a level physical
1/251
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
252 Terms
what is pressure?
the force exerted by gas molecules in a given volume
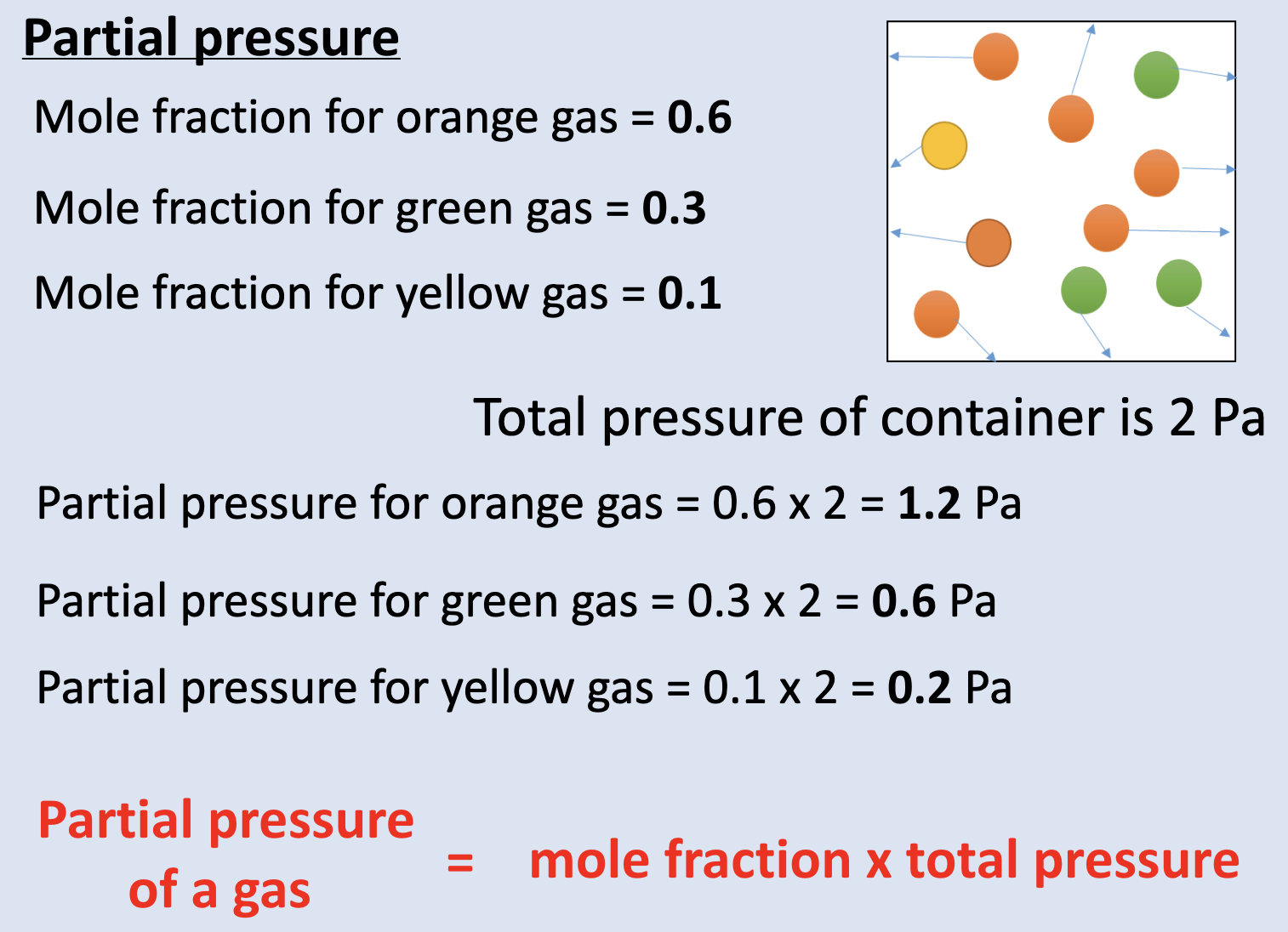
what is partial pressure?
the portion of the total pressure exerted by the presence of a single type of gas molecule
give the eqn for the mole fraction:
no. of moles of a gas / total no. of moles of gas

