Nutri Sci 10S 2nd Midterm
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
4 absorptive processes for Nutrient Absorption
Passive diffusion, facilitated diffusion, active absorption, endocytosis
Protein Structure
Amino Acids are bounded by peptide bonds that form polypeptides which fold to form 3 dimensional shapes and make proteins
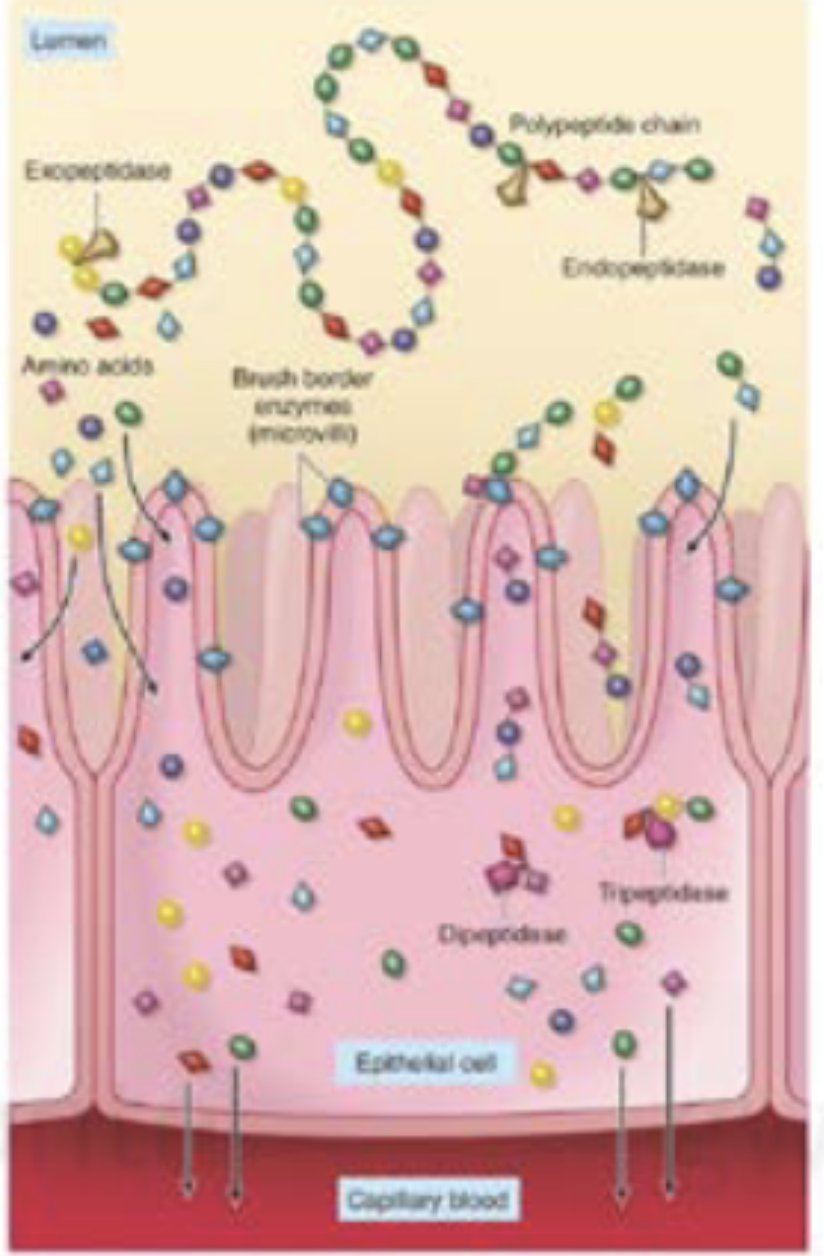
Enzyme Functions of Trypsin and Salivary Amylase: Where is it located?
Salivary amylase - located in the mouth and breaks down starch into shorter chains of glucose.
Trypsin - Located in the pancreas which breaks proteins and polypeptides into shorter polypeptides.
