6.1.2 Heat, q, and Work, w
1/41
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
Heat
energy in transition from a region of high temperature to a region of low temperature
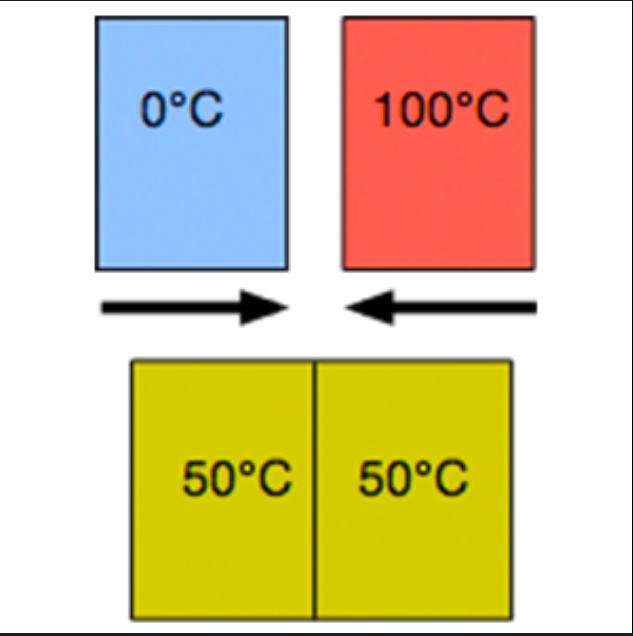
When will heat flow between its system and its surroundings?
when the system isn’t in thermal equilibrium w/ its surroundings
Thermal Equilibrium
when the sys. and the surr. have the same temp.
What is q?
heat
When is q > 0?
When heat is transferring from surroundings → system
rxn absorbs heat from surroundings
endothermic
When is q < 0? What is its name?
-q
When heat is transferring from system → surroundings
rxn releases heat into surroundings
exothermic
What 2 things can happen when a particle is heated? How does the heat affect each scenario?
temp. increases
energy added goes into moving the particles
phase change
occurs at a constant temp.
energy goes into breaking bonds
increases potential energy of molecules
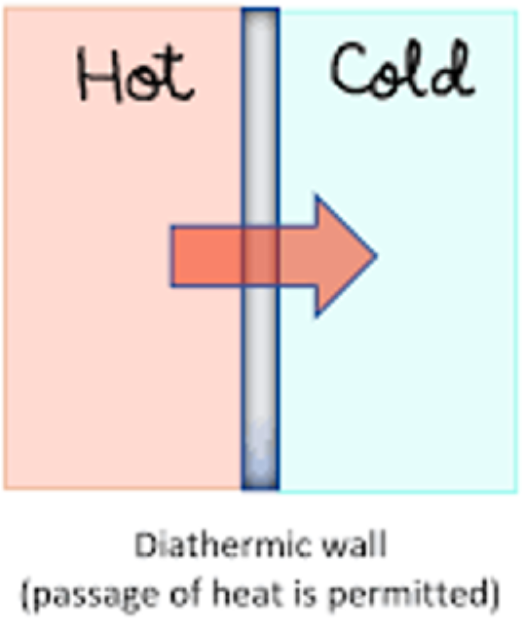
Diathermic
a boundary that permits heat to be transferred between the system and its surroundings
Adiabatic
a boundary that prohibits heat from being transferred between the system and its surroundings
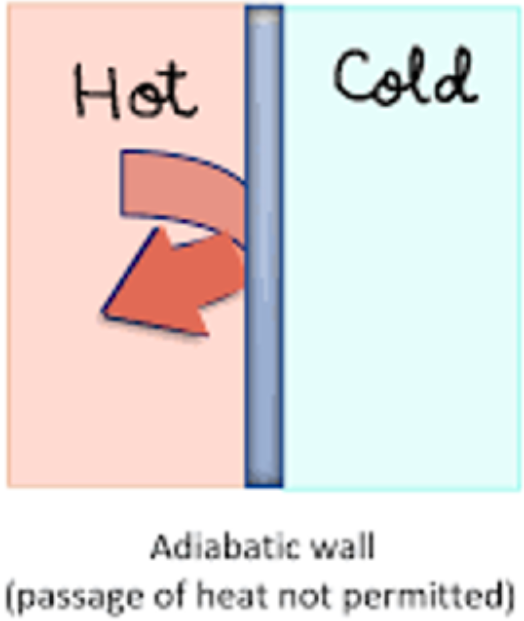
If a process is adiabatic, what does it tell you about heat?
