Comprehensive A-Level Biology: Cell Structure, Function, and Microscopy Techniques
1/171
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
172 Terms
What is the minimum distance between two objects where they can still be viewed as separate?
The minimum distance is determined by the wavelength of light in a light microscope and the wavelength of the electron beam in an electron microscope.
How is magnification calculated?
Magnification = size of the image / size of the real object.
What type of microscope uses a beam of electrons to create an image?
Transmission Electron Microscope (TEM) and Scanning Electron Microscope (SEM).
What is the purpose of calibrating a microscope?
To align the stage micrometer and eyepiece graticule for accurate measurement.
What is the scale of a typical eyepiece graticule?
The scale is usually 1mm long with 100 divisions, so each division is 10μm.
What should you do when drawing a specimen under a microscope?
Draw in pencil, title the diagram, and state the magnification used.
What is a technique that uses different chemical stains to color various parts of a cell?
Staining technique, e.g., using crystal violet and acetic orcein.
How does a Transmission Electron Microscope (TEM) work?
A beam of electrons passes through the sample to create an image, focused using electromagnets in a vacuum.
What are the advantages of using a TEM?
Highest resolving power and high magnification.
What are the disadvantages of using a TEM?
Requires extremely thin specimens, complex staining methods, and the specimen must be dead.
What can you see with a TEM?
A 2D image of details within organelles, such as cristae in mitochondria and grana in chloroplasts.
What does a Scanning Electron Microscope (SEM) provide?
A 3D image of the surface of cells and organelles.
What are the advantages of using a SEM?
High magnification and the ability to view 3D images.
What are the disadvantages of using a SEM?
Requires extremely thin specimens, complex staining methods, and the specimen must be dead.
What is the role of electromagnets in electron microscopy?
They are used to focus the beam of electrons onto the sample.
What is the significance of using a vacuum in electron microscopy?
A vacuum is required to prevent electron scattering by air molecules.
What is the difference between a light microscope and an electron microscope?
Light microscopes use visible light to magnify specimens, while electron microscopes use electron beams for higher resolution.
What is the main limitation of light microscopes compared to electron microscopes?
Light microscopes have lower resolving power due to the longer wavelength of light.
What is the function of a stage micrometer?
It is a microscope slide with a ruler/scaled bar etched into it for calibration purposes.
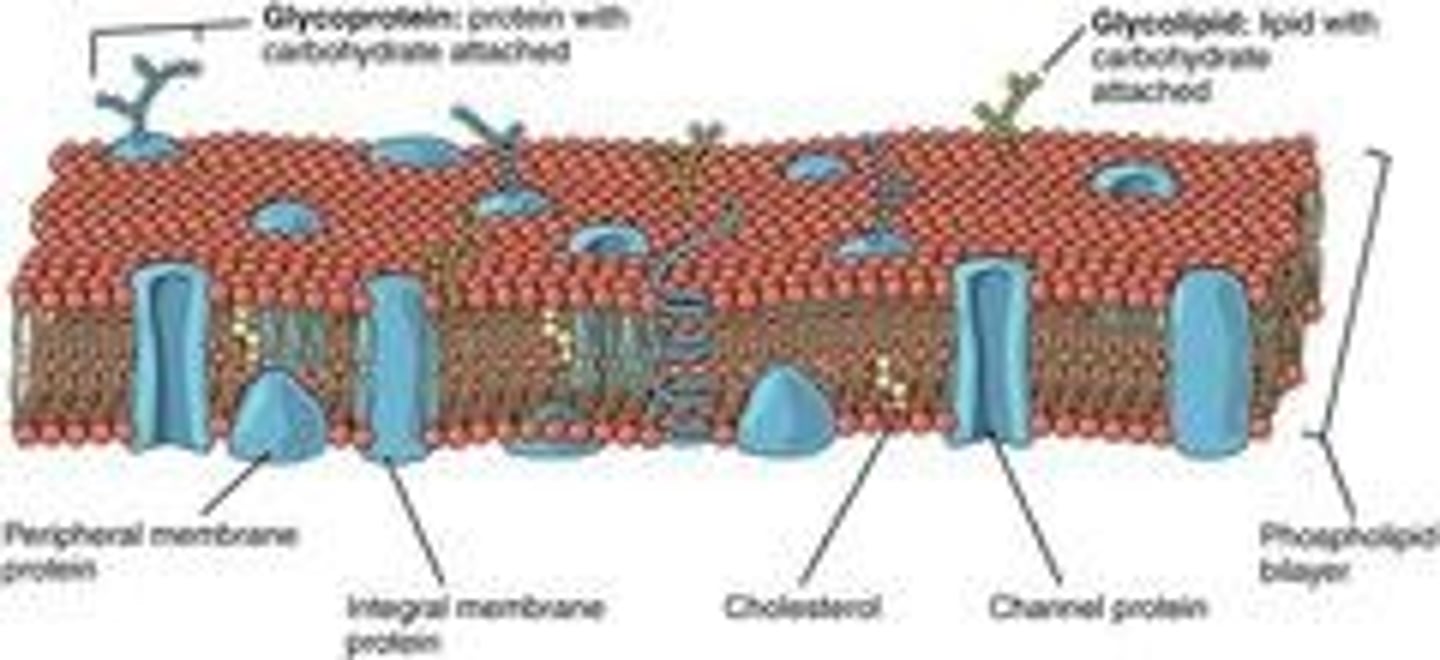
What is the structure of the plasma membrane?