The Digestion and Absorption of Proteins
The Central Dogma
Information in an organism, that decides and organism, and describes everything that it can do, is encoded in the DNA.
DNA is then copied into RNA, which is very similar, it has exactly the same sequence as DNA, but it can easily be changed,
From the information in the RNA, you can then make protein by reading the sequence of bases in the RNA three at a time and three code for one amino acid.
The Central Dogma
Information from Nucleic acids cannot be back-Engineered. Where Nucleic acids can be turned into proteins
Steps of Protein Synthesis: Transcription and Translation
In the nucleus the blueprint, or code, for the protein is copied or transcribed from the DNA gene into a molecule of messenger RNA (mRNA).
The mRNA takes the genetic information from the nucleus to structures called ribosomes in the cytosol, where proteins are made.
In the cytosol, transfer RNA (tRNA) reads the genetic code and delivers the needed amino acids to the ribosome to form a polypeptide chain.
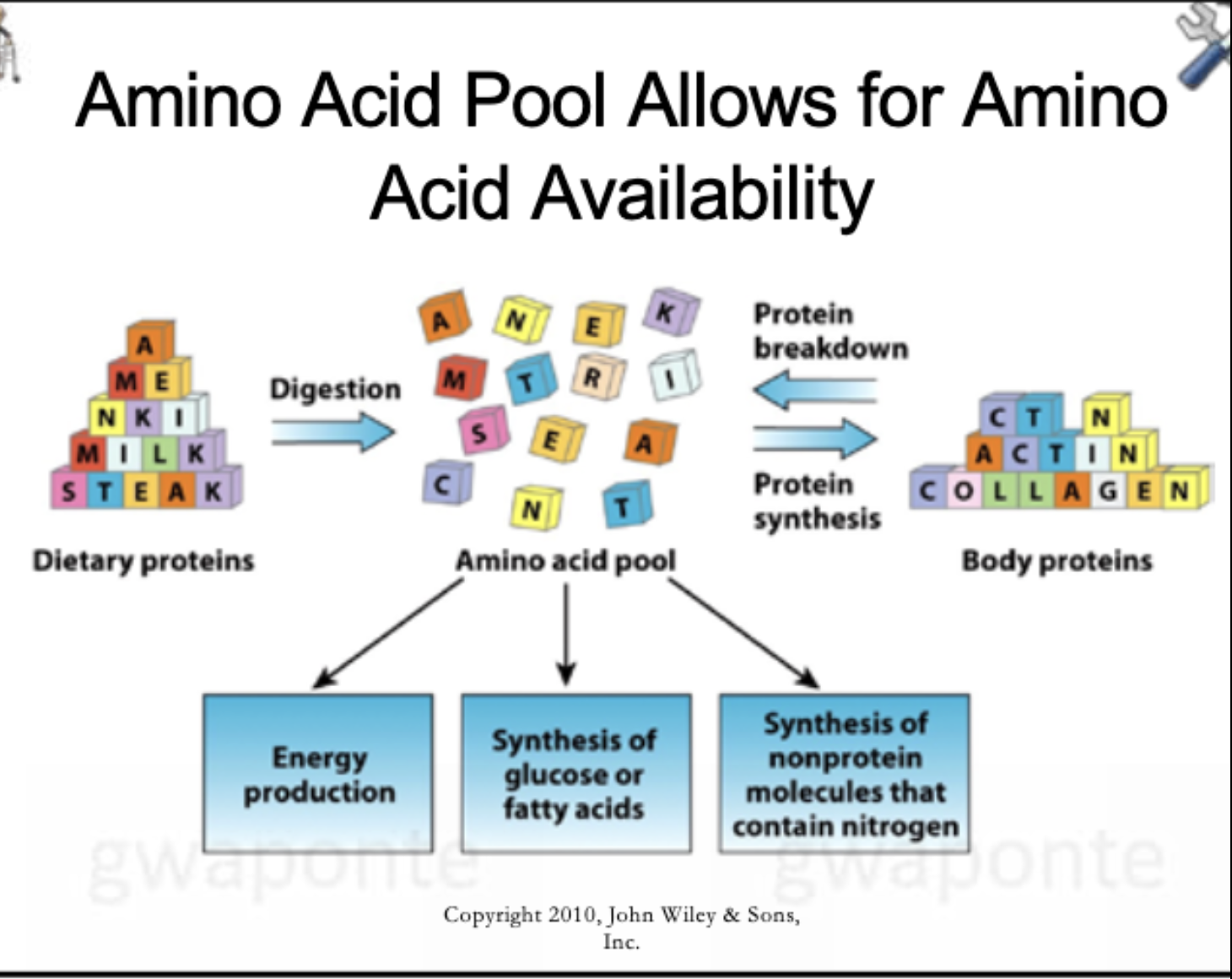
Amino Acid Pool Allows for Amino Acid Availability
Limiting Amino Acids
Our bodies use amino acids in a specific ratio to each other, so if a person doesn’t get enough of one of them to match with the rest, the rest can only be used at a level to balance with that low one. The amino acid that is deficient in the diet is the limiting amino acid.
