human phys exam 2
1/133
Earn XP
Description and Tags
Ch 6: Nervous System
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
134 Terms
What is the anatomical division of the nervous system?
central nervous system: brain & spinal cord
peripheral nervous system: nerves outside of the brain & spinal cord
What is the functional division of the nervous system?
sensory (afferent) division: carries impulses from receptors to CNS; detects internal & external stimuli
motor (efferent) division: carries impulses from CNS to effectors
somatic nervous system (SNS): control voluntary movements (e.g., skeletal muscles)
autonomic nervous system (ANS): controls involuntary functions (e.g., smooth muscle, cardiac muscle)
sympathetic: “fight or flight”
parasympathetic: “rest and digest”
what are neurons?
cells in the nervous system that sense environmental changes, process information, integrate (communicate changes to other neurons), command body responses (motor)
what are glial cells?
insulates, supports, and nourishes neurons
can also communicate, receive and send signals
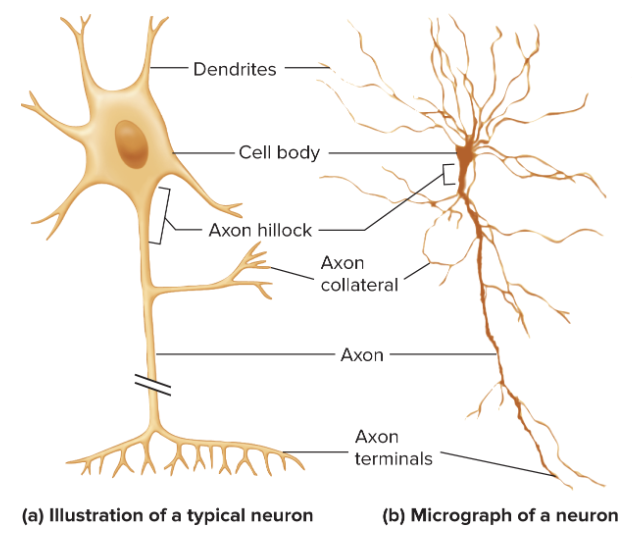
what is the structure of neurons?
dendrites: receives signals and conducts impulses toward cell body
dendritic spines: increases surface area of dendrites
cell body: contains nucleus
axon hillock: beginning of axon
axon: conduct nerve impulses to other neuron
axon collateral: branches from axon
axon terminal: releases neurotransmitter
varicosity: bulges that store & release neurotransmitters
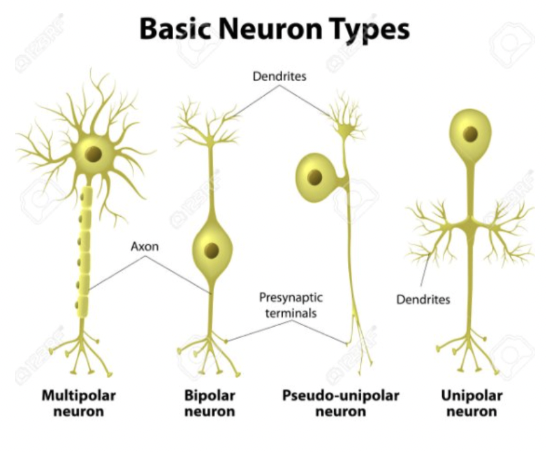
what are the type of neuron shapes?
multipolar neuron: most common; found in CNS, motor neurons
bipolar neuron: least common; found in eyes; two processes
pseudo-unipolar neuron: 1 process, but looks like 2 processes
unipolar neuron: 1 process
*pseudo-unipolar & unipolar are similar—both sensory neurons
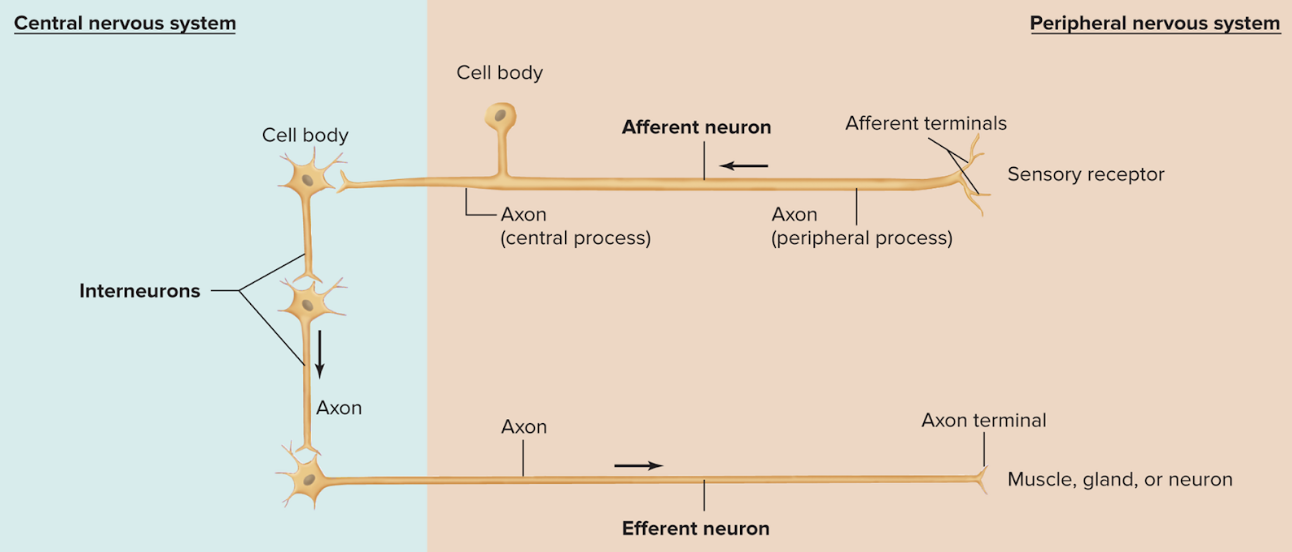
What are the three types of neurons? what is the order for signaling pathway?
