Applied Microbiology Lab Midterm
5.0(2)
Card Sorting
1/44
Earn XP
Description and Tags
Last updated 4:37 PM on 10/31/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
1
New cards
What Biosafety Level lab are we in?
Biosafety Level II lab
2
New cards
Lab Apparel/PPE
- lab coat with top two buttons buttoned at minimum
- long pants cover the whole skin area
- long hair tied back. ponytail tucked in back of lab coat
- safety goggles
- closed toe and closed heel shoes: no sandals, clogs, mules, or crocs
- long pants cover the whole skin area
- long hair tied back. ponytail tucked in back of lab coat
- safety goggles
- closed toe and closed heel shoes: no sandals, clogs, mules, or crocs
3
New cards
Safety procedures
- no food, drink, gum, or smoking
- no putting on makeup
- all belongings should be in the cubby (backpack, computer, etc.)
- only thing at your station should be manual, pen, pencil, sharpie.
- lab coat and writing utensils should be in Ziplock bag when not in use.
- no putting on makeup
- all belongings should be in the cubby (backpack, computer, etc.)
- only thing at your station should be manual, pen, pencil, sharpie.
- lab coat and writing utensils should be in Ziplock bag when not in use.
4
New cards
How-to put-on PPE
1. Wash hands
2. Put on gloves right after washing hands
3. Disinfect goggles with disinfectant and put them on
4. Put on lab coat (button at least 2 buttons)
5. Wipe down tabletop with disinfectant
2. Put on gloves right after washing hands
3. Disinfect goggles with disinfectant and put them on
4. Put on lab coat (button at least 2 buttons)
5. Wipe down tabletop with disinfectant
5
New cards
How-to- take- off PPE
1. wipe down tabletop with disinfectant
2. disinfect gloves with disinfectant
3. unbutton lab coat
4. remove gloves and dispose of
5. remove lab coat
6. wash hands thoroughly
7. remove goggles and disinfect
2. disinfect gloves with disinfectant
3. unbutton lab coat
4. remove gloves and dispose of
5. remove lab coat
6. wash hands thoroughly
7. remove goggles and disinfect
6
New cards
DISPOSAL- Biohazard box
- gloves only in red biohazard box (NOT in black trash can or sharp containers)
- Contaminated items (paper towels used for cleaning the bench or bacteria spills need to be disposed in red biohazard box in the front
- Contaminated items (paper towels used for cleaning the bench or bacteria spills need to be disposed in red biohazard box in the front
7
New cards
DISPOSAL- Regular trashcan
- all contaminated paper towels (paper towels used to dry hands), petri dishes sleeves, cotton swab package
8
New cards
DISPOSAL- Biohazard bags at back
- contaminated plates (cannot exceed half full, tell TA)
- contaminated enteropluri test tubes
- contaminated GN-ID A & B Test strips
- contaminated enteropluri test tubes
- contaminated GN-ID A & B Test strips
9
New cards
DISPOSAL- Small bench top sharps container
- contaminated razor blades, syringe needles, microscopic slides, covering slips, Kimwipes and cotton swabs
10
New cards
DISPOSAL- Benchtop at the back
- contaminated tubes (separate liquid(left) and solid(right) tubes
- transfer your tubes from the white rack into the colored racks separately
- all labels need to be removed from the tubes, and disposed into biohazard box
- spray the white rack with disinfectant, and put them back in order next to the wall
- transfer your tubes from the white rack into the colored racks separately
- all labels need to be removed from the tubes, and disposed into biohazard box
- spray the white rack with disinfectant, and put them back in order next to the wall
11
New cards
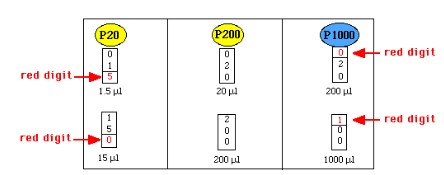
Micropipetting and the Basic Steps to Pipetting
- measures in volume units of microliters (uL)
- uses volumes between 0.5uL to 1000uL
- uses disposable tips that should be discarded in sharps containers
STEPS TO PIPETTING:
1) Select a pipette based on the volume needed
2) Adjust the volume on the pipette to the desired amount
3) Attach tip snugly
4) Depress plunger to first stop
5) Place tip in fluid and slowly release plunger, leaving the tip in the fluid a few seconds after the plunger is fully released.
6) Place the tip in the receiving tube, then eject the sample slowly by depressing the plunger to the second stop.
7) Discard the tip using the ejector into a sharp's container.
- uses volumes between 0.5uL to 1000uL
- uses disposable tips that should be discarded in sharps containers
STEPS TO PIPETTING:
1) Select a pipette based on the volume needed
2) Adjust the volume on the pipette to the desired amount
3) Attach tip snugly
4) Depress plunger to first stop
5) Place tip in fluid and slowly release plunger, leaving the tip in the fluid a few seconds after the plunger is fully released.
6) Place the tip in the receiving tube, then eject the sample slowly by depressing the plunger to the second stop.
7) Discard the tip using the ejector into a sharp's container.
12
New cards
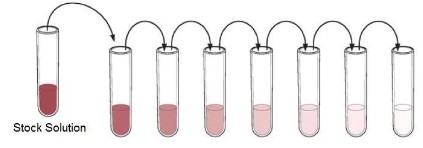
Serial Dilutions
- A dilution is performed by taking a portion (called an aliquot) of one solution and adding it to an appropriate amount of liquid (diluent) in order to lower the concentration of whatever is in the solution.
- A series of subsequent dilutions is called a serial dilution.
- the only thing that is changing in all of these solutions is concentration.
