Solid State 4thY
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Facets
Natural and flat on the atomic level
Pyroelectricity and piezoelectricity
Pyroelectricity (tourmaline)
Separation of electrical charges in a crystal by change of temperature
(Anisotropic charge only in certain directions, i.e. on certain crystal faces)
Piezoelectricity (quartz),
Generation of electrical charge in response to stress
Anisotropy of physical properties due to periodic structures
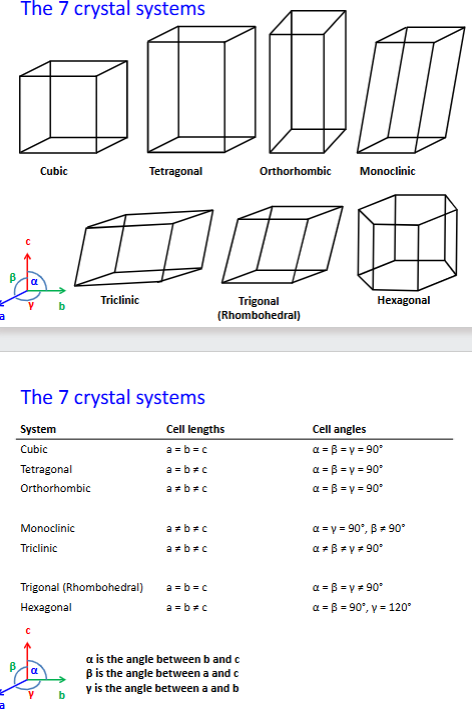
name and describe the 7 crystal systems