What is an Amino Acid Pool?
An accumulation of amino acids in the liver and blood that adjusts to meet the body’s need for protein and amino acids.
Key Cellular Organelles
Rough endoplasmic reticulum, smooth endoplasmic reticulum, Nucleus, Ribosomes
Inter-relationship between Metabolism
Metabolism is related to physiology and behavior
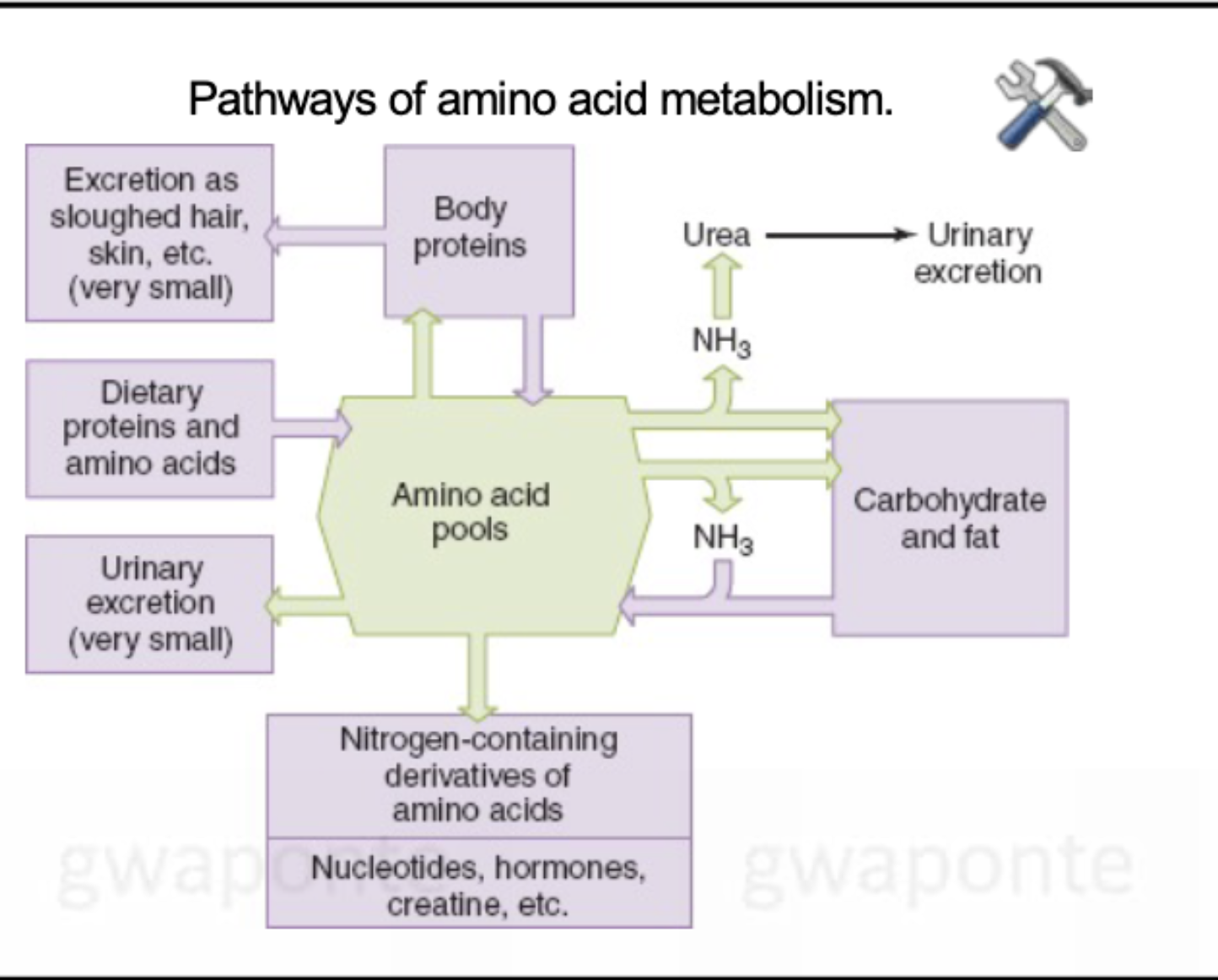
Pathways of amino acid metabolism
Amino Acids: Transamination
When a nonessential amino acid is not available from the diet, it can be made in the body by the process of transamination.
Deaminoation and Synthesis of a Nonessential Amino Acid
The deamination of an amino acid produces ammonia (NH3) and a keto acid.
Given a source of NH3 the body can make nonessential amino acids from keto acids.
Energy Production from Protein
Amino group is removed by deamination.
Deamination of some amino acids produces three-carbon molecules that can be used to synthesize glucose, via gluconeogenesis
Deamination of some amino acids results in two-carbom molecules that form acetyl-CoA, which can enter the citric acid cycle or be used to synthesize fatty acids.
High-energy electrons from the breakdown of amino acids are transferred to the electron transport chain where energy is trapped and used to produce ATP and water
Urea Synthesis (using CO2)
Amino acids are deaminated before they can be metabolized to produce ATP or used the synthesize glucose or fat.
The amino group forms toxic ammonia
Ammonia is converted into urea in the liber.
