Chemistry: Reversible reactions
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
What are reversible reactions?
Chemical reactions that can go either forward or backwards
What happens in the different directions?
The forward reaction forming the products
The reverse reaction forming the reactants
What do you use when writing the equation?
Use ⇌
Where can reversible reactions be seen?
In some hydrated salts, those that contain water of crystallisation
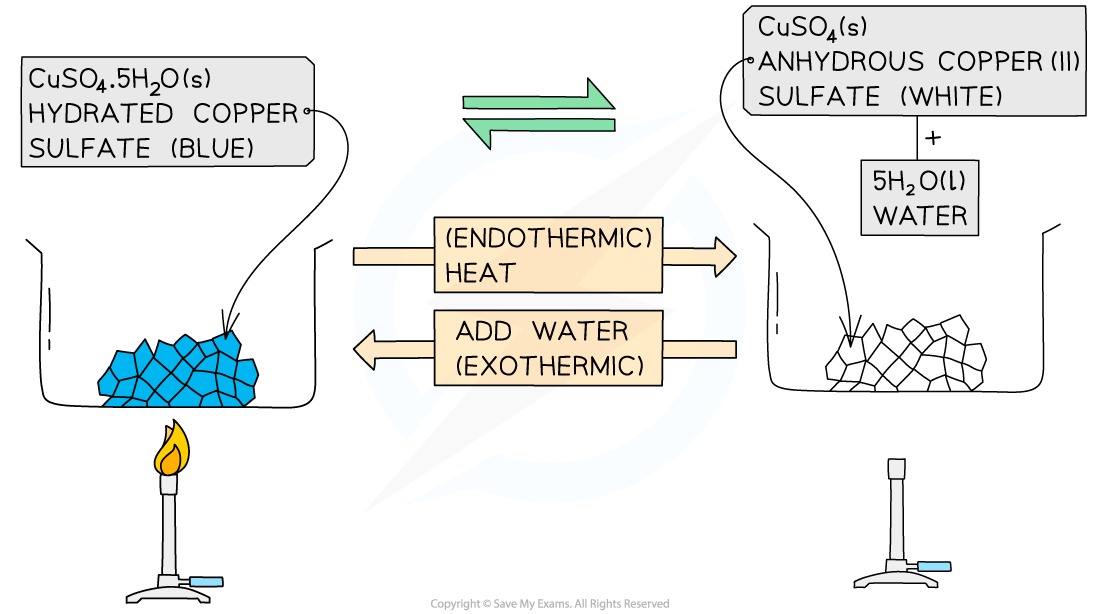
What is an example?
hydrated copper(II) sulfate ⇌ anhydrous copper(II) sulfate + water
CuSO4•5H2O ⇌ CuSO4 + 5H2O
What happens in forward reaction and what reaction is it?
Blue crystals
The hydrated salt can be heated / dehydrated to form anhydrous copper(II) sulfate, CuSO4
This reaction is endothermic as energy is taken in to remove the water
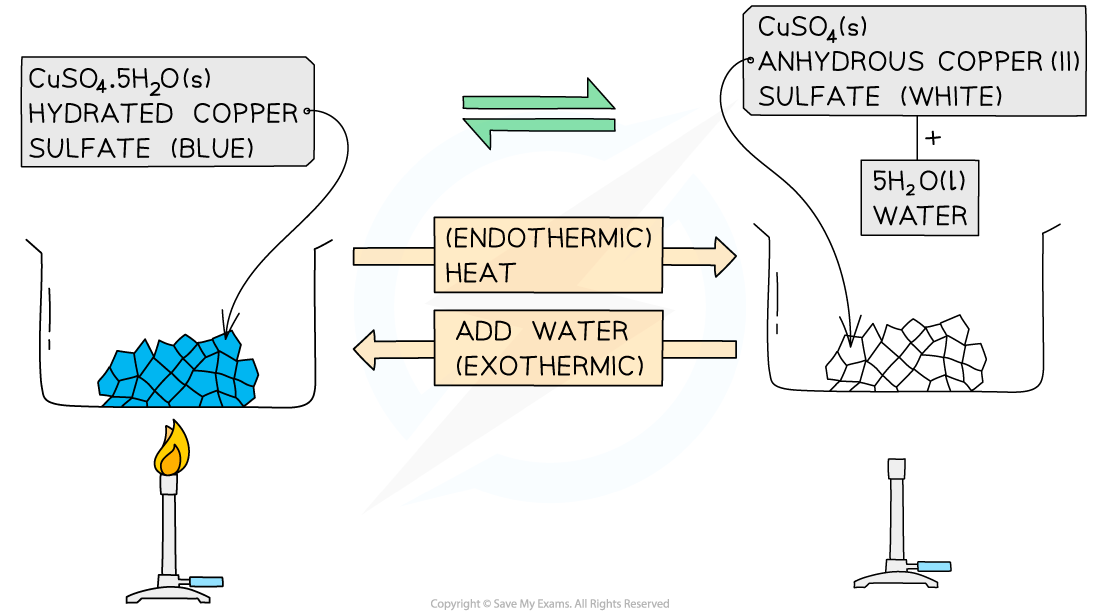
What happens in backward reaction and what reaction is it?
