk populations targeted
entero-toxigenic E. coli (ETEC): toxico-infection
entero-pathogenic E. coli (EPEC): infection/invasion
entero-invasive E. coli (EIEC): infection/invasive
diffuse-adhering E. coli (DAEC): infection/invasive
entero-aggregative E.coli (EAEC): infection
Shiga toxin-producing E. coli (STEC) and the entero-hemorrhagic E. coli (EHEC): infection/invasion; toxico-infection
strains are differentiated into subgroups by three antigens on cell: somatic (LPS; O), flagellar (H), and capsule (K) antigens
EHEC, STEC
over 100 serotypes of STEC, with some EHEC-type organisms within STEC group
EHEC are differentiated by characteristic bloody (frank) diarrhea
STEC and Shiga toxin (verotoxin)
nearly identical to shillega dysenteria stx toxin (phage encoded toxin genetics)
leads to post-acute disease sequelae HUS and TPP
O157 and non-O157 STEC: E. coli O157: H7, O103, O111, O45, O145, O121
these 7 STEC serogroups treated as adulterants in fresh/raw non-intact beef products (ground beef, gyro, beef, tenderized, or enhance beef products)
other characteristics
capable of growth at higher temperatures (> 44.5 °C)
enzyme glucuronidase is made
can grow in presence of bile and some other detergents
inability to use some CHO versus other E. coli
high acid tolerance (pH 4.0-4.5)- growth
may survive even lower pH conditions in foods
no more heat tolerant than other E. coli and Salmonella
sources of STEC in foods
animal-derived foods: E. coli maintain reservoirs in bovine GI tract (commensal)
beef, dairy breeds
calves have higher carriage rates
dairy breeds of greater concern for carriage, likely conversion of carcass meat into ground beef
recovered from game animal carcasses, feces
fecal matter cross-contamination of produce-irrigating waters, produce production systems
human-human; fecal/oral transmission
foodborne disease outbreaks and beef
1992: outbreak which identifies E. coli O157 as foodborne pathogen
1993: Jack-in-the-Box (Pacific NW): 179 cases, fatalities- leads to mandatory adulterant status by USDA and pushes HACCP forward for USDA-inspected products in 1999
2007: Topp’s ground beef: 40 cases, over 1 M lbs recalled
2013: Coco Loco’s (5 confirmed cases, 5 non-confirmed cases): under-cooked ground beef, E. coli O157:H7
2014: Wolverine Meats: 12 cases, 1.8 M lbs recalled
2015: Chipotle Restaurants (2 outbreaks, E. coli O26)
other outbreak food vehicles
dairy: non-pasteurized milk
produce: leafy greens (spinach, lettuce), walnuts, cucumbers, raw apple ciders, sprouts
drinking water
further processed foods: pizza rolls with pepperoni, cookie dough, flour (GM, E. coli O121, O26)
recent outbreaks in US: romaine lettuce (2019, 2018, 2017): lettuce grown western US (AZ, CA)
another way to be exposed
direct contact with fecal matter, contaminated dust, bedding- fairs and petting zoos
multiple outbreaks in US surrounding
NC (2011): cases of disease at state fair (2 mo- 62 years old in pt)
wash hands thoroughly after handling livestock, adults, and children
hemorrhagic colitis, STEC disease
mild diarrhea (non-bloody) to severe bloody diarrhea and post-acute syndromes also
incubation period: 2-12 days (3-4 is most common): colonization of gut occurs
abdominal pain, cramps is common; symptoms resolve typically within 7-10 days
hemolytic uremic syndrome (HUS): most common in young children but can affect teens, young and older adults
thrombotic thrombocytopenia (TPP): adults
low incidence; low disease risk
similar symptoms pf HUS but with bleeding in brain and other organs in body
disease dose: ~10 CFU has been reported (generally thought <100 CFU)
fatality is rare, but can occur in severe cases: estimated rate 1%
pathogenic elements, EHEC
intimin and tir: allows for formation of pedestal-type intimate attachment of producing E. coli and other enterics
encoded genetically on locus of Enterocyte Effacement (LEE)
attaching/effacing (A/E) lesions: Effacement (removal) of GI microvilli
60 MDa plasmid (pO157): enterohemolysin, catalase
shiga toxins
the STEC and EHEC produce stx1 and/or stx2
either chromosome or phage-encoded
multiple variant forms of stx2 spread throughout EHEC, EPEC, Citrobacter spp., and other Escherichia spp.'