what does the sum of the mole fractions always = ?
1

what is Kp? what is it deduced from?
the equilibrium constant calculated from partial pressures for a system at constant temperature
deduced from the eqn for a reversible reaction occurring in the gas phase
give the eqn for partial pressure:
partial pressure = mole fraction x total pressure
how do you write a Kp expression and deduce its units?
same as Kc - instead of using [], use () and write pp before each thing (to show it’s a partial pressure)
deduce units by cancelling (same as Kc)
how does temperature affect Kp?
if the temperature change causes the equilibrium to shift right, Kp increases
if the temperature change causes the equilibrium to shift left, Kp decreases
how does a catalyst affect Kp?
no effect!
why might Kp have no units?
moles of products = moles of reactants (so units cancel)
what is an acid?
acid = H+ donor
what is a base?
base = H+ acceptor
what does acid-base equilibria involve?
transfer of protons
what denotes a strong acid?
acid which completely ionises in water to give H+ (e.g. HCl → H+ + Cl-)
what is a concentrated acid?
acid w/ a larger amount of acid particles in a given volume
what is the formula used to calculate pH?
pH = -log10 [H+]
what is the formula used to calculate [H+] ions?
[H+] = 10-pH
give the general eqn for acids in aqueous soln:
HA + H2O → H3O+
OR HA → H+ + A-
what is a monoprotic acid? give 2 examples:
acid which donates one proton for every acid molecule
so [H+] = 1 mol dm-3
e.g. HCl, HNO3
what is a diprotic acid? give an example:
acid which donates 2 protons for every acid molecule
so [H+] = 2 mol dm-3
e.g. H2SO4
what is a triprotic acid? give an example:
acid which donates 3 protons for every acid molecule
so [H+] = 3 mol dm-3
e.g. H3PO4
give the expression for the ionic product of water, Kw:
Kw = [H+][OH-]
explain why [H2O] is not shown in the Kw expression:
[H2O] is (almost) constant
explain why the value of Kw increases as the temperature increases (2)
equilibrium is endothermic
equilibrium shifts to the RHS to minimise/oppose temp increase (so more H+ and OH- formed)