A phospholipid bilayer with embedded intrinsic and extrinsic proteins.
What surrounds the nucleus?
A double membrane called the nuclear envelope with nuclear pores.
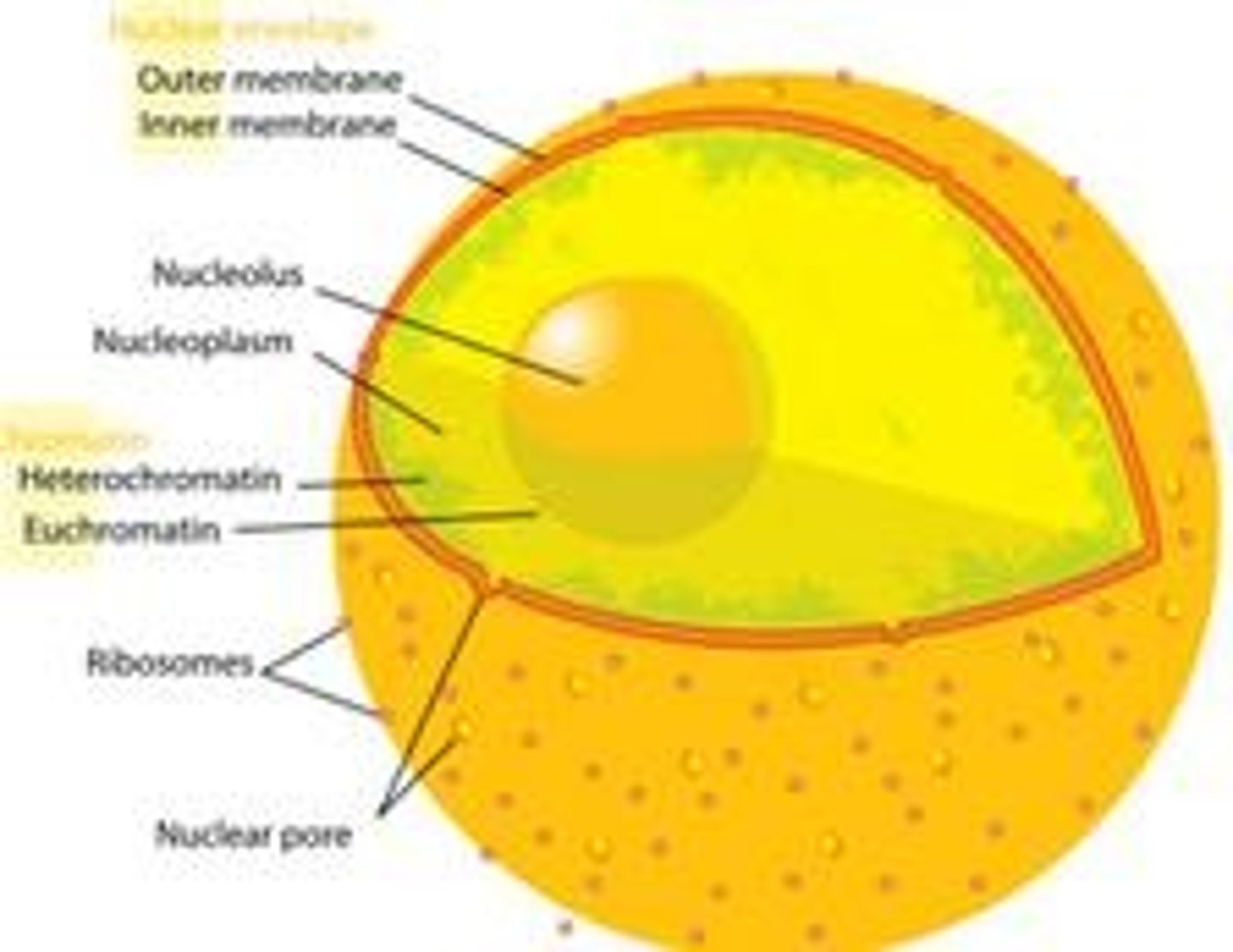
What is contained within the nucleus?
Chromosomes with proteins bound, linear DNA, and a nucleolus.
