PM-138 Skills for Medical Sciences
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
61 Terms
What is the difference between consent for clinical care and research?
Clinical - inform patients; protect clinicians; often implied
Research - inform participants; protect participants; arguably more rigorous
What is informed consent?
A process in which patients are given important information, including possible risks and benefits, about a medical procedure or treatment, genetic testing, or a clinical trial. This is to help them decide if they want to be treated, tested, or take part in the trial
What is research governance?
Overseen by a number of regulatory bodies and advisory group

What is the Declaration of Helsinki (1964)?
Expands the 10 principles of the Nuremberg Code with particular reference to:
• Identifies minimum information to be given to competent subjects
• Caution if dependent relationship exists
• Caution if possibility of duress
• Special attention paid to individual information needs and
methods of delivery
Describe the updated version of the Declaration of Helsinki (2013)
• This version is very empowering, aims to strengthen standards internationally-but especially relating to developing countries with limited resources
• Inclusion of under-represented groups
• Compensation and treatment must be in place for those harmed
• Access to new interventions where no proven one exists, following advice and informed consent
• Use of innovative consent methods i.e. recordings, videos etc.
• Giving feedback to participants after the research-demystifying and respectful
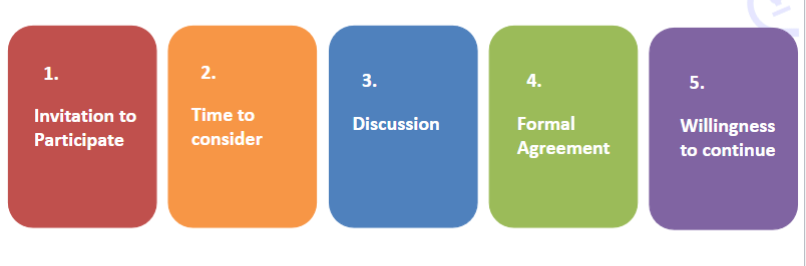
Recall the process of consent
What is the Human Tissue Act 1961 about?
Human Tissue Act established the Human Tissue
Authority – to regulate removal, storage, use and
disposal of human tissueHuman Tissue Act 2004 – effective 1st September
2006Based on a lack of objection to tissue removal from deceased for research or education
Consent and licensing are underlying principles of the Act; HT Act (Scotland) 2006 – no licences
Stipulates that consent is legally required to store and use “relevant material” for “scheduled purpose” of research (some exemptions)
Consent requirements differ for tissue from the living and the deceased
DNA theft – new offence
Criminal penalties for breach of the Act – fine, 3 years
imprisonment or both
What are the Human Tissue Acts (HTA) regulated sectors?
Anatomy
Bone marrow donation from children / lack of
capacityHuman application – cells / tissues (e.g. Skin,
stem cells, bone marrow, bone)Living organ donation assessment
Post mortem
Public display
Research
Organ donation and transplantation
Define these terms; ‘scheduled purpose’ and ‘relevant material’
Scheduled Purpose: Covers all activities or purposes
under the Human Tissue Act for which consent is requiredRelevant Material: material that is made of, or
includes, human cells other than gametes (eggs and sperm)
What activities are covered by the Human Tissue Act?
HT Act uses the term “scheduled purposes” to cover all
activities that fall under the ActResearch is a scheduled purpose – “removal, storage
and use of relevant material for research into
disorders, or the functioning of, the human body”All scheduled purposes legally require consent
Give a general description of ‘scheduled purposes’
Anatomical examination
Determining cause of death
Efficacy of drug or treatment after death
Obtaining information about a person which may be relevant to others
Public display
Research in connection with disorders, or functioning of the human body
Transplantation
When is consent not needed?
Consent is not needed for the following purposes if tissue or samples are obtained from the living. It MUST however be obtained if they relate to a deceased individual
Clinical audit
Education/training (including research techniques)
Performance assessment
Public health monitoring
Quality assurance
What are the relevant material under the Human Tissue Act 2004?
The definition of relevant material in the Act is:
Section 53: Relevant material:
1. In this Act, "relevant material" means material, other than gametes, which consists of or includes human cells.
2. In this Act, references to relevant material from a human body do not include embryos outside the human body, or hair and nail from the body of a living person

What is excluded from the HTA?
Excludes
Gametes
Acellular material e.g. serum, plasma (depending on preparation)
Embryos created outside the body
Cell lines
Extracted cellular components including DNA
Hair and nails from the living
What are the offences under the HTA?
