Immune System & Pathogen Avoidance
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
what are some of the first line defences
skin
enzymes
lysozymes
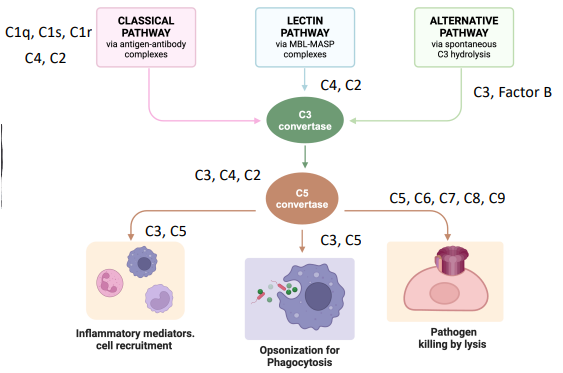
what are the 3 pathways for complement
classical pathway
lectin pathway
alternative pathway
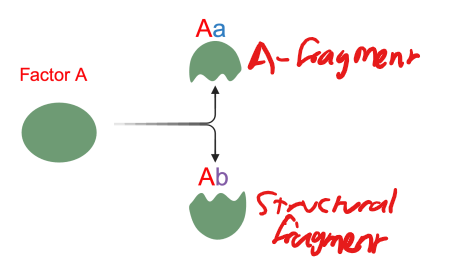
what are proenzymes
enzymes that must be cleaved to be activated, 2 fragments
structural - helps form other complexes
A-fragment - inflammatory mediators
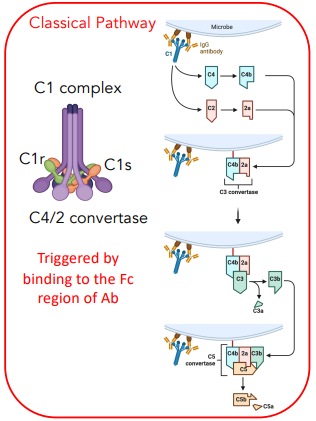
explain the classical pathway
consists of the C1 complex (C1q, C1r, C1s)
binds to microbe, converts C4 → C4b, C2 → 2a
C4b & 2a form C3 convertase
converts C3 → C3a + C3b
C3 convertase + C3b = C5 convertase
converts C5 → C5a + C5b
explain the lectin pathway
MBP complex (MASP1/2)
binds to microbe, converts C4 → C4b, C2 → 2a
C4b & 2a form C3 convertase
converts C3 → C3a + C3b
C3 convertase + C3b = C5 convertase
converts C5 → C5a + C5b
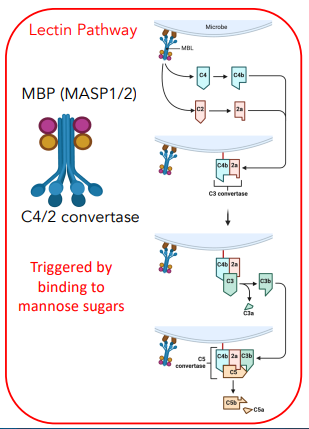
how are the classical & lectin pathway different in activation
classical = triggered by binding to Fc region of antibody
lectin = triggered by binding to mannose sugars
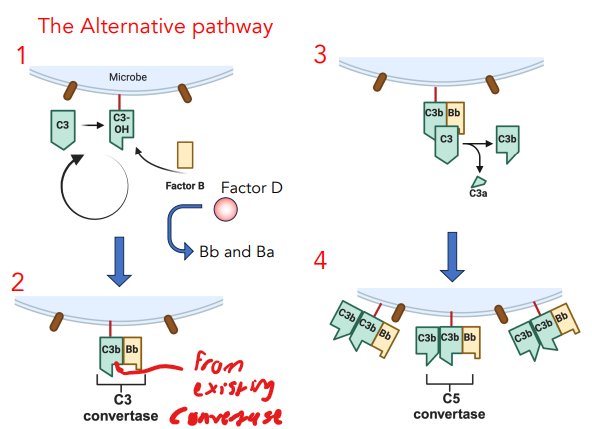
explain the alternative pathway
C3 spontaneously undergoes hydrolysis
binds factor B, cleaved by Factor D → Bb + Ba
C3b + Bb = C3 convertase
converts C3 → C3b + C3a
C3b + C3b + Bb = C5 convertase
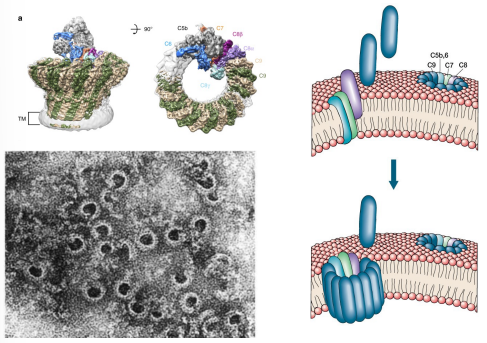
explain how the membrane attack complex works
C5 convertase recruits C5-C9
proteins are recruited in a “circular” fashion hence forming pores in the membrane of the microbe
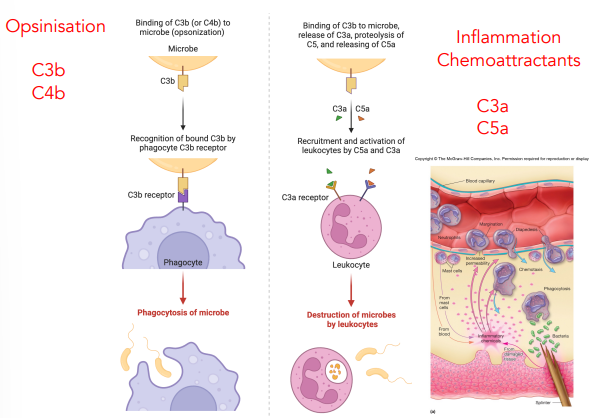
explain how opsonization works