What is the function of the nucleolus?
To synthesize ribosomes.
What occurs at the site of transcription in the nucleus?
Primary mRNA splicing.
What is the role of nuclear pores?
To allow movement of substances to and from the cytoplasm.
What are cilia?
Hair-like projections out of cells that can be mobile or stationary.
What is the function of mobile cilia?
To help move substances in a sweeping motion.
What is the structure of mitochondria?
Double membrane with inner membrane folded into cristae and a fluid-filled center called the matrix.
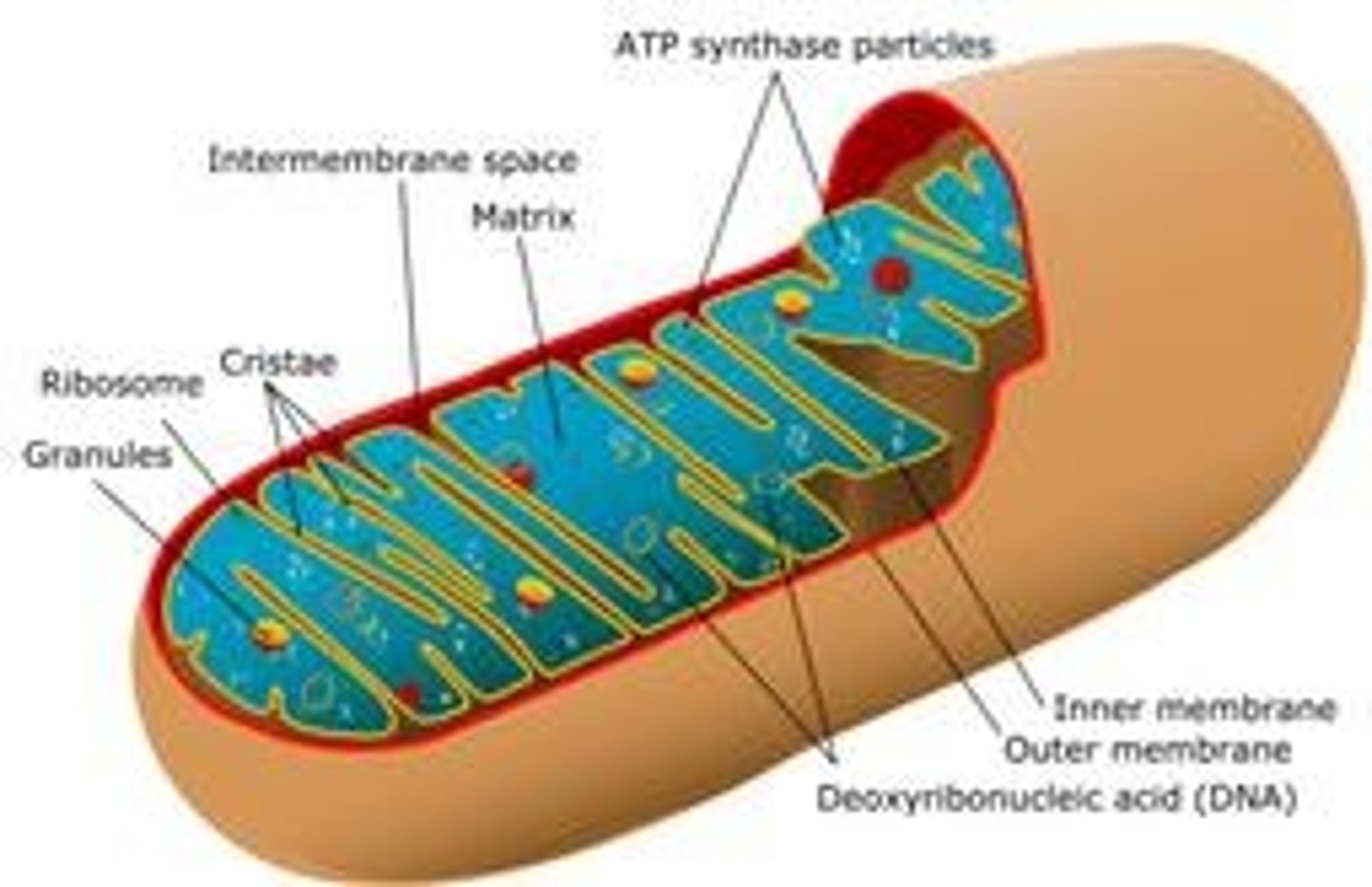
What is the primary function of mitochondria?
Site of aerobic respiration.
What is the structure of flagella?
Whip-like structures made of microtubules that occur in pairs.
What is the function of flagella?
To provide locomotion.
What is the Golgi apparatus made of?
Stacks of membranes creating flattened sacs called cisternae, surrounded by vesicles.