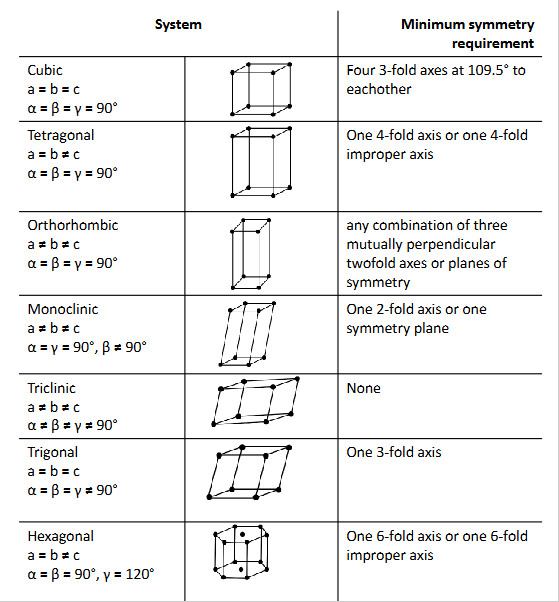
symmetry elements of each crystal system
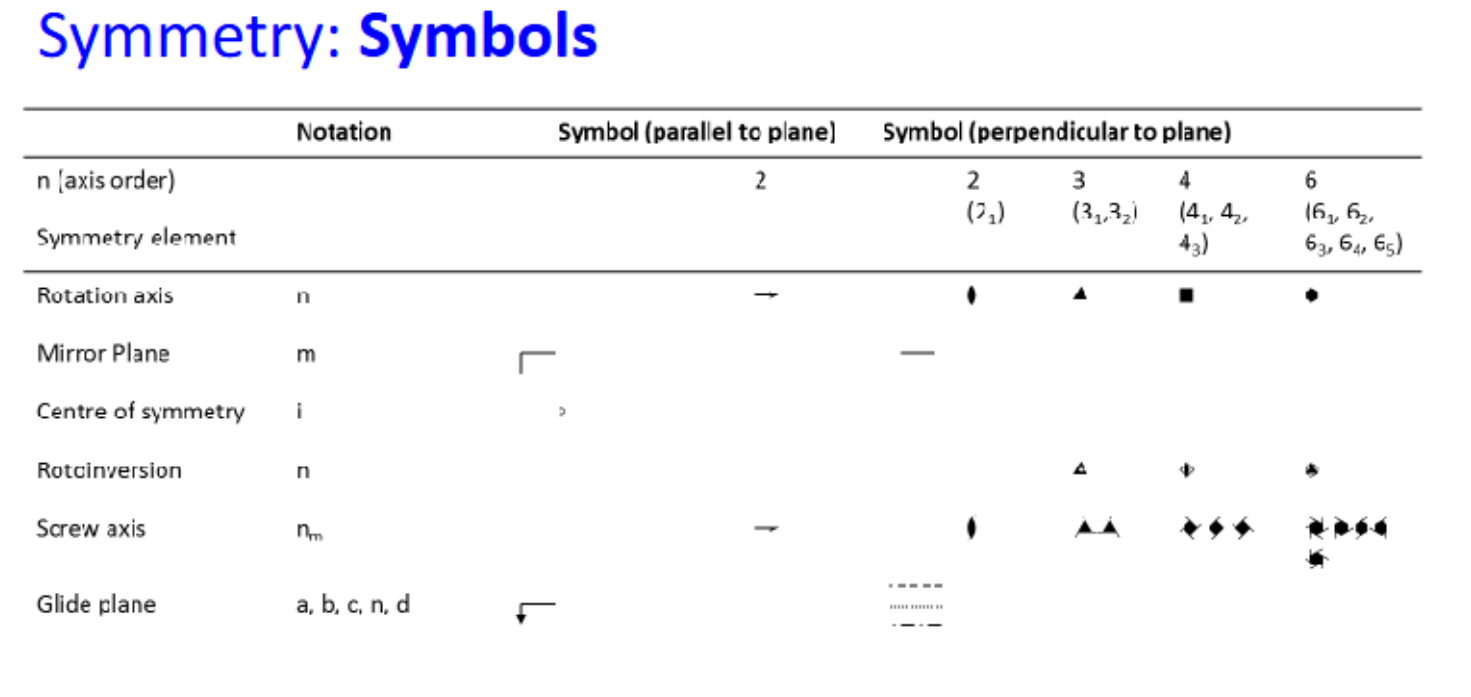
What is the restriction on the order of rotation
There is a restriction on n, the order of rotation
2, 3, 4, 6 ONLY (1 also but doesn’t really count)

notation for Schoenflies and Hermann-Mauguin



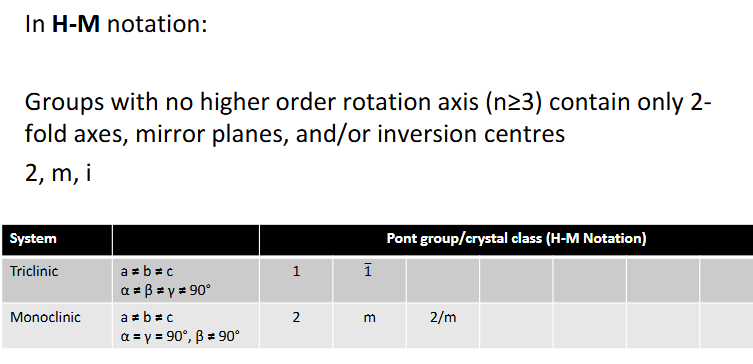

H-M notation





Face definition
A face is a flat surface which
bounds the crystal in a specific
direction
Edge definition
Two intersecting non-parallel
faces define an edge.
