Hydride Reductions
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Hydride Reduction
1) NaBH4/EtOH
or
2) LiAlH4/ether/H3O+
-aldehyde and ketone reactants yield ____
-carbonyl reduction cant produce ___
- primary and secondary alcohols
- tertiary alcohols
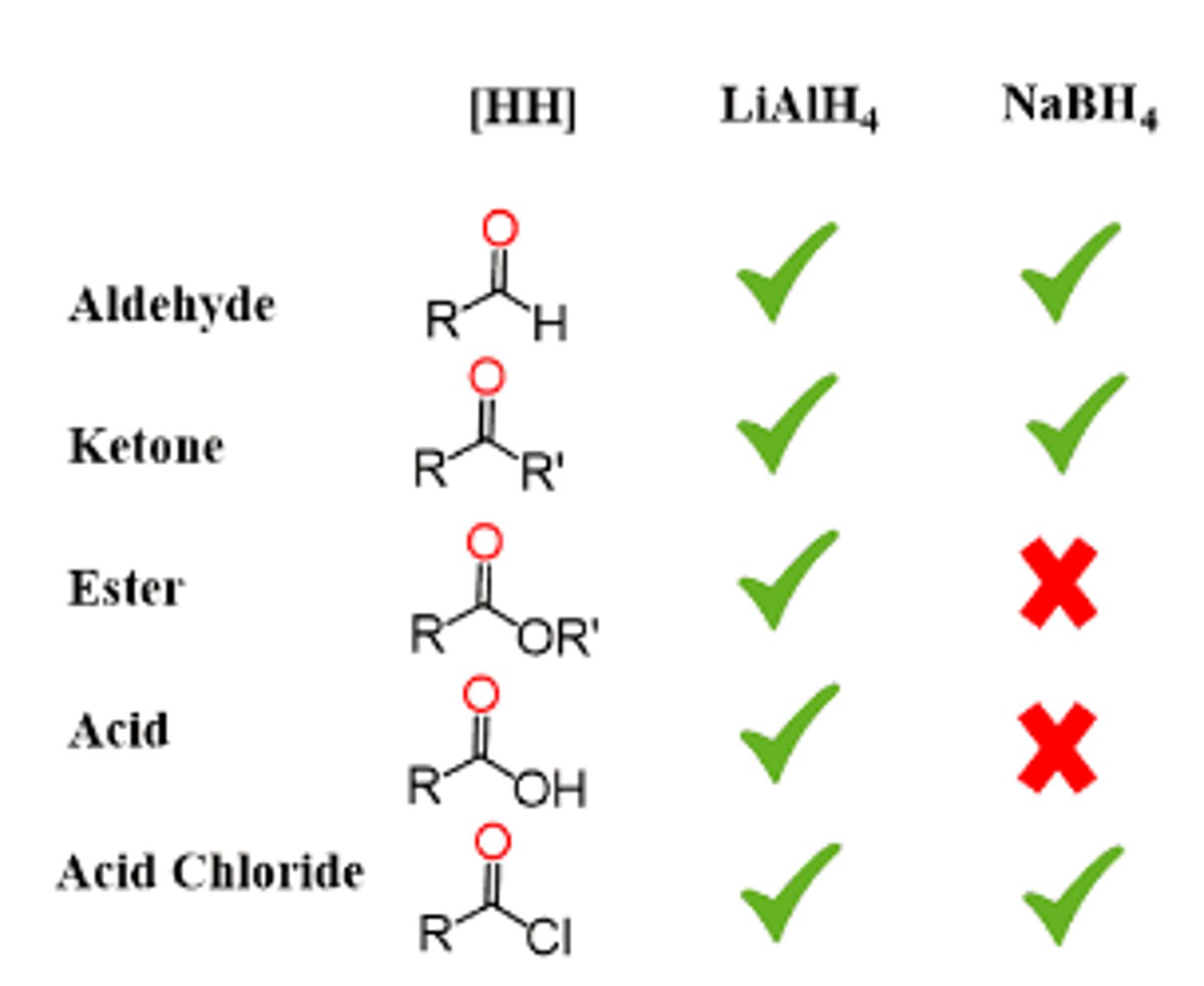
NaBH4 vs LiAlH4
- NaBH4 can reduce aldehydes, ketones
- LAH (more powerful) can reduce