lecture 3: diffusion, osmosis and toxicity
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
13 Terms
nanoscale pores
responsible for transporting molecules in and out of a cell membrane
diffusion
essential biological process
transports O2 and CO2 into/out of the bloodstream, ions across cell membranes etc.
Occurs due to random thermal motion of molecules, similar to motion in a gas
Higher temperature (higher thermal motion) leads to faster diffusion
cause of diffusion
Due to random thermal motion; molecules are bouncing into something
Net motion; characterised by a root mean square distance
Motion of the di-molecule can be characterised by this
Occurs from a region of high concentration towards a region of lower concentration
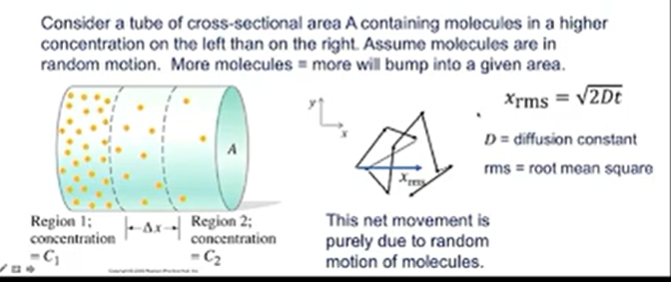
diffusion equation; fick’s law
provides the rate of diffusion
the greater the difference in concentration per unit distance, the greater the flow rate (i.e. the rate of diffusion J)

alveolar sac structure