∵ no heat is transferred ∴ q = 0
Synonym for fusion?
melting!
Constant Pressure Heating Curve for a Pure Substance
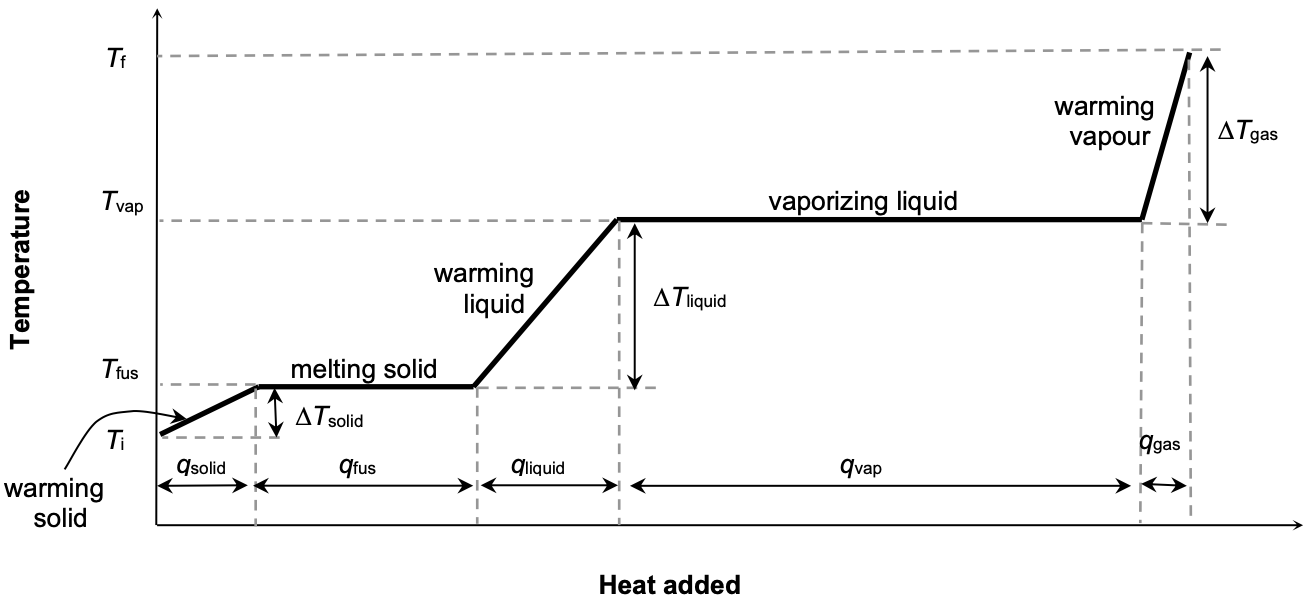
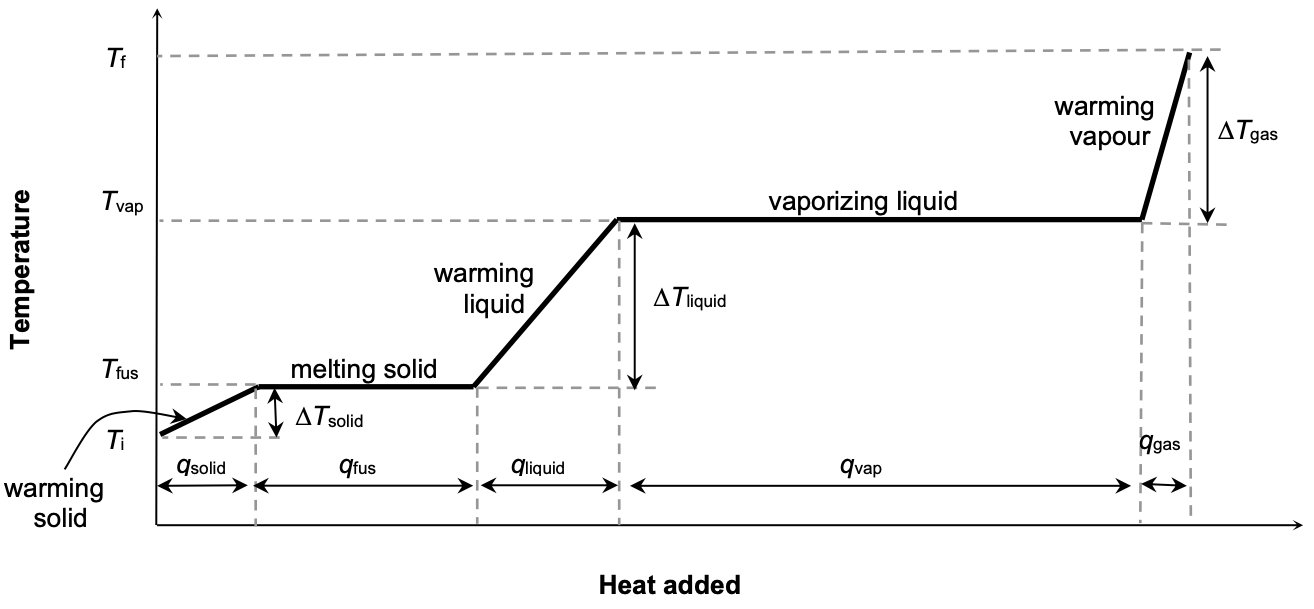
By studying the graph above, we observe that:
temp. of system remains constant while system undergoes a phase change
heat of vaporization (qvap) is typically much greater than the heat of fusion (qfus).
What information about the substance can we obtain from heating curve?
The heat capacities of solid, liquid and gas can be obtained from the slopes of the “warming (diagonal) lines”
The heat of fusion (qfus) and the heat of vaporization (qvap) can be obtained from the horizontal portions of the heating curve.
C
heat capacity
C
\frac{q}{\Delta T}
Csolid
\frac{q_{solid}}{\Delta T_{solid}}
Cliquid
\frac{q_{liquid}}{\Delta T_{liquid}}
Cgas
\frac{q_{gas}}{\Delta T_{gas}}
qsolid
heat absorbed by solid
qliquid
heat absorbed by liquid
qgas
heat absorbed by gas
∆Tsolid
Tfus - Ti
∆Tliquid
Tvap - Tfus
∆Tgas
Tf - Tvap
Heat Capacity
C
the amount of heat required to raise the temperature by 1 oC or 1 K
Average Heat Capacity
C=\frac{q}{\Delta T}
q = ?
C∆T
A temp. change in K is equal to the temp change in…
oC
\frac{\Delta T_{}}{K} = what in degrees celcius?
\frac{\Delta t_{}}{^{o}C}+273.15
or add 273.15 to celcius to get kelvin
Heat Capacity of an Object Units
J K⁻¹
Heat Capacity of an Pure Substance Units
J K⁻¹ g⁻¹ or J K⁻¹ mol⁻¹
What is “c”? What are its units?
specific heat capacity
J K⁻¹ g⁻¹
What must be constant during heating occurs?
either:
pressure
volume
Cv
heat capacity for heating at constant volume
Cp
heat capacity for heating at constant pressure
Why do complex molecules tend to have higher molar heat capacities?
when a substance is heated, the added energy is absorbed by atoms or molecules
if you ↑ the # of ways these entities can absorb energy, molar heat capacity ↑