C3b on microbes can bind to C3b receptors on phagocytes
induced endocytosis → phagocytosis
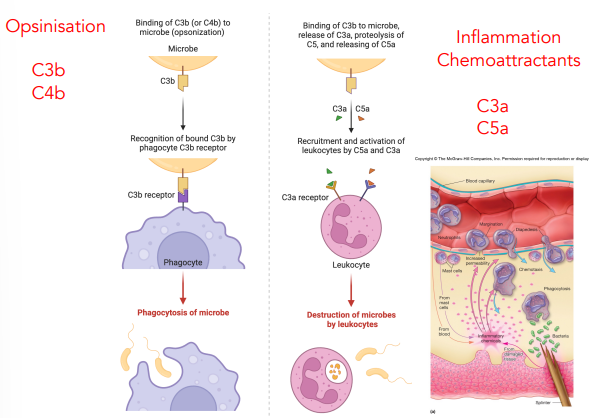
explain chemoattractants
C3a & C5a recruit & activate leukocytes
both can bind to receptors on leukocytes
leukocytes can destroy microbes
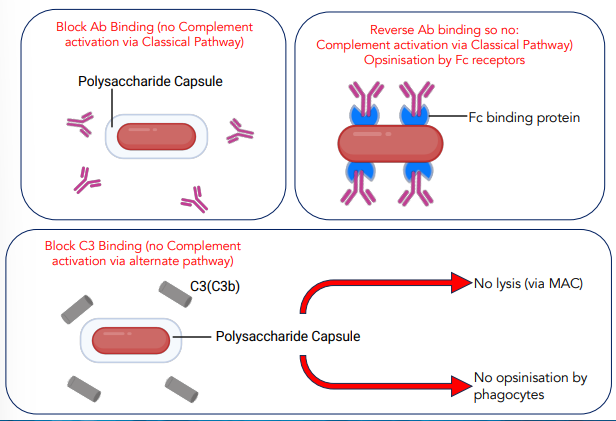
what are some ways that microbes can avoid complement
possessing a capsid, physically blocks complement
Fc binding proteins, binds to antibody Fc hence preventing complement action
how are pathogens detected by the immune system
pattern recognition receptors (e.g. toll-like receptors)
recognize different PAMPs
all drives NF-kB transcription factor
what is NF-kB
a transcription factor responsible for synthesis of pro-inflammatory mediators
TNF-α
IL-6
Pro-IL-1β
describe the process of interleukins synthesis
parts of pathogen are recognised by PRRs, downstream phosphorylation of NF-kB
translocate into nucleus
promotes gene expression for pro-interleukins
pro-form = inactive
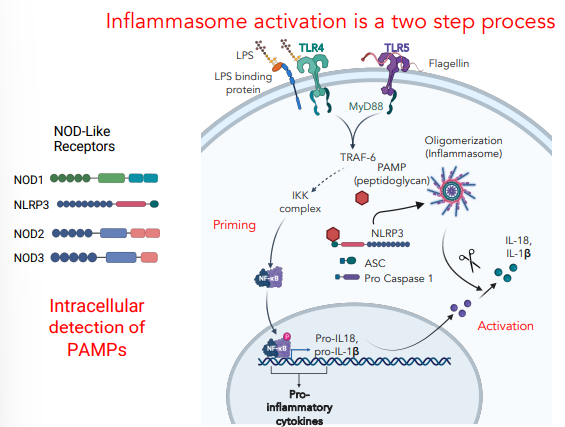
describe the process of inflammasome activation
PAMPs invade cell → recognised by nod-like receptors
causes oligomerization of nod-like receptors, ASC & pro caspase 1
this forms inflammasome (this activates pro caspase)
inflammasome cleaves & activates pro-interleukins
how can bacteria interfere with the inflammasome activation
intercept signaling events
degrade NF-kB subunits
what is some information on NK cells
contain granules with cytolytic molecules
perforin & granzyme B
can also produce inflammatory mediators
interferon-y
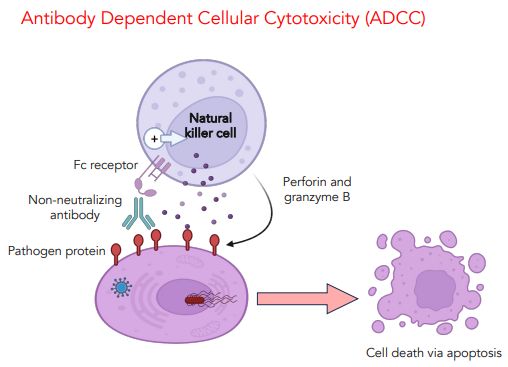
what is antibody dependent cellular cytotoxicity