Urea can safely travel in the blood and is filtered out of the blood by the kidney and eliminated from the body in the urine.
Nitrogen Balance
B = I - (U + F + S)
B = Nitrogen balance
I = nitrogen intake
U = urinary nitrogen
F = fecal nitrogen
S = loss of nitrogen through the skin
Proteins in the Diet
Recommended protein intake
Adult RDA = 0.8 grams / kilogram body weight
Infant RDA = ~ 1.5 grams / kilogram body weight
Elderly 1.0 to 1.2 g/kg
Depends on quality of protein
3 Steps of Nitrogen Balance
Positive Nitrogen Balance, Nitrogen Equilibrium, Negative Nitrogen Balance
Positive Nitrogen Balance
Situations when positive nitrogen balance occurs: Growth, pregnancy, recovery stage after illness/injury, athletic training resulting in increased lean body mass, increased secretion of certain hormones, like insulin, growth hormone, and testoterone.
Nitrogen Equilibrium
Situation when nitrogen equilibrium occurs: Healthy adults meeting protein and energy needs.
Negative Nitrogen Balance
Situations when negative nitrogen balance occurs: Inadequate intake of protein, inadequate energy intake, conditions like fevers, burns, and infections, bed rest (for several days), deficiency of essential amino acids, increased protein loss, increased secretion of certain hormones.
Protein Nitrogen Balance
A pregnant woman adds proteins so she has a positive nitrogen balance
A healthy person who is neither gianing nor losing nitrogen is in nitrogen equilibrium
A person who is severely ill and losing protein has a negative nitrogen balance.