What is the function of the Golgi apparatus?
To modify proteins and lipids, and package them into vesicles.
What is the structure of lysosomes?
Vesicles containing hydrolytic enzymes.
What is the function of lysosomes?
To hydrolyze pathogens and cell waste products.
What is the cytoskeleton?
A network of fibers found within the cytoplasm, consisting of microfilaments, microtubules, and intermediate fibers.
What are the functions of microfilaments?
Responsible for cell movement.
What are the functions of microtubules?
Responsible for creating a scaffold-like structure and providing mechanical strength.
What is the rough endoplasmic reticulum (RER)?
A system of membranes with bound ribosomes, often continuous with the nucleus.
What is the smooth endoplasmic reticulum (SER)?
A system of membranes with no bound ribosomes.
What is the function of the RER?
Site of protein synthesis (translation).
What is the function of the SER?
To create, store, and transport lipids and carbohydrates.
What is the structure of a cell wall?
Made of polysaccharides; cellulose in plants, chitin in fungi, and peptidoglycan in bacteria.
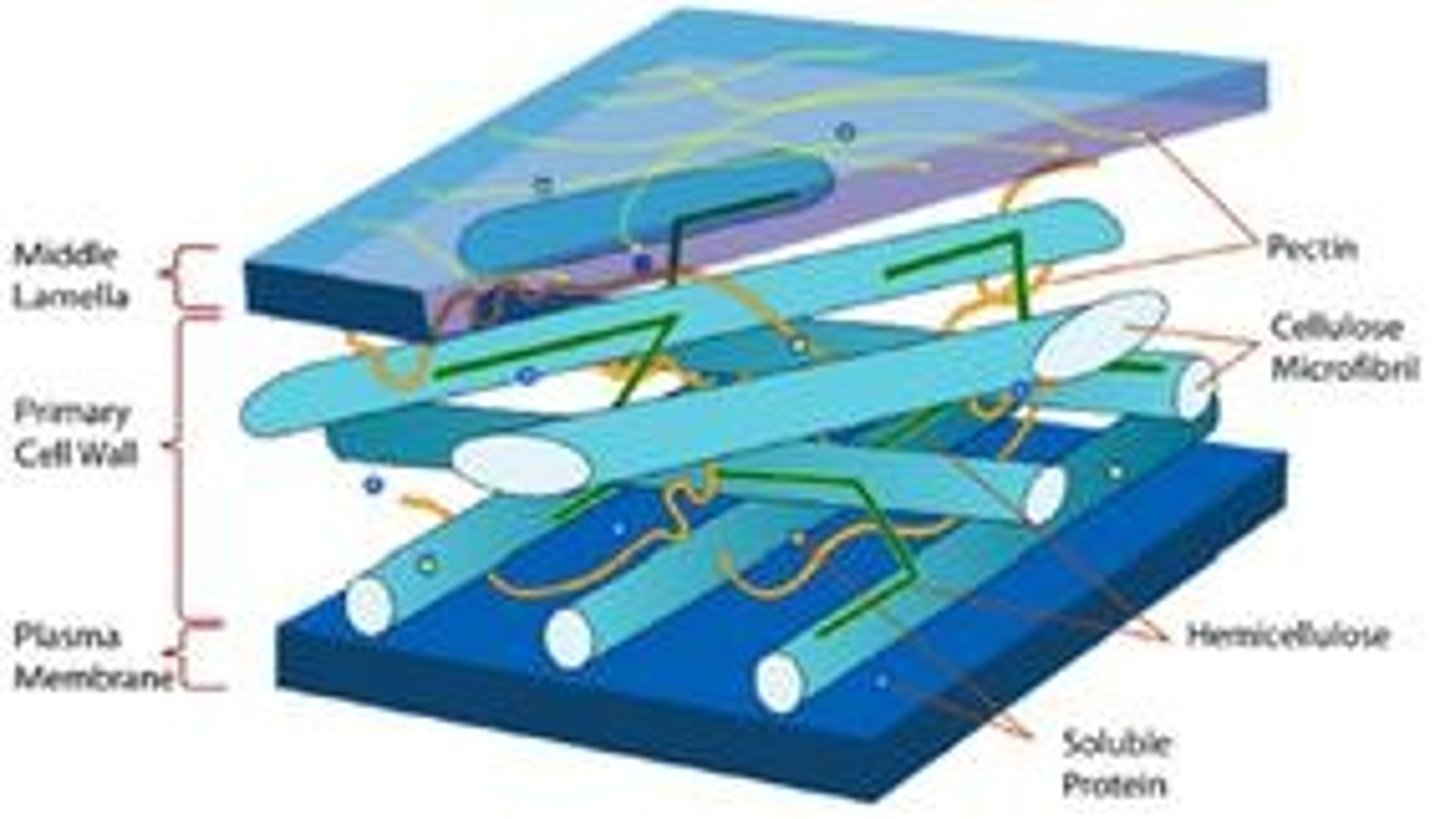
What is the function of the cell wall?