Lung contains about 600 million alveoli (SA - 70m2)
Alveolar membrane between air and blood is thin, typically 0.5 μm
Roughly spherical in shape
Oxygen molecules diffuse across membrane rapidly approx. 0.25 s
Oxygen concentration in blood leaving lungs is almost equal to the concentration in alveolus
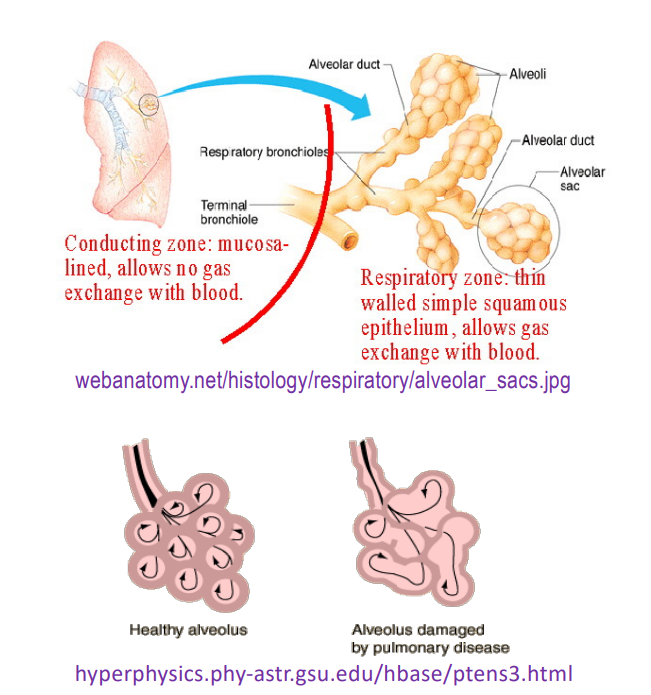
diffusion example; oxygen in blood

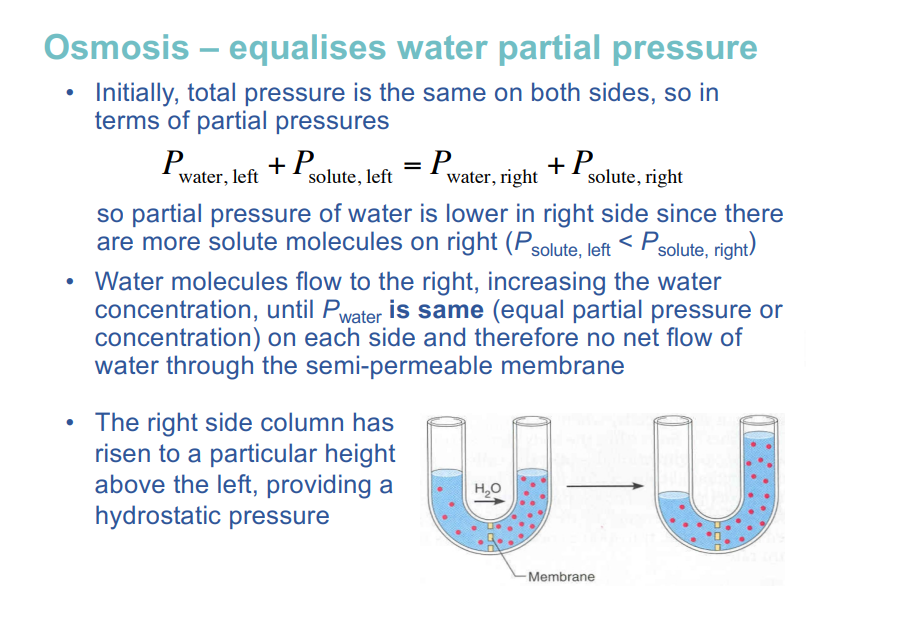
osmosis
osmosis is the movement of water molecules from a solution with a high concentration of water molecules to a solution with a lower concentration of water molecules, through a cell's semi permeable membrane
osmotic concentration/osmolarity
Dissolve sugar or salt (solute) in water (solvent)
The osmotic concentration, or osmolarity, is;
The number of moles of solute particles, or osmoles, per litre of solution (osmol/L)
Osmole (or Osm or osmol) is a non-SI unit defining;
The number of moles of particles that contribute to the osmotic pressure
