BIOC 4331 Lecture 17
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
What is V₀ in enzyme kinetics?
The initial rate/initial velocity of product formation (measured at the start, before significant product builds up).
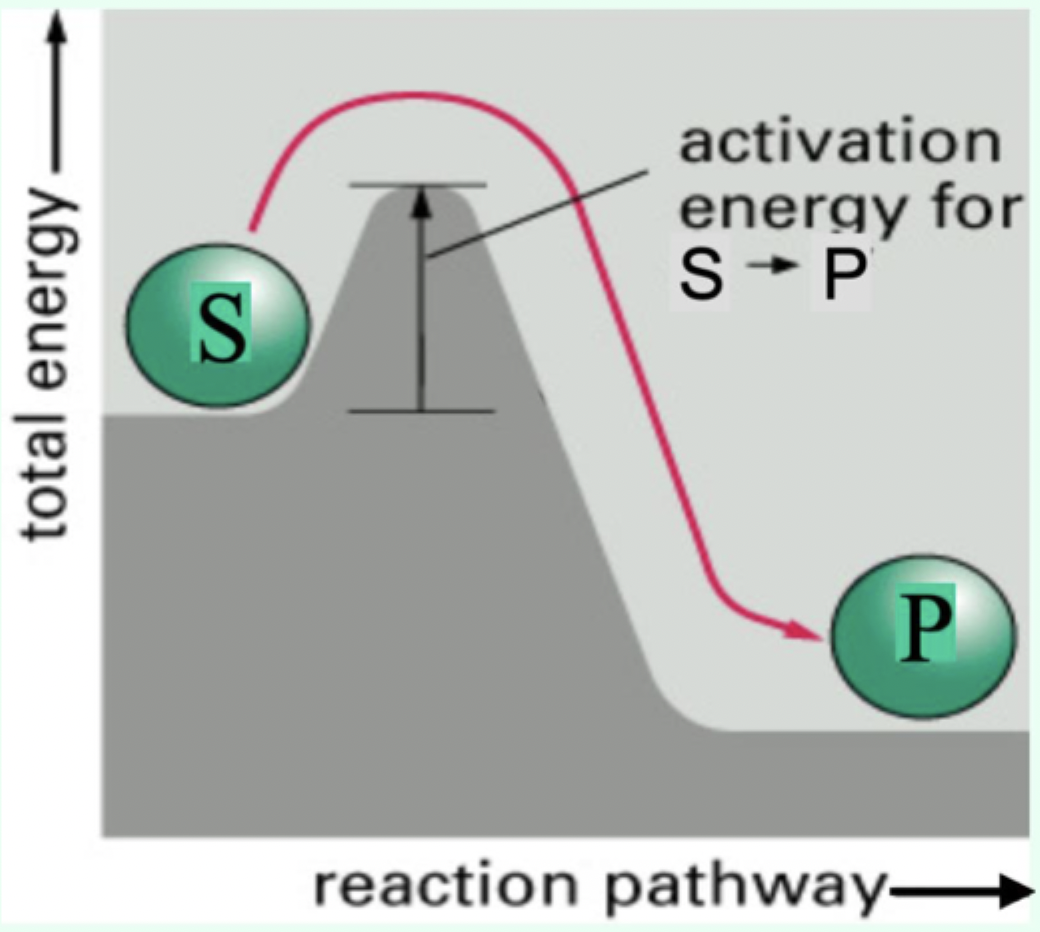
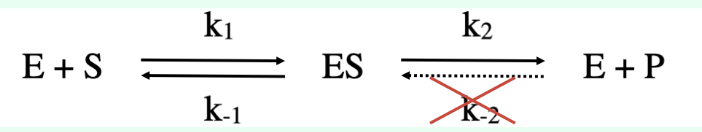
Why is the step ES → E + P treated as irreversible in Michaelis–Menten kinetics?
Measurements are taken under initial velocity conditions, where:
[P] ≈ 0 (no product has accumulated yet)
The reverse reaction (E + P → ES) is negligible
Therefore, ES proceeds forward to form product
![<p>Measurements are taken under <strong>initial velocity conditions</strong>, where:</p><ul><li><p><strong>[P] ≈ 0</strong> (no product has accumulated yet)</p></li><li><p>The reverse reaction (<strong>E + P → ES</strong>) is negligible</p></li><li><p>Therefore, ES proceeds forward to form product</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/d5200d5c-fd74-45bd-824e-0773c7725280.png)

What is kcat?