How much does nitrogen make up in the body?
makes up approximately 16% of the weight of an amino acid. Therefore, nitrogen intake multiplied by 6.25 provides an estimate of protein intake. A crude estimate of the % protein content of food is obtained from N x 100/16 or N x 6.25
Estimates of Protein Quality in Digestibility
Apparent digestibility = N eaten - N fecal / N eaten x 100
Protein Efficiency Ratio
PER (protein efficiency ratio) = live weight gain / crude protein intake. If a particular diet does not support growth, then a PER of zero will be recorded (zero weight gain / any amount of protein intake = zero)
Factors that affect protein quality
Digestibility
Amino acid content
Toxins
Form of amino acids
How do you measure quality of a protein?
Isocaloric diets - Test diet and control diets have the same caloric density
Pair feeding - The caloric intake between subjects on the control and those subjects on the test diet are equal.
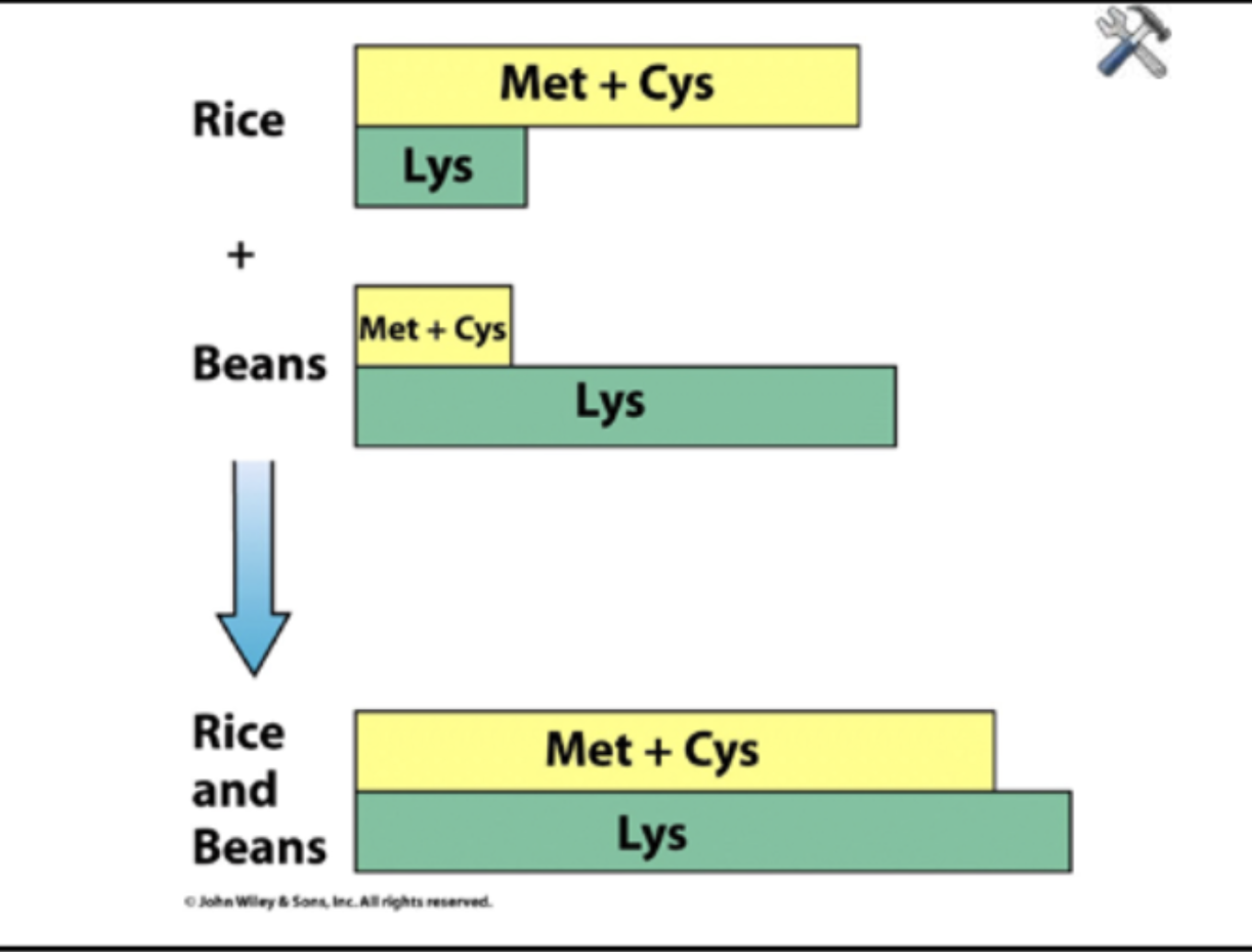
Protein complementation
Combining plant protein sources to achieve a better amino acid balance than each would have alone. Like legumes: peas, beans, lentils, peanuts, alfalfa. Grains: wheat, rice, corn, oats, rye
Golden Rule in Lecture 8
What you eat doesn’t enter your body unless it is biologically available
Protein Efficiency Ratio
PER (protein efficiency ratio) = live weight gain / crude protein intake.
