BIOS 301: L4 Carbs
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
Carbohydrates
Cn(H2O)n
Produced in plants from CO2 and H2O via photosynthesis
Functions: energy source and energy storage, structural components of cell walls and exoskeletons (chitin), informational molecules in cell to cell signaling
Can be covalently linked with proteins and lipids
Oxidation of sugars is the central energy-yielding pathway in most non-photosynthetic cells!
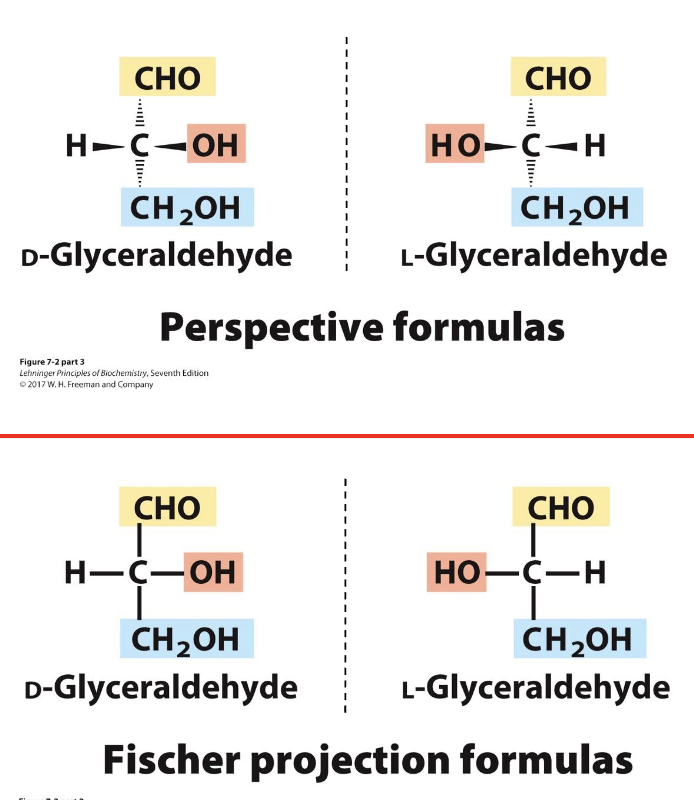
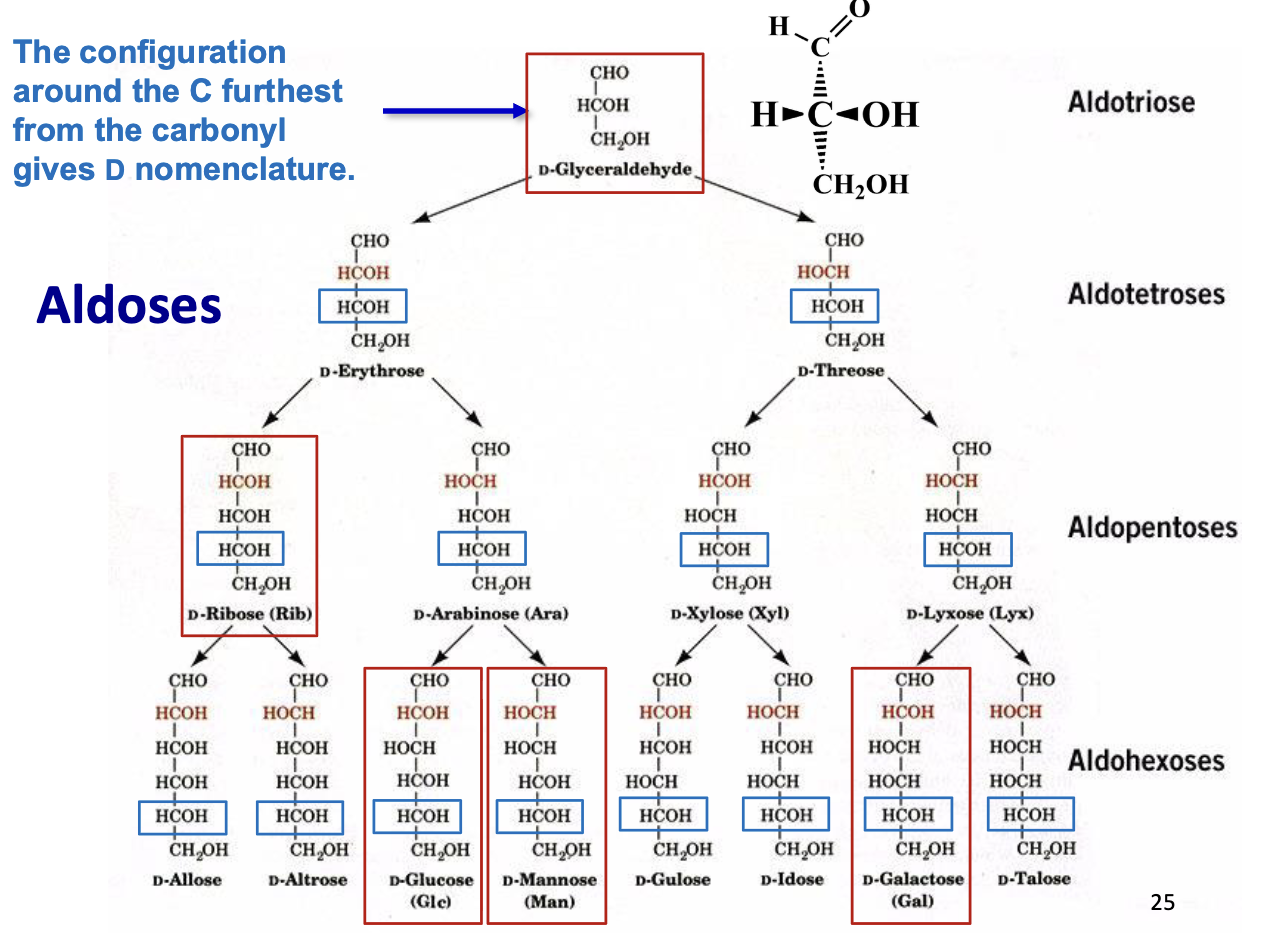
Fischer Projections – Chiral Compounds
Purpose: Represent chiral carbohydrates in 2D
Perspective rules:
Horizontal bonds: pointing toward you
Vertical bonds: pointing away from you
Useful for quickly visualizing stereochemistry of sugars
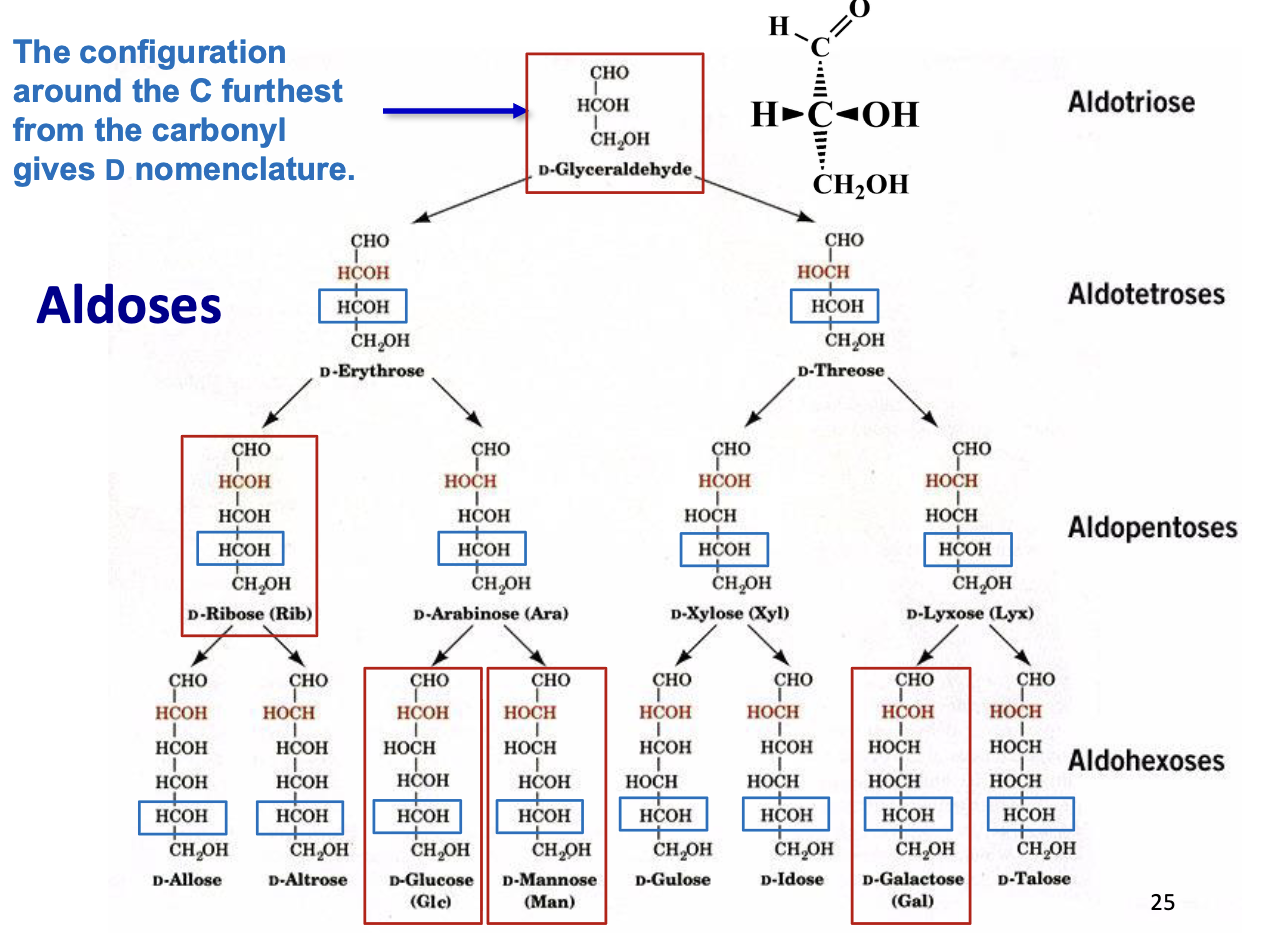
Carbohydrate Nomenclature
Basic naming: number of carbons + -ose
3C → triose, 4C → tetrose, 5C → pentose, etc.