explain why water is neutral:
[H+] = [OH-] - dissociation of each water molecule gives 1 H+ and 1 OH-
a pH meter is calibrated using a calibration graph - to create the calibration, the pH meter is used to measure the pH of separate sons, each w/ a known, accurate pH.
suggest why the pH probe is washed w/ distilled water between each of the calibration measurements (1)
either:
different solns must not contaminate each other
to wash off any residual soln/substance (which could interfere w/ the reading)
a pH meter is calibrated using a calibration graph - to create the calibration, the pH meter is used to measure the pH of separate solns, each w/ a known, accurate pH.
the calibrated pH meter is used to monitor the pH during a titration of HCl w/ NaOH - explain why the vol of NaOH soln added between each pH measurement is smaller as the end point of the titration is approached (1)
to avoid missing the endpoint (mention large pH change if pH change is large)
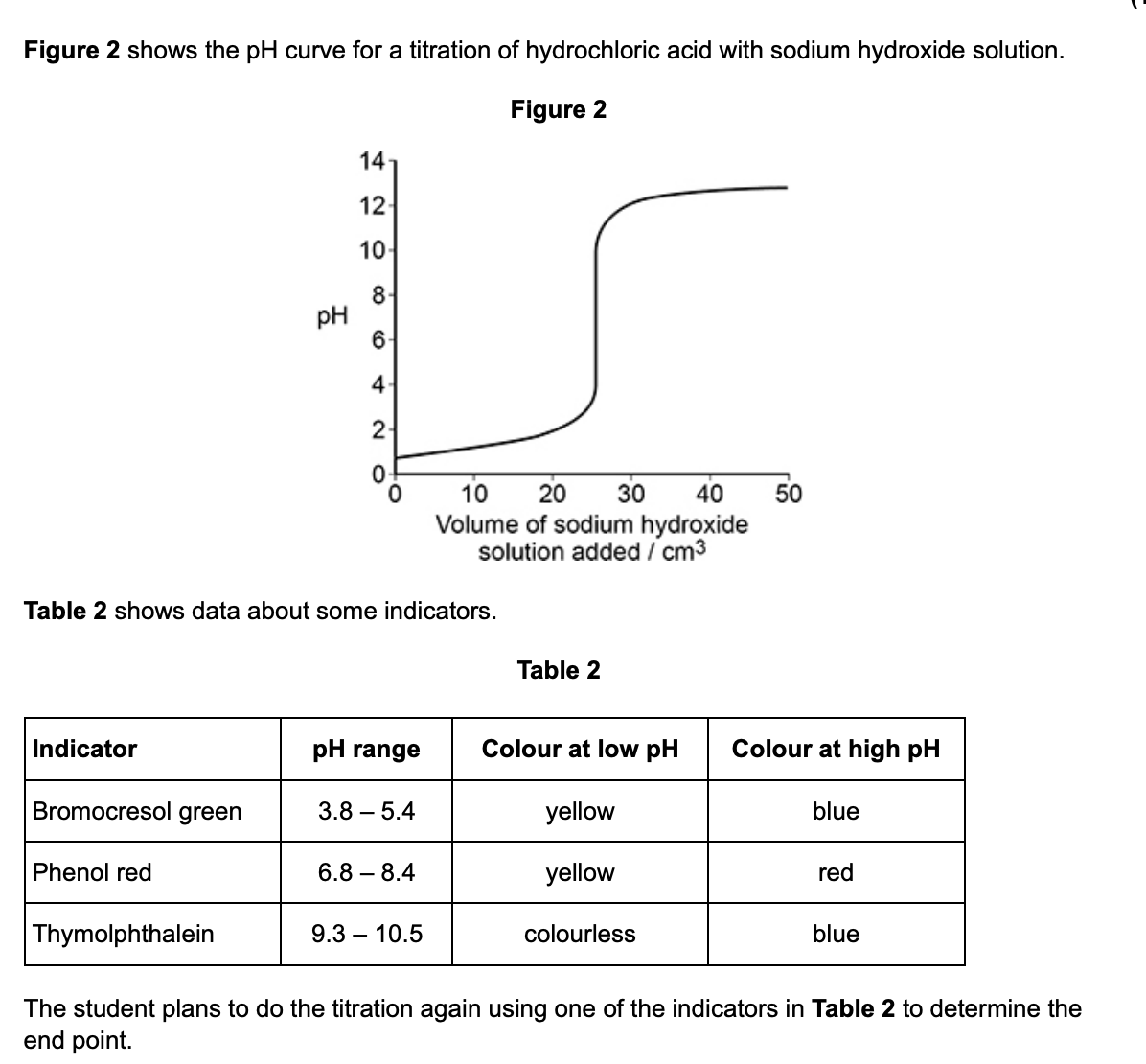
state why all 3 of the indicators in Table 2 are suitable for this titration (1)
all have a colour change/pH range w/in the steep/vertical part of the titration curve
for pure water at 40oC, pH = 6.67.
a student thought that the water was acidic - explain why the student was incorrect (1)
[H+] = [OH-]
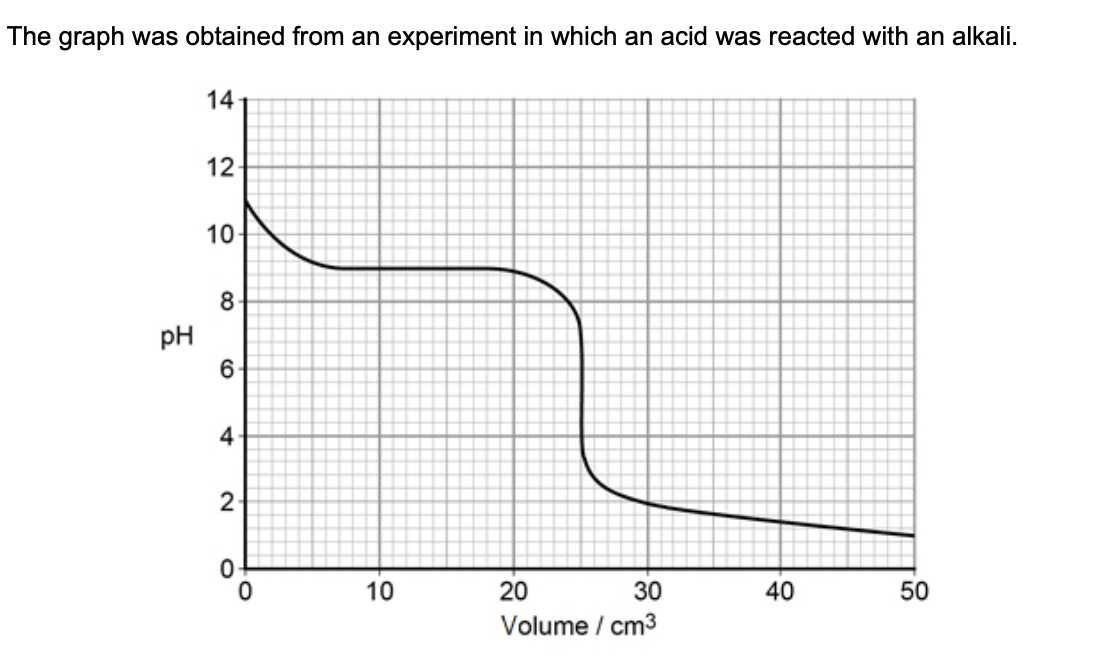
suggest briefly a practical procedure that a student could use to obtain data from which the curve in the graph could be plotted (3)
place a fixed vol of alkali in a flask or beaker
add acid in small portions from a burette
stir and use a pH meter to record the pH after each addition of acid
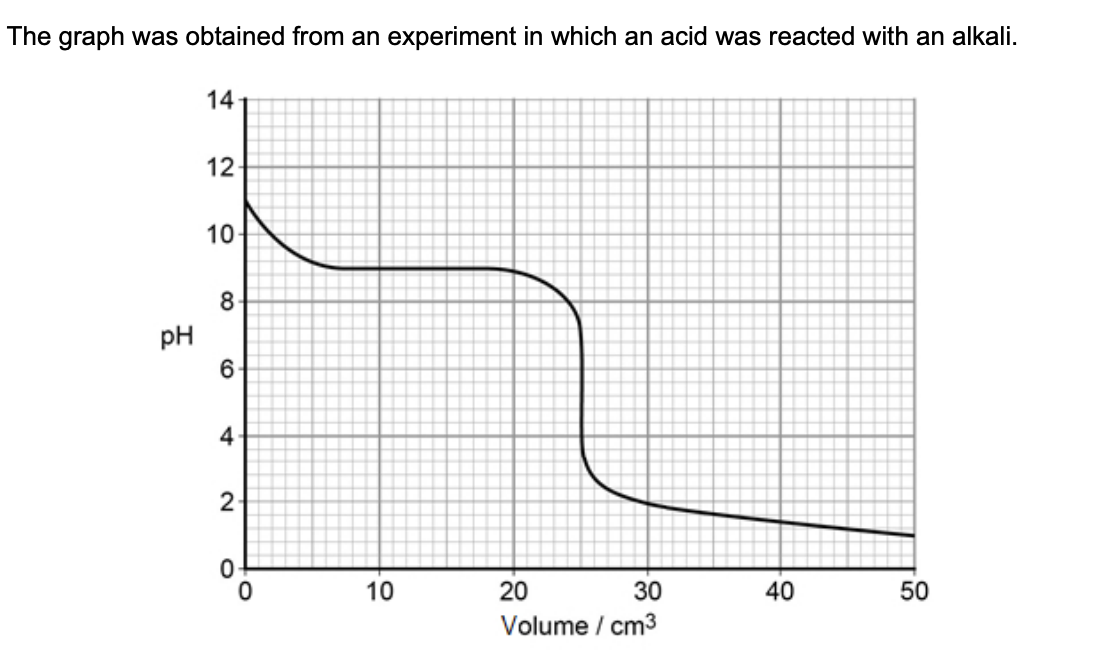
the student was provided w/ samples of 3 different indicators - suggest how the practical procedure could be refined by the student to identify the most suitable indicator (2)
repeat the experiment w/ each indicator
select the indicator that changes colour rapidly when the pH changes from about 7 to 4
explain briefly why a pH meter should be calibrated before use (1)
over time/after storage meter does not give accurate readings
