AQA GCSE Chemistry - Equations
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
Avogadro's number
There are 6.02 x 10²³ particles of a substance in 1 mole.
The mole
moles = mass/relative atomic mass
Concentration in g/dm³
concentration = mass (g)/volume (dm³)
Concentration in mol/dm³
concentration = moles (mol)/volume (dm³)
or
moles = concentration x volume (in dm³)
Gas volumes
1 mole of any gas has a volume of 24dm³
volume (dm³) = moles x 24dm³
Converting from cm³ to dm³
1dm³ = 1000cm³
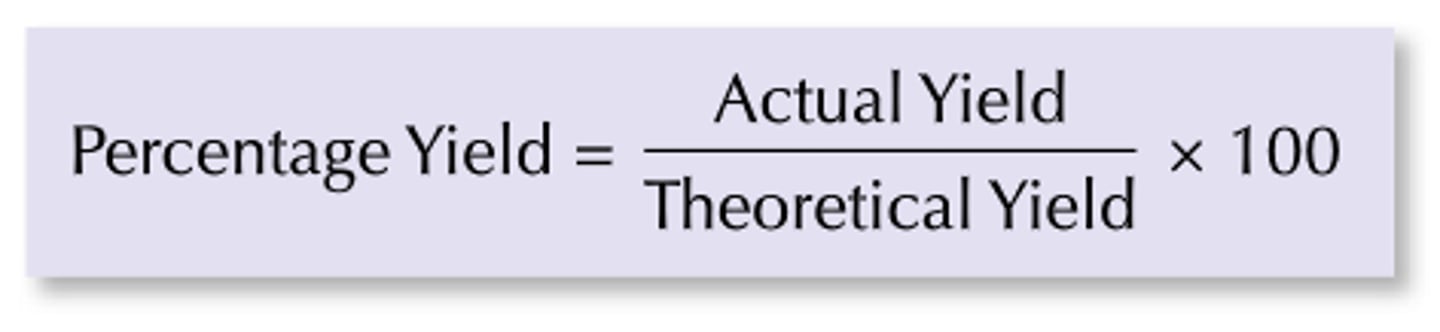
Percentage Yield
(mass of product actually made/maximum theoretical mass of product) x 100
Atom Economy
(relative formula mass of desired product from equation/sum of relative formula masses of all reactants from equation) x 100
Calculating energy changes
Energy change in a reaction = Total energy needed to break the bonds in reactants - Total energy released when bonds in the products are formed
Rate of reaction
mean rate of reaction = quantity of reactant used/time taken
mean rate of reaction = quantity of product formed/time taken
(Quantity usually measured as a mass or volume)
Chromatography
Distance moved by substance/distance moved by solvent