Functional groups:
Aldehyde → aldose
Ketone → ketose
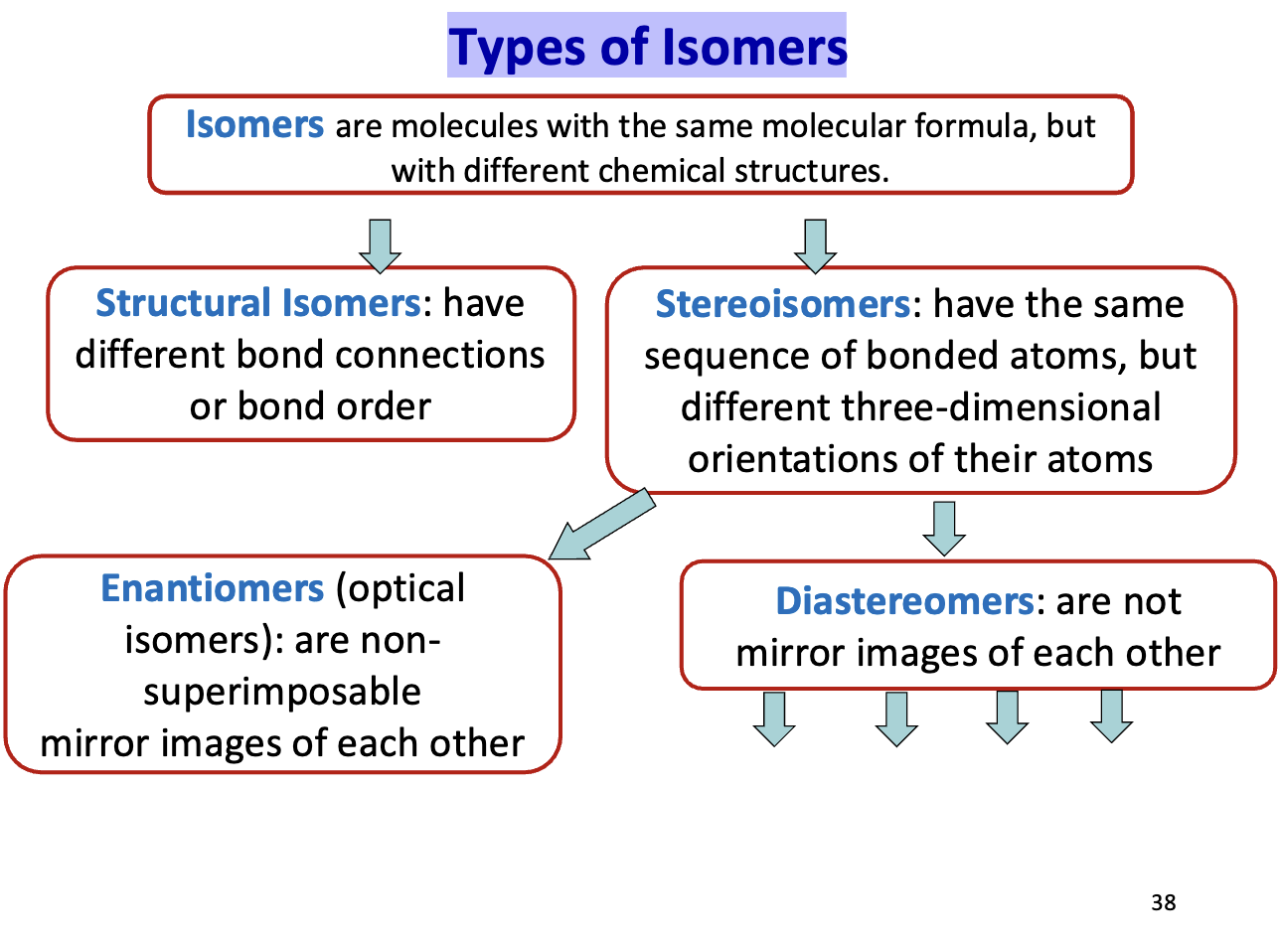
Carbohydrates can undergo extensive isomerization:
Structural isomers (different connectivity)
Stereoisomers (different spatial arrangement)
Structural Isomers – Aldose vs Ketose
Aldose: carbohydrate with an aldehyde group
Ketose: carbohydrate with a ketone group
Structural isomers: same molecular formula, different functional group placement
Stereoisomers – Diastereomers & Epimers
Diastereomers: stereoisomers that are not mirror images (≠ enantiomers)
Epimers: a type of diastereomer differing at only one chiral center
Epimers ≠ enantiomers
Have different physical properties