E.g. NaCl dissociates into Na+ and Cl= ions, so 1 mole/L of NaCl gives 2 osmol/L of solute particles
Glucose does not dissociate, so 1 mole/L of glucose gives 1 osmol/L of glucose
osmosis via a semi permeable membrane
spontaneous movement driven by diffusion
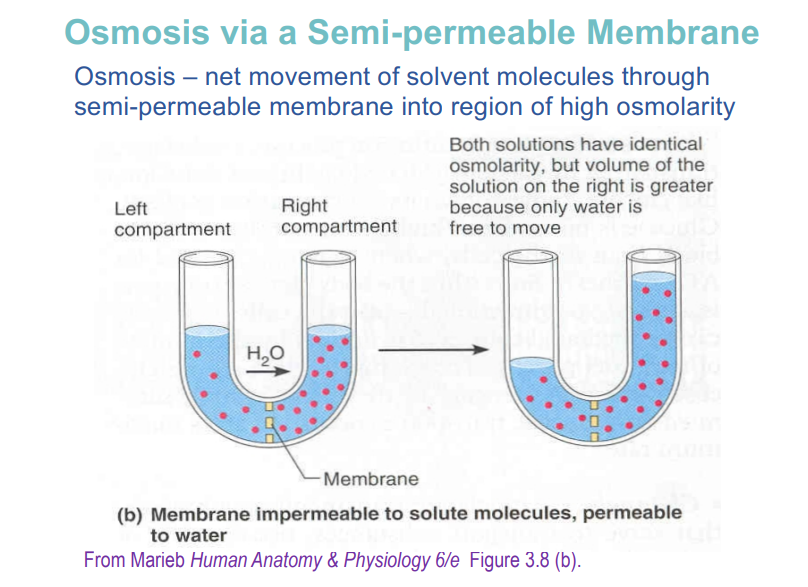
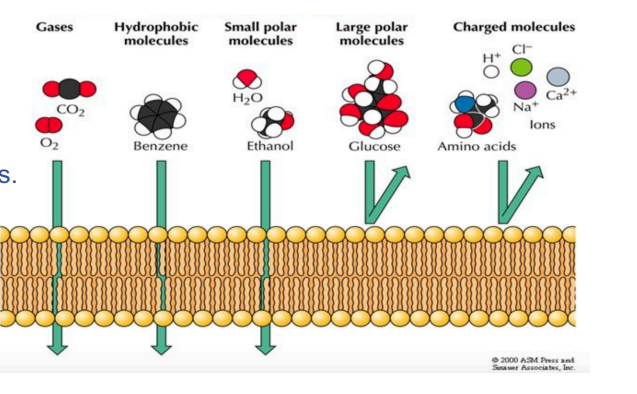
semi permeable membrane
Barrier that blocks some molecules, but lets others through
Typically lets through polar molecules (like water), but blocks bigger polar or charged molecules, like proteins or ions
Common feature in many biological membranes
osmosis and equalising of water pressure
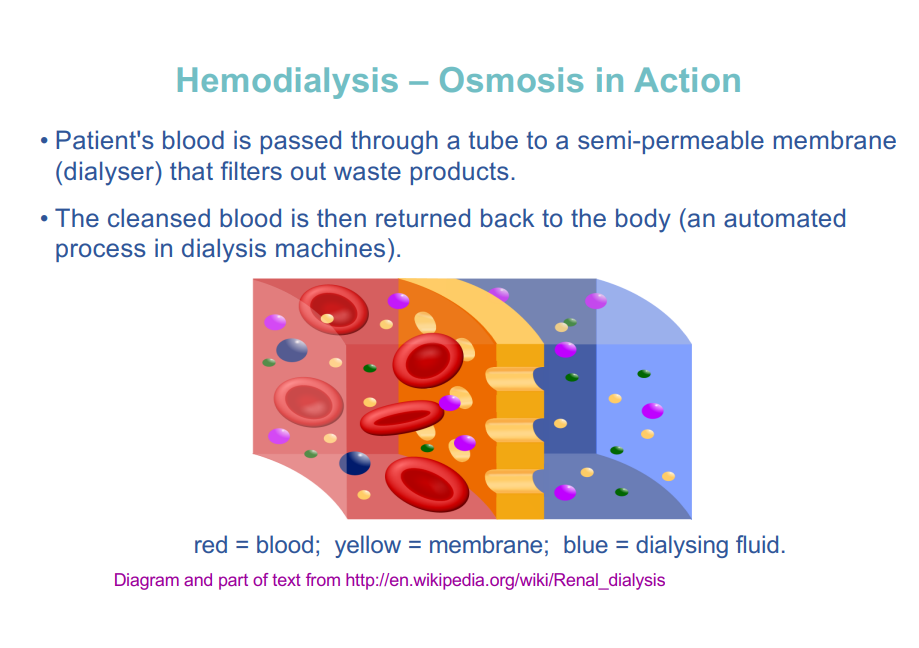
hemodialysis
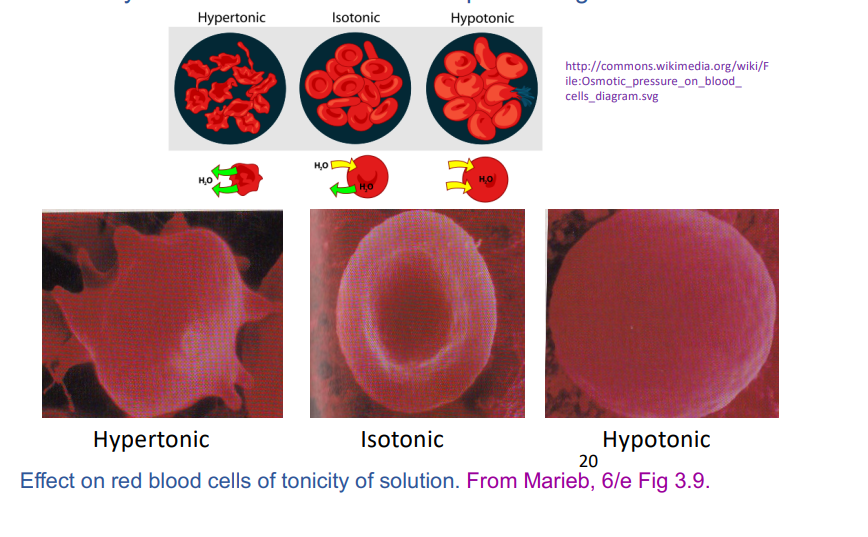
tonicity
the effect a solution has on the volume of a cell due to osmosis, specifically the relative concentrations of solutes that cannot freely cross the cell membrane, leading to water movement and cell swelling or shrinking
a measure of the osmotic pressure gradient