This is usually seen as white crystals / powder
Adding water to the anhydrous salt forms the hydrated copper(II) sulfate pentahydrate, CuSO4•5H2O
This reaction is highly exothermic
How should reversible reactions be done?
in CLOSED SYSTEM to prevent products from escaping/reactants from outside reacting to allow for reverse reaction for equilibrium
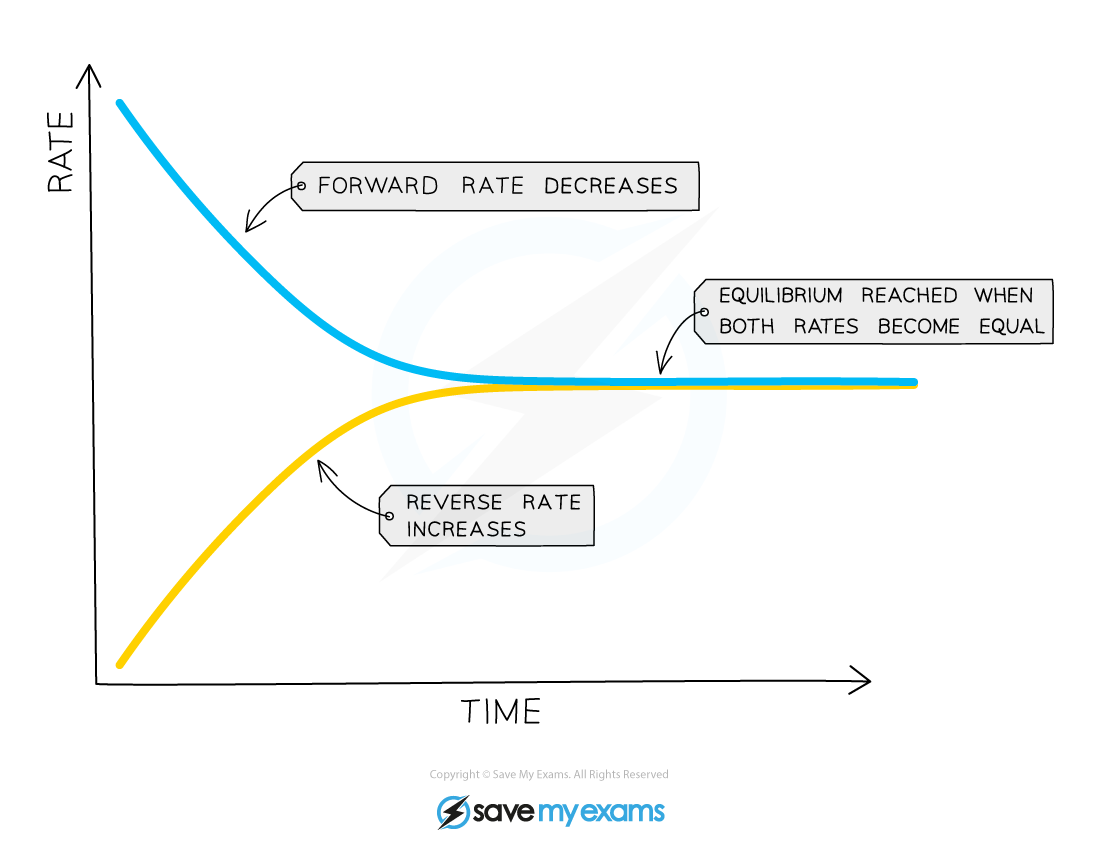
What is equilibrium?
When the rate of the forward reaction equals the rate of the reverse reaction.
