AP Bio Unit 1 Test
1/115
Earn XP
Description and Tags
Chapters 2-5
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
116 Terms
Acid
Substance that lowers the pH of a solution by releasing H+ ions
Anion
A negatively charged ion, forms ionic bonds with cations
Aqueous solution
Solution that can be dissolved in water
Atom
Smallest unit of an element that cannot be broken down into smaller parts
Atomic nucleus
Center of an atom, holds most of the mass, where protons and neutrons are found
Atomic number
Number of protons in nucleus of an atom
Base
A substance that raises the pH by reducing the H+ concentration of a solution
Capillary action
When a liquid “climbs” up a tube, caused by a balance of cohesive and adhesive forces in the liquid
Cation
Positive ion, forms ionic bonds with anions
Compound
Fixed ratio of two or more elements that make up a substance
Dissociation
Molecules break down into ions (water into H3O+ and OH-)
Electron
Negatively charged subatomic particle
Element
Material that cannot be broken down further by chemical reactions
Heat of vaporization
Heat at which a substance turns from a liquid to a gas
Hydration shell
Sphere of water molecules that surround molecules of a solute when dissolved in water
Hydrogen bond
Bond formed between two atoms from two different polar molecules with a partial positive or negative charge
Hydrogen ion
H+, released by acids, lowers the pH of a solution
Hydrophilic
Polar substance with an affinity for water
Hydrophobic
Nonpolar substance that HATES water
Hydrophobic interactions
When nonpolar molecules cluster together instead of dissolving in water, this is due to them disrupting hydrogen bonds and water molecules trying to push them out of the way
Hydroxide ion
OH-, released by bases, raises the pH of a solution
Inorganic
Substances that are not organic, do not contain carbon hydrogen bonds
Ion
At atom of the same element with a different number of electrons, giving it a positive (cation) or negative (anion) charge
Ionic bond
A bond between a cation and an anion where electrons are transferred from one ion to the other
Ionization
The process of an atom becoming an ion by gaining or losing electrons
Isotope
An atom of the same element with a different number of neutrons; radioactive isotopes decay and can be used in the medical field for imaging
Mass number
Number of protons and neutrons in an atom
Matter
Something that takes up space and has mass
Molecular formula
Shows the exact amount of atoms that make up a molecule
Molecule
Two or more atoms bonded with each other
Neutron
Subatomic particle with no charge, contributes to the weight of an atom
Nonpolar covalent bond
Bond where electrons are shared equally because of similar or equal electronegativity in the atoms
Octet rule
Atoms like to fill their outer shells with 8 electrons and will interact with other atoms to do this
Orbital
3D space where an electron can be found most of the time
pH
Measure of how acidic or basic a solution is, relates to the concentration of H+ ions
Polar covalent bond
Bond where electrons are shared but not equally because one atom is more electronegative
Proton
Subatomic particle with a positive charge
Radioactive tracer
When a molecule is replaced with a radioactive isotope so scientists can see the path it took previously (ex. Carbon-14)
Solute
Substance that is dissolved
Solvent
Substance that dissolves other substances
Specific heat
Amount of energy required to change the temperature of 1g of a substance by 1 degree Celsius
Structural formula
A chemical formula that shows the amount of atoms, their arrangement, and their bond structure in a molecule
Surface tension
Makes the surface of water feel like a solid, measures how difficult it is to break the surface of a liquid, due to cohesive forces of polar substances
Valence
Bond capacity of an atom
Valence electrons
Electrons in the outer shell of an atom, can be shared with or transferred to another atom
Ground state
Electrons in this state are closer to the nucleus and have less energy
Excited state
Electrons in this state are further from the nucleus and have more bonding potential. They are also less stable
Mixture
Two or more substances that are physically combined but not chemically bonded
Hydroxyl formula
-OH
Hydroxyl class name
Alcohol
Hydroxyl characteristics
Polar, forms hydrogen bonds with water
Carboxyl formula
-COOH
Carboxyl class name
Carboxylic acid
Carboxyl characteristics
Acts as an acid, polar, hydrophilic
Amino formula
-NH2
Amino class name
Amine
Amino characteristics
Acts as a base, polar, hydrophilic
Phosphate formula
PO4-2
Phosphate class name
Organic phosphate
Phosphate characteristics
Polar, hydrophilic, releases energy when reacting with water
Carbonyl formula
C = O
Carbonyl class name (found at the end)
Aldehyde
Carbonyl class name (found in the middle)
Ketone
Carbonyl characteristics
Associated with sugars, polar, hydrophilic
Methyl formula
-CH3
Methyl class name
Methylated compound
Methyl characteristics
Nonpolar, affects gene expression, affects shape and function of sex hormones
Sulfhydryl formula
-SH
Sulfhydryl class name
Thiol
Sulfhydryl characteristics
Two react to stabilize protein structures
Carbohydrate elements
CHO
Carbohydrate function
Short term energy storage, structure
Carbohydrate monomer
Monosaccharide
Carbohydrate polymer
Polysaccharide
Carbohydrate examples
Glycogen, glucose, lactose
Carbohydrate functional group
-OH hydroxyl
Lipid elements
CHO
Lipid function
Long term energy storage, insulation and protection, make up cell membranes and hormones
Lipid monomer
Fatty acids
Lipid polymer
Tricylglycerol
Lipid examples
Fats, oils, waxes, steroids, phospholipids
Lipid functional groups
-COOH carboxyl and -OH hydroxyl
Protein elements
CHON(S)
Protein function
enzyme for catalysis, provides structural support, aids in movement and cellular communication
Protein monomer
Amino acids
Protein polymer
Polypeptides
Protein examples
Amylase, pepsin, hemoglobin, keratin, insulin
Protein functional groups
-NH2 amino, -COOH carboxyl
Nucleic acid elements
CHONP
Nucleic acid monomer
Nucleotides
Nucleic acid polymer
Polynucleotides
Nucleic acid examples
DNA, RNA
Nucleic acid functional groups
PO4-2 phosphates
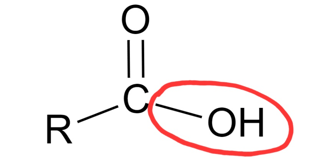
What is this?
Hydroxyl
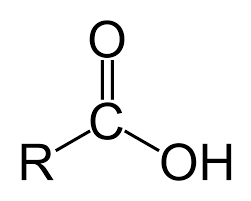
What is this?
Carboxyl
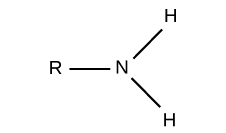
What is this?
Amino
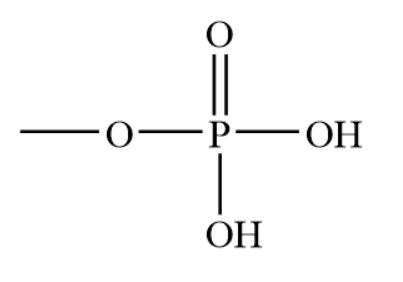
What is this?
Phosphate
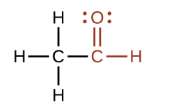
What is this?
Carbonyl (Aldehyde)
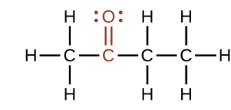
What is this?
Carbonyl (Ketone)
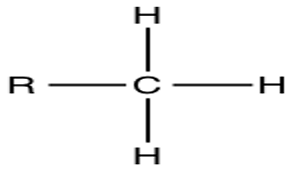
What is this?
Methyl