The turnover number: max catalytic activity; # of substrate molecules converted to product per enzyme per unit time at substrate saturating conditions [S].
![<p>The <strong>turnover</strong> number: max <strong>catalytic</strong> activity; # of substrate molecules <strong>converted to product per enzyme per unit time</strong> at <strong>substrate</strong> <strong>saturating</strong> <strong>conditions</strong> [S].</p>](https://knowt-user-attachments.s3.amazonaws.com/9756b309-5554-4614-b471-656fecbd382e.png)

What is the relationship between k₂ and kcat?
k₂ = kcat (used interchangeably).

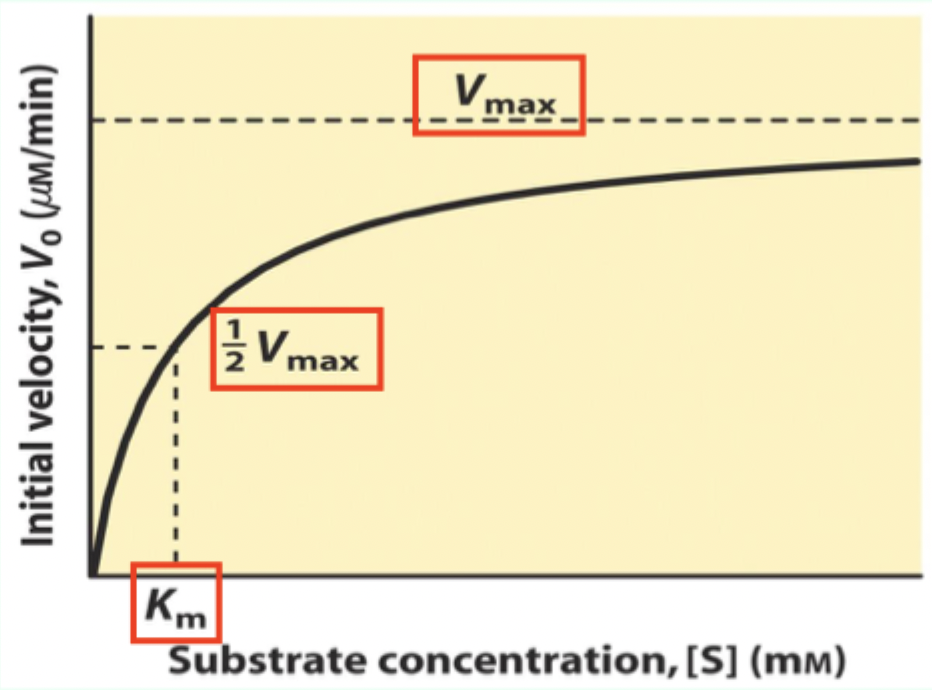
What does Vmax represent?
The maximum velocity achieved at very high (effectively infinite) substrate concentration.