aldehydes, ketones, esters, and carboxylic acids
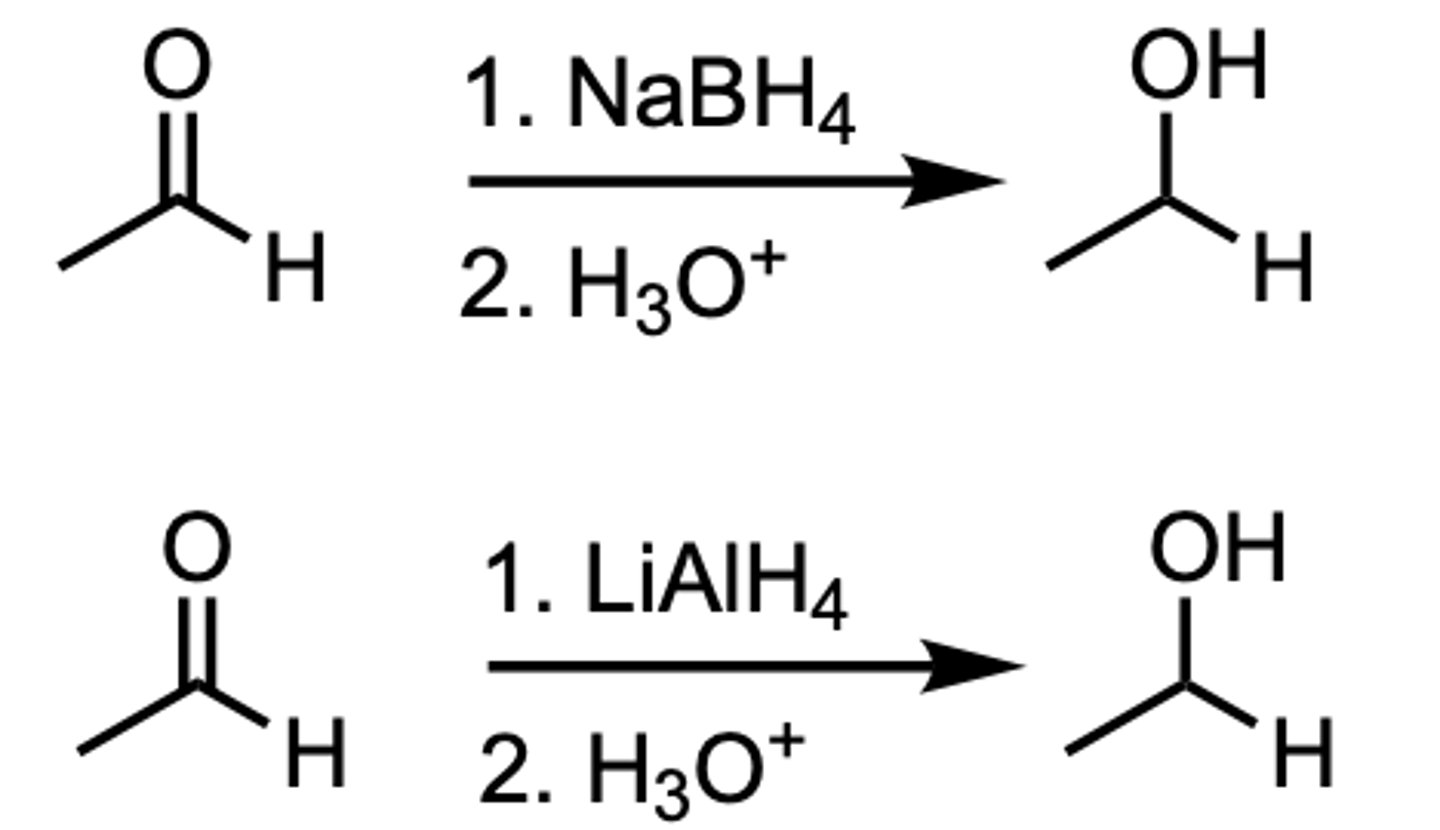
Reduction of an Aldehyde to a ___
1 ̊Alcohol
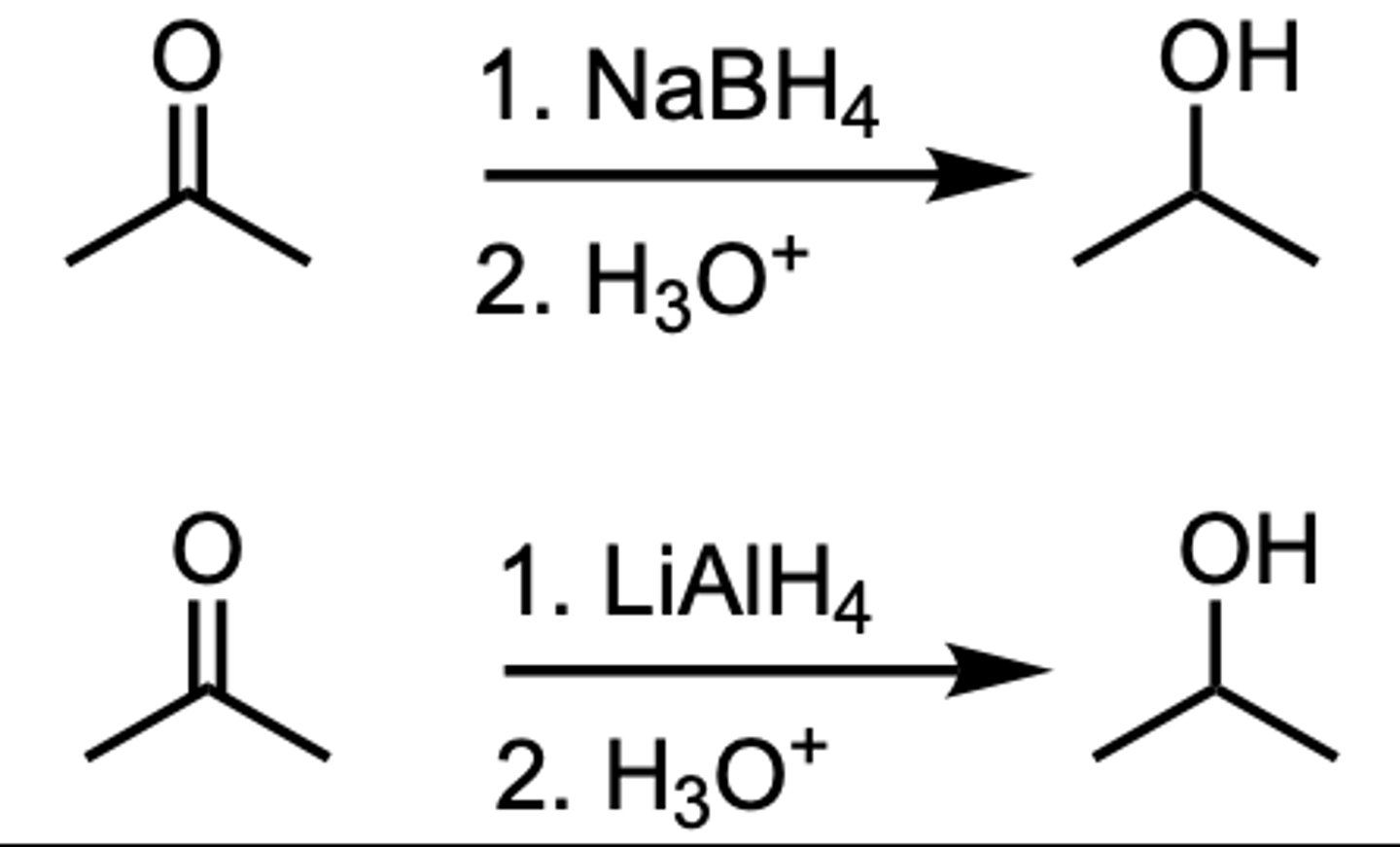