when an antibody binds to a pathogen, the Fc receptor on NK cells can bind to the antibody
this triggers the degranulation of perforin & granzyme B
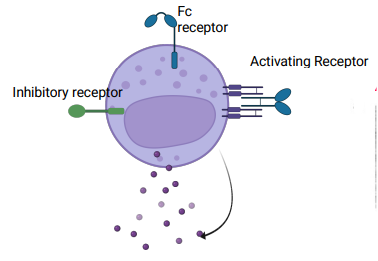
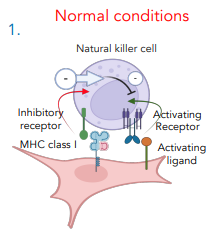
how do NK cells function normally
contain an inhibitory & activating receptor
inhibitory receptor bind MHC I receptors
this suppresses the activity of the NK cell
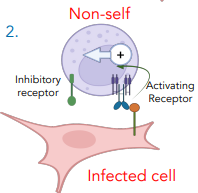
how do NK cells function against non-self pathogens
pathogens DON’T expression MHC I, thus activating receptor is triggered
degranulation occurs, pathogen dies
pathogens downregulated MHC I expression by interfering with the biochemical pathway
how can pathogens evade NK cells
express MHC I like molecules
block cell expression of activating ligands
what are the contents of granules in eosinophils, basophils, mast cells
vasodilators - prostaglandins, leukotrienes
degrading enzymes - proteases, lipases, etc.
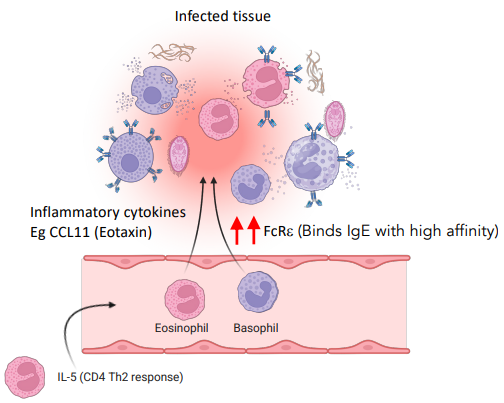
how do basophils & eosinophils function
circulate in the body, when moved to tissues via cytokines, they bind to pathogens via IgE receptors
release histamine, vasodilators.
what are some cells of the adaptive immune system
B cells - antibody production
CD4+ T cell - maturation of adaptive response
CD8+ T cell - recognition & removal of viral host cells
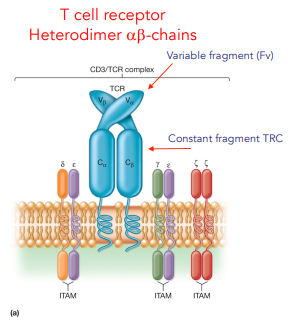
explain the design of the T cell receptor
2 chains α & β, each chain has 2 domains
proximal = constant
distal = variable (binding)
explain the design of the B cell receptor
pair of heterodimers (heavy + light chains)
constant Fc region
variable Fv region
associates with other protein for signaling clonal exp.