Provides structural strength and prevents cells from bursting due to osmotic pressure.
What are plasmids?
Loops of DNA found in prokaryotic cells.
What is the role of calcium ions in cells?
Muscle contraction and nerve impulse transmission.
What is the role of sodium ions in cells?
Co-transport and reabsorption of water in the kidney.
What is the role of potassium ions in cells?
Stomatal opening and nerve impulse transmission.
What is the function of hydrogen ions in cells?
Chemiosmosis, pH regulation, and translocation.
What type of bonds allow water molecules to stick together?
Hydrogen bonds
What is the role of water as a solvent?
Water dissolves polar or charged molecules, making them hydrophilic.
What happens to the internal temperatures of plants and animals in fluctuating external temperatures?
They remain constant.
What is cohesion in the context of water?
The sticking together of water molecules via hydrogen bonds.
How does water function as a coolant?
It has a high specific heat capacity and high latent heat of vaporization.
What is condensation in chemistry?
The joining of two smaller monomer units to form a chemical bond with the release of water.
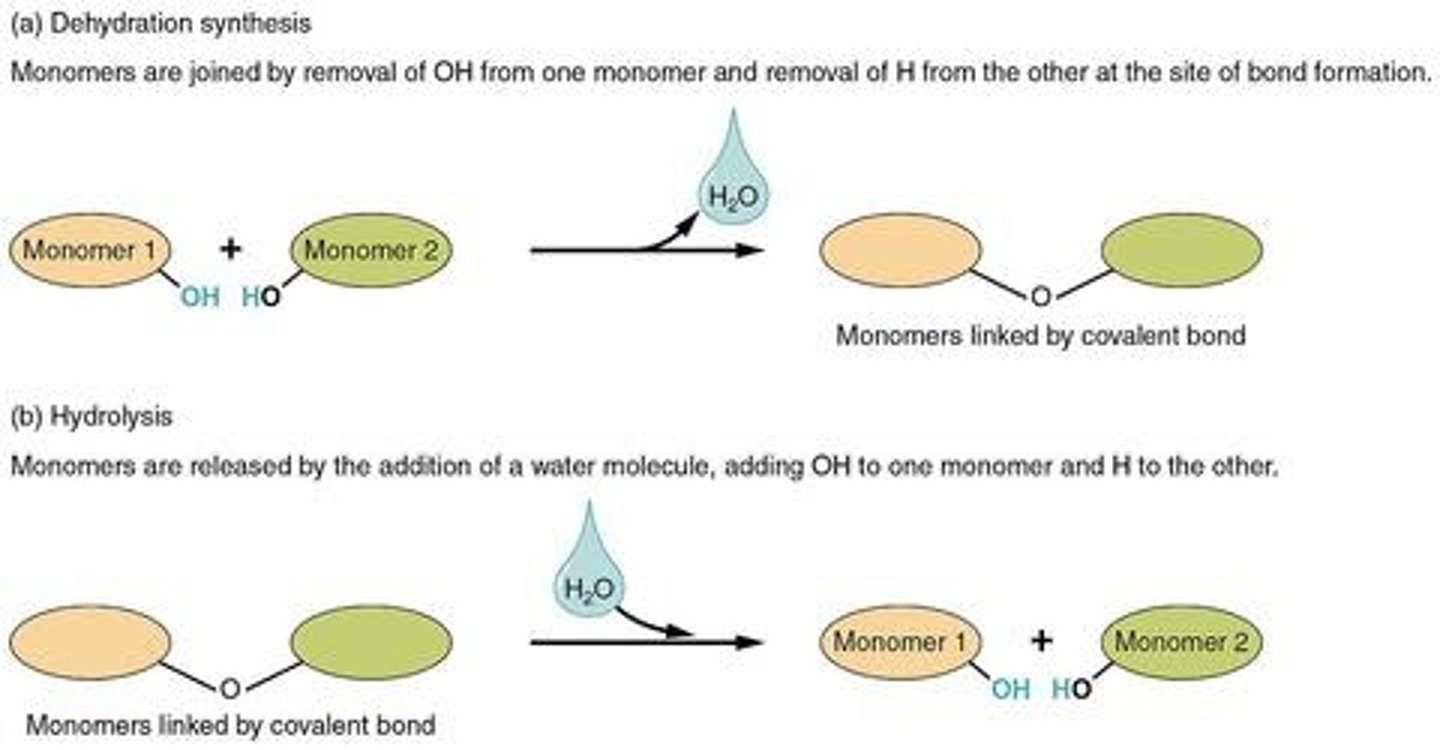
What is hydrolysis?