Removing, storing or using relevant material for Scheduled Purposes
without appropriate consentStoring or using relevant material donated for a Scheduled Purpose for
another purposeHolding bodily material with the intention of its DNA being analysed for research without consent
Carrying out licensable activities without holding a licence from the HTA
Trafficking in human tissue for transplantation purposes
What are the licensable activities under the HTA?
The HTA licenses and inspects organisations which:
Carry out anatomical examination
Carry out post-mortem examination
Remove ‘relevant material’ from a deceased person
Store ‘relevant material’ for a ‘scheduled purpose’
Store anatomical specimens
Publicly displays a body or relevant material from a
deceased person
Relevant material, including commercially sourced tissues and samples from healthy individuals, can only be stored for research under the governance of:
HTA licence or NHS REC approval (university approval not sufficient)On expiry of NHS REC approval for a study involving relevant material, a HTA licence is required to legally continue storage of any residual material.
What are the exceptions to HTA licensing?
REC approved (or pending) projects
Material pending transfer (<1 week)
Material pending processing to render acellular (<1 week)
Supplied by HTA licensed tissue banks
Person died over 100 years ago
Short term storage of samples prior to analysis is NOT an exemption
Consent for HTA licensing is required to undertake what?
• Remove, store and use relevant material from the living
• Remove, store and use relevant material from a dead body
• Store and use dead bodies
• Consent must be ‘appropriate’ and ‘valid’
Describe how appropriate consent is applied towards the living and deceased

What are the exemptions to consent?
• Relevant material is from the living AND anonymised AND part of a NHS REC approved project
• Material collected before 1st September 2006 (existing holding)
• >100 years since death
• Imported tissue - good practice prevails
Is DNA considered relevant material?
No
What is DNA theft?
“DNA Theft” - holding bodily material with intent to analyse DNA without qualifying consent subject to certain exceptions e.g:
• material is from the living, anonymised and approved by an
NHS REC
• 100 years since death
• existing holding – pre-Sept 1st 2006
• medical diagnosis
• criminal investigations
• Penalty - Fine, 3 years imprisonment or both
What does ‘protected’ mean?
People are not allowed to discriminate, harass or victimise another person because they have any of the protected characteristics.
Who has responsibility under the Equality Act?
Education providers (Schools, FHE colleges and Universities)
Service providers
Government departments
Employers
Providers of public functions
Associations and membership bodies
Transport providers
Who is protected under the Equality Act (2010)?
Everyone in Britain is protected by the Act.
The “protected characteristics” under the Act are (in alphabetical order): Age, Disability, Gender, Marriage & civil partnership, Pregnancy and maternity, Race, Religion and belief, Sex, Sexual orientation
What is the difference between equality and equity?
Equality is treating everyone the same
This only really works if everyone starts from the same place and needs the same help
Equity is giving everyone what they need to be successful
Equity appears unfair, but it actively moves everyone closer to success by “levelling the playing field”
List the reasons why incorporating EDI is essential in clinical trials
-It leads to improved health for all communities, as UK clinical trials often fail to reflect the full diversity of the population
It produces more robust data demonstrating more clearly either the universal benefit of a drug or treatment, or its differential effect in different communities
It creates jobs, boosts local economies, and supports the life sciences sector as a key strategic pillar in the UK economy
What is explicit and implicit bias?
Explicit Bias:
Usually directed toward a group of people based on what is being perceived.
An explicit stereotype regarding gender may be “All males enjoy watching sports”
Implicit Bias:
Operate outside of a person's intentional, conscious cognition. It is the attribution of particular qualities to a member of a certain group unconsciously.
What is a ‘stereotype threat’?

Define research
systematic investigation into and study of materials and sources in order to establish facts and reach new conclusions; investigate systematically
Define (bio)medical research
Basic research (also called bench science or bench research) applied research, or translational research conducted to aid and support the development of knowledge in the field of medicine
What is clinical research and what does it determine?
Branch of healthcare science that determines the safety and effectiveness (efficacy) of:
• medications
• devices,
• diagnostic products
• treatment regimens intended for human use.
These may be used for prevention, treatment, diagnosis or for relieving symptoms of a disease
What are observational studies?
Participants are assessed for biomedical or health-related outcomes. May receive diagnostic, therapeutic and standard care interventions but are not assigned to specific groups by the investigator.
What are interventional trials?