human and livestock disease
2e: pig edema
holotoxin (protein): one A unit and 5 B-units
binds to specific renal cell surface lipid receptor and is then imported
induces failure of intoxicated cells to complete translation (protein synthesis) by halting tRNA successful binding in ribosomes- leads to cell death
pathogenic E. coli control
proper cooking of foods (where applicable)
fully cooked meats, pasteurized dairy
ground, non-intact beef: 160 °F internal temperature (USDA recommended)
other validated antomicrobial interventions to reduce, prevent growth
wash produce with chlorinated water in home, restaurant
good personal hygiene
sanitation in process environment, restaurant
Salmonella enterica
facultatively anaerobic regular rods, oxidase negative, mesophilic infectious/invasive pathogen
catalase-negative, urease-negative, produce lysine decarboxylase, reduce H2S and precipitate sulfur in presence of iron to FeS
can ferment glucose, but not lactose or sucrose; can use citrate as carbon source
infectious/invasive pathogen of humans and animals
typhoidal salmonellae: S. Typhi, paratyphi
non-typhoidal salmonella
some salmonella flagellated (peritrichous), but some non-flagellated (S. Gallinarum, S. Pullorum)
adulterant in RTE foods, fully cooked meat/poultry (zero-tolerance)
identified by CDC as #1 cause of human foodborne bacterial disease
disease incidence, outbreaks
estimated ~1.4 million cases per year
animal- and plant-derived foods
numerous serovars involved in differing outbreaks
globally spread but differing serovars tend to dominate in differing regions
fresh and further-processed foods
Salmonella taxonomy
two species with genus: S. enterica, S. bongori
within S. enterica, six sub-species:
I: enterica
II: salamae
IIIa: arizonae
IIIb: diarizonae
IV: houtenae
VI: indica <3
separated by likenesses in biochemical and genomic attributes
serovars identified and differentiated (O, H, K antigens)
Salmonella serovars
serological identification rests on differentiation of somatic, capsular, and flagellar antigens (Kaufmann-White scheme)
capsule: Typhi, Paratyphi, Dublin
flagellar: most salmonellae, but some non-flagellated
somatic: all (major, minor LPS components)
serovars were often used as species names prior to revision in serotyping and nomenclature
>2500 known serovars (and counting…)
reservoirs of salmonella
livestock (meat, milk), poultry animals (poultry meat, eggs)
identification of multiple other transmission vehicles/exposure sources:
irrigation water, drinking water
plant-derived foods
companion, livestock animals (feces, urine)
salmonella growth
can grow optimally at 32-38 °C, bur reports of low and high-temperature growth also exist
prior cold, heat exposure can lead to adaptation of cell
no growth, but long-term survival in frozen foods reported
can remain viabke in dehydration/low a_w foods for long time periods, resisting heat application (also can remain infectious)
tolerance to acidity at pH 4.5 and alkali up to pH 9.5 (optimal at neutral pH)
acetic acid may be more functional vs. lactic acid
three genetically encoded acid tolerance systems allows increasing acid tolerance as lower pH environments encountered
mild pH reduction: ATP consuming homeostasis, log-phase ATR
moderate (pH 5.5-4.3): stationary phase inducible ATR
hard (pH 5.0-4.3): rpoS inducible system
mild acid shock result in cross-protection to subsequent heat exposure (changes in membrane lipids, metabolic outputs, chaperone proteins activated to protect DNA, RNA, other critical systems)
foodborne Salmonellosis
typically caused by non-typhoidal salmonellae: invasive pathogen
incubation period ranges from 8-72 hr, with symptoms running 2-7 days (typically self-limiting)
fever, abdominal pain, frequent diarrhea, vomiting, headache
acute disease is more common, but fatalities occur, or post-acute systemic/chronic sequelae are reported
Reiter’s syndrome, reactive arthritis
septicemia or bacteremia may also occur
antibiotics are not typically prescribed, and most cases require fluids, rest
typhoid fever (enteric fever)
S. Typhi, Paratyphi A
incubation period runs 1 week to 2 months; severe diarrhea and constipation can occur
rash may occur; septicemia can occur
asymptomatic carrier state can occur following decline of clinical symptoms (gallbladder infection sub-clinical)
Mary Mallon (Typhoid Mary)
pathogenesis, non-typhoidal Salmonella
attachment of Salmonella to GI cells allows subsequent uptake into the GI cell (invasion)
GI cell membrane ruffles, resulting in endocytosis (uptake/invasion) of Salmonella into GI epithelials
may be followed by GI programmed cell death (Salmonella-directed)
pathogen can also survive in phagocyte acidic phagosome (membrane-bound small compartment entrapping Salmonella)
some serovars contain virulence plasmid(s)
siderophores (Fe-acquisition from host); vaccines for animals have targeted these surface-located proteins
controlling Salmonella
cooking: pathogen not excessively heat-tolerant
heat tolerance increased in low a_w environments
cooking of some produce items (e.g., nuts)
high-fat foods provide enhanced Salmonella protection (e.g., chocolate, peanut butter)
sanitation, pathogen-reducing interventions on food animal carcasses, produce
pre-harvest interventions: competitive exclusion bacteria, vaccinations, animal handling practices
pathogen source tracking in live animal, food process environment
Shigella spp.