Examples:
D-Mannose and D-Glucose → epimers (differ at 1 chiral center)
D-Galactose and D-Glucose → epimers
D-Mannose and D-Galactose → diastereomers (differ at >1 chiral center, not epimers)
D-Glyceraldehyde Structure
D-Ribose (Rib) Structure
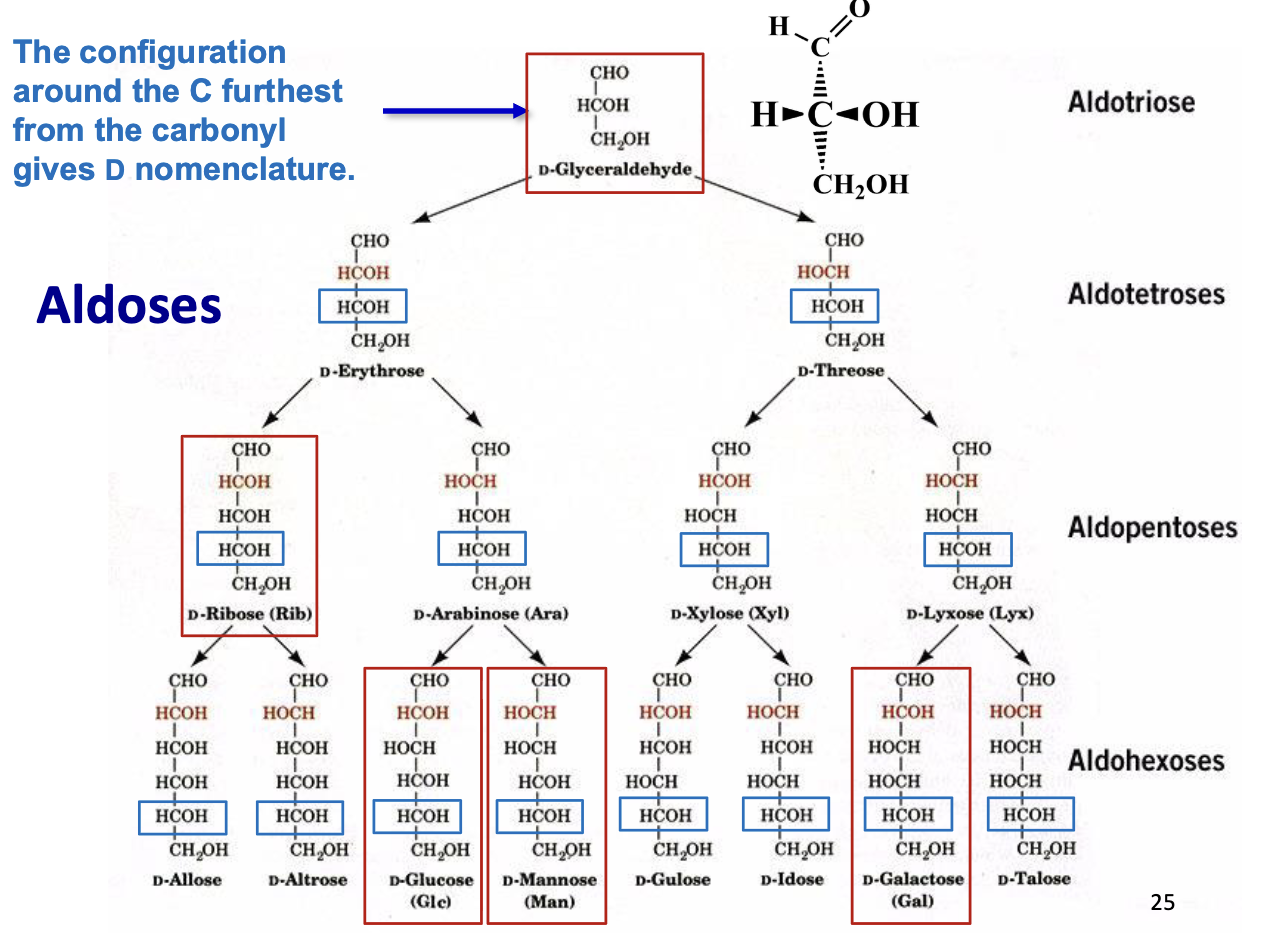
D-Glucose (Glc) Structure
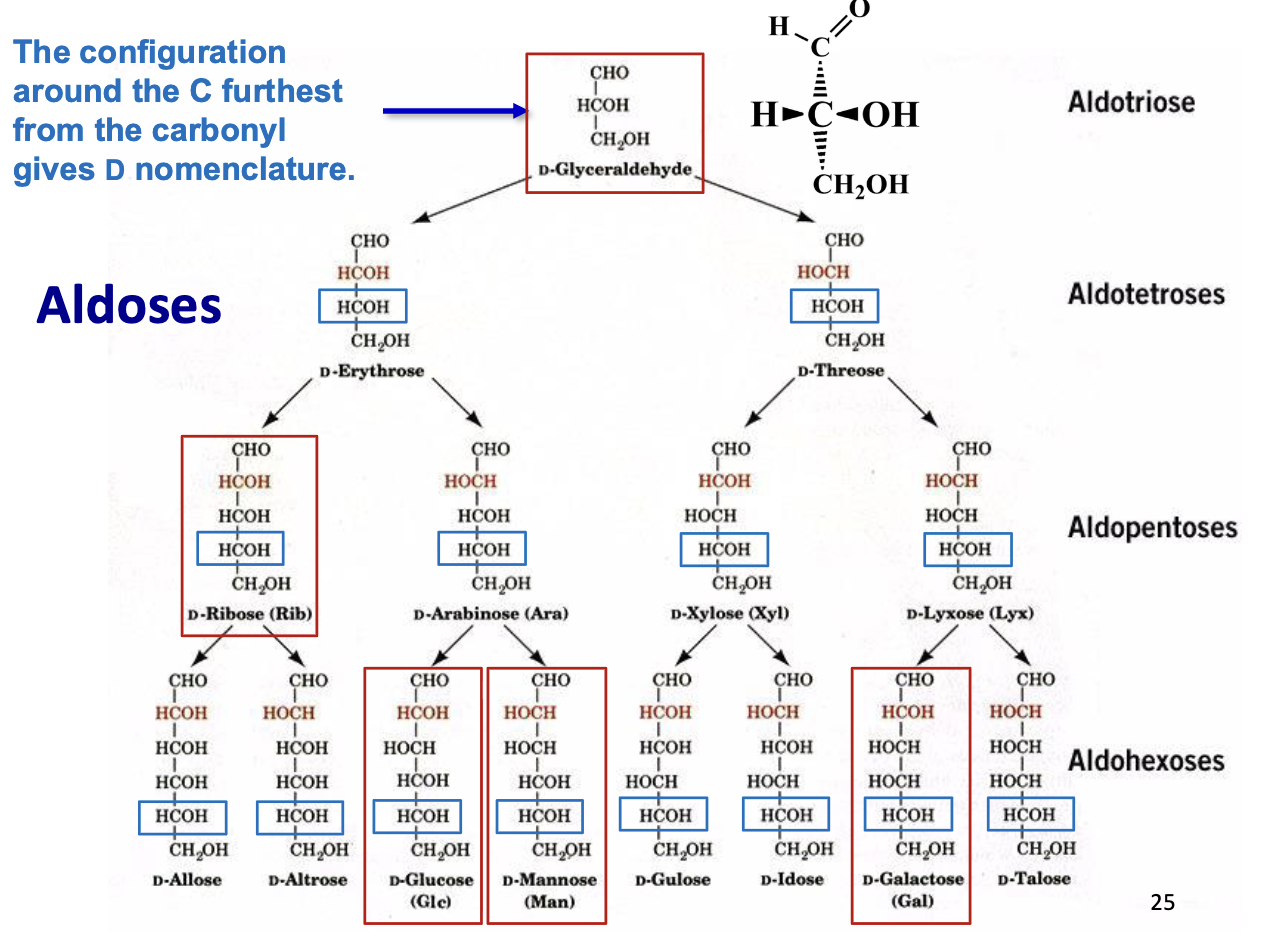
D-Mannose (man) Structure
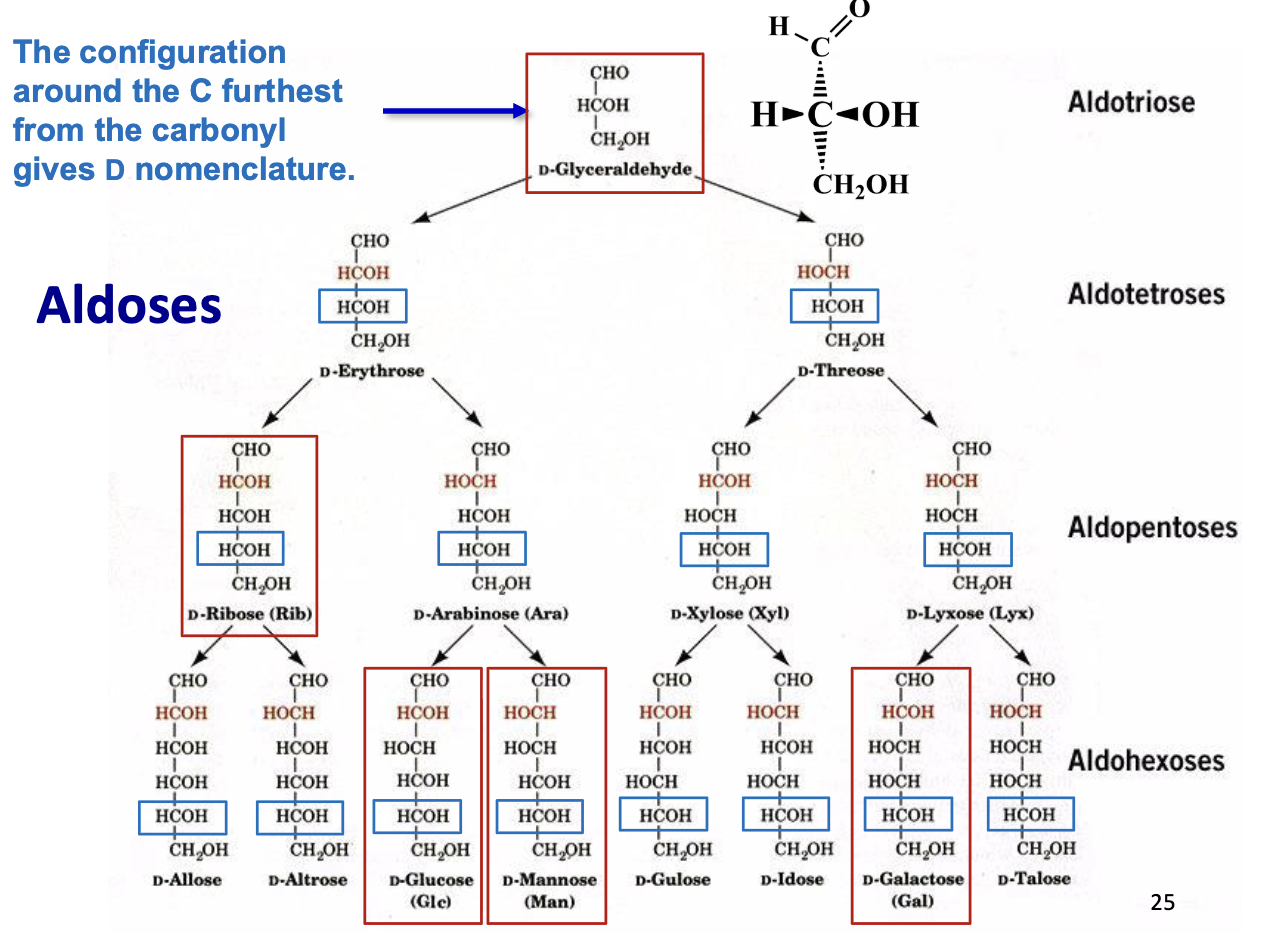
D-Galactose (Gal) Structure
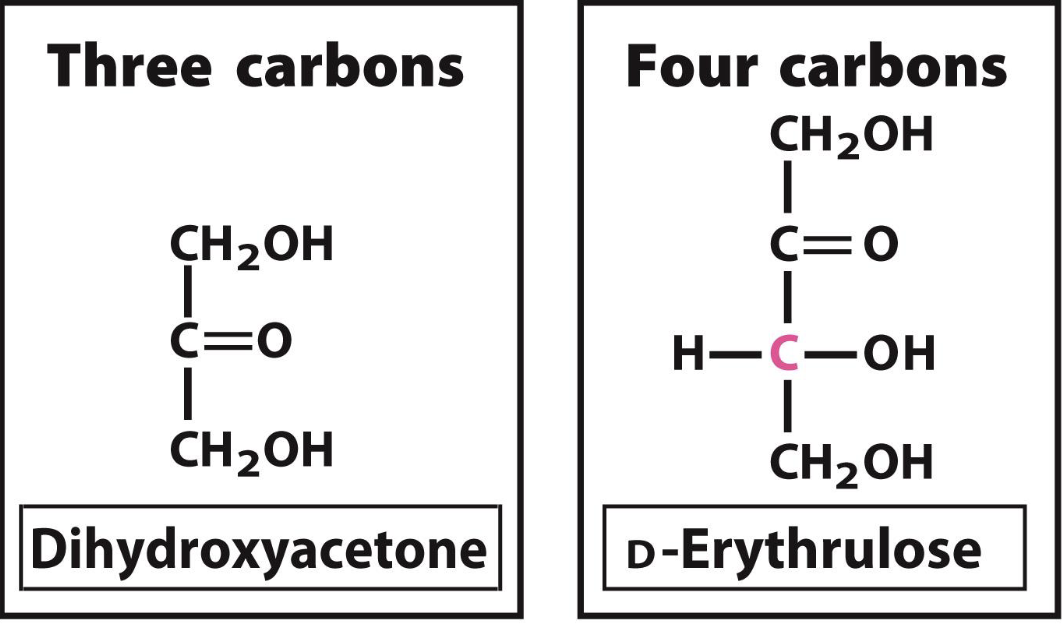
Dihydroxyacetone
a d ketoses
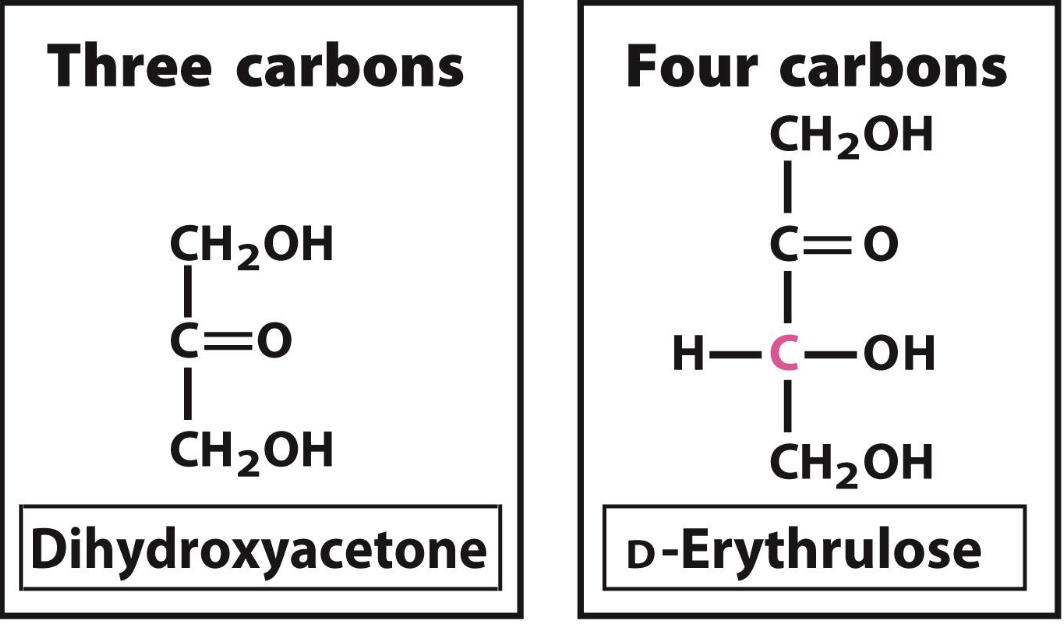
D-Erythrulose
D ketoses
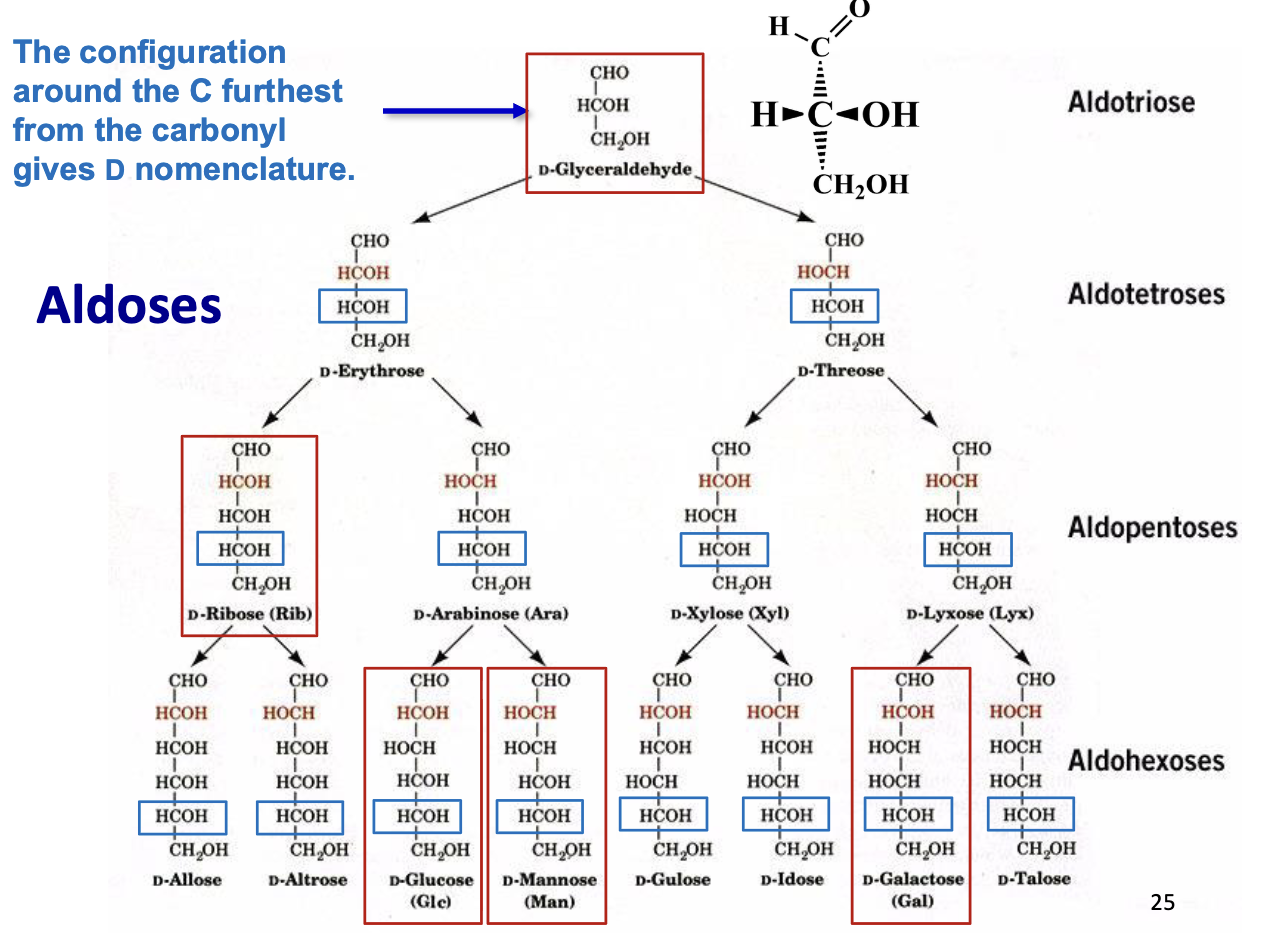
Aldoses
