The Structure and Function of Macromolecules
- Macromolecules
- Are large molecules composed of smaller molecules
- Are complex in their structure
- Most macromolecules are polymers, built from monomers
- Four classes of life’s organic molecules are polymers
- Carbohydrates
- Proteins
- Nucleic acids
- Lipids
- A polymer
- A long molecule consisting of many similar building blocks called monomers
- Specific monomers make up each macromolecule
- E.g. amino acids are the monomers for proteins
- The Synthesis and Breakdown of Polymers
- Monomers form larger molecules by condensation reaction called dehydration synthesis
- Polymers and disassemble by hydrolysis
- Addition of water molecules
- Although organisms share the same limited number of monomer types, each organism is unique based on the arrangement of monomers into polymers
- An immense variety of polymers can be built from a small set of monomers
- Carbohydrates
- Serve as fuel and building material
- Include both sugars and polymers
- Starch, cellulose, etc.
- Sugars
- Monosaccharides
- Are the simplest sugars
- Can be used for fuel
- Can be converted into other organic molecules
- Can be combined into polymers
- Can be linear
- Can form rings
- Disaccharides
- Consist of two monosaccharides
- Are joined by a glycosidic linkage
- Polysaccharides
- Are polymers of sugars
- Serve many roles in organism
- Storage Polysaccharides
- Starch
- Is a polymer consisting entirely of glucose monomers
- Is the major storage form of glucose in plants
- Glycogen
- Consists of glucose monomers
- Is the major storage form of glucose in animals
- Cellulose
- Is a polymer of glucose
- Has different glycosidic linkages than starch
- Difficult to digest
- Cows have microbes in their stomach to facilitate this process
- Chitin
- Is found in the exoskeleton of arthropods
- Can be used as surgical thread
- Lipids
- Are the one class of large biological monomers that do not consist of polymers
- Share the common trait of being hydrophobic
- Fats
- Constructed from two types of smaller molecules
- A single glycerol and usually three fatty acids
- Vary in length and number and location of double bonds they contain
- Saturated fatty acids
- Have the maximum number of hydrogen atoms possible
- Have no double bonds
- Unsaturated fatty acids
- Have one or more double bonds
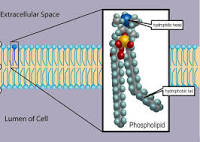
- Phospholipids
- Have only two fatty acids
- Have a phosphate group instead of third fatty acid
- Structure
- Consists of a hydrophobic “head” and hydrophobic “tails”
- Results in a bilayer arrangement founds in cell membranes
- Steroids
- Lipids characterized by a carbon skeleton consisting of four fused rings
- Cholesterol
- Found in cell membranes
- Is a precursor for some hormones
- Proteins
- Proteins have many structures, resulting in a wide range of functions
- Proteins do most of the work in cells and acts as enzymes
- Proteins are made of monomers called amino acids
- Enzyme
- Type of protein that acts as a catalyst, speeding up chemical reactions
- Polypeptides
- Polymers of amino acids
- A protein consists of one or more polypeptides
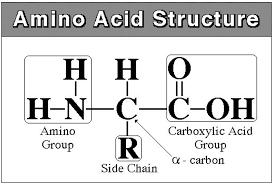
- Amino acids
- Are organic molecules possessing both carboxyl and amino groups
- Differ in their properties due to differing side chains, called R groups
- Linked by peptide bonds
- Protein Conformation and Function
- A protein’s specific conformation (shape) determines how it functions
- Four Levels of Protein Structure
- Primary structure
- Unique sequence of amino acids in a polypeptide
- Secondary structure
- Folding or coiling of the polypeptide into a repeating configuration
- Includes the a helix and β pleated sheet
- Tertiary Structure
- Overall three-dimensional shape of a polypeptide
- Results from interactions between amino acids and R groups
- Quaternary structure
- The overall protein structure that results from the aggregation of two or more polypeptide subunits
- Sickle Cell Disease
- Results from a single amino acid substitution in the protein hemoglobin
- What determines Protein Conformation
- Protein conformation depends on the physical and chemical conditions of the protein’s environment
- Temperature, pH, etc.
- Denaturation is when a protein unravels and loses its native conformation
- The Protein Folding Problem
- Most proteins
- Probably go through several intermediate states on their way to a stable conformation
- Denaturated proteins no longer work in their unfolded conditions
- Proteins may be denaturated by extreme changes in pH or temperature
- Chaperonins
- Protein molecules that assist in the proper folding of other proteins
- X-ray crystallography
- Used to determine a protein’s three-dimensional structure
- Nucleic Acids
- Store and transmit hereditary information
- Genes
- Are the units of inheritance
- Program the amino acid sequence of polypeptides
- Are made of nucleotide sequences of DNA
- DNA
- Deoxyribonucleic acid
- Stores information for the synthesis of specific proteins
- Found in the nucleus of the cell
- Functions
- Directs RNA synthesis
- Transcription
- Directs protein synthesis through RNA
- Translation
- Structure
- Nucleic acids exist as polymers called polynucleotides
- Each polynucleotide
- Consists of monomers called nucleotides
- Sugar + phosphate + nitrogen base
- Nucleotide monomers
- Made up of nucleosides (sugar + base) and a phosphate group
- Nucleotide polymers
- Are made up of nucleotides linked by the -OH on the 3’ carbon of one nucleotide and the phosphate on the 5’ carbon of the next
- Gene
- The sequence of bases along a nucleotide polymer
- DNA double helix
- Have two polynucleotides that spiral around an imaginary axis
- Form a double helix
- Consists of two antiparallel nucleotide strands
- A, T, C, G
- The nitrogenous bases in DNA
- Form hydrogen bonds in a complementary fashion
- A with T only
- C with G only
- DNA and Proteins as Tape Measures of Evolution
- Molecular comparisons
- Help biologists sort out the evolutionary connections among species