sensory/afferent neuron: conveys info from PNS to CNS; unipolar or pseudounipolar; cell body in PNS
interneurons: lies within CNS; connects afferent and efferent neurons; generally multipolar; allows one sense to activate multiple pathways
motor/efferent neuron: conveys info away from CNS to effector cell; multipolar; cell body in CNS
order: sensory —> interneurons —> motor neurons
what is the difference between gray matter and white matter?
gray matter: composed of densely packed cell bodies; located outside
white matter: composed of bundles of myelinated axons; located inside
what are the types of glial cells in the CNS?
oligodendrocytes
microglia
ependymal cell
astrocyte
what is the oligodendrocyte?
CNS glial cell that forms myelin sheath in CNS axons; white matter
what is microglia?
CNS glial cells that acts as phagocytes to remove dead cells/pathogens and provide immune defense
what is ependymal cell?
CNS glial cell that lines brain ventricles & central canal of spinal cord
produces cerebrospinal fluid (CSF)
what are astrocytes?
maintains blood brain barrier
nourishes neurons w/ glycogen
reuptake of neurotransmitters
repair + regenerate neurons
communicate changes in blood
regulation of blood in extracellular brain
maintain ion concentrations (K+)
what are schwann cells?
PNS glial cells that forms myelin sheath
what is the blood brain barrier?
protective, selective barrier that separates the bloodstream from the brain tissue
structure: Blood → Endothelial cell with tight junctions → Basement membrane → Astrocyte end-feet → Brain tissue
what is electrical potential vs. voltage?
electrical potential: charge difference across the cell membrane at one point
voltage: difference in electrical potential between two points.
what factors influence ion movement across the membrane?
concentration gradient (diffusion)
ion channels (leak channels, gated channels)
equilibrium potential (influenced by electrical gradient)
what is the purpose of the Nernst equation?
calculates equilibrium potential of ONE ion across a membrane
does not consider permeability
what is the Goldman-Hodgkin-Katz (GHK) equation?
calculate the actual membrane potential considering MULTIPLE ions
used to calculate the resting membrane potential bc multiple ions (K+, Na+, Cl-) contribute to the resting membrane potential
takes in account ions’ concentration gradients & relative permeabilities
What is the resting membrane potential? What contributes to the resting membrane potential?
-70mV → excess positive ions outside of membrane
more Na+ and Cl- outside of cell
more K+ inside of cell
Na+/K+ pump: 3 Na+ out, 2K+ in
K+ leaky channel: move K+ out of cell down its gradient

Why is the equilibrium potential not reached?
need to consider multiple contributing ions (Na+, K+, Cl-)
nernst equation does not factor permeability of each ion
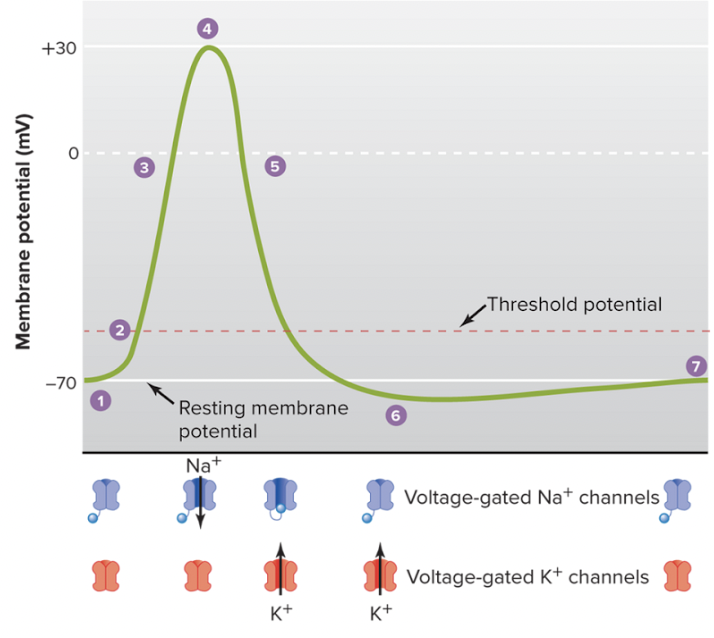
Describe the phases of action potential?