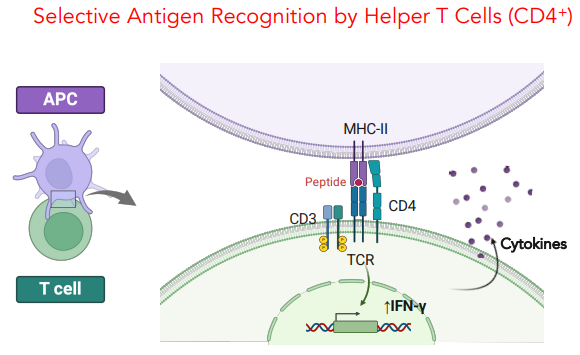
describe selective antigen recognition by Helper T cells (CD4+)
CD4+ binds to APC via MHC II (stabilized by CD28 ligand)
triggers CD3 phosphorylation
causes expression of IFN-y which is released
describe selective antigen recognition by cytotoxic T cells (CD8+)
T cell binds to antigen-MHC I complex on abnormal cells
stabilized by CD28 ligand
delivers granzymes & perforin to cell
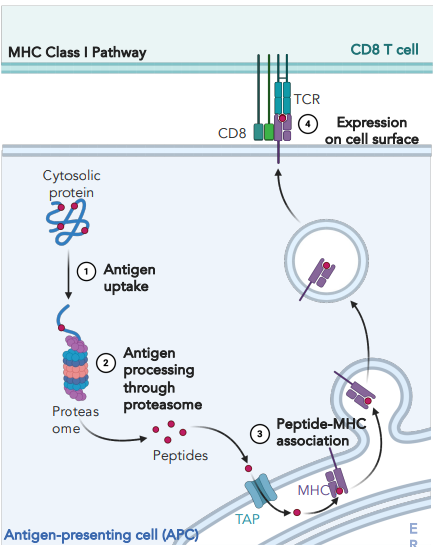
explain how antigen-MHC I complexes are formed
cytosolic protein is broken down by proteasome
peptides are transported to ER by TAP transporter
empty MHC I is loaded with peptide
complex is transported to surface via vesicle
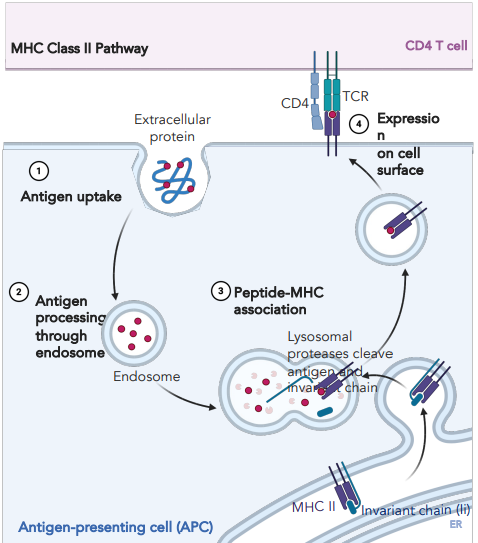
explain how antigen-MHC II complexes are formed
endocytosis of extracellular protein, processed through endosome
MHC II with pre-loaded invariant chain is dispatched
endosome and vesicle with MHC II combine
invariant chain is displaced by peptide
vesicle is transported to surface
how can pathogens interfere with MHC I/II pathways
preventing processing (proteasome)
preventing transport of vesicle
describe co-stimulation in T cells
CD28 ligand is expression on naive T cells