Carbohydrates with an aldehyde functional group (–CHO)
Carbonyl is at the end of the molecule (C1)
Named based on carbon number:
3C → aldotriose
6C → aldohexose
Examples: glucose, ribose
D-Fructose
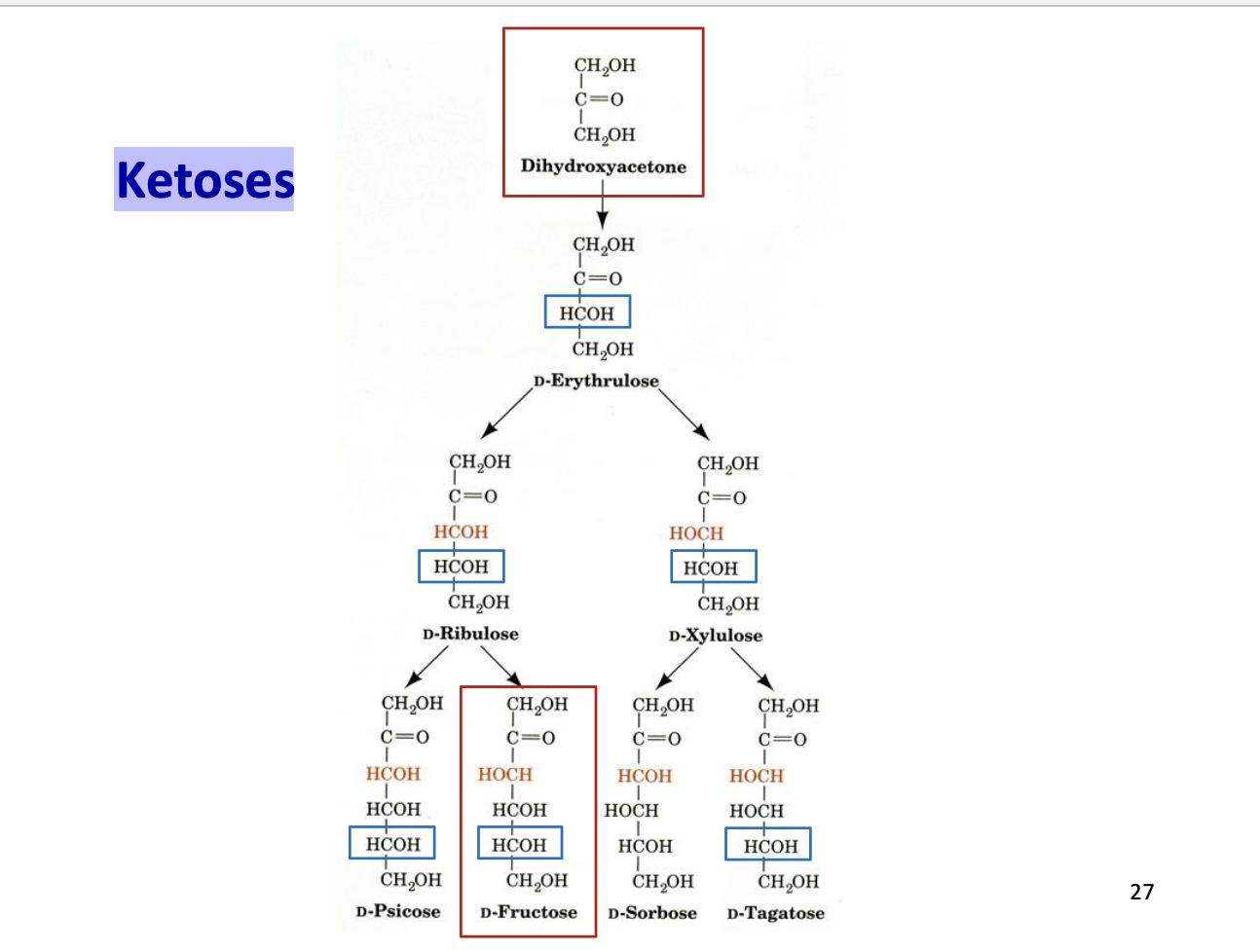
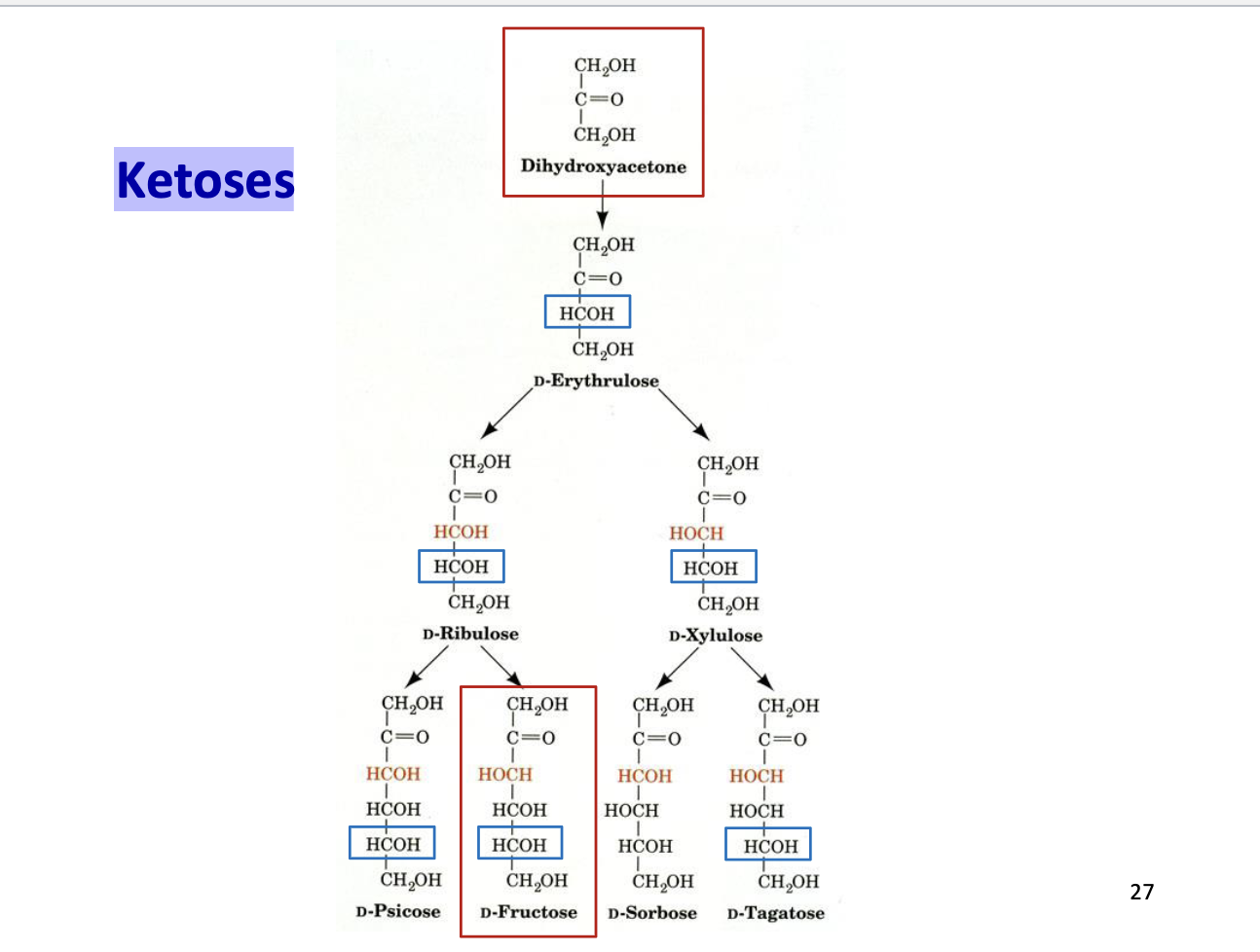
Ketoses
Carbohydrates with a ketone functional group (C=O)
Carbonyl is usually at C2 (middle of the chain)
Named based on carbon number:
3C → ketotriose
6C → ketohexose
Examples: fructose, ribulose
Reducing Sugars
Definition: sugars with a free aldehyde at the anomeric carbon (C1)
Can reduce metal ions:
Cu²⁺ → Cu⁺ (Fehling’s test)
Ag⁺ → Ag⁰ (Tollens’ test)
Allows detection of reducing sugars like glucose