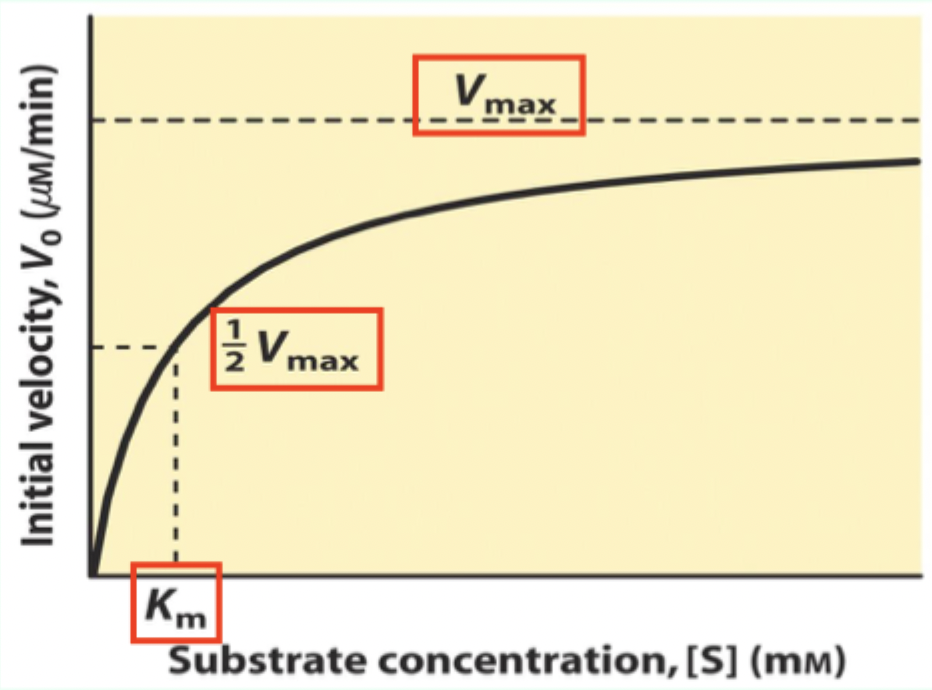
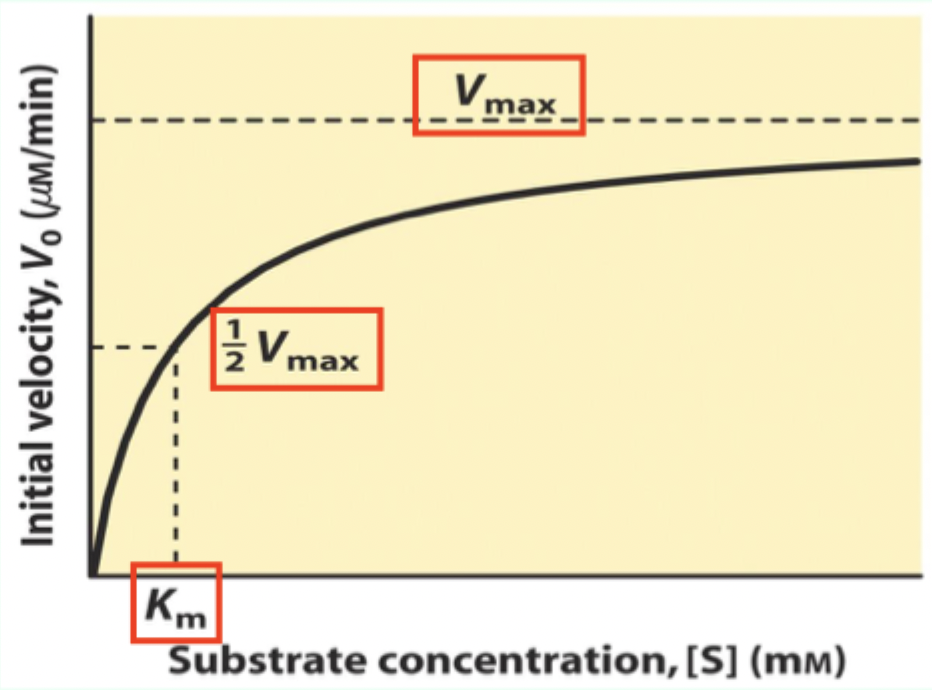
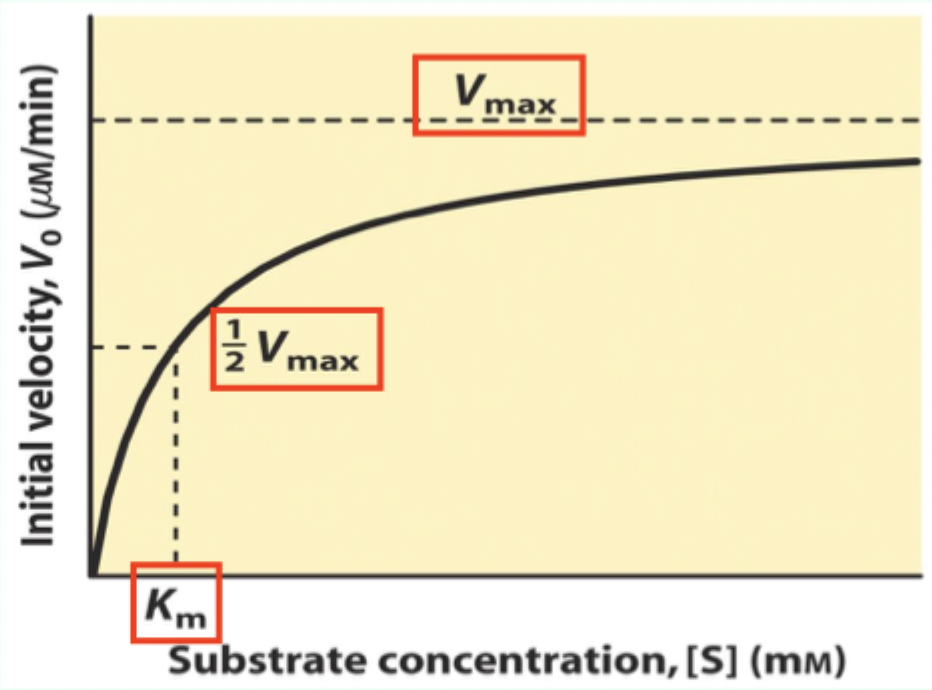
What is Km on a Michaelis–Menten plot?
