General Science Fall Exam
1/43
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Newtons 1st Law
objects in motion, stay in motion objects that rest, stay at rest, until an unbalanced force acts upon it.
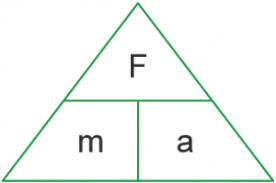
Newton’s 2nd law
F=MA Force (N) = mass(kg) X acceleration(m/s 02)

Newtons 3rd law
for every action there is an equal and opposite reaction.
Interia
resistance to change in motion
Derived
An object at rest remains at rest, and an object in motion remains in motion at constant speed and in a straight line unless acted on by an unbalanced force.
Acceleration
The rate at which velocity changes
How do you find acceleration?
A= final speed— initial speed /time
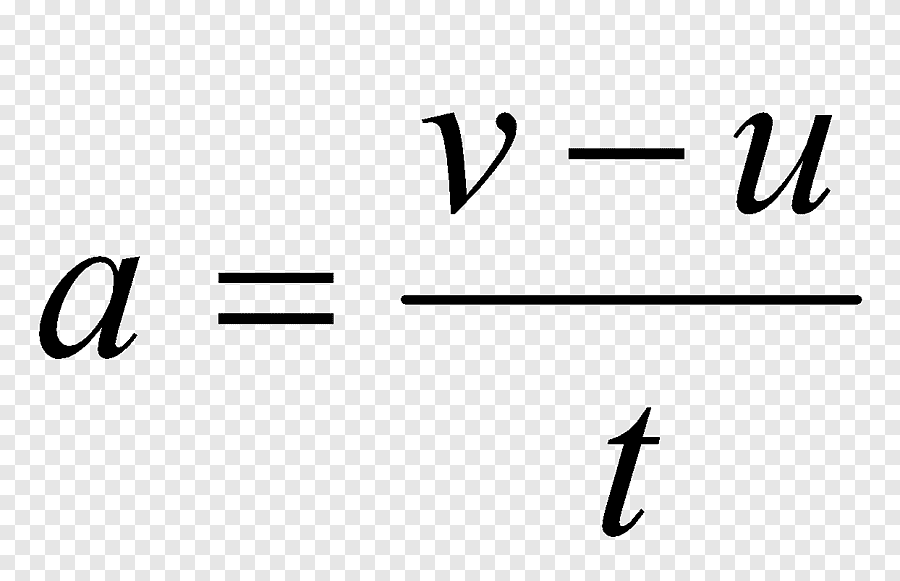
Example of constant speed
Carousel, the speed stayed constant, but the direction stays the same.
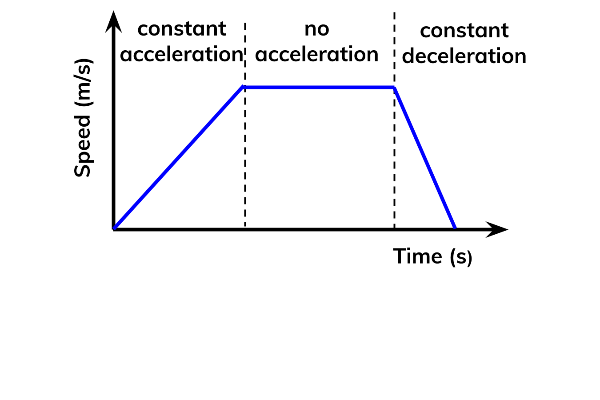
Graph Acceleration
Remember positive accl, no accl, and decrease accl.

Units of speed
Kl p/h, ft p/sec, mi (miles) p/h, or meters p/sec
How to calculate distance speed or time
Cover up letter you need to find to see the calucation
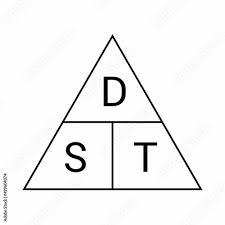
The distance an object travels per unit of time.
Speed
The steepness of a graph
Slope
Speed or speed in a given direction
Velocity
The rate in which velocity changes
acceleration
A value that falls directly in the middle of other related values
Average
A letter or symbol that represents a number that can change
Variable
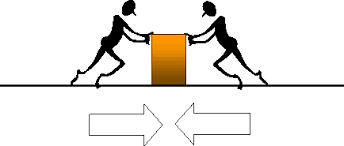
Net Force - how do you calculate if both arrows on N push against each other?
You subtract
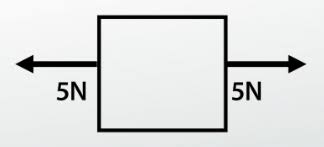
Net force - how would you calculate if both arrows are the same amount of N (newtons)?
balaced object no motion
Exothermic Reaction
Energy released lowering in temp.
Endothermic Reaction
Bonds of reactants breaking releasing energy as heat.
Physical change
Bending, crushing, cutting, melting, freezing, and boiling.
Chemical Change
Rusts forms, changes in color, bubbles
solvent
what is dissolving the solute
Solute
What is being dissolved by the solvent
What is the positively charged subatomic particle?
Proton
What is the negatively charged subatomic particle?
Neutron
What 2 subatomic particles are about the same size?
Protons & Neutrons
What do we call the first column on elements?
Alkali metals
Where are metals located on the periodic table?
Left
Soure to taste
Acid
Bitter to tatse
Base
Low on the pH scale
Acid
High on the pH scale
Base
What is the correct number of electrons in the first 3 shells?
8
Who said electrons have exact paths>
Niles Bohr
Who said the atoms was mostly empty space?
Ernest Rutherford
Who said the atoms was a solid positive material with negative spots?
Thomason
What kind of problems can develop from using synthetic materials such as plastic?
Negative impacts
Chemicals and resources that are made by humans are called __?
Natural resources
What kind of mixture is spaghetti and meatballs?
Heterogenous mixture
Cutting, melting, bending, or crushing are examples of what kind of change?
Physical change
What kind of change is the formation of rust?
Chemical
Which pieces of information does a chemical equation include
Reactants, products, & coefficients