contains an immune tyrosine activating motif (ITAM)
triggers phosphorylation of signaling kinases
induces signaling for cell survival
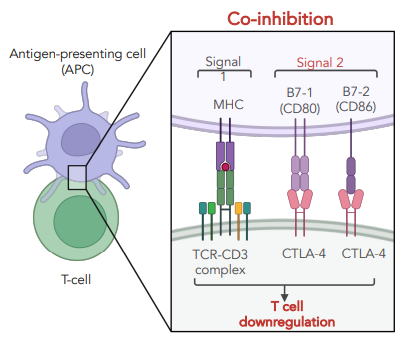
how do you stop T cell proliferation
CTLA-4, has higher affinity for CD80/CD86
contains an ITIM motif (inhibitory) competitor for CD28
upregulated after activations
dephosphorylates signaling tyrosine kinases
what are PD-1 & TIM3 checkpoints
receptors that act as phosphatases (dephosphorylates) activated kinases
limits effector function
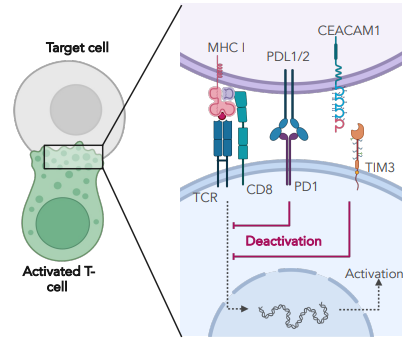
how might chronic infection result in sustained expression of checkpoint molecules
if an infection cannot be cleaned up, the constant activation of T cells also drives the constant activation of inhibitory signals'
thus T cells cannot be restimulated
infection persists
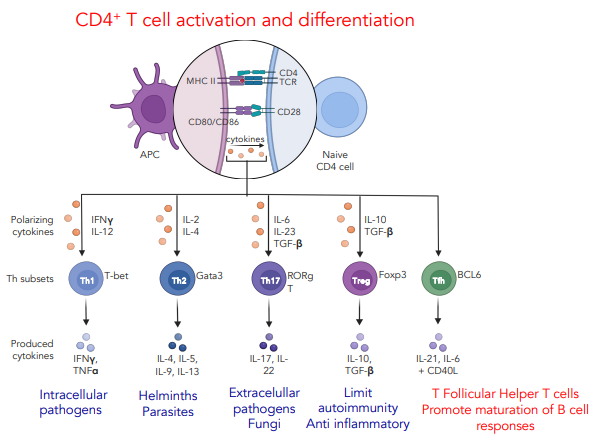
what is the role of cytokines in CD4+ T cell function
different cytokines stimulate different T cell responses
what are T follicular helper cells
specialised CD4+ T cells which help B cells produce antibodies
provide costimulation (CD4 & CD40L)
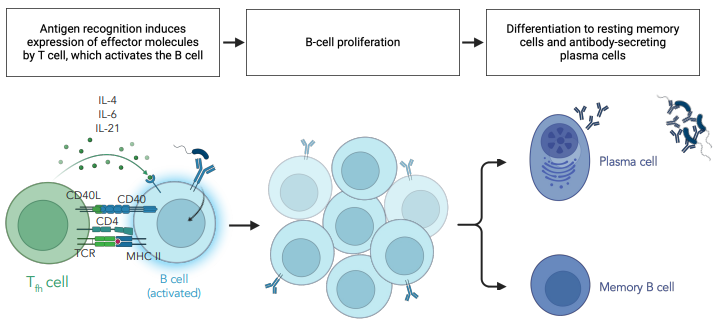
what is the general process of B cell activation via Tfh cells