The Michaelis–Menten (M–M) constant; Km is the [S] where V₀ = 0.5 Vmax
![<p>The <strong>Michaelis–Menten (M–M) constant</strong>; <strong>K<sub>m</sub></strong> is the [S] where V₀ = 0.5 Vmax</p>](https://knowt-user-attachments.s3.amazonaws.com/0766689f-301e-4ace-8507-b8ba7f6b073d.png)
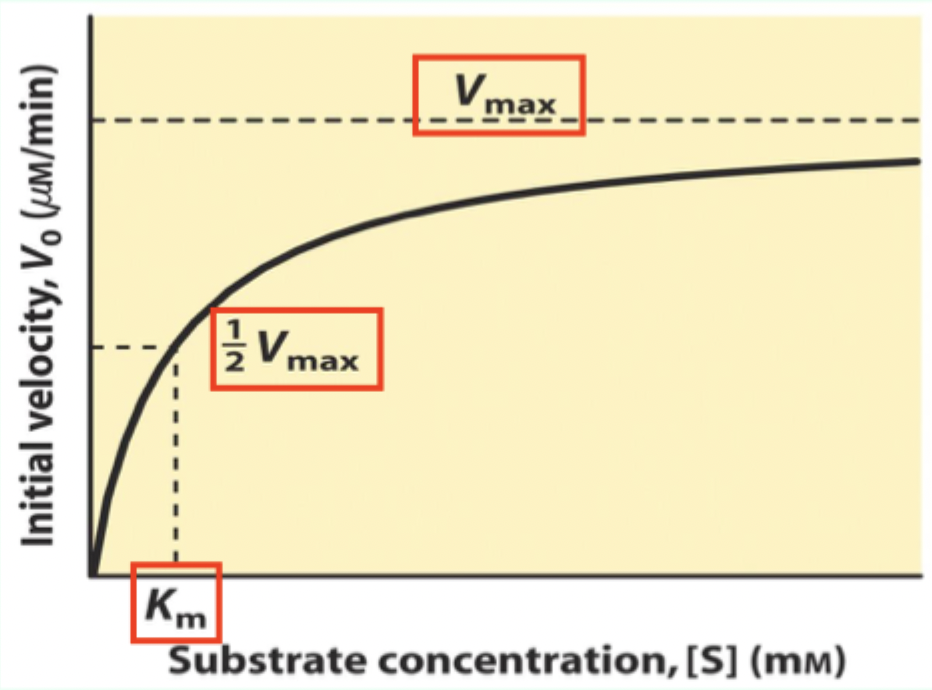
What does Km measure about enzyme–substrate interaction?
A measure of affinity: lower Km = tighter substrate binding (higher affinity).