The breaking of a chemical bond between two bonded monomers by adding a water molecule.
What are monosaccharides?
Small, single units that act as building blocks to create larger molecules, such as glucose.
What are the structural isomers of glucose?
Alpha glucose and beta glucose.
What is the difference between alpha glucose and beta glucose?
In alpha glucose, the H group is at the top of the hexose ring on carbon 1; in beta glucose, the OH group is at the top.
What are disaccharides?
Carbohydrates formed from two monosaccharides joined by a glycosidic bond through a condensation reaction.
What is maltose?
A disaccharide formed from two alpha glucose molecules.
What is lactose?
A disaccharide formed from beta glucose and galactose.
What is sucrose?
A disaccharide formed from alpha glucose and fructose.
What is cellulose?
A polysaccharide in plant cell walls formed by the condensation of beta-glucose.
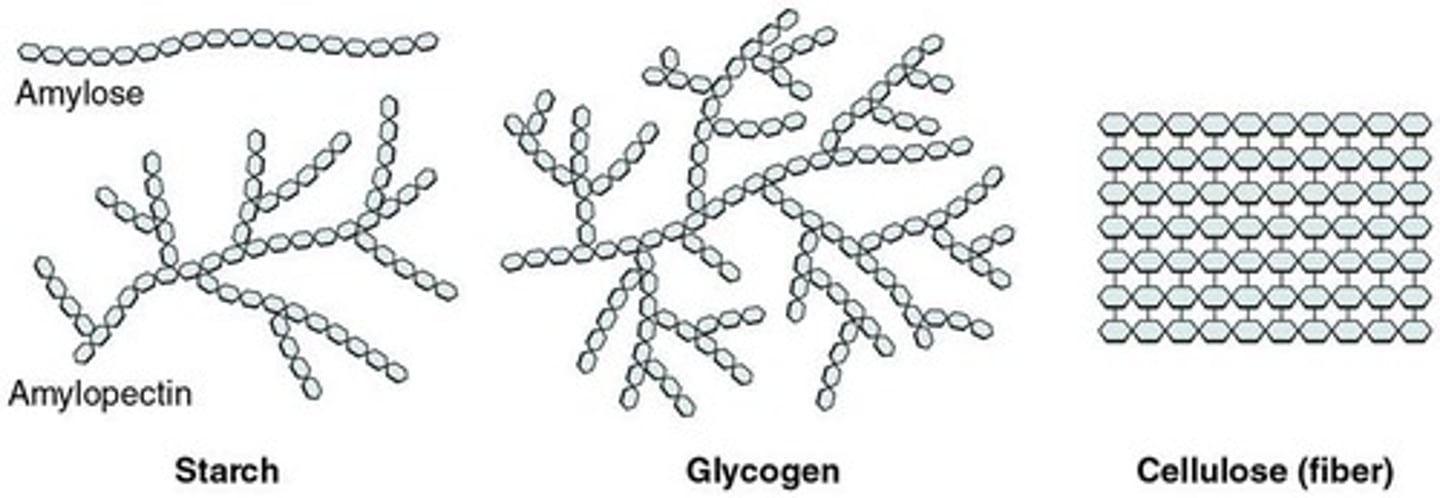
What is glycogen?
A polysaccharide in animals formed by the condensation of alpha-glucose.
What is starch?
A polysaccharide in plants formed by the condensation of alpha-glucose.
What are the two polymers contained in starch?
Amylose (1-4 bonds) and amylopectin (1-4 and 1-6 bonds).
What are the properties of amylose?
It is coiled to form a helix and joined via 1-4 glycosidic bonds.
What are the properties of amylopectin?
It is branched, insoluble, and has a large surface area for rapid hydrolysis of glucose.
Why is starch insoluble?
It is a large molecule, which does not affect osmosis.
What is the significance of the spiral structure of starch?
It allows for compact storage, fitting a lot into a small space.
What is the primary component of starch?
Polysaccharide composed of alpha-glucose.
What type of bonds provide stability and rigidity to cell walls?
Large number of hydrogen bonds between chains.
What is a triglyceride composed of?
One molecule of glycerol and three fatty acids.

What type of bonds are formed in triglycerides?
Three ester bonds.
What is the solubility characteristic of triglycerides?
They are non-polar and insoluble in water (hydrophobic).
What is the structure of fatty acids?
Long hydrocarbon chains with a carboxyl group at one end.
What is a sterol?
A type of lipid with four carbon rings and a hydroxyl group.
What is the role of cholesterol in cell membranes?
Decreases fluidity at high temperatures and increases fluidity at low temperatures.
What are phospholipids composed of?
One molecule of glycerol and two fatty acids.