what denotes a weak acid? give the eqn for this:
only slightly dissociate in aq soln
HA ⇌ H+ + A-
what denotes a weak base?
dissociates only slightly in aq soln
give the expression for Ka, the acid dissociation constant:
Ka = [H+][A-] / [HA]
give the expression for pKa:
pKa = -log10 (Ka)
give the expression for calculating Ka from pKa:
Ka = 10-pKa
how does pKa vary w/ the strength of the acid?
the larger the pKa, the weaker the acid
the smaller the pKa, the stronger the acid
explain how Ka varies w/ the strength of an acid according to the PoE:
the bigger the Ka, the further the PoE lies to the RHS and the stronger the acid
the smaller the Ka, the further the PoE lies to the LHS and the weaker the acid
give the assumption we make for weak acids and give the expression derived from this:
[H+] = [A-]
so Ka = [H+] / [HA]
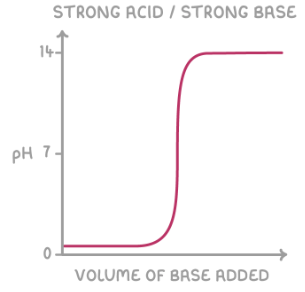
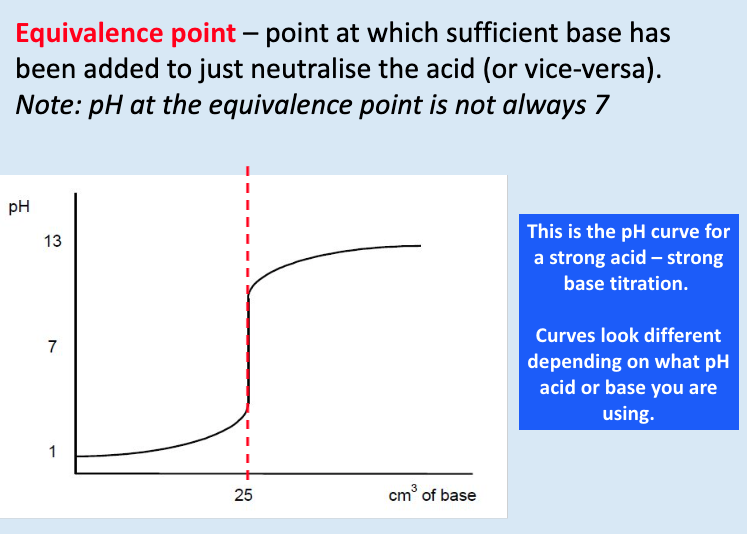
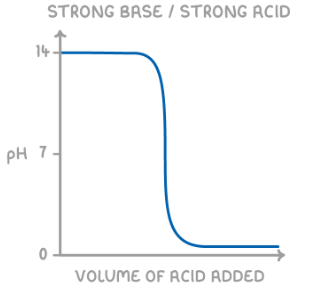
give the shape of the titration curve for a strong base added to a strong acid:
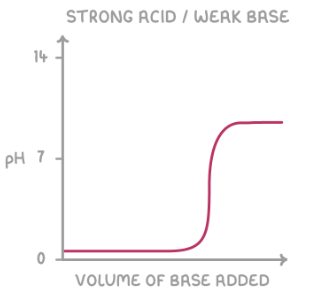
give the shape of the titration curve for a weak base added to a strong acid:
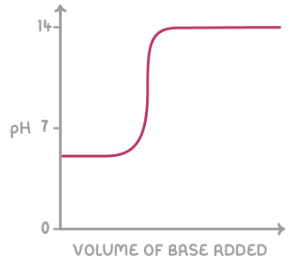
give the shape of the titration curve for a strong base added to a weak acid:
give the shape of the titration curve for a weak base added to a weak acid:
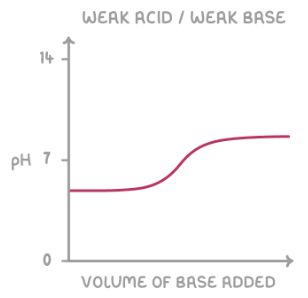
what is the equivalence point?
point at which sufficient base has been added to just neutralise the acid/base
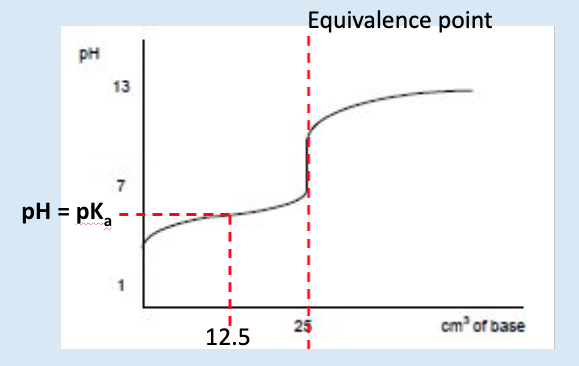
how can we calculate pKa from a titration curve?
pH at half neutralisation = pKa
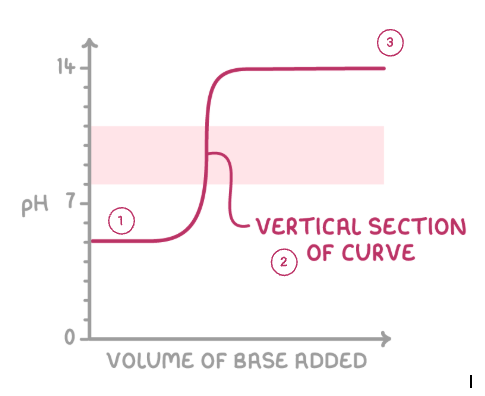
explain the general shape of a pH curve:
pH changes little as acid is in XS
equivalence point - point at which sufficient base has been added to just neutralise the acid
pH changes little as base in XS
give the general shape of a pH curve if an acid is added to a base:
what do you need to take into account when choosing a suitable indicator?
should change colour over a narrow range and at the vertical section of the curve
give the method for calibrating the pH probe:

give the method for measuring the pH of the mixture of acid and alkali:

give the general eqn for the reaction of a weak acid w/ a strong base - give the assumption we cannot make:
HA + OH- → A- + H2O
we cannot make the assumption that [H+] = [A-]
what is a buffer soln?
soln which maintains an approximately constant pH, despite dilution or addition of small amounts of acid or base
what is an acidic buffer - what do they contain and why do they work?
maintain pH below 7
made from a weak acid (HA) and a soluble salt of the acid (A-)
dissociation of acid is at equilibrium (HA ⇌ H+ + A-)
describe what happens when a small amount of base is added to an acidic buffer:
OH- react w/ H+ in buffer, removing some H+
the PoE for the dissociation of HA ⇌ H+ + A- (change eqn to specific Q) moves to the right to replace the H+ which reacted w/ the OH-
OH-+ HA → H2O + A-
this keeps the pH almost constant

describe what happens when a small amount of acid is added to an acidic buffer?
H+ conc increases so PoE of HA ⇌ H+ + A- shifts left
soluble salt of the acid provides A- to react with added H+ so pH remains roughly unchanged
A- + H+ → HA

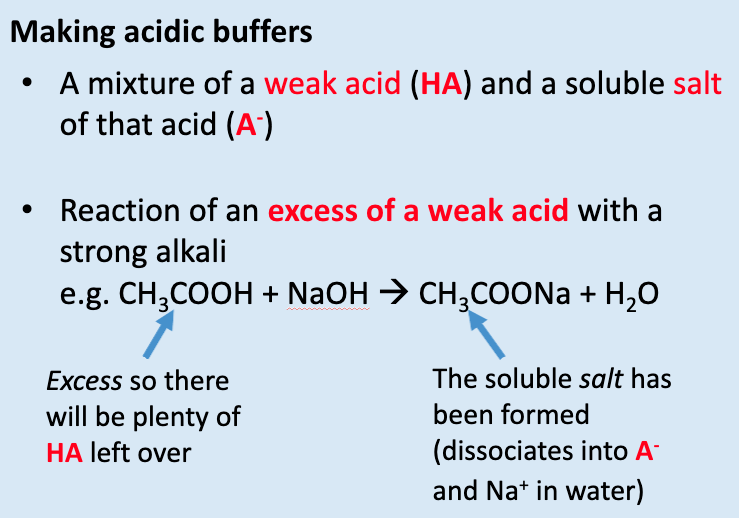
give the 3 methods of making acidic buffers:
mixture of weak acid and soluble salt of acid
add XS of weak acid to strong alkali
½ neutralise acid to make buffer w/ pH = to pKa
what is a basic buffer? what do they contain?
maintains pH above 7
mixture of weak base and soluble salt of that base
dissociation of base at equilibrium
B + H2O ⇌ BH+ + OH-
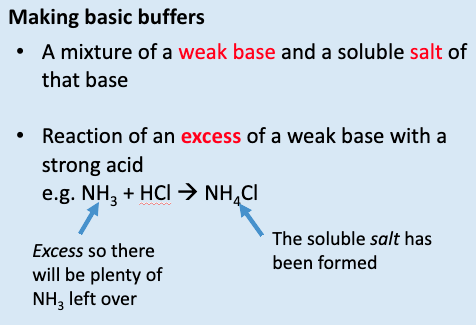
give 2 methods of making basic buffers:
mixture of a weak base and soluble salt of that base
reaction of an XS of a weak base w/ a strong acid
what happens when a small amount of acid/base is added to the basic buffer?

what is the standard electrode potential (Eθ)?
p.d. measured under standard conditions when the ½ cell is connected to a standard hydrogen electrode
a measure of where the PoE lies for each ½ cell
how can we relate electrode potentials to PoE?
the more +ve the electrode potential, the further right the PoE
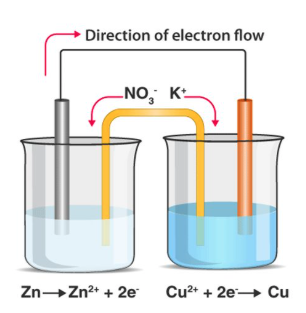
how are ½ equations always written for standard electrode potentials?
always written as reduction (see image for e.g.)