- The only difference between the stock tube and the final dilution (the tube on the far right) is how much "stuff" is in the solution.
- Looking at it , this should make sense. Thesolution on the far left (called the stock solution) is a very dark pink. With each dilution, the
color of the solution is considerably lighter, representing the lower concentration of each diluted solution. This is because each time a dilution is performed, a small amount of that "stuff" is transferred to the next tube.
- A series of subsequent dilutions is called a serial dilution.
- the only thing that is changing in all of these solutions is concentration.
- The only difference between the stock tube and the final dilution (the tube on the far right) is how much "stuff" is in the solution.
- Looking at it , this should make sense. Thesolution on the far left (called the stock solution) is a very dark pink. With each dilution, the
color of the solution is considerably lighter, representing the lower concentration of each diluted solution. This is because each time a dilution is performed, a small amount of that "stuff" is transferred to the next tube.
13
New cards
Labeling Method
- Petri dish is labelled on the bottom rather than on the lid. Write close to the edge of the bottom of the plate to preserve area to observe the bacteria culture after it has incubated.
- Culture tube is labelled on the glass rather than on the cap. Write close to the bottom edge of the cap to preserve area to observe the bacteria culture.
**Labels should include lab section, group initials, type of media, bacteria name, and the date.
- Culture tube is labelled on the glass rather than on the cap. Write close to the bottom edge of the cap to preserve area to observe the bacteria culture.
**Labels should include lab section, group initials, type of media, bacteria name, and the date.
14
New cards
Growth Media
- refers to a liquid, or gelatin, which supports the growth of microorganisms
15
New cards
Liquid Media- NUTRIENT BROTH
- mostly water with dissolved peptones and some beef extract. (Peptones are short chains of amino acids)
16
New cards
Solid Media
- are prepared by adding agar to liquid medium
- adding agar to nutrient broth makes nutrient agar (NA)
- is necessary for the isolation of individual colonies
- can be set in petri dishes (plates)
- can be set in tubes (solidifies at an angle) which is referred to as a slant
- agar solidifies at 40 degrees C. (can't incubate at high temps w/out agar liquifying)
- adding agar to nutrient broth makes nutrient agar (NA)
- is necessary for the isolation of individual colonies
- can be set in petri dishes (plates)
- can be set in tubes (solidifies at an angle) which is referred to as a slant
- agar solidifies at 40 degrees C. (can't incubate at high temps w/out agar liquifying)
17
New cards
Sterilization
- treatment that eliminates all forms of life, including heat resistant spores and viruses
18
New cards
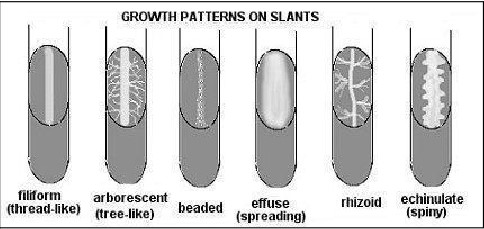
Bacterial Colonies
- a collection of many bacteria (several million) on a surface such as agar.
- bacterial growth on slants can also vary greatly
- bacterial growth on slants can also vary greatly
19
New cards
Different Streak patterns
a. tri- streak Isolation streak)
b. lawn streak
b. lawn streak
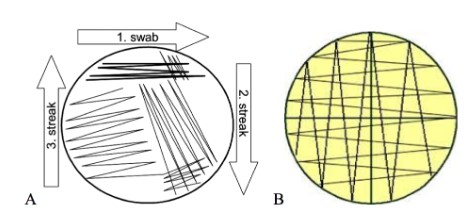
20
New cards
Bacteria Morphology
Bacilli- rods
Cocci- spheres
Streptococci- chains of spherical cells
Diplococci- pair of spherical cells
Staphylococci- clusters of spherical cells
Cocci- spheres
Streptococci- chains of spherical cells
Diplococci- pair of spherical cells
Staphylococci- clusters of spherical cells
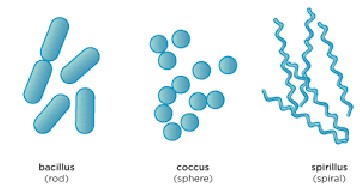
21
New cards
Gram Stain
Gram Positive - purple color (thick peptidoglycan
Gram Negative- pink (thin peptidoglycan)
- gram stain process= crystal violet-> iodine solution-> rinsed w/ethyl alcohol
- RESULTS:
Bacillus licheniformis: large gram positive (purple rod)
Escherichia coli: small gram negative (pink rod)
Gram Negative- pink (thin peptidoglycan)
- gram stain process= crystal violet-> iodine solution-> rinsed w/ethyl alcohol
- RESULTS:
Bacillus licheniformis: large gram positive (purple rod)
Escherichia coli: small gram negative (pink rod)
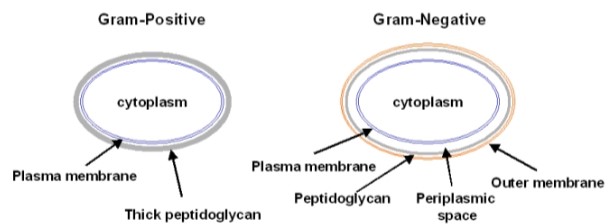
22
New cards
Capsule Stain
Capsule: an envelope having a gelatinous consistency
Procedure for visualizing the bacterial capsule- India ink (negative stain) THE BACKGROUND IS STAINED BUT NOT THE CELL
Procedure for visualizing the bacterial capsule- India ink (negative stain) THE BACKGROUND IS STAINED BUT NOT THE CELL