Modern methods: colorimetric or electrochemical tests
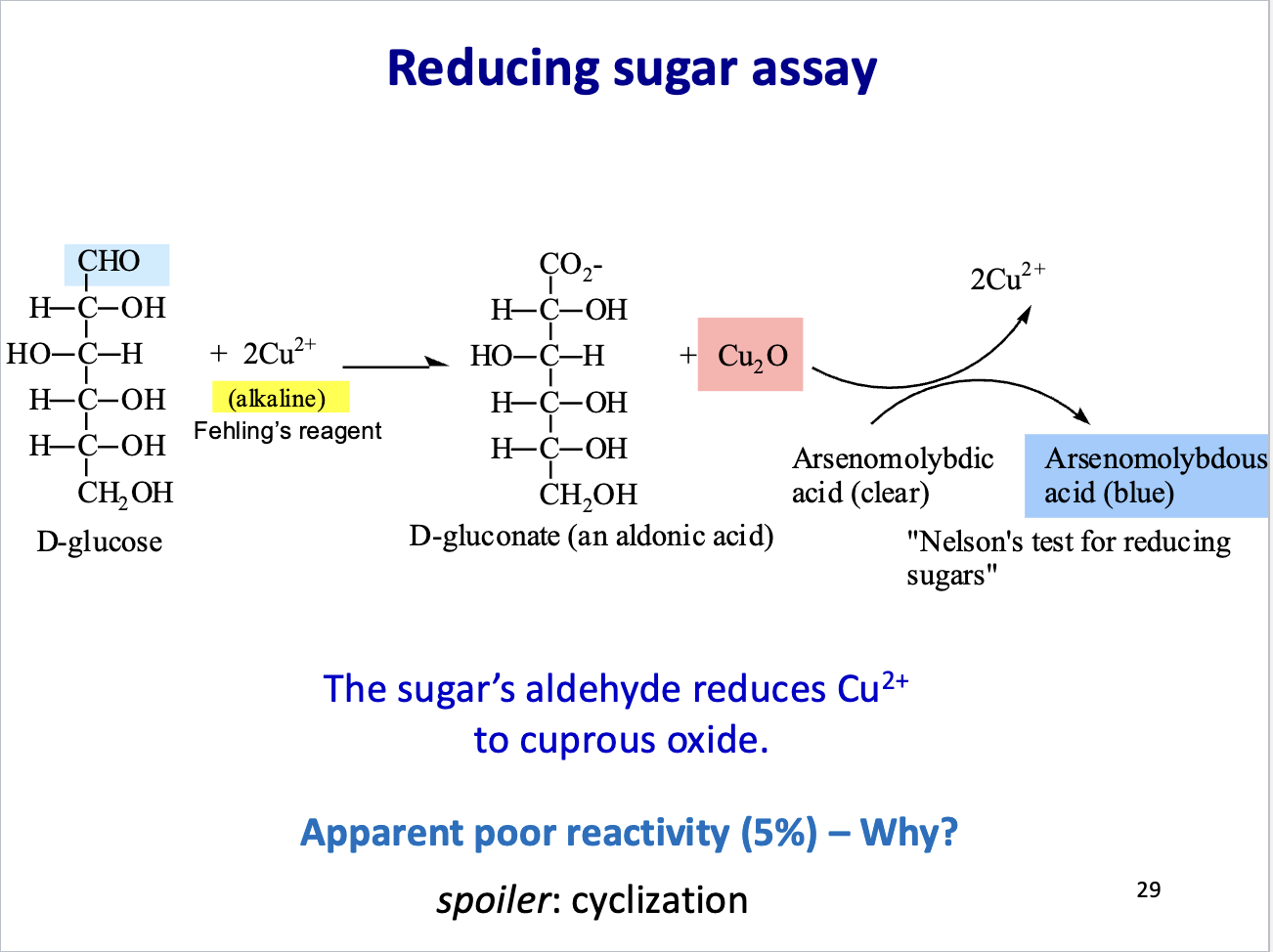
Reducing sugar assay
Reducing sugars have a free aldehyde (or sometimes ketone) group that can donate electrons to metal ions like Cu²⁺ or Ag⁺, reducing them to Cu⁺ (forms a brick-red precipitate) or Ag⁰ (silver mirror). The color change shows sugar is present.
Nelson’s Test (Color Development)
To make the result easier to measure with a lab instrument (spectrophotometer), a second step is added:
The Cu2O produced in the first step reacts with Arsenomolybdic acid.
This converts the clear acid into Arsenomolybdous acid, which has a distinct blue color. The intensity of this blue color tells you how much sugar was originally in the sample.
Why is reducing sugar assay a slow reaction?
Cyclization In an aqueous solution (like your blood or a lab beaker), glucose doesn't mostly exist in that straight-chain "linear" form shown on the left. Instead:
Over 99% of glucose molecules fold into a ring structure (pyranose).
The aldehyde group is "hidden" or "locked" inside the ring (as a hemiacetal).
Only the small fraction (~1%) that is in the open-chain form at any given moment can react. As that 1% is used up, the rings slowly open to provide more, but the "instant" availability of the reactive group is very low.