Reduction of a Ketone to a ___
2 ̊Alcohol
Reduction of a Carboxylic Acid to a ___
1 ̊Alcohol
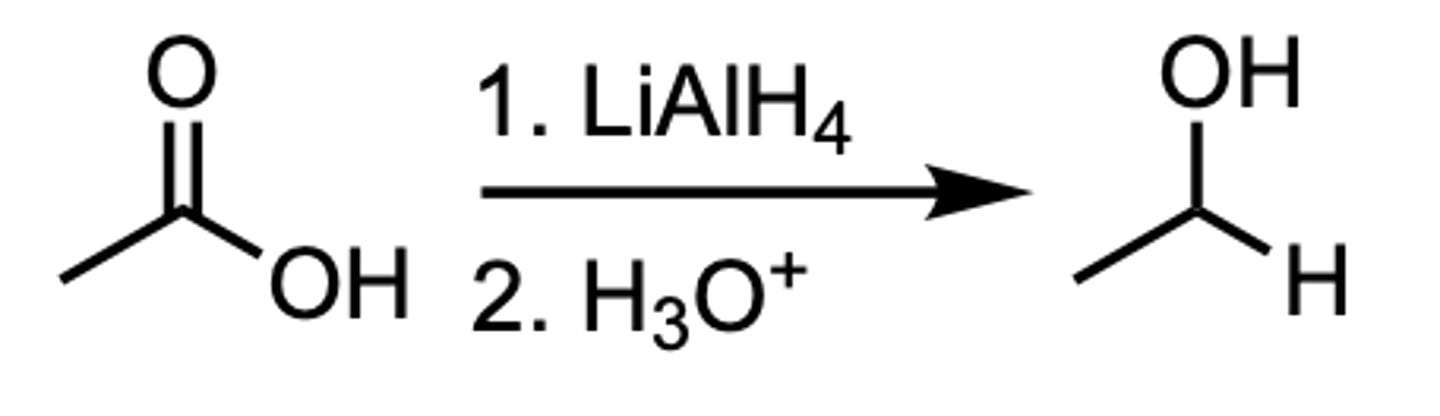
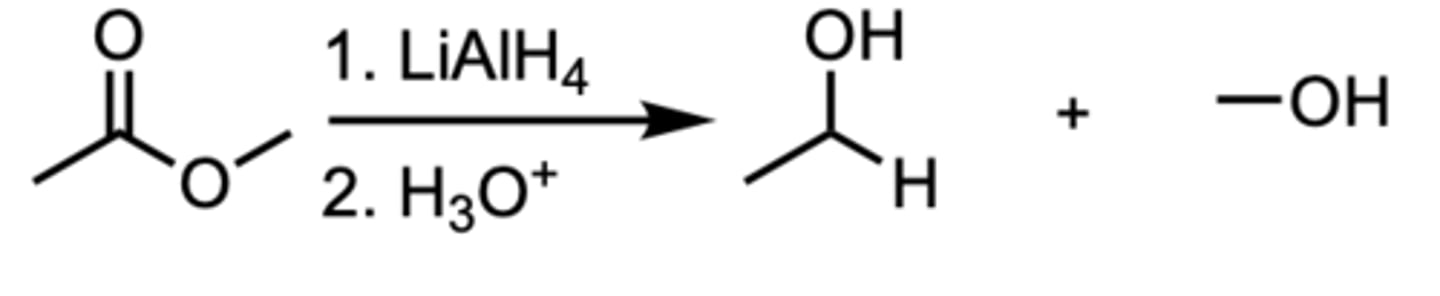
Reduction of an Ester to a ___ Alcohol