what can we use to measure the Eθ for a single ½ cell?
standard hydrogen electrode = electrode consisting of H2 gas in contact w/ H+ ions on a platinum surface
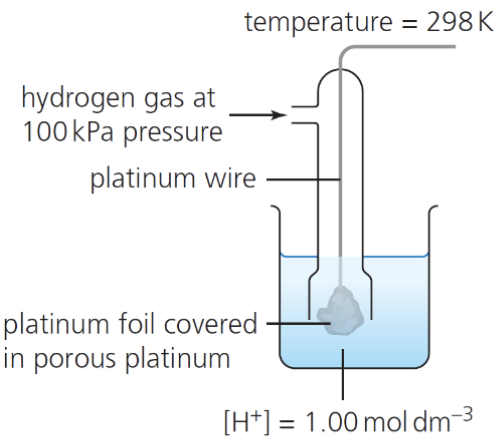
describe how we can use the standard hydrogen electrode to measure the Eθ for a single ½ cell:
H2 gas flows over the inert Pt electrode to establish the equilibrium 2H+ (aq) + 2e- ⇌ H2 (g)
the standard hydrogen electrode is connected to the ½ cell under investigation under standard conditions
the Eθ of the standard hydrogen electrode is 0.00 V by definition
so we can determine the Eθ by using the formula Ecell = Eright - Eleft
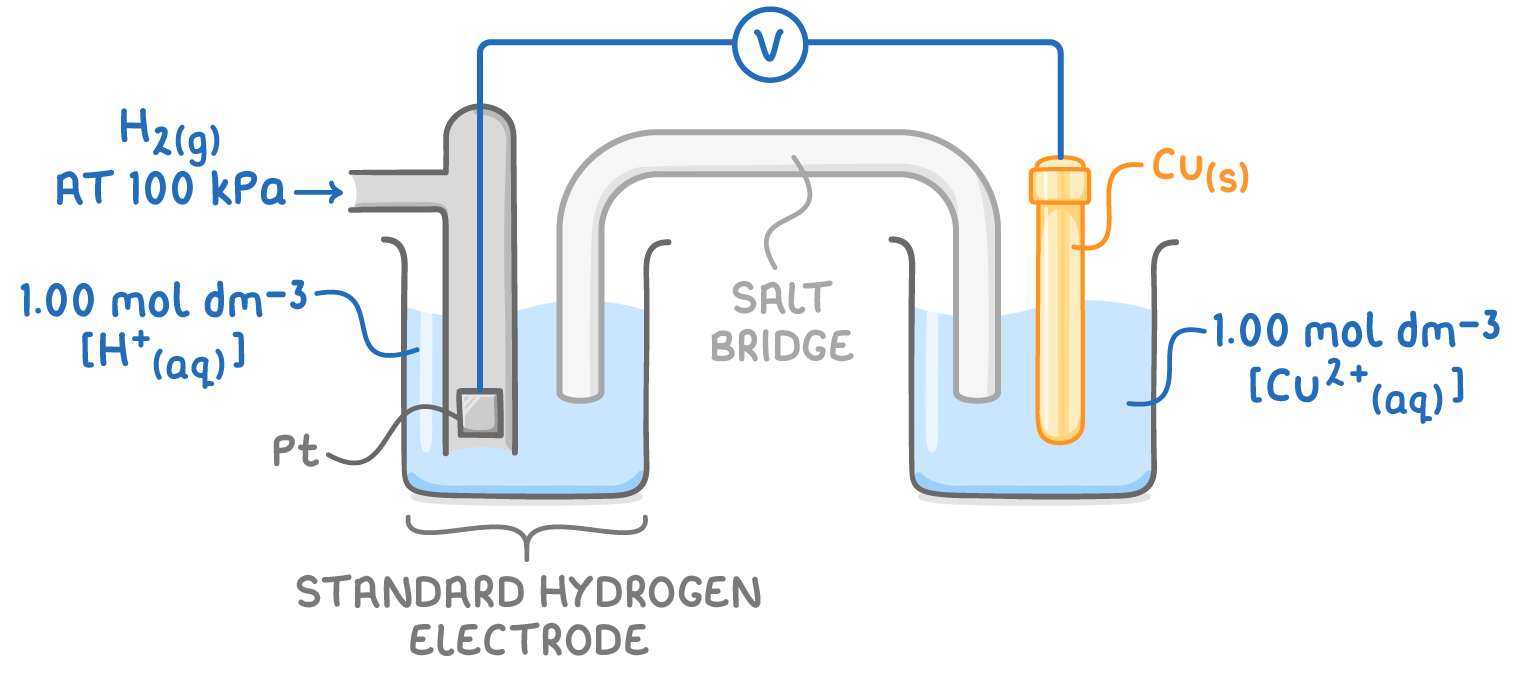
state the standard conditions necessary when using the standard hydrogen electrode:
298 K
H2 gas at 100 kPa
all solns have a conc of 1 mol dm-3
which side is the standard hydrogen electrode always positioned on?
LHS - regardless of Eθ values of other cells
give the formula used to calculate the EMF (Ecell) of a cell:
Ecell = Eright - Eleft
give the symbols used in cell notation and state what they represent:
comma for those in the same state!

which species is placed closest to the salt bridge || when writing cell notation?
species w/ the highest oxidation state (i.e. oxidised species)
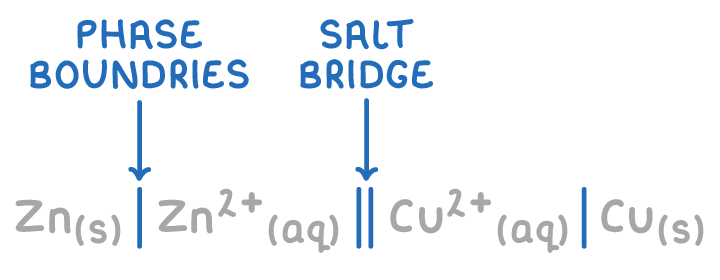
on which side of the salt bridge || do you write each species in cell notation?
as seen/given in a diagram:
-ve ½ cell (oxidised) on LHS
+ve ½ cell (reduced) on RHS
why is Pt used as the contact electrode in the standard hydrogen electrode?
inert
give and explain the cell notation for the standard hydrogen electrode being used to measure the EMF of a Cu/Cu2+ ½ cell:
Pt (s) | H2 (g) | H+ (aq) || Cu2+ (aq) | Cu (s) :
species w/ highest oxidation state closest so salt bridge
| separate species w/ diff states
Pt (s) included - used as a contact: solid contact must be used if there are only aq/gaseous species

you are provided with the Daniell cell referred to in part (b), including a Zn electrode of known mass - briefly outline how you could carry out an experiment to confirm your answer to part (b) (3)
allow cell to discharge until [CuSO4] = 0.5 mol dm-3
confirm using colorimetry
weigh mass of Zn electrode before and after experiment
![<ul><li><p>allow cell to discharge until [CuSO<sub>4</sub>] = 0.5 mol dm<sup>-3</sup></p></li><li><p>confirm using colorimetry</p></li><li><p>weigh mass of Zn electrode before and after experiment </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/07b94776-d6dd-4cbb-9f6e-5fa11fa6a424.png)