resting membrane potential (-70 mV)
stimulus causes membrane to reach threshold voltage (-50 mV)
depolarization: voltage-gated Na+ channels open causing influx of Na+ into the cell; membrane potential becomes more positive
at +30 mV: inactivation of Na+ channels & delayed opening of voltage-gated K+ channels
repolarization: voltage-gated K+ channel open causing K+ to leave the cell; helps membrane return towards -70mV resting potential
hyperpolarization (-80 mV): delayed closing of K+ channel causes membrane to be below resting membrane potential
voltage-gaated K+ channel closes & resting membrane potential is restored
what is the all-or-none principle?
applies to action potential
any stimulus that is strong enough to meet threshold potential will generate same strength action potential
strength of stimulus does not affect strength of action potential
what is the refractionary period?
refractionary period: regulates # of action potentials that can occur during repolarization & hyperpolarization
absolute refractionary period: NO action potential can occur during refractionary period bc Na+ channel is inactivated
relative refractionary period: action potential can occur during hyperpolarization if strong enough to reach threshold bc Na+ channels are closed, not inactivated
how is an action potential self-propagating?
local depolarization from Na+ influx at one segment of the membrane triggers depolarization in the adjacent Na+ channels of the next segment
refractory period ensures action potential moving in one direction
myelination allows depolarizing current to travel longer distance and speeds up conduction
what is myelin?
insulator on axons that allows for fast propagation of action potential
insulation stops leakages of ions → causes longer effective distance of ions to activate next segment of Na+ channels (aka longer depolarization)
formed by oligodendrocytes (CNS) and schwann cells (PNS)
what is salutatory conduction?
axon potentials appears to jump from node to node due to Na+ channels located there
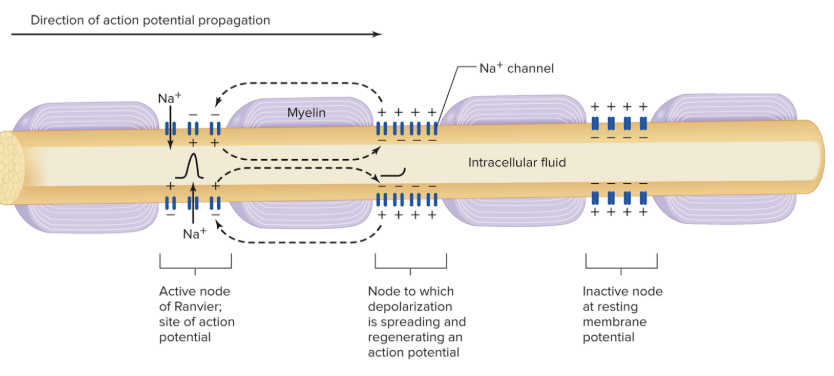
what are graded potentials? what are the key characteristics?
changes in membrane potential that are confined to relatively small region of plasma membrane (short-distance signaling)
direction: stimulus can cause depolarization or hyperpolarization
strength of stimulus affects intensity of graded potential
short distance propagation from leaky membrane + unmyelinated axon
summation of graded potentials can lead to action potentials
what are the types of graded potential summations?