B cell endocytosis of antigen & receptor
antigen is loaded on MHC II, transported to surface
Tfh cell binds via MHC II, CD4 & CD40L
releases cytokines allowing expansion
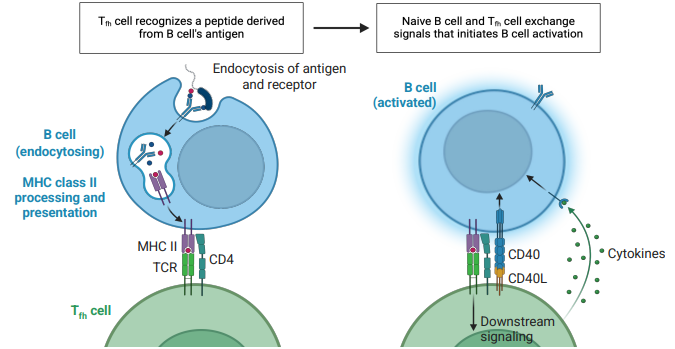
explain B cell activation in the lymph node
lymph node is split into a B cell & T cell area
B cell expresses CCR7 chemokine attracting Tfh cells
Tfh cell expresses CXCR5 chemokine attracting B cell
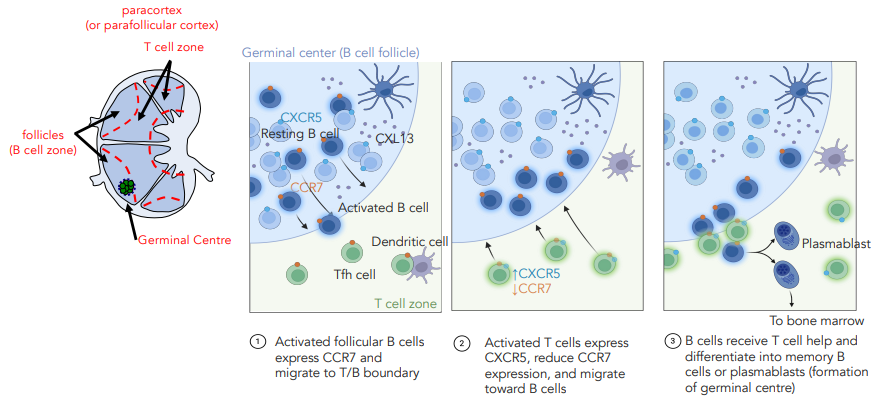
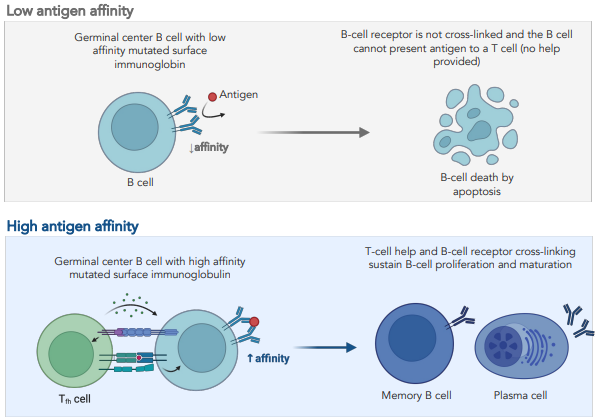
what is affinity maturation
introduce mutations within Ig gene regions
if mutations increase affinity cell get selective survival
i.e. if B cell cannot bind antigens no survival signals
if B cell binds induces survival signals & proliferation
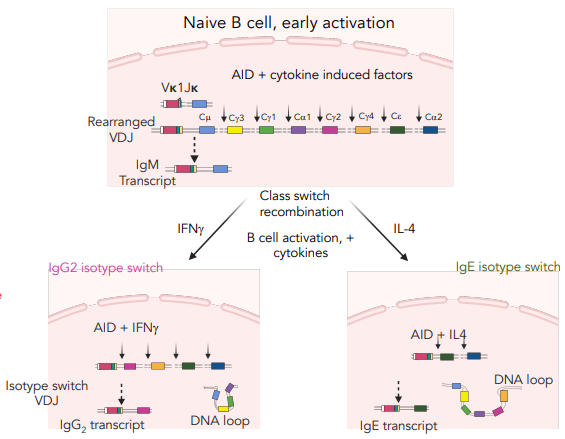
what is isotype switching
after VDJ recombination for the Fv region, AID & cytokine induced factors can alter the Fc region of an antibody
based on presence of interferon/interleukin
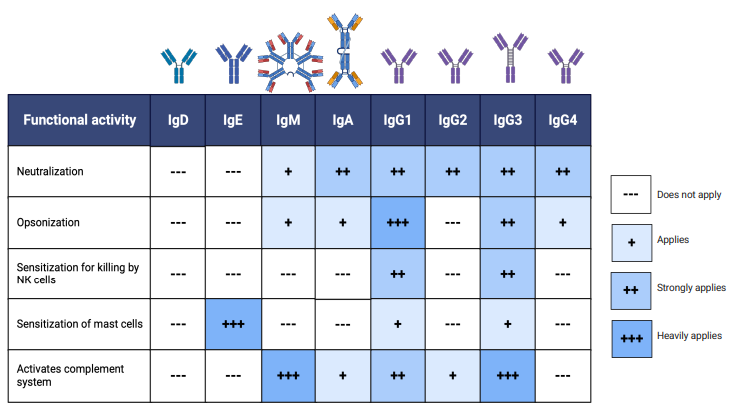
what are some qualities of different antibodies
how can pathogens evade antibody responses
blocking Ab binding (capsule)
reverse Ab binding (receptors for Fc region)
antigen variation (genetic mutations)