If a particular diet does not support growth, then a PER of zero will be recorded (zero weight gain/any amount of protein intake = zero)
Golden Tool
What you eat doesn’t enter your body unless it is biologically available
Proteins that May Harm Certain Individuals Phenlyketonuria
PKU is an inherited condition attributed to a defective gene.
Aspartame, a suger substitute, contains phenylalanine.
Protein Deficiency
Protein-energy malnutrition (PEM) is used to refer to the continuum of protein deficiency conditions ranging from mostly protein deficiency, called kwahiorkor, to overall energy deficiency called marasmus. Most protein-energy malnutrition is a combination of the two.
Protein-Energy Malnutrition Kwashiorkor and Marasmus
Kwashiorkor - Severe protein deficit often accompanied by infections or other diseases. Characteristics: Mild to moderate weight loss, maintenance of some muscle and subconscious fat, growth impairment, rapid onset, fatty liver.
Marasmus - Severe energy and protein deficient. Characteristics: Severe weight loss, wasting of muscle and body fat, severe growth impairment, develops gradually.
Pros and Cons of Vegetarian Eating
Types of vegetarian diets: semi-vegetarian, lacto-ovo vegetarian, vegan
Health benefits vs. health risks: Less fat, saturated fat, and cholesterol, restrictive diets may lack nutrients, careful planning needed for children, pregnant women.
Nutrients Supplied by Plant and Animal Foods
Animal produces provide sources of protein, B vitamins and minerals such as iron, zinc and calcium. However, animal products are low in fiber and can be high in fat. Plant sources of protein are also a good source of B vitamins, iron, zinc, fiber and calcium, but in less absorbable forms.
Food Protein Allergies
Food proteins (allergens) cause an immune response, creating an allergic reaction. IgE most commonly produced anaphylaxis. 8 foods account for 90% of all food allergies like peanuts, tree nuts, milk, eggs, fish, shellfish, soy, and wheat.
Too Much Protein
Heart disease, obesity, cancer, osteoporosis, protein and amino acids supplements (generally not needed with risks unknown)
Protein Excess: Hydration and Kidney Function
When protein intake increases above the amount needed, so does the production of protein breakdown products such as urea, which much be eliminated from the body by the kidneys. To do this, more water must be excreted in the urine, increasing water losses.
Protein Excess: Kidney Stones
The increase in urinary calcium excretion associated with high-protein diets has led to speculation that a high protein intake may increase the risk of kidney stones.
Protein Excess: Heart Disease and Cancer
High-protein diets is related to more to the dietary components that accompany animal versus plant proteins. Typically high protein diets are also high in animal products; this dietary pattern is high in saturated fat and cholesterol and low in fiber.
Celiac Disease
Celiac disease, more casually referred to as wheat or gluten intolerance, found in wheat, rye, and barley.