temporal summation: consecutive depolarization events occurring at same location but diff times before neuron is able to return to resting membrane potential
spatial summation: depolarization events simultaneously occurring at diff locations but at same time
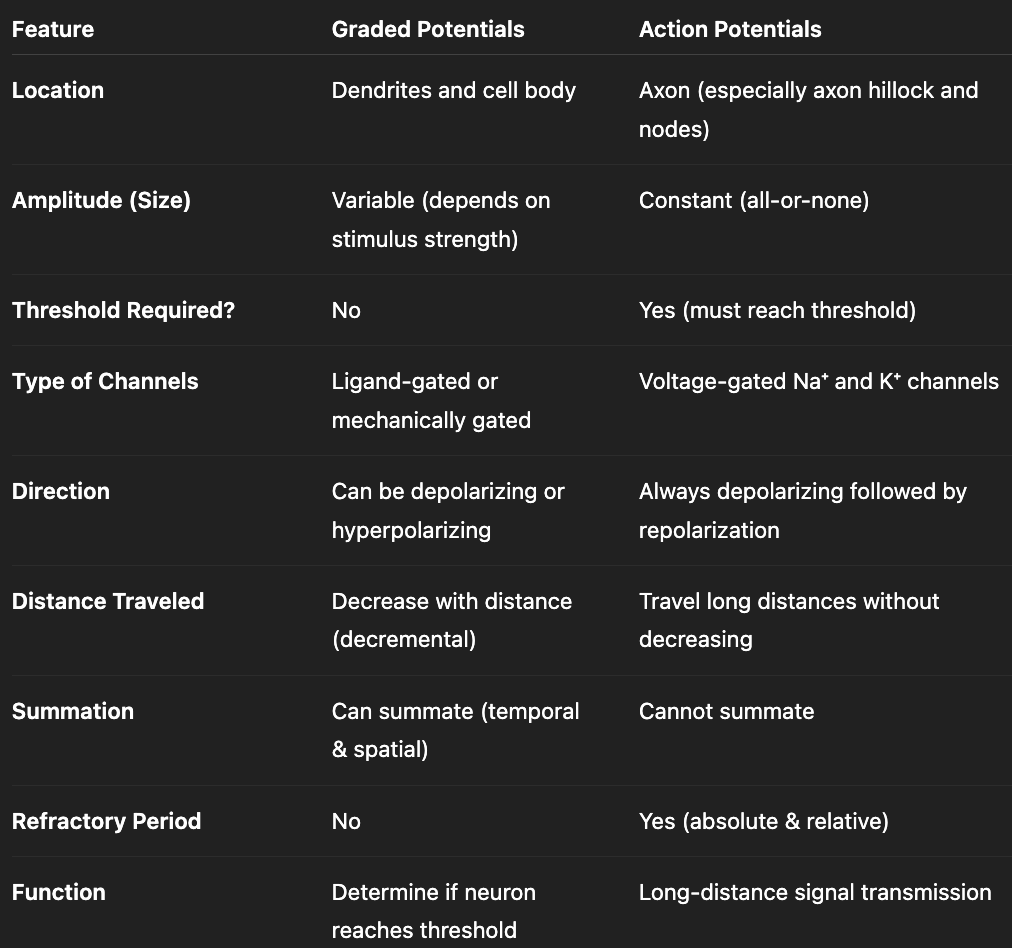
compare graded and action potentials
what is length constant? why might they differ between neurons?
distance that ions travel until their voltage reaches 37% of original value
increase in axon diameter = increase length constant
myelination = increase length constant
what is neuropathy? what are examples?
damage or dysfunction of nerves that impairs their ability to transmit signals properly
ex: multiple sclerosis, guillain-barre syndrome, lead poisoning
what is multiple sclerosis (MS)
chronic autoimmune disease where immune system destroys myelin sheath in CNS
what is Guillain-Barre Syndrome?
autoimmune disorder where immune system destroys myelin sheath of peripheral nerves (PNS)
triggered by virus
results in paralysis
can be reversible
what is lead poisoning?
causes demyelination and ion channel dysfunction
what is a synapse?
refers to region spanning from axon button (in presynaptic neuron) to dendrite (in postsynaptic neuron)
what is the synaptic cleft?
space between pre and post synaptic neuron
what is the difference between excitatory and inhibitory synapse?
excitatory: bring membrane potential closer to threshold
inhibitory: bring membrane potential further from threshold
what is the difference between chemical and electrical synapse?
chemical: release of neurotransmitters to synaptic cleft; delayed depolarization
electrical: plasma membrane of pre and post synaptic neurons are joined by gap junctions; allows for local currents to flow directly across junction → rapid communication, in-sync depolarization
What are the steps from action potential to neurotransmitter release?
action potential reaches presynaptic terminal
depolarization opens voltage-gated Ca2+ channels
influx of Ca2+ in presynaptic terminal
Ca2+ directly activates synaptotagmin (protein involved in neurotransmitter release) → activates SNARE protein → leads to vesicle docking and fusion
exocytosis—vesicle releases neurotransmitter into synaptic cleft
neurotransmitter diffusion across synaptic cleft and binds to receptors on postsynaptic membrane
postsynaptic potential generated
termination of signal: reuptake/diffusion/inactivation
Limitations on the maximum action potential firing frequency of neurons are determined by ____.
length of refractory periods
ionotropic vs. metabotropic postsynaptic receptors
ionotropic: opening ion channel
metabotropic: G-protein coupled receptor that results in metabolic processes
how does botulism/botox work?
destroys SNARE protein → no vesicle docking + fusion → no acetylcholine release from motor neuron → no muscle action potential → no DHP activation → no Ca2+ release from SR → no contraction
results in muscle weakness/paralysis
How can medication manipulate neurotransmitter regulation?
neurotransmitter synthesis
storage
release
agonist/antagonist
reuptake
neurotransmitter breakdown
How can neurotransmitter concentration be regulated in a synaptic cleft?