regular rods, facultatively anaerobic mesophiles, oxidase-negative, non-motile
unlike other enterics, not able to use lactose
genus contains 4 species: S. dysenteriae, S. boydii, S. flexneri, and S. sonnei
nearly identical to E. coli genetically
capacity of stx-carrying phages to infect E. coli from Shigella
difficulty human infectious disease; human are only reservoir of organism
cause of foodborne shigellosis (mild self-limiting disease) ranging to bloody acute diarrhea and bacillary dysentery (more severe, possibility fatal)
foodborne disease
transmission via fecal/oral route following poor personal hygiene practices and spread of contaminated feces
disease symptoms vary greatly between cases but generally include severe diarrhea (species-specific) and potential for heavy blood, mucosal stools (bacillary dysentery)
watery diarrhea + pathogen colonization of colon leads to mucous cells sloughing off
fatality rate varies with severity of disease; in dysentery fatality rate ranges 10-15%
infective dose approximately 10-200 cells
pathogen can survive for long periods in frozen food, allowing for distribution and retention of possible infectivity
Shigellosis
incubation: 8-50 hr, with duration lasting 5-7 days in acute cases
disease occurs most frequently in younger children (<6 years old)
Shiga toxin produced by isolates; can produce HUS and other chronic sequelae
fever occurs drives extensive GI colonization
estimated as 6th most frequent cause of human foodborne disease (500,000 cases annually)
pathogenesis of Shigella
enterotoxin production by bacterium
invasion capacity, similar to pathogenic E. coli
Shiga toxin production
incubation of organism at physiological temperature induces invasion capabilities
prevention/control
proper cooking of foods, refrigeration to prevent growth
good personal hygiene of process facility employees
FDA Food Code (2017 edt.) lists Shigella as reportable for restauranteurs and recommends restriction from food handling duties for at least 3 days post-exposure
Yersinia spp.
family Enterobacteriaceae; infectious zoonotic pathogen; oxidase-negative regular rod that can oxidize/ferment glucose (facultative anaerobe)
type species: Y. pestis (cause if bubonic, pneumonic plague) (named for A. Yersin who discovered agent as plague cause, 1894)
18 species and multiple subspecies
foodborne species of concern in food safety: Y. enterocolitica, Y. pseudotuberculosis
Y. enterocolitica exhibits psychotropics capacity with growth at 4 °C and survival in cooked foods enhanced over raw foods
cooking enhances nutrient utility to organism
fewer competitors
does not survive proper cooking so fresh and post-process contaminated foods at highest risk (pork and other animal-derived foods including shellfish)
capable of growth from pH 4-10, with alkali tolerance high
Yersinia enterocolitica physiology, typing
most isolates are Voges-Proskauer+ (indicates capacity to produce and metabolize acids into non-acidic metabolites)
many isolates use D-xylose, trehalose but most cannot produce beta-D-glucosidase (releases glucose from oligomers)
typing based on O-antigen (e.g., 4/O:3) and H-antigen profiles
urease+
Yersiniosis
typically acute diarrheal disease with higher incidence in young children (<5 years)
symptoms in young children include diarrhea, vomiting, fever and abdominal pain
older children may present with symptoms mirroring appendicitis
infectious dose estimated 10^4-10^6 cells; incubation period ranges from 1-11 days post-consumption
clinical symptoms may persist for few days up to 3 weeks
chronic sequelae can occur, with reactive arthritis most commonly reported (other include hyperthyroidism, Graves’ disease, goiter, Hashimoto’s thyroiditis)
bacteremia can lead to higher mortality rate (30-60%) post-acute disease
CDC reported 1% of yersiniosis-identified outbreaks in US were caused by Y. pseudotuberculosis from 1996-2007
symptoms, diseases
enterocolitis, pharyngitis
autoimmune: arthritis, uveitis, myocarditis, Hashimoto’s, Grave’s Disease
septicemia, pneumonia, endocarditis, peritonitis, meningitis, eye infections
sources of pathogenic Yersiniae
CDC estimates ~117K cases per year with about 35-40 deaths
disease fits definition of zoonoses (animal- human transmission)
animal-derived foods (meat, milk, byproducts)- undercooked or raw
swine tonsils: carriage site for serotype 4/O:3 Y. enterocolitica
tripe, chitterlings, tongue may also be contaminated
proper sanitation in processing, home can reduce contamination, spread to consumers
pathogenesis elements
invasive pathogen (similar to Salmonella)- modulated by invasin protein and host cell for internalization
enterotoxin production - role in disease unclear
flagella- not produced in lab medium at physiological temperature but recovered from patients
phospholipase production- hemolysis of blood cells
urease production: acid protection
pYV: plasmid-encoded protection factors against immunity and phagocytosis