Corner definition
Three or more intersecting
non-parallel faces define a
corner
Zone definition
Three or more faces sharing
parallel edges define a zone
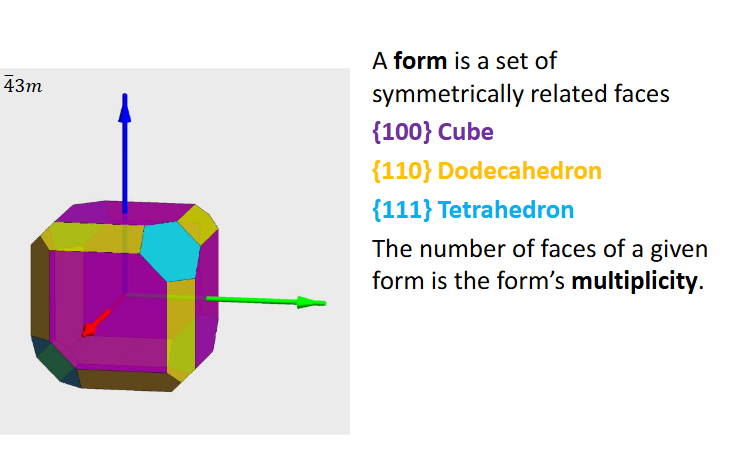
Form and multiplicity definition
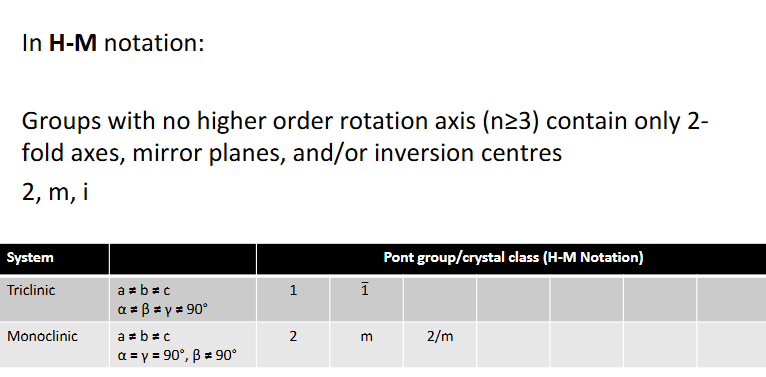
Groups with no higher order rotation axis (n≥3) contain what and what crystal structures are these?
For groups with only one higher order rotation axis what is the order of labels and which crystals are these?
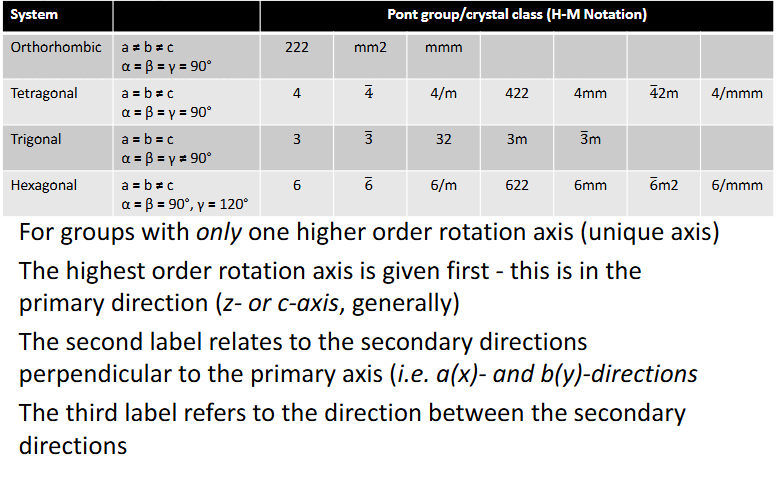
For groups with multiple higher order rotation axes, what system is this and in what order would the labels be in?

Forms have names:
Pedion:
Pinacoid:
Sphenoid:
Prism:
Pyramids:
Forms have names:
Pedion: A plane
Pinacoid: Two parallel planes
Sphenoid: Two intersecting planes, related by a two-fold axis
or mirror plane
Prism: Multiple (3, 4, 6, 8, or 12) intersecting faces
whose edges are parallel to zone axis
Pyramids: Multiple intersecting faces which share a corner
Open vs closed forms
Closed forms define/enclose a volume of space – cube,
tetrahedron etc. Open forms do not (need other forms to chose a shape)
Cubic forms for 111, 110 and 100
Tetrahedron or octahedron for 111, rhombic dodecahedron for 110 and cube for 100.