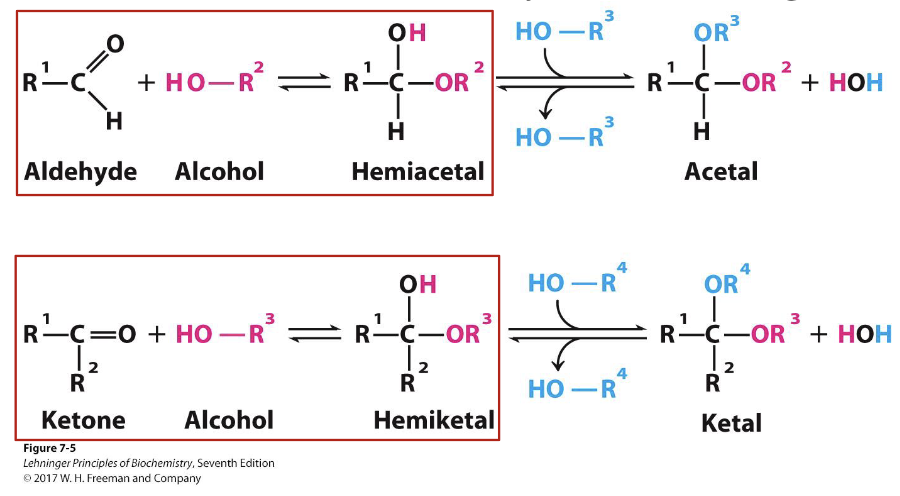
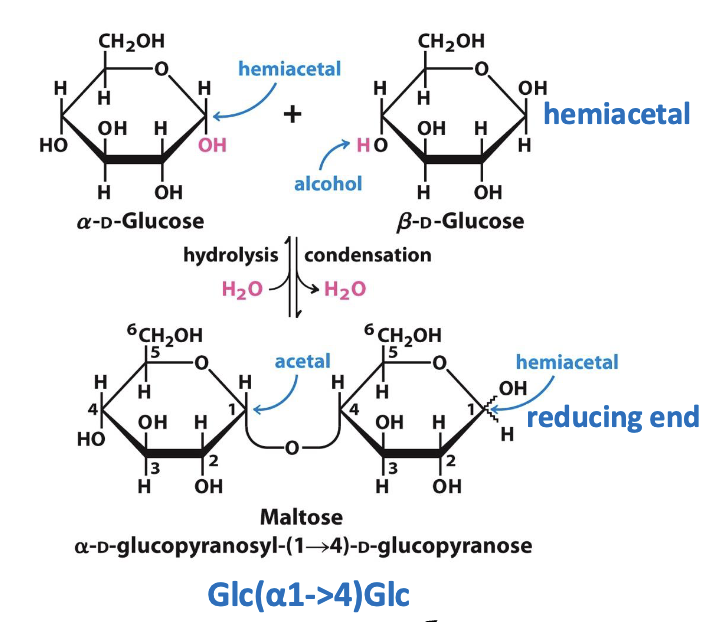
Hemiacetals & Hemiketals – carbohydrate reactivity
Aldehydes/ketones are electrophilic. Alcohols are nucleophilic.
Aldehyde + alcohol → hemiacetal
Ketone + alcohol → hemiketal
These reactions let sugars cyclize into rings.
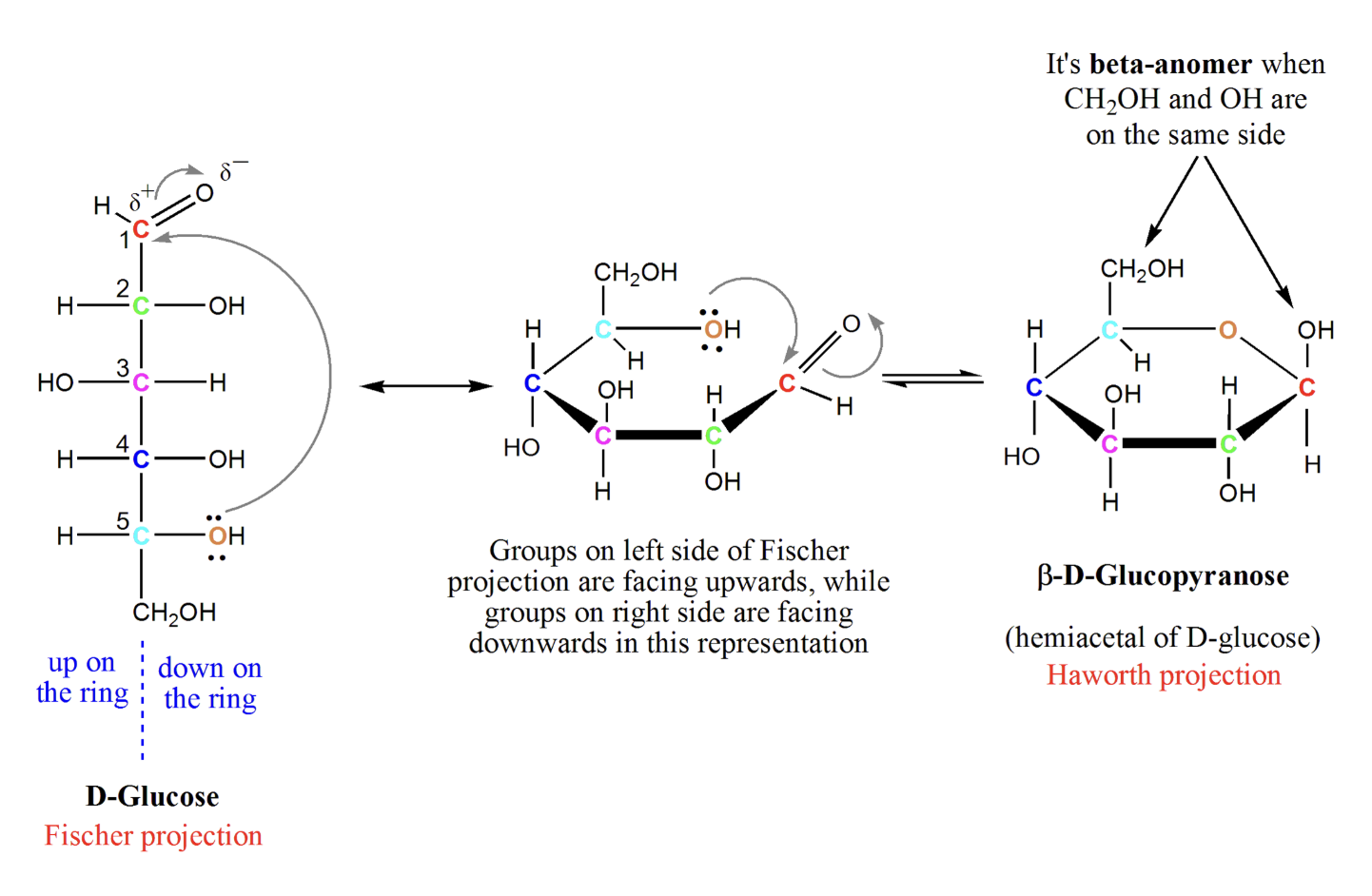
Formation of pyranose ring (hemiacetal)
A sugar’s aldehyde reacts with an internal alcohol (usually on C5) → forms a hemiacetal → sugar cyclizes into a 6-membered pyranose ring. The anomeric carbon becomes newly chiral.