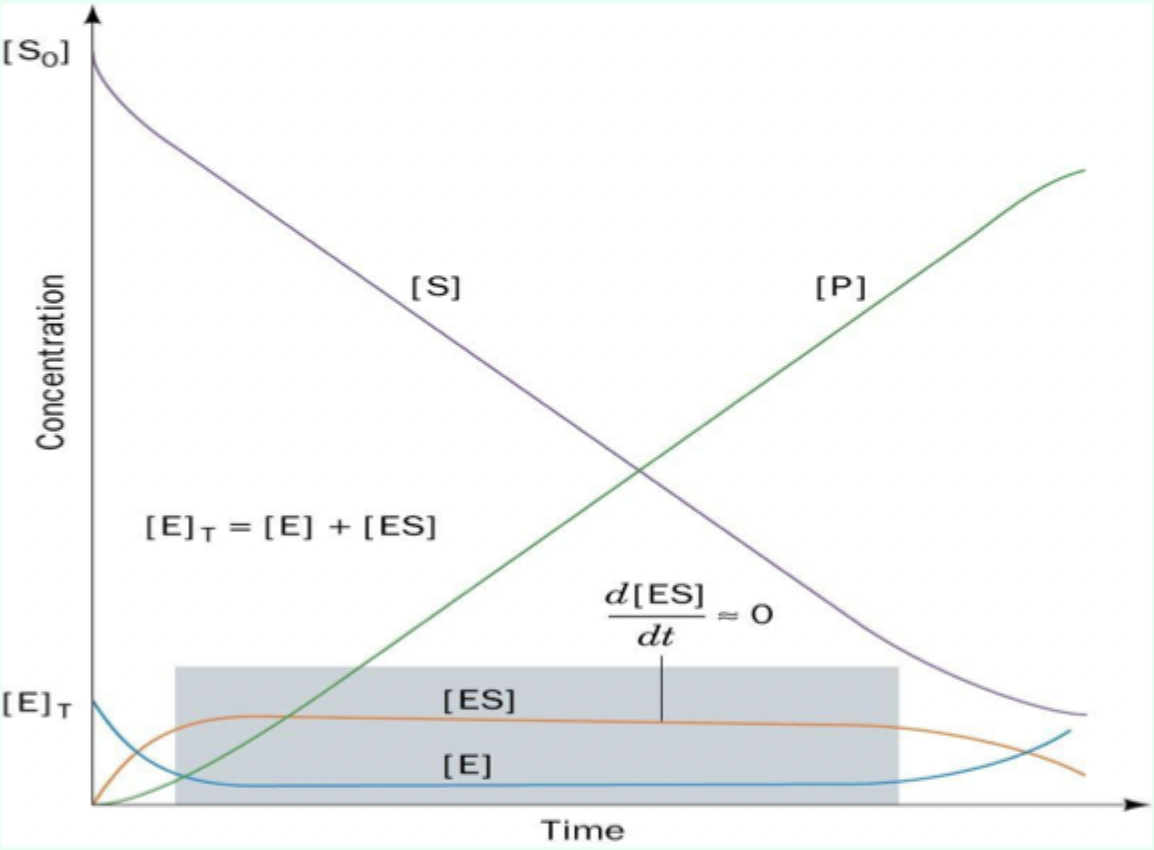
Steady state assumption (what becomes constant?)
After a short time, [ES] is effectively constant (steady state), so d[ES]/dt ≈ 0.
![<p>After a short time, <strong>[ES] </strong>is effectively constant (<strong>steady state</strong>), so d[ES]/dt ≈ 0.</p>](https://knowt-user-attachments.s3.amazonaws.com/9e4f8f01-d47b-4d4f-842f-c41182a0643a.png)
What does kcat/Km represent physically?
Specificity constant: Equivalent to the rate constant for the reaction between free enzyme and free substrate; a measure of enzyme efficiency.
What’s the diffusion-controlled upper limit for kcat/Km? And what are those enzymes called?
~10⁸–10⁹ M⁻¹ s⁻¹; “Almost perfect catalysts” (diffusion-limited).
Enzymes with kcat/Km near 10⁸–10⁹ M⁻¹ s⁻¹ are called what?
“Almost perfect catalysts” (diffusion-limited).
What is a double reciprocal (Lineweaver–Burk) plot?
A plot of 1/V₀ vs 1/[S] that yields a straight line.
![<p>A plot of <strong>1/V₀ vs 1/[S]</strong> that yields a <strong>straight</strong> line.</p>](https://knowt-user-attachments.s3.amazonaws.com/f6e95b7c-e038-4755-82e9-757ebe5b8dee.png)
What are the units of kcat?
s⁻¹
What are the units of Km?
Concentration (M), because substrate is reported as a concentration.