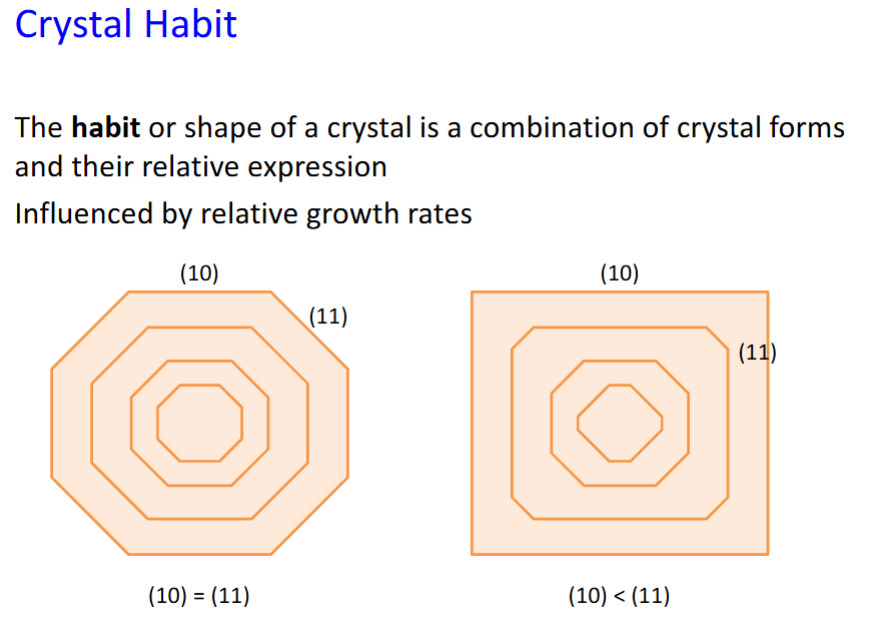
Crystal habit
for the square case (11) grows faster, filling in the corners
Different nucleation processes
Homogenous (same phase), heterogeneous (different phases, nucleation on a foreign object or solid like a seed crystal in solution), these are both primary and secondary nucleation occurs when nucleation is induced by exiting crystals of the same substance
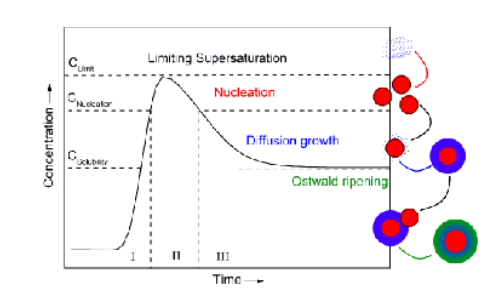
Draw a graph that describes nucleation and explain how traversing this graph differently can change the outcome
If you go up the slope quickly most material is consumed in nucleation (and hence not available for growth) and if slowly only a few nucleation seeds are formed, if you po over this full graph quickly small crystals are formed and slowly big crystals are formed.
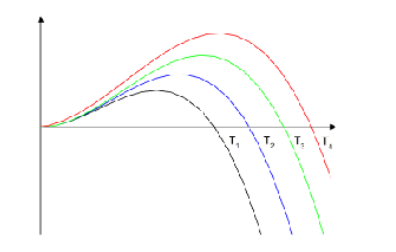
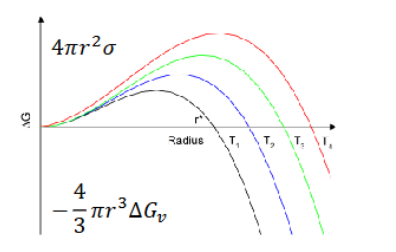
Explain what this graph shows and fill in missing details
Critical nucleus size results from interplay between volume and surface energy. Increased stability with volume due to bonding in the volume (scales with r³) and reduced stability due to interface costing energy (scales with r²). At lower radius the interfacial component dominates, as size is increased the volume overcomes the interfacial energy and makes the cluster more stable, the maximum is the critical size where the cluster cannot redissolve in solution.
Change in chemical potential equation of nucleation
Formation of eumorphic vs hopper crystals and assembly of interconnected cubes.
Formation of hopper crystals occurs above a certain threshold of supersaturation (eumorphic below this supersaturation), above this threshold, because cubic growth is limited by the incorporation of ions into the surface, the only way to incorporate more ions from the supersaturated solution is then to create new surfaces. Because the edges of the first cubic nucleus point in three directions into a highly supersaturated solution, they can grow faster than the flat centers of its faces. Because the edges of the first cubic nucleus point in three directions into a highly supersaturated solution, they can grow faster than the flat centers of its faces. In this way, they will serve as a point for the secondary nucleation of a new cubic crystal. The dynamics of hopper growth then shows up as an assembly of interconnected cubes leading to rapid consumption of the supersaturation in a very short time