Participants are assigned to specific groups, by investigators, to receive one or more (or no) interventions so that they may be assessed for biomedical or health-related outcomes evaluated
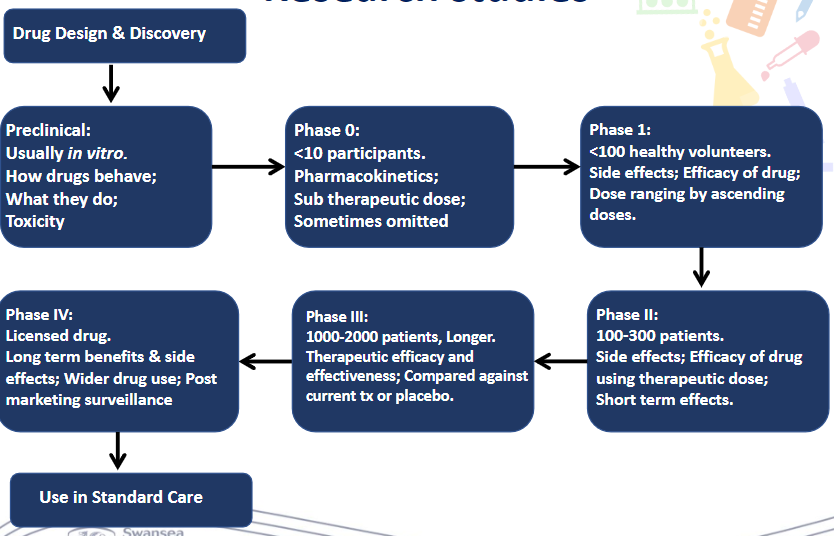
Recall the phases in research studies
Define the following terms; treatment, placebo, randomisation, blinding, power calculation
• Treatment: Active drug or device in a clinical trial
• Placebo : Does not contain the active drug or treatment in the trial
• Randomisation: Randomly allocates patients to groups e.g. treatment, placebo
• Blinding: Concealing the group allocation to one or more participant in a clinical trial
• Power Calculation: The number of participants required to show a statistical effect (based on pilot study data)
What are the different types of clinical studies?
Open label trial
• Both Researcher and participant know the treatment arm
• May or may not be randomised
• What problems could arise from this kind of trial?
Single Blinded trial
• The participant does not know the treatment arm
• The researcher does know the treatment arm
• What problems could arise from this kind of trial?
Double blinded trial
• Neither the participant, nor the researcher, know the treatment arm
• Randomisation and codes are held by a third party
• Groups are unblinded after date collection and analysis
• What problems could arise from this kind of trial?
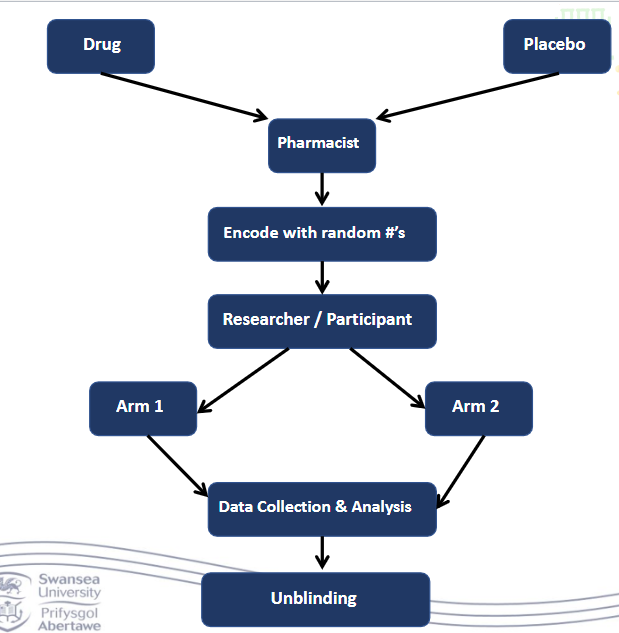
What happens in the randomised control trial ‘The Gold Standard’?