reuptake
enzymatic degradation
diffusion
what is an adequate stimulus?
preferred stimulus that sensory receptor is most sensitive to
requires least amount of energy to activate receptor
what is the 3 step process of the somatosensory system?
reception: receptor detects stimulus
transduction: convert stimulus into signal (aka receptor potential)
perception: process + interpret signal in brain
what is a receptor potential?
graded potential that occurs in sensory receptors in response to stimulus
converts stimulus into electrical signal
can trigger action potential if reaches action potential
what is the difference between sensation and perception?
sensation: sensory info that does not reach consciousness
perception: person’s awareness of sensation (interpretation)
what is modality?
definition: type of sensation encoded by a receptor; corresponds to the type of stimulus energy it is specialized to detect
mechanoreception: touch, pressure, vibration
thermoreception (temp): heat, cold
nociception (pain): injury, extreme temp
chemoreception: smell, taste
photoreception (vision): light, color
how does signal transduction relate to modality?
diff signal transduction mechanisms lead to different modalities bc different pathways are activated
What are the two ways that receptor potentials can activate action potentials
direct activation—depolarization from receptor potential triggers action potential in same neuron (e.g., mechanoreceptors, nociceptors, thermoreceptors)
indirect activation—receptor potential triggers neurotransmitter release to initiate action potential in separate neuron (e.g., photoreceptors, chemoreceptors)
what is adaptation? what are the two types of adaptors?
adaptation: diminishing activity in response to repeated or sustained stimuli
phasic receptors: fast-adapting; burst of action potential when stimulus is applied and removed; receptor potential does not persist when stimulus is applied
e.g., slight touch (clothes on body), slight temperature (stepping in shower), smell
tonic receptors: slow-adapting; sustained signaling
e.g., pain, vision
what is acuity?
degree of sharpness/precision to which you can sense a stimulus
how does two-point discrimination demonstrate acuity?
two-point discrimination: test that determines minimum distance at which a person can perceive two simultaneous touches as separate points instead of one.
tests how well the nervous system can distinguish closely spaced stimuli
How does receptive field size impact acuity?
receptive field: area of skin that activates a single sensory neuron
smaller receptive field = neuron covers small area → stimuli activate diff neurons → high acuity
large receptive field = neuron covers large area → stimuli activate same neuron → brain perceives as one stimulus → low acuity
How does dendrite density impact acuity?
high dendrite density (receptors) → high acuity
low dendrite density (receptors) → low acuity
what does localization of a stimulus depend on?
size of receptive field
degree of receptive field overlap
what is receptive field overlap?
definition: when neighboring sensory neurons cover some of the same area of sensory space → single stimulus can activate more than one sensory neuron
**helps localize stimulus by comparing neuron firing activity
what is lateral inhibition?
definition: when neuron directly under the stimulus fires strongly and suppresses neighboring weaker responses; leads to increasing contrast between signals, which improves localization/acuity
how does rate of adaptation indirectly influence acuity?
tonic receptors (slow adaptation) → sustained signal → increase acuity
how does myelination indirectly influence acuity?
myelination → sustained signal → increase acuity
what is synesthesia? what are the two common types?
synesthesia: stimulating one sense triggers involuntary experiences in another; crossing in signaling pathways where modalities are perceived in different pathway
grapheme: letters/numbers have colors
chromesthesia: music and sound triggers color
what factors influence the intensity of stimulus?
recruitment of diff # of afferent neurons
frequency of firing
magnitude of receptor potentials
what is the somatosensory system?
detects and processes sensations arising from the body (soma)—including skin, muscles, joints, and internal tissues
allows body to perceive touch, pressure, vibration, temperature, pain, and body position (propioception)
what is the difference between S1 and S2?
S1 (primary somatosensory cortex): bulk of info; perceives sensation in the moment
S2 (secondary somatosensory cortex): processes memory of sensation
what is homunculus?
distorted miniature representation of the human body used to map sensory/motor cortex areas based on proportional brain dedication
hands, mouth, and eyes are depicted large bc lots of sensory receptors
what are significant structures in spinal cord vertebrae?