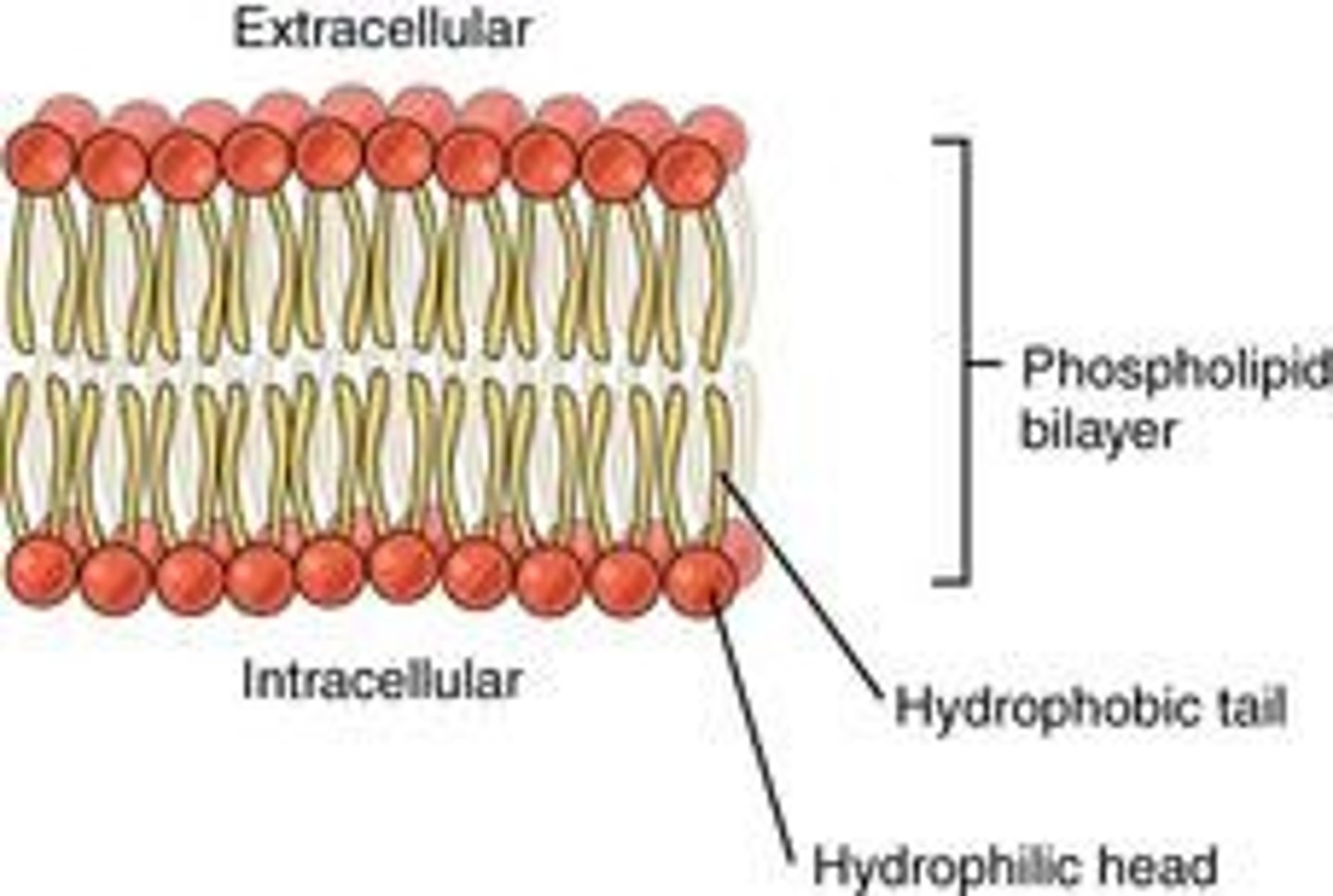
What makes phospholipids polar molecules?
The hydrophilic phosphate 'head' and the hydrophobic lipid 'tail'.
What is the monomer subunit of a protein?
Amino acid.