• Participants are allocated at random to receive one of several interventions
• One of these is usually the standard of comparison or a control
• Control may be standard practise, a placebo or no intervention
Give a brief overview of the timeline of humans in research from 4th century B.C. till 1962
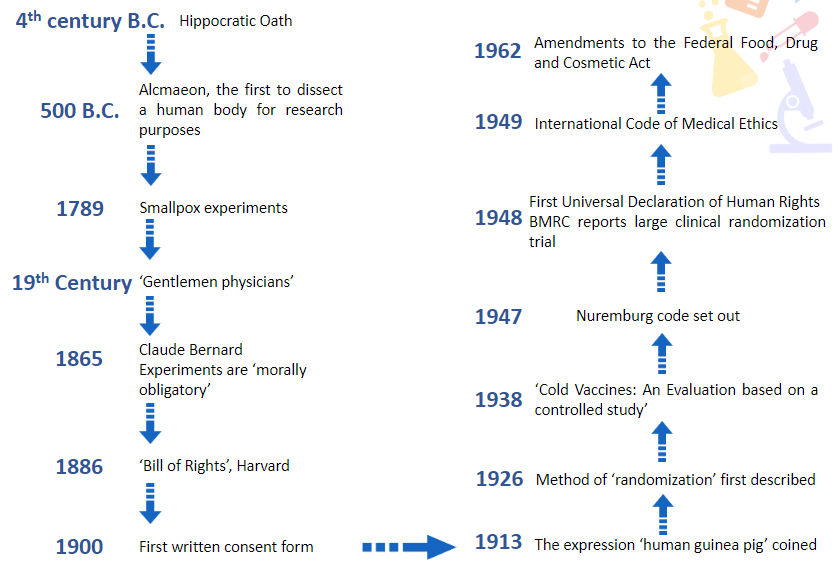
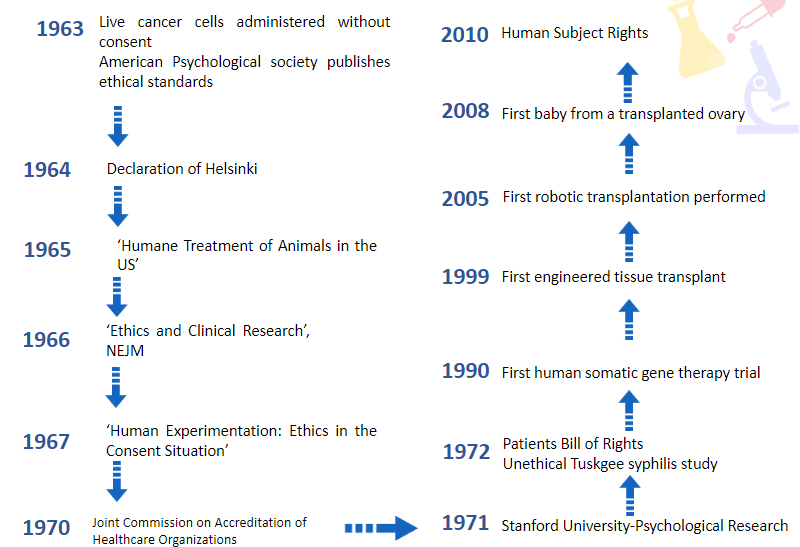
Give a brief overview of the timeline of humans in research from 1963 till 2010
Define ‘ethics’
Moral principles that govern a person’s behaviour or the conducting of an activity
The branch of knowledge that deals with moral principles
What are the rights of study participants?
1. Right to informed consent
- In a form legible to them
- Must be able to retain the information
2. Right to refuse the study, or to withdraw at any time
3. Right to be told reasonable risks and possible benefits of the study
4. Right to be told of any costs or compensation associated with the study
5. Right to have a contact in the research team
6. Right to be given all options available to them, including those not part of the study
What happened in the Tuskgee Syphilis study?
• >100 men died
• Countless others suffered insanity, blindness and long-term illnesses
• 1975: $10 million out of court settlement to participants, wives, widows and offspring.
• Last participant died in 2004
What is the CIA mind control program?
• 1953-1973
• ‘Project MKUltra’, undertaken by the US Central Intelligence Agency
• Intended to identify and develop drugs to torture and weaken individuals, particularly those in the Soviet Union
• At least 80 institutions, including universities, hospitals, prisons and pharmaceutical companies.
• Completely illegal and unethical.
• No informed consent or a follow up data – files rumoured to have been destroyed in 1973
What happened in Nazi Human Experimentation during the 1940s?
• Carried out in Nazi concentration camps on thousands of individuals
• Survival of Personnel trials
• Hypothermia trials
• Pressure trials
• Potable seawater
• Development of poisons
• ‘Twins’ experiments
• Racial and ideological tenets of Nazi world view
What happened during the Nuremberg trials (1945-1949)?
• Held in Nuremburg, Germany.
• Brought to trial senior Nazi officers for war crimes
• 12 were sentenced to death.