ventral horn = larger wings
dorsal horn = smaller wings
ventral root
dorsal root
dorsal root ganglion
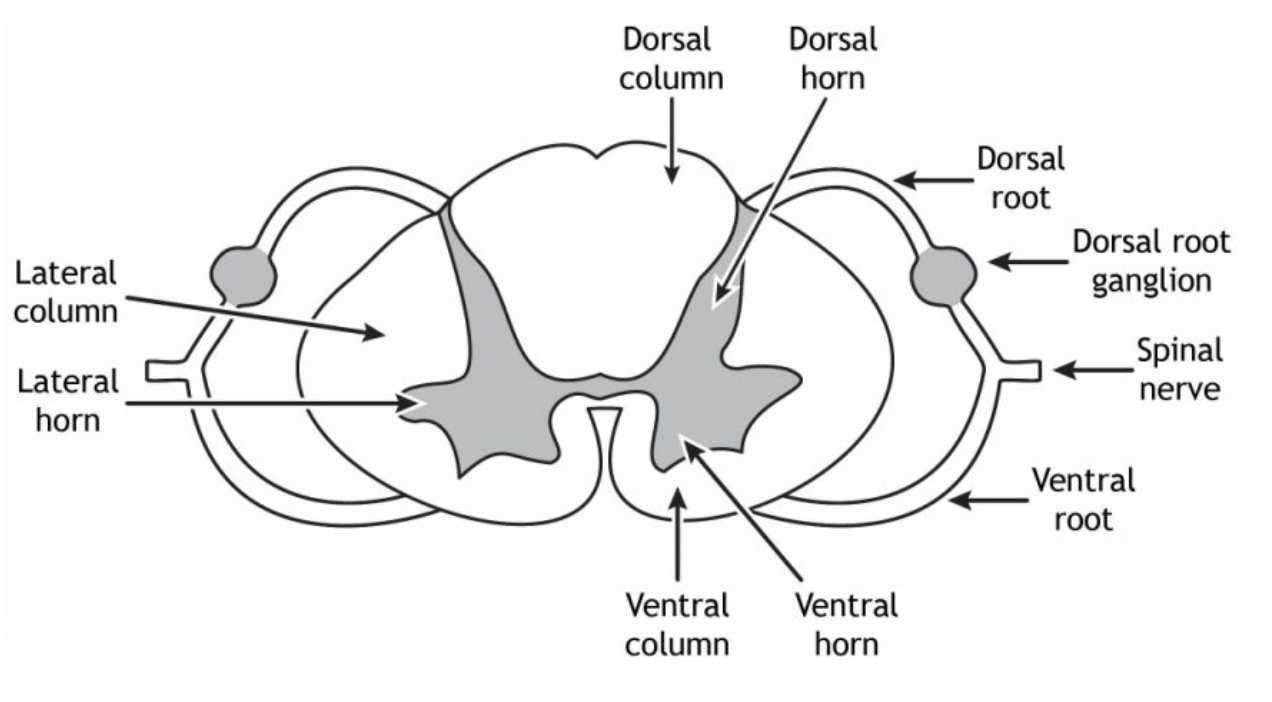
where do afferent signals enter?
dorsal root
where do motor/efferent signals exit?
ventral root
what is the dorsal root ganglion?
cluster of sensory neuron cell bodies
what is the pathway from stimulus to spinal cord?
Stimulus → Receptor potential → Action potential → Peripheral afferent fiber → Dorsal root ganglion (cell body) → dorsal root
what are the two types of somatosensory pathways? how do they differ?
ascending anterolateral pathway: immediately synapse in spinal cord → crossover in spinal cord → ascend to brain through anterolateral column (side) → ascends to thalamus → reaches somatosensory cortex
dorsal column pathway: ascend to brain through dorsal column (back) → synapse in brain → crossover in brainstem → ascends to thalamus → reaches somatosensory cortex
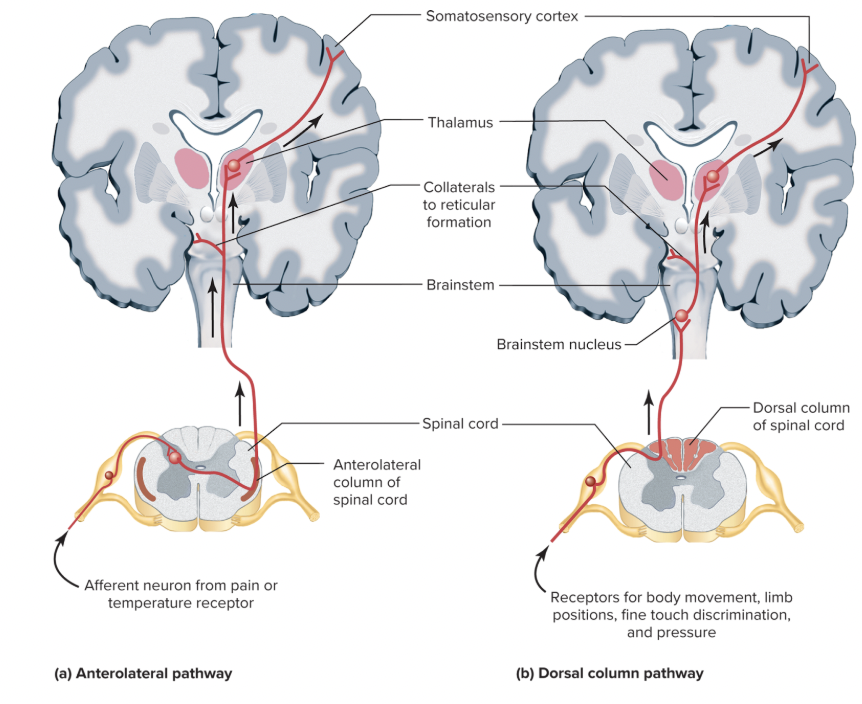
what is the full somatosensory pathway, from sensory to motor command?