use data from the table above to explain why Au+ ions are not normally found in aq soln (2)
Eθ of Au+ bigger than Eθ of H2O
so Au+ ions oxidise H2O

use data from the table to predict and explain the redox reactions that occur when iron powder is added to an XS of aq silver nitrate (3)
Eθ of Ag+ bigger than Eθ of Fe2+
Eθ of Ag+ bigger than Eθ of Fe3+
so Ag+ oxidise Fe → Fe2+ and → Fe3+

give the features of non rechargeable cells and explain why they are necessary:
porous separator - allows ions to move/flow/transfer
carbon rod - made from graphite to allow e- to flow
zinc - container for cell (gets used up)
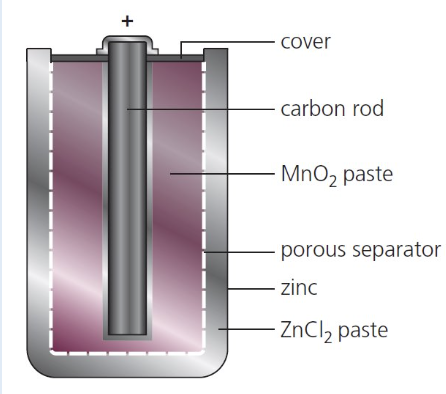
give an example of a non rechargeable cell:
either:
alkaline battery
dry cell
suggest why a non rechargeable cell often leaks after being used for a long time (1)
Zn oxidised