Brought into creation ‘The Nuremburg Principles’
What is the Nuremberg code?
10 principles which must be adhered to in order to ‘satisfy moral, ethical and legal concepts’
What are the principles of the Nuremberg code?
1. Voluntary consent
2. Yield fruitful results for society
3. Designed and based on results of animal experimentation
4. Avoid all unnecessary physical and mental suffering and injury
5. No experiment should be conducted where there is a reason to believe that death or disabling injury will occur
6. Humanitarian importance
7. Proper preparations and adequate facilities
8. Conducted by scientifically qualified persons at all study stages
9. Participants can bring the experiment to an end
10. Scientist in charge must be prepared to terminate at any stage
Recall the major advances in basic research that depends on animal experimentation.

Recall the medical breakthroughs following animal experiments
• Physiology of digestion
• Insulin
• Electrocardiogram (ECG)
• Penicillin
• Hepatitis B
• Yellow fever
• Polio vaccine
• Molecular structure of DNA
What are the advantages of animals in science?
• Alternative to humans
• Genetic and systemic similarities between animals and humans (95% of our DNA shared with mice)
• Animals suffer from similar diseases to humans including cancer, TB, influenza and asthma
What are the limitations of animals in science?
• Ethical issues of animal use
• Expensive
• Time-consuming
• Humans are in some ways physiologically different to animal models, e.g., CYP enzyme expression, important for drug metabolism, varies among species
• Unrealistically high doses used
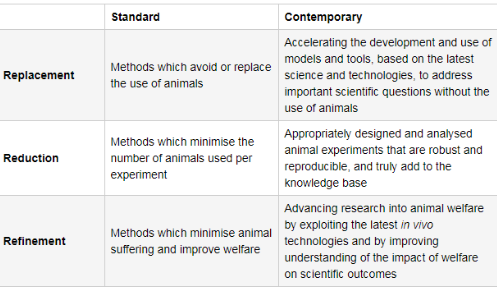
What is the 3Rs principle?
• The Reduction, Replacement and Refinement of animals in research
• Framework for humane animal research
• Integrated into national and international legislation regulating the use of animals in scientific procedures
What is the NC3Rs?
• The National Centre for the Reduction, Replacement and Refinement of Animals in Research/Testing
• UK government funded
• Collaborate with universities, industry, research councils and regulatory authorities
What is replacement?
• Replace or avoid the use of animals
• Human volunteers, tissues and cells (e.g.,HeLa cells)
• Established animal cell lines
• Immature forms of vertebrates, or invertebrates, such as Drosophila and nematode worms to supplement research
• Mathematical and computer (in silico) models
• In collaboration with Innovate UK, NC3Rs are establishing a world-leading non-animal technologies (NATs) sector in the UK
What is reduction?
Taking measures to use a smaller number of animals, to obtain
information from fewer animals or more information from the same number of animals
• Power calculations
• Culture cells from a single animal
• Save all samples
Give an example of reduction
• Easton et al., Durham University
• Multiple trials with single rat, no handling in between
• Reduced no. rats required
• Redesigned apparatus with GSK – no. mice reduced by 50% (8/expt)
• Multiple applications for models of cognitive decline – reduced variation, insights into age-related memory decline in TASTPM mouse model
What is refinement?
• Refinement: improving experiments, housing and care to reduce suffering throughout animals’ lives
• Example: Certain methods of handling can cause mice stress
• This may influence their behaviour and physiology, possibly leading to variability within/between studies
• Therefore, handling mice to minimise stress is not only beneficial for experimental results but also animal welfare
What are the UK regulations on research using animals?
• Animal research in the UK is strictly regulated
• The laws on research using animals are set out in the Animals (Scientific Procedures) Act 1986.
• Home Office - enforces the laws, including regulations on housing, environment, welfare, care, and health
• Permission to work with animals is granted by the Home Office by license only under very specific conditions
• Home Office’s inspection system ensures that rules are not violated
What is the Home office licence?
Three licenses are required by the ASPA before testing on animals is permitted:
• personal license for each person carrying out procedures on animals
• project license for the programme of work
• establishment license for the place at which the work is carried out
What are the ARRIVE guidlines?
• Animal Research: Reporting of In Vivo Experiments
• Initiative to improve the design, analysis and reporting of animal research
– maximising information published and minimising unnecessary studies
• Published in the online journal PLOS Biology in June 2010
• Endorsed by scientific journals, major funding bodies and learned societies