Receptor → sensory neuron (afferent pathway) → dorsal root ganglion → dorsal root → synapse at dorsal horn → ascends to brain → thalamus (relay center) → somatosensory cortex → motor cortex (generates motor response) → motor command travels down descending motor tract → motor neuron synapses in ventral horn → exits spinal cord via ventral root → muscle contracts
what is the somatosensory reflex arc pathway
*does not require brain (e.g., knee jerk reflex)
Receptor → sensory neuron → dorsal root → dorsal horn → interneuron → motor neuron (ventral horn) → ventral root → muscle
what are dermatomes? how do they relate to pathologies?
areas of skin innervated by nerves in a segmented fashion; location of sensation is where the spinal cord will receive the signal
ex: shingles—dormant virus in dorsal root ganglion; rash follows form of dermatome
ex: spinal cord injury—loss of sensation in body can indicate which vertebrae has been injured
how do mechanoreceptors work?
direct activation due to lipid tension (membrane bilayer pulling receptor)
direct linkage via. intracellular and extracellular proteins (proteins pulling receptor)
indirect activation via. a system that activates the channel
how does threshold stimuli relate to sensitivity to touch?
low threshold = require little mechanical force to open mechanically-gated ion channels → high sensitivity (detect extremely subtle touch)
how does receptive field size influence sensitivity and acuity?
small receptive field size → high sensitivity + acuity
detects fine detail
large receptive field size → broad detection + lower acuity
usually detects pressure or vibration
how does adaptation affect touch?
phasic (rapidly adapting) → movement, texture, vibration
tonic (slowly adapting) → grip control, maintain continuous awareness of objects in hand
what are nociceptors?
pain receptors that respond to tissue damage, chemical, mechanical or thermal stimulation
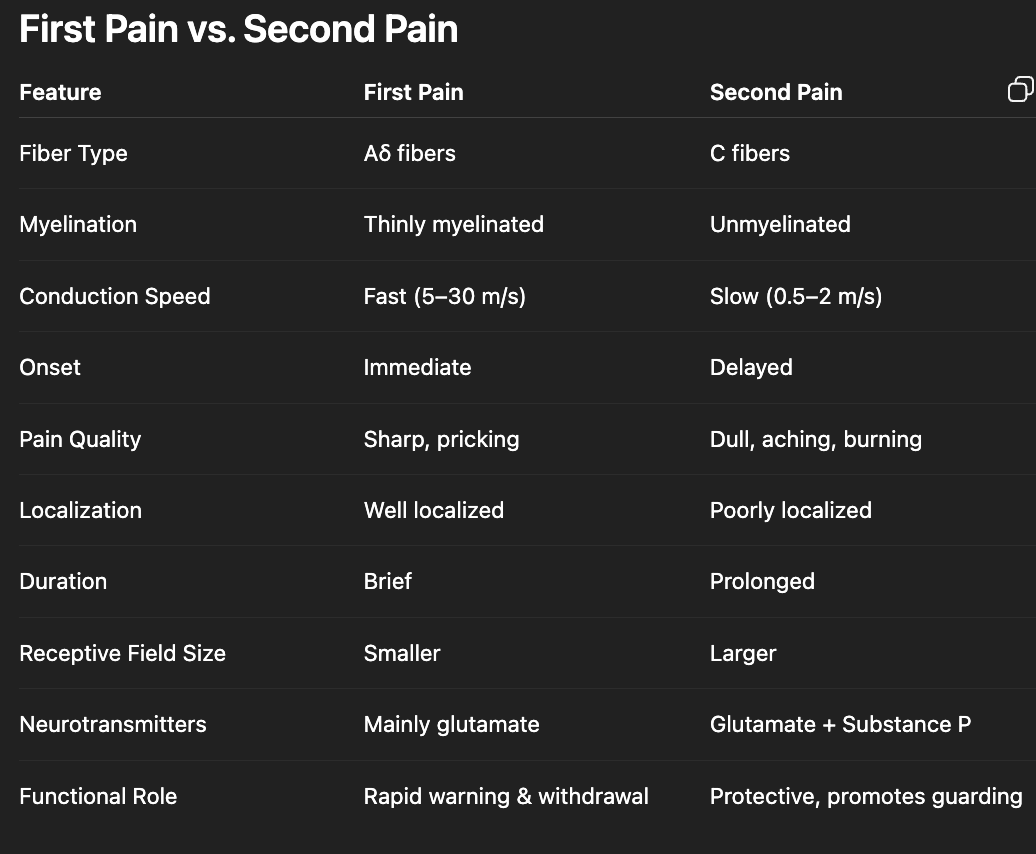
what are the types of nociceptor fibers?