1 ̊Alcohol
Reduction of an Ester to an Aldehyde
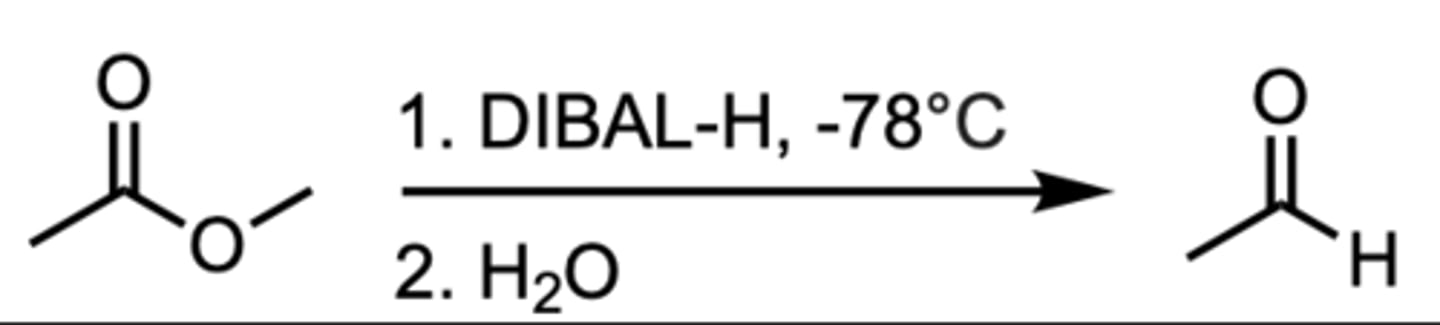
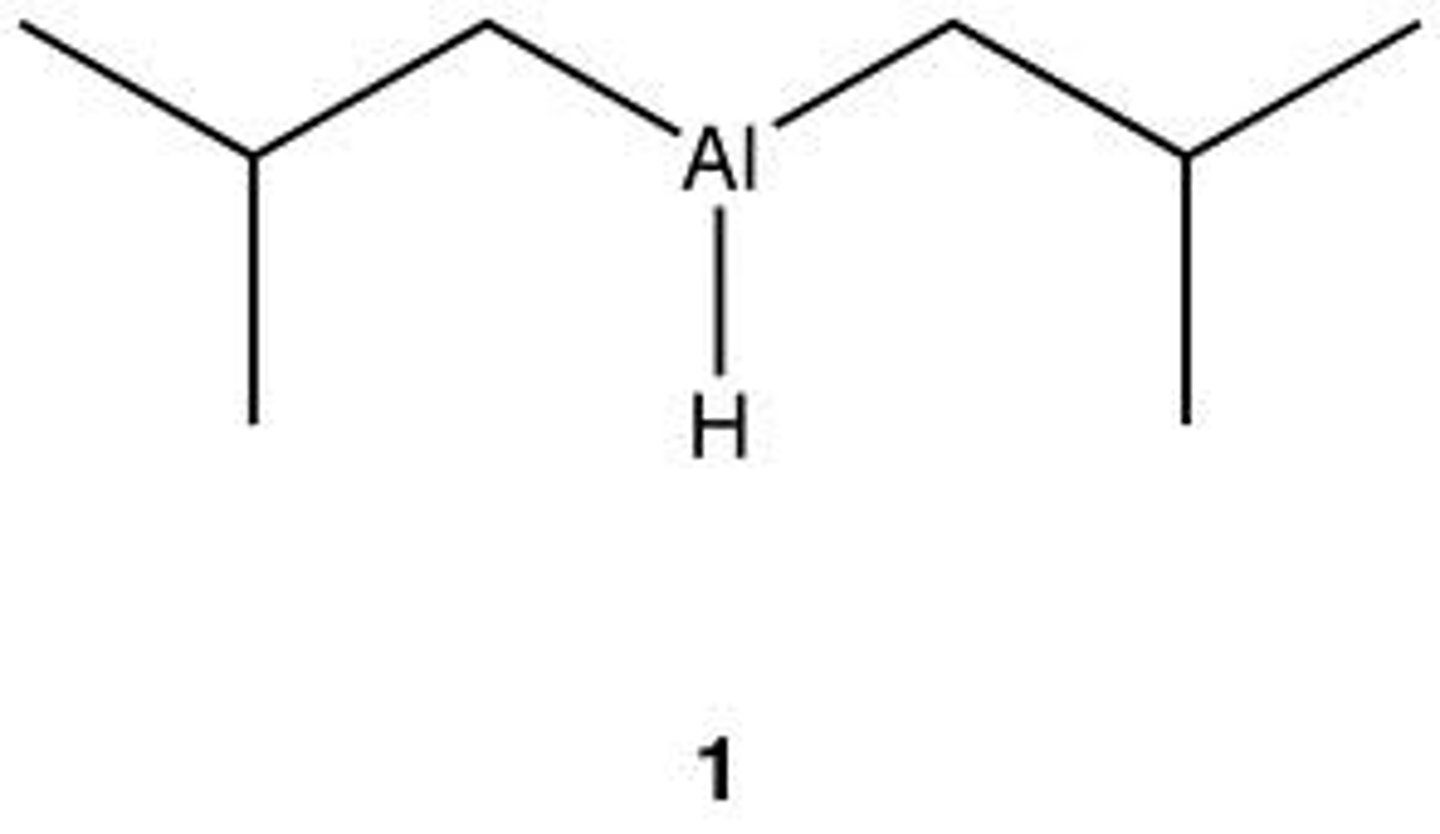
DIBAL-H
Reduction of an Acyl Chloride to a __ Alcohol