What is the haber process?
manufacturing ammonia by combining nitrogen from the air and hydrogen
Describe this process in relation to reaching equilibrium
At the start of the reaction, only nitrogen and hydrogen are present
This means that the rate of the forward reaction is at its highest, since the concentrations of hydrogen and nitrogen are at their highest
As the reaction proceeds, the concentrations of hydrogen and nitrogen gradually decrease
So, the rate of the forward reaction will decrease
However, the concentration of ammonia is gradually increasing and so the rate of the backward reaction will increase
Ammonia will decompose to reform hydrogen and nitrogen
In a closed system, the two reactions are interlinked and none of the gases can escape
So, the rate of the forward reaction and the rate of the backward reaction will eventually become equal and equilibrium is reached
Changes in what can be used to predict the changes to the position of equilibrium?
Temperature
Pressure
When does the position of the equilibrium shifts right?
when the forward reaction is favoured
This means that there is an increase in the amount of products formed
When does the position of the equilibrium shifts left?
when the reverse reaction is favoured
So, there is an increase in the amount of reactants formed
If the temperature of the reaction increases, where will the equilibrium shift?
The equilibrium will shift in the direction of the endothermic reaction
If the temperature of the reaction decreases, where will the equilibrium shift?
The equilibrium will shift in the direction of the exothermic reaction
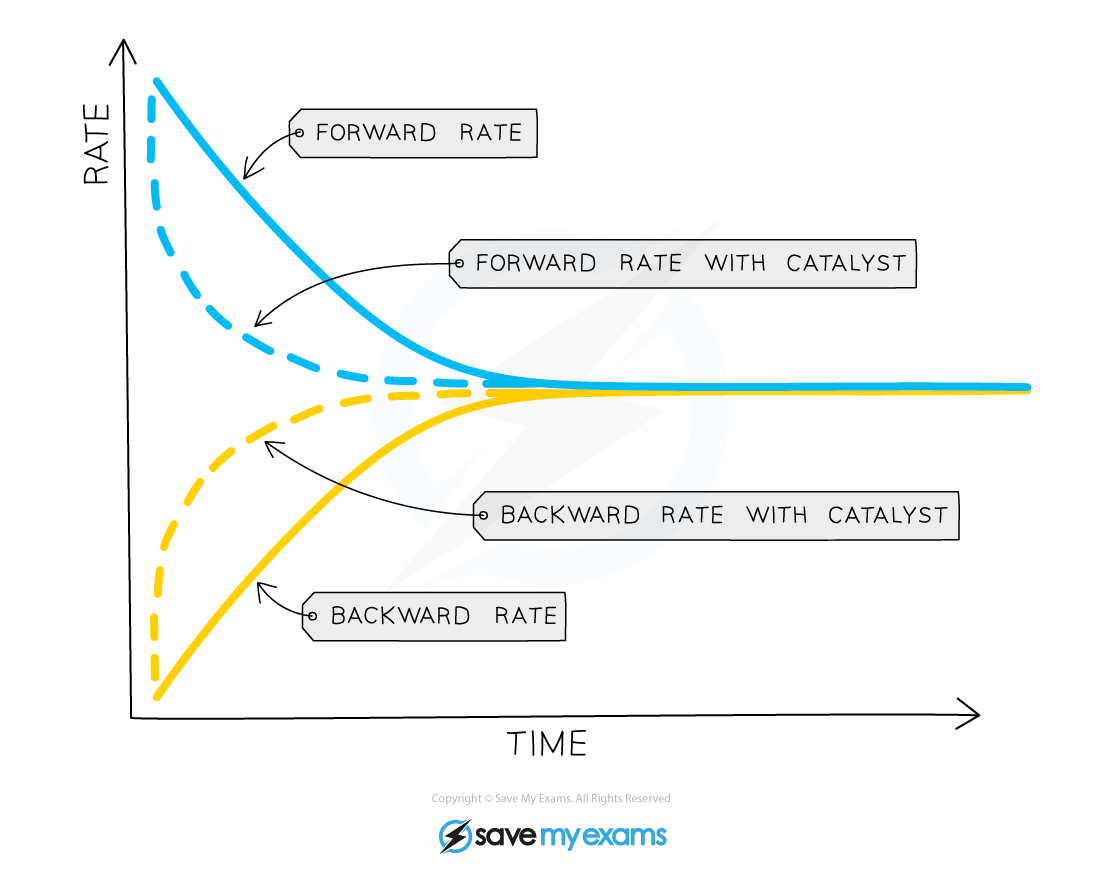
What is the effect of a catalyst on the equilibrium?
not affect the position of equilibrium but it does increase the rate at which equilibrium is reached
As catalyst increases the rate of both the forward and backward reactions by the same amount (by providing an alternative pathway requiring lower activation energy)
As a result, the concentration of reactants and products is nevertheless the same at equilibrium as it would be without the catalyst