Aδ (delta) fibers:
light myelinated, fast conducting →first pain
smaller receptive field size → increased acuity → sharp, localized pain
higher intensity pain for shorter period of time
polymodal nociceptors (C fibers):
unmyelinated, slow conducting → second pain (dull, aching, throbbing, nonlocalized pain)
lower intensity pain for prolonged time
what is unique about sleeping nociceptors?
activated by chemicals released during tissue damage
located on C fibers (slow, chronic, dull second pain)
ex: washing hand under water w/ cut
what neurotransmitters are involved in nociception?
glutamate: primary fast transmitter; released from Aδ and C fibers
substance P (neuropeptide): produce slower, prolonged excitation for chronic pain signaling; released mainly from C fibers
what are A-alpha fibers
associated with tonic (slow adaptation) in muscle
what are A-beta fibers?
associated with fast adaptation (phasic) in touch
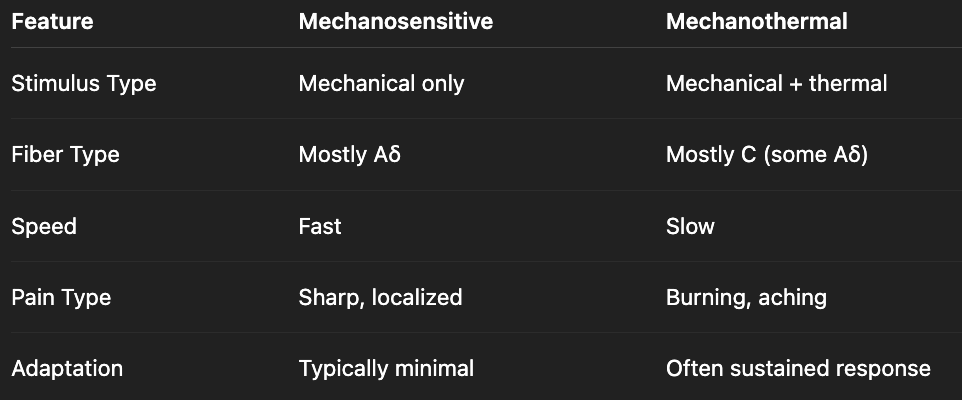
what is the difference between mechanosensitive and mechanothermal receptors?
what is the difference between anesthesia and analgesics?
anesthesia: blocks all sensations
analgesic: blocks pain signaling
what are the subcategories of anesthesia?
general anesthesia: induces total unconcsciousness; affects entire body
local anesthesia: numbs specific small area
what are common forms of analgesics?
opiods/oppiates
cox inhibitors
what is hyperalgesia vs. hypoalgesia?
hyperalgesia: increases pain sensitivity
hypoalgesia: decreases pain sensitivity
what is the difference between first and second pain? how is it coded?
Describe the pain transduction pathway and how it relates to perception of pain.
Tissue injury → releases chemical signals (e.g., prostaglandins, histamine)
Nociceptor activation
Aδ and C fibers transmit signal
Synapse in dorsal horn
Cross in spinal cord
Ascend via anterolateral column
Thalamus relay
Cortex processes → conscious pain
How is hypoalgesia naturally activated?
stress → release serotonin + norepinephrine → releases opioids (e.g., beta-endorphins) → prevents release or binding of pain neurotransmitters from nociceptors (glutamate and substance P)
How is hyperalgesia naturally activated?
after tissue injury, inflammatory mediators are released (e.g., prostaglandins, histamine, cytokines) which lowers activation threshold of nociceptors → nociceptors fire more easily?
why does opioid addiction occur?
opioids bind to opiate receptors and inactivate GABA-ergic neuron → inhibits GABA release → allows dopamine release
what is referred pain?
Pain that is perceived at a location different from the actual site of tissue injury.
can occur due to convergence at the same spinal cord segment or shared neurons
ex: left arm pain during heart attack
what are eicosanoids?
lipid signaling molecules desrived from arachidonic acid that regulate inflammation, pain, immune responses, blood clotting
(e.g., prostaglandins, thromboxanes, leukotrienes)
what is the COX-1 pathway in nociception?
tissue injury releases inflammatory mediators (e.g., histamine, cytokines)
inflammatory mediators bind to GPCR on nociceptor
Gq activates PLC → cleaves PIP2 to IP3 + DAG → IP3 increases Ca2+, DAG activates PKC
activation of PLA2 causes release of arachidonic acid
COX-1 converts arachidonic acid into prostaglandins & thromboxanes
sensitizes nociceptors & lowers activation threshold, leading to increased excitability and hyperalgesia
Compare COX-1 and COX-2