Photosynthesis
Energy from the sun

Carbohydrates
Macronutrient - 4 kcal/g
Carbon, oxygen, hydrogen - carbon-hydrate: C + H2O, CHO
Glucose: C6H12O6
Carbohydrates functions
Provides energy, spares protein, Provides sweetness, Provides fiber, enhances functions of many proteins
Carbohydrate Types
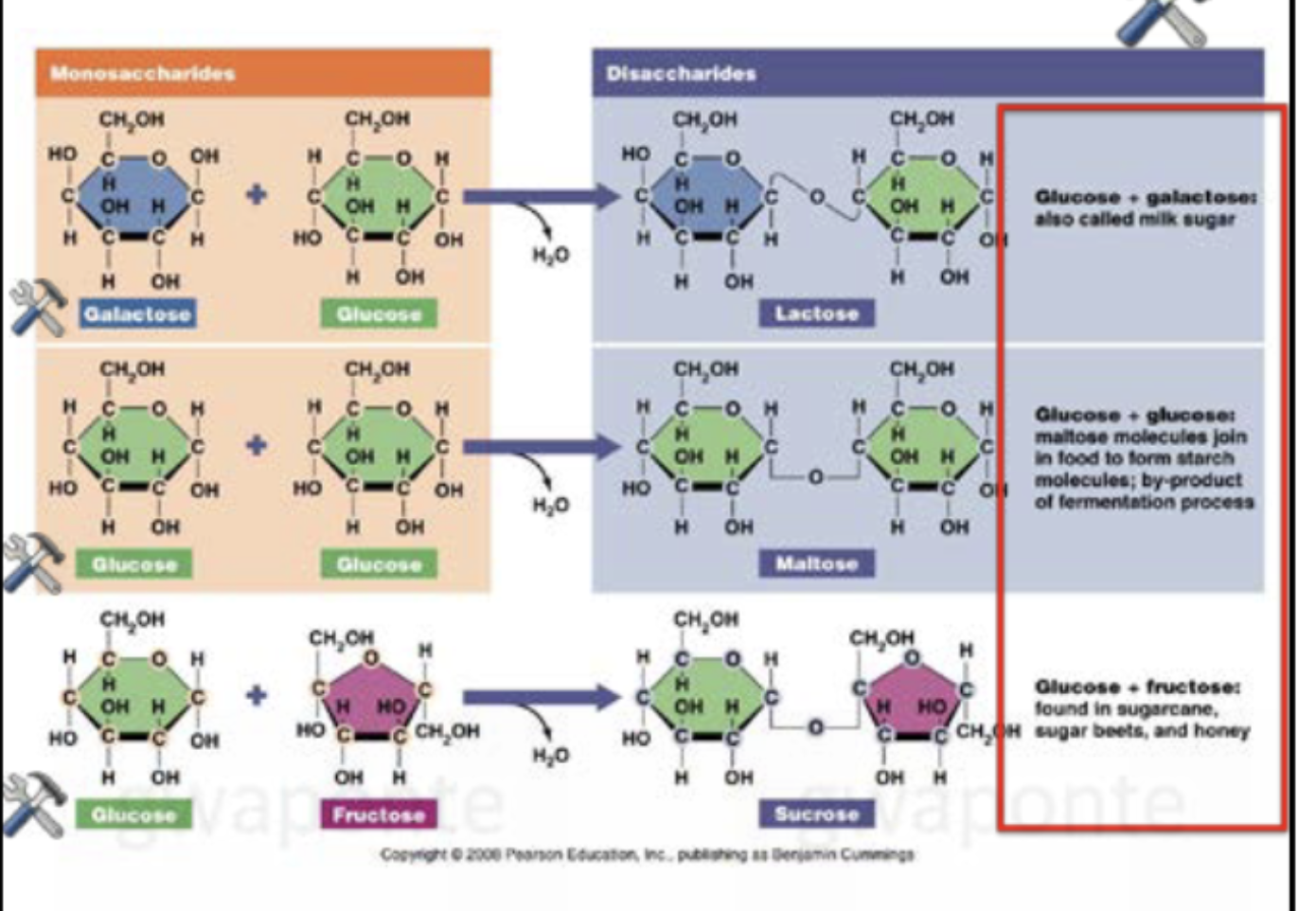
Simple: Monosaccharides (glucose, fructose), disaccharides (sugar and glucose + fructose)