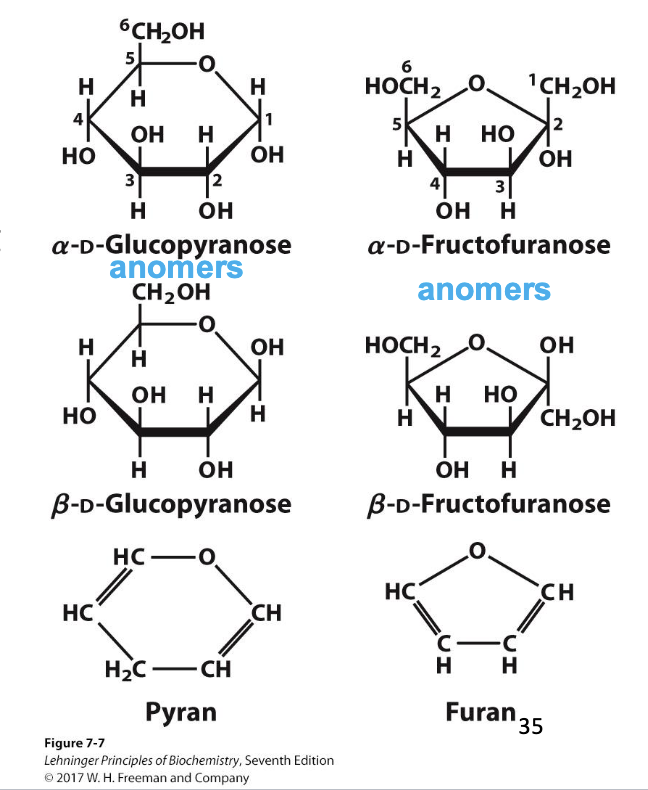
pyranose
A 6-membered sugar ring that contains an oxygen atom. Highlights that sugars can cyclize into 6-membered rings.
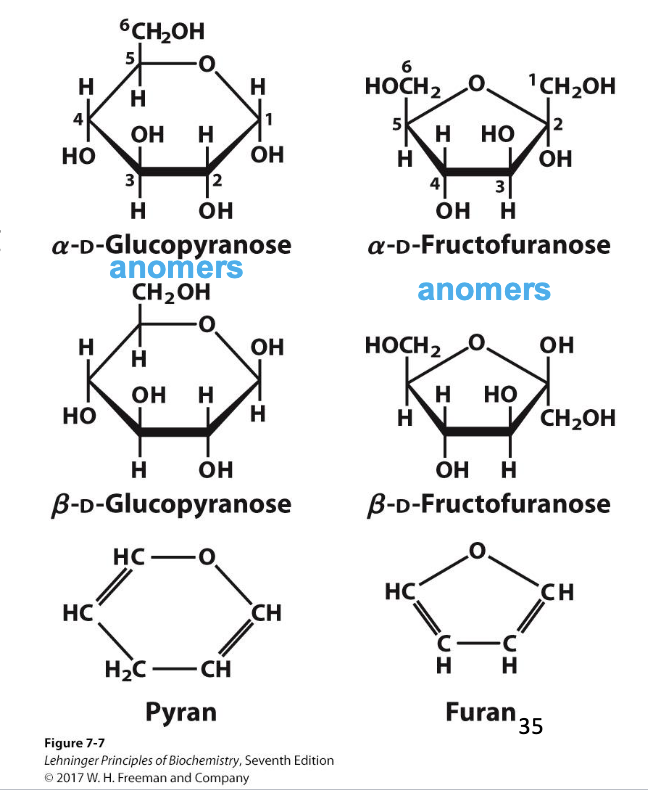
furanose
A 5-membered sugar ring that contains an oxygen atom. Highlights that sugars can cyclize into 5-membered rings.
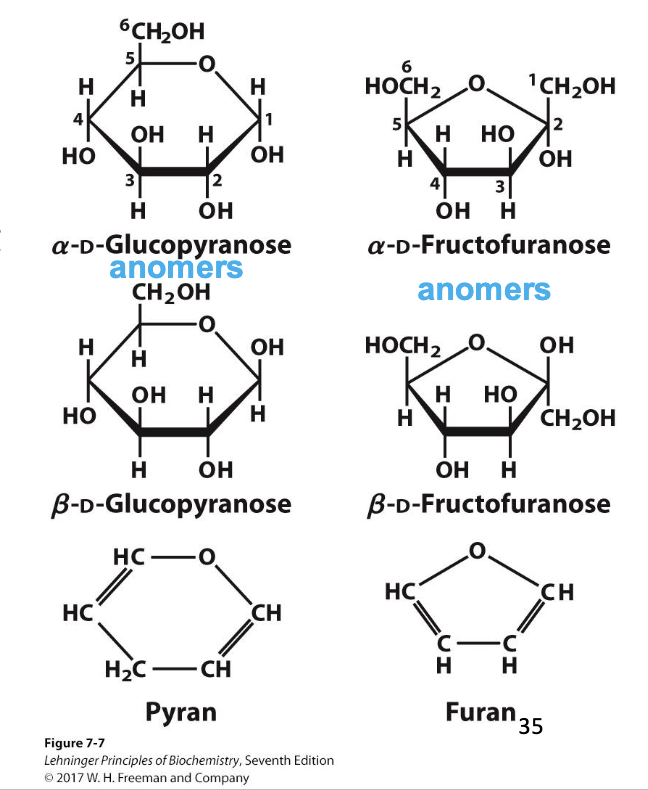
anomer
The new chiral carbon formed from the former carbonyl carbon during ring formation. Highlights that cyclization creates a new stereocenter at the anomeric carbon.
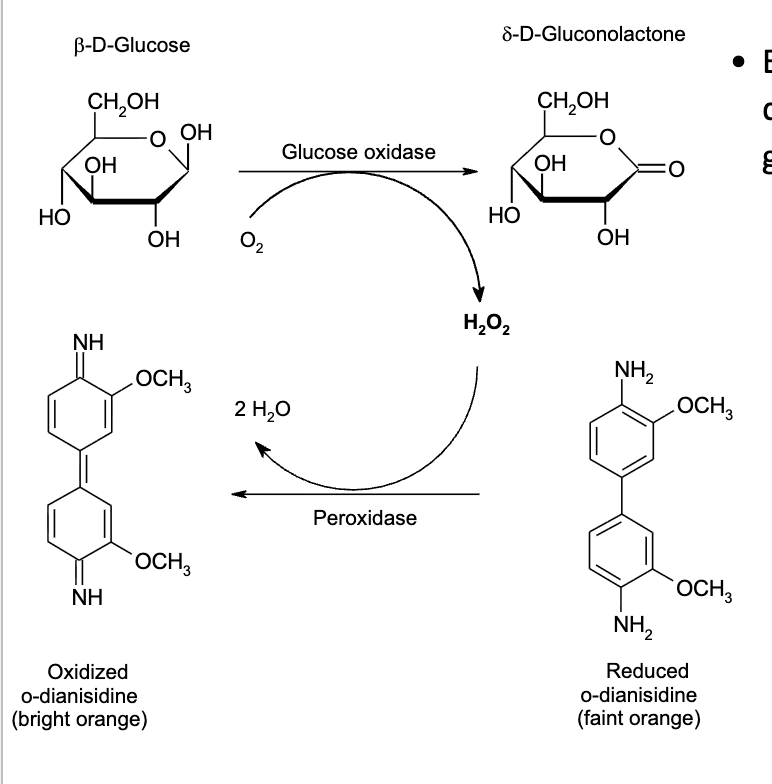
Colorimetric Glucose Analysis
Enzymatic methods are used to
quantify reducing sugars such as
glucose.
– The enzyme glucose oxidase
catalyzes the conversion of
glucose to glucono-
-lactone and
hydrogen peroxide.
– Hydrogen peroxide oxidizes
organic molecules into highly
colored compounds.
– Concentrations of such
compounds are measured
colorimetrically.
Pyranose rings favor chair or boat conformations?
A pyranose is a monosaccharide (sugar) that exists as a six-membered cyclic hemiacetal ring, comprising five carbon atoms and one oxygen atom.
Favors chair
chair confirmations require energy to flip.
Types of Isomers
Types of Diastereomers

The Glycosidic Bond
Two sugars join via a glycosidic bond: anomeric carbon + hydroxyl group
The bond is an acetal, more stable & less reactive than a hemiacetal
Second sugar’s hemiacetal = reducing end
Anomeric carbon in glycosidic bond = nonreducing end
Disaccharides named by linkage (e.g., α-D-glucopyranosyl-(1→4)-D-glucopyranose = maltose)
Nonreducing Disaccharides
Formed when two anomeric carbons join via a glycosidic bond
Product has two acetal groups, no hemiacetals
No reducing ends → nonreducing sugar
The orientation of the sugars can be switched