1 ̊Alcohol
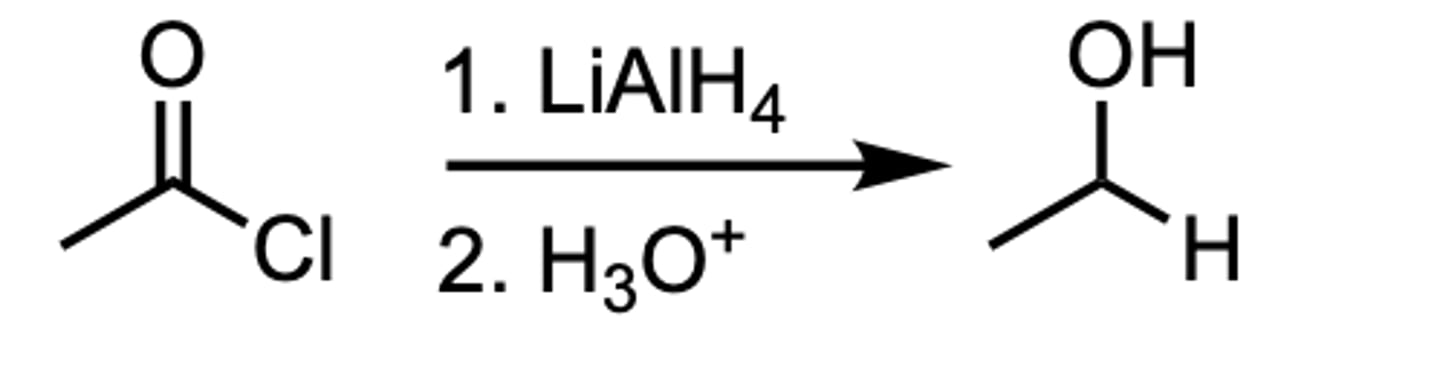
Reduction of an Acyl Chloride to an Aldehyde

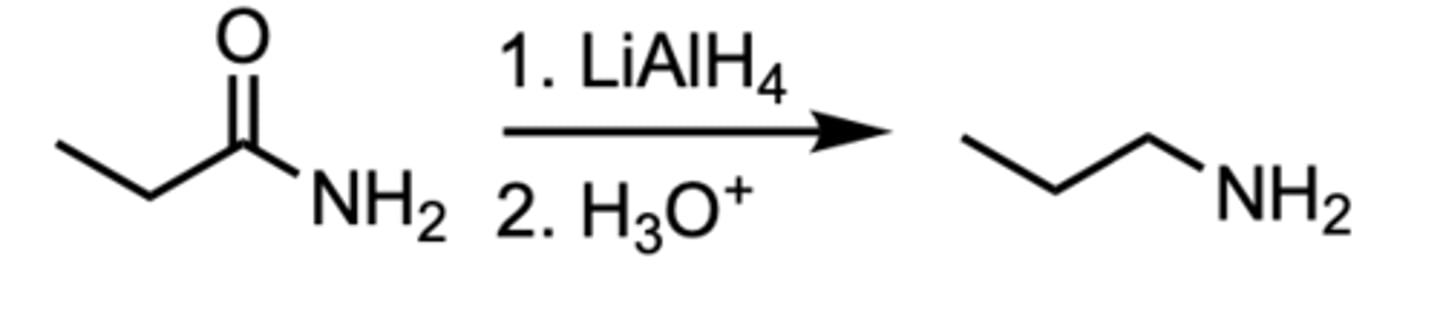
Reduction of an Amide to an ___
Amine