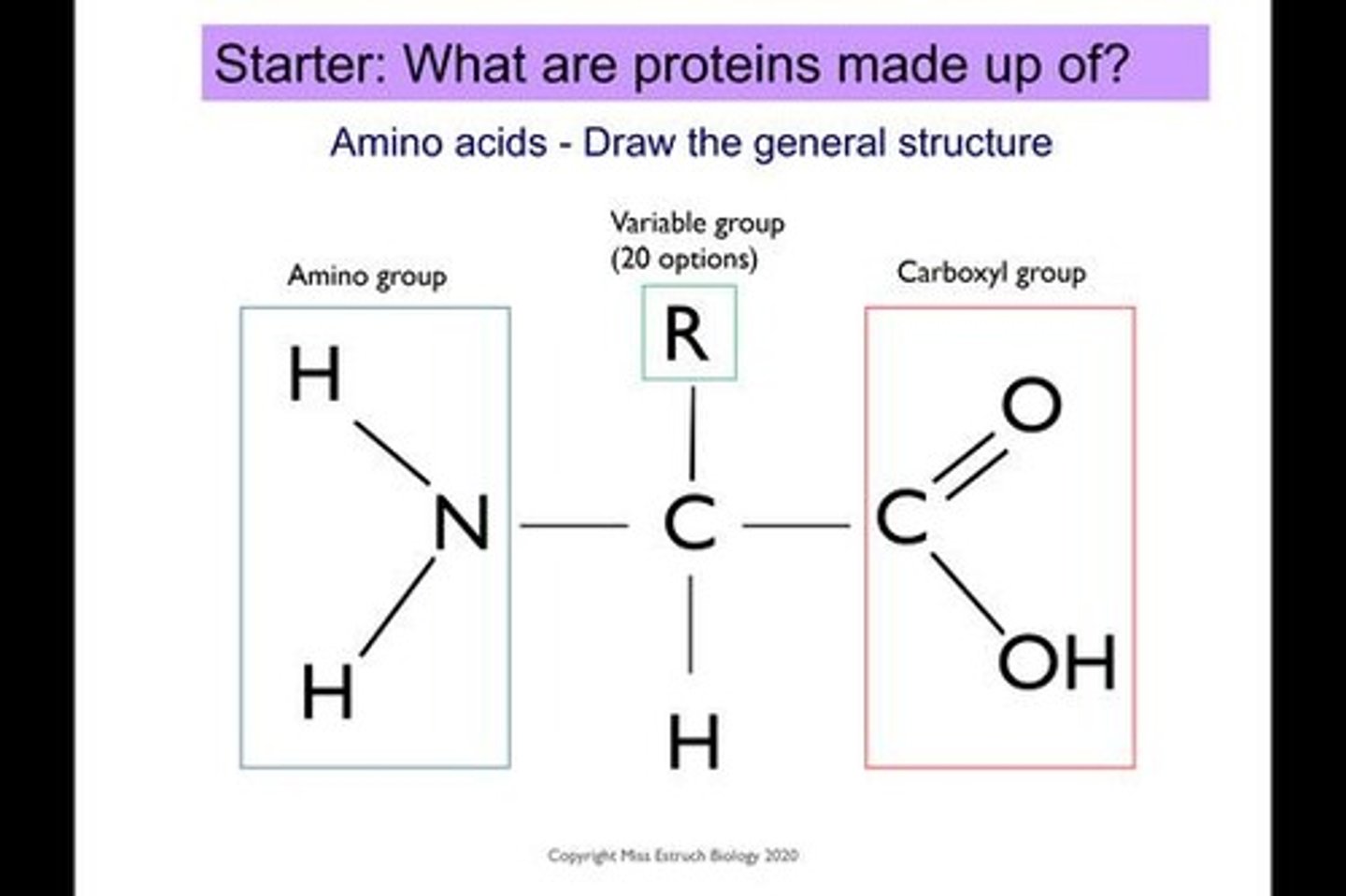
What groups are found in an amino acid?
Carboxyl group, amine group, and R group.
What type of bond joins amino acids in a protein?
Peptide bonds.
What is an alpha helix?
A coiled shape in the secondary structure of proteins held by hydrogen bonds.
What is a beta-pleated sheet?
A folded shape in the secondary structure of proteins held by hydrogen bonds.
What type of bond forms in the tertiary structure of proteins?
Ionic bonds, hydrogen bonds, and disulfide bonds.
What is a quaternary protein structure?
A protein made up of more than one polypeptide chain.
What is a globular protein?
A protein with a spherical shape, often with a prosthetic group.
What is a fibrous protein?
A protein with long twisted strands that provide structural strength.
What is the test for reducing sugars?
Add excess Benedict's solution, heat, and observe color change to green/yellow/orange/brick red.
What indicates the presence of starch in a test?
The solution turns blue-black when iodine is added.
What is the test for lipids?
Add ethanol, shake, then pour into water; a white emulsion indicates presence.
What is the test for proteins?
Add biuret solution; it turns purple in the presence of protein.
What is a non-reducing sugar?
A sugar that cannot reduce Cu²⁺ in Benedict's solution without hydrolysis.
What is the role of biosensors in biochemical tests?
They provide quantitative results for the concentration of substances.
What is the formula for calculating the Rf value?
Rf = distance moved by solute / distance moved by solvent
What is a nucleotide?
The monomer of DNA and RNA, consisting of a pentose sugar, a phosphate group, and a nitrogenous base.
What are the nitrogenous bases found in DNA?
Adenine, guanine, cytosine, and thymine.
What are the nitrogenous bases found in RNA?
Adenine, guanine, cytosine, and uracil.