Complex: (3 or more monosaccharides), oligosaccharides, polysaccharides (starch, fiber, glycogen)
Monosaccharides
Glucose, fructose, galactose
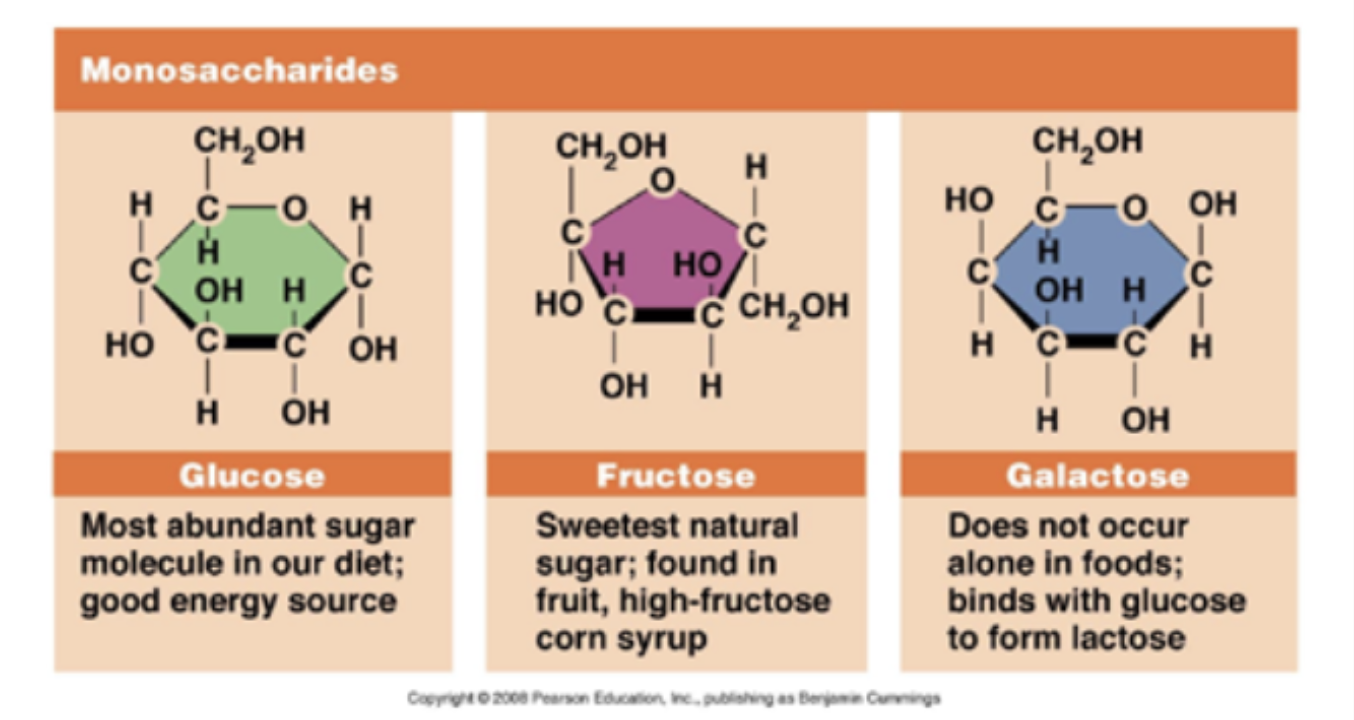
Structures of monosaccharides to Disaccharides
Lactose Intolerance
Not enough lactase in the SI to digest lactose
Symptoms: diarrhea, bloating, intestinal gas