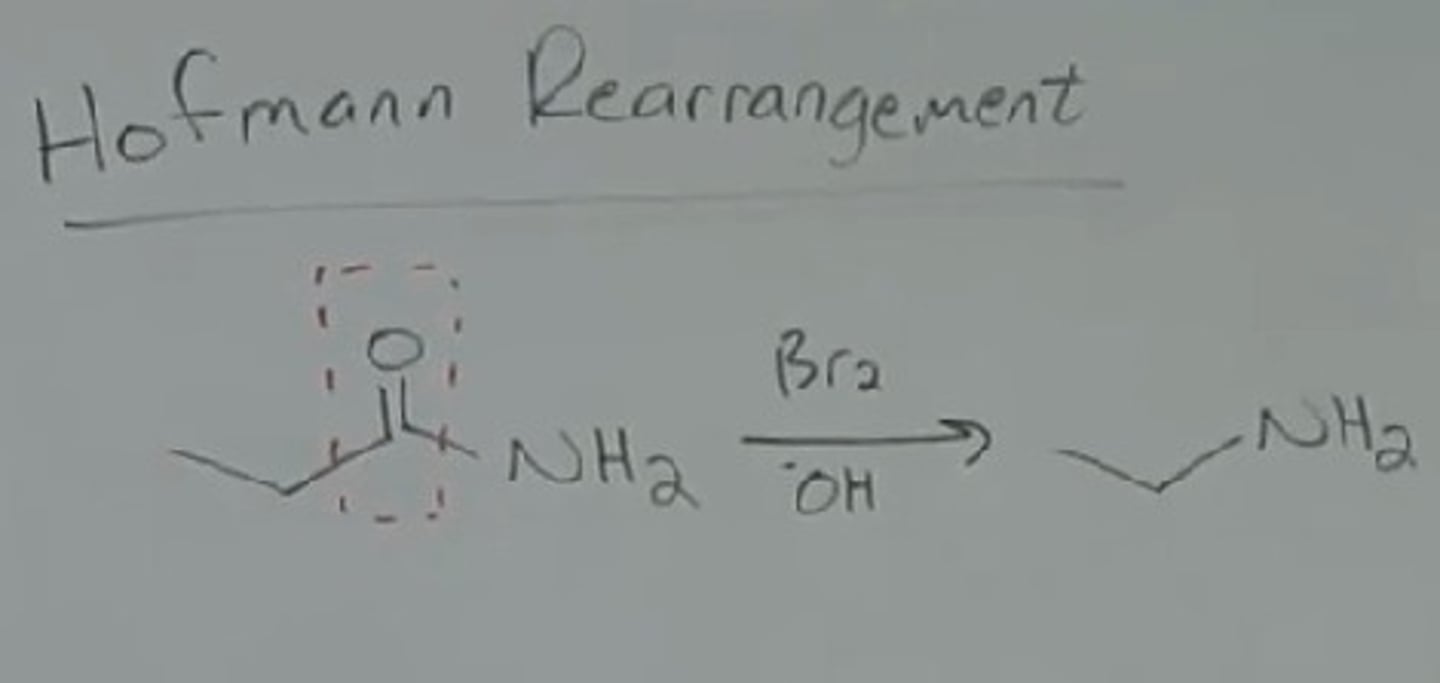
Hofmann Rearrangement
1 ̊ amide -> 1 ̊ amine with one fewer carbon atom
1) Br2
2) NaOH
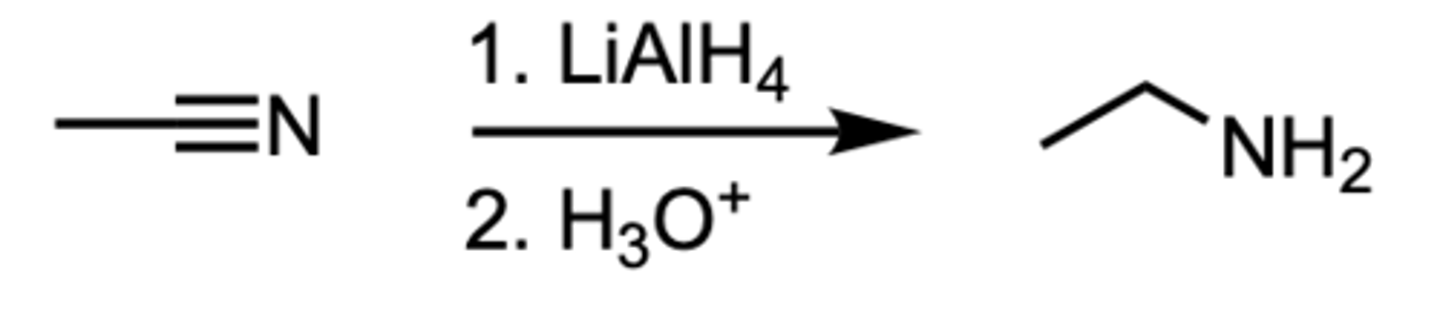
Reduction of a Nitrile to an ____
Amine