give an example of a rechargeable cell and give the 2 ½ eqns associated w/ it:
lithium ion:
Li+ + CoO2 + e- ⇌ LiCoO2 (+ve electrode)
Li+ + e- ⇌ Li (-ve electrode)
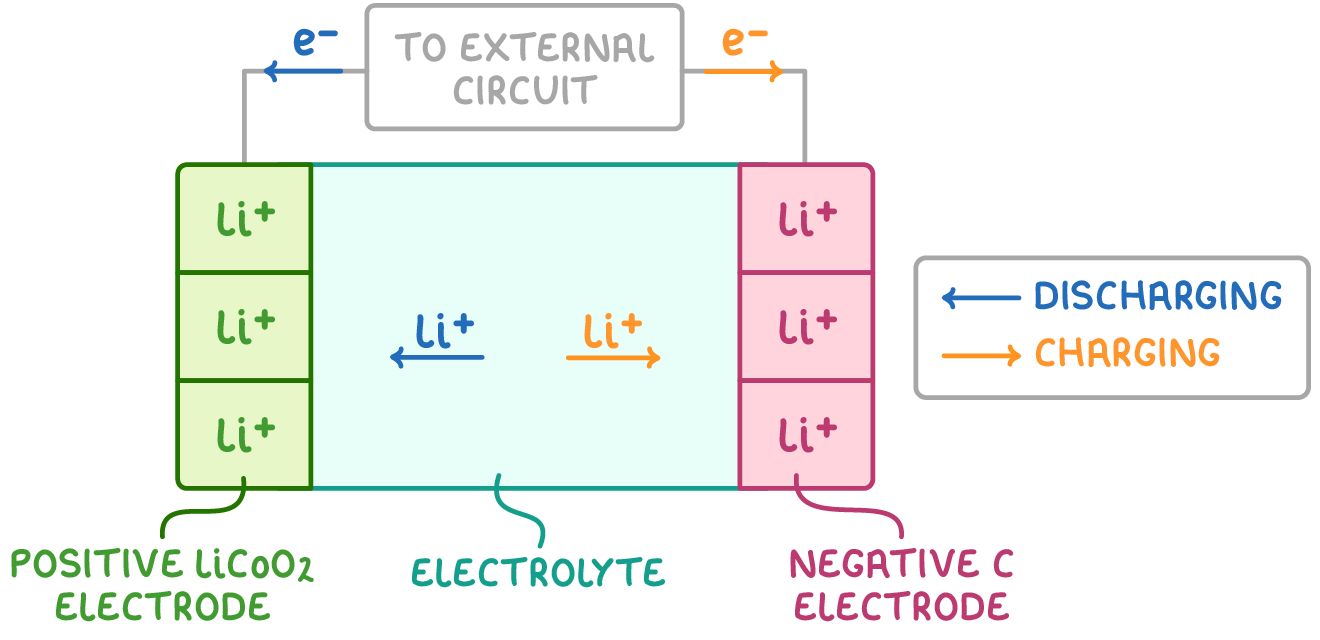
give the overall eqn for the reaction that occurs in a lithium-ion cell:
Li + CoO2 ⇌ LiCoO2
for a rechargeable reaction, what do the different directions of the reaction apply to?
forward reaction = discharge (being used)
backward reaction = recharge (being charged)
give the 3 types of cell:
non rechargeable (irreversible)
rechargeable (reversible)
fuel cells
describe how an alkaline hydrogen-oxygen fuel cell works:
at the +ve electrode, ½ O2 reacts w/ H2O to produce 2OH- and 2e- - O2 is reduced
OH- diffuse across membrane
at the -ve electrode, H2 enters and reacts w/ OH- to produce 2H2O
e- travel along circuit to +ve electrode - process repeats as long as there is a constant supply of fuel
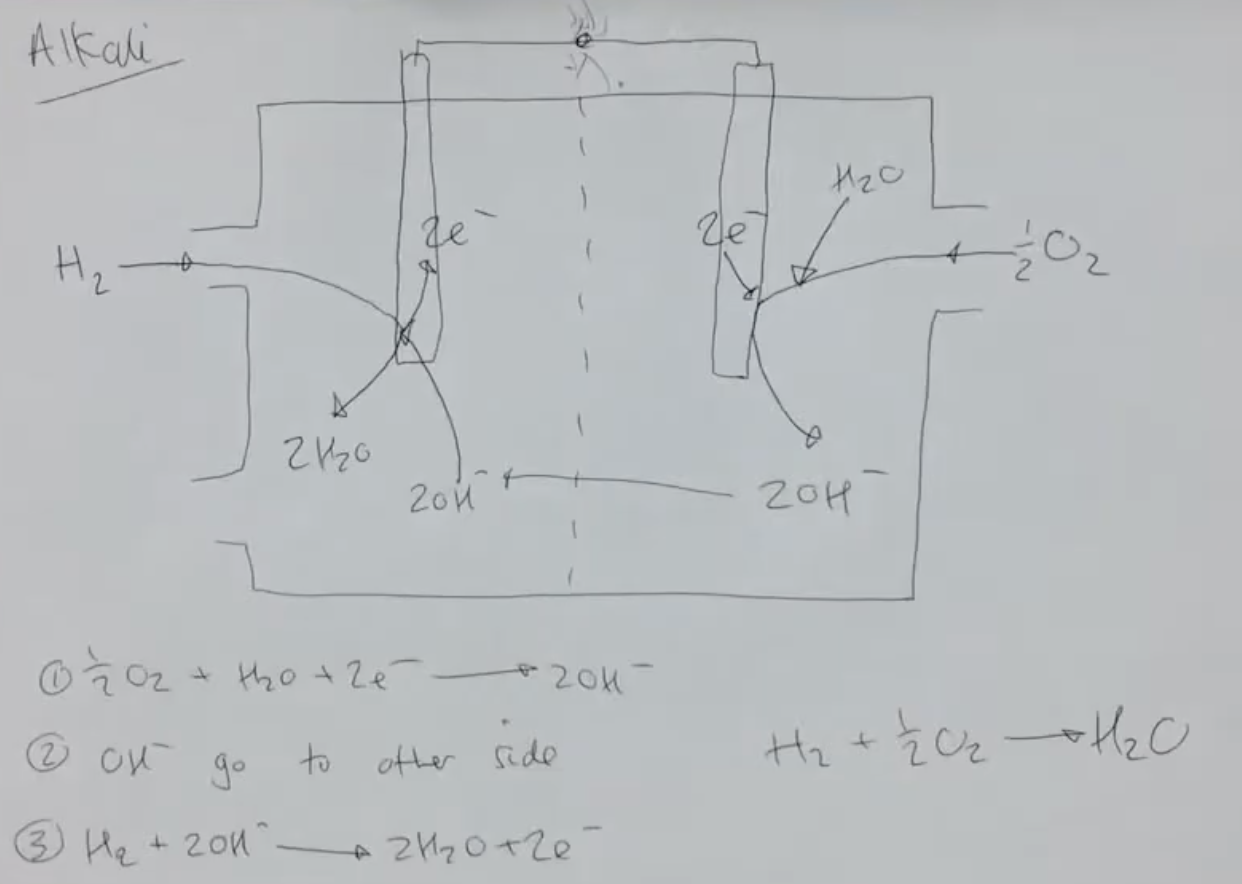
give the eqn for the reaction at the -ve electrode of an alkaline hydrogen-oxygen fuel cell:
H2 + 2OH- → 2H2O + 2e-
give the eqn for the reaction at the +ve electrode of an alkaline hydrogen-oxygen fuel cell:
O2 + 2H2O + 4e- → 4OH-
give the overall eqn for the reaction that occurs in an alkaline hydrogen-oxygen fuel cell:
2H2 + O2 → 2H2O
give a benefit of using non-rechargeable cells:
cheap
give a drawback of using non-rechargeable cells:
difficult to dispose of/waste issues
give 3 benefits of using rechargeable cells:
less waste as metal compounds reused
cheaper in the long run
lower environmental impact
give 2 drawbacks of using rechargeable cells:
need E source to recharge
some waste at end of useful life
give 3 benefits of using hydrogen fuel cells:
converts more of the available E from combustion of H2 into KE of car
no pollutants (CO2, SO2 etc.) as only waste product is H2O
efficient
no need to recharge
give 4 drawbacks of using hydrogen fuel cells:
need constant fuel supply
H2 is flammable and explosive
high cost
difficult to source H2 (usually made using fossil fuels)
suggest 1 advantage of using methanol, rather than hydrogen, in a fuel cell, for use in cars (1)
methanol (is liquid so) can be stored easily or transported easily
allow more energy can be produced from 1 cm3 of methanol (liquid) than from 1 cm3 of hydrogen (gas)
suggest why the recharging of a lithium cell may lead to the release of CO2 into the atmosphere (1)
electricity from recharging the cell may come from power stations burning fossil fuel
describe an electrochemical cell:
2 half cells - electrodes submerged in solns of own ions
half cells connected by high resistance voltmeter and salt bridge

what is the purpose of the high resistance voltmeter?
measures p.d. between electrodes - this is the EMF/Ecell
typically e- would flow from more reactive to less reactive metal but high resistance inhibits e- flow to maintain conc of ions in soln
what is the salt bridge?
filter paper soaked in saturated soluble salt soln e.g. potassium nitrate
OR a tube containing unreactive ions in an agar gel
what is the purpose of a salt bridge?
ions in the ionic substance in the salt bridge movem of ions between electrodes through the salt bridge
to complete and maintain an electric circuit
how does a salt bridge work?
to maintain electrical neutrality:
-vely charged ion moves to +ve ½ cell via salt bridge
+vely charged ion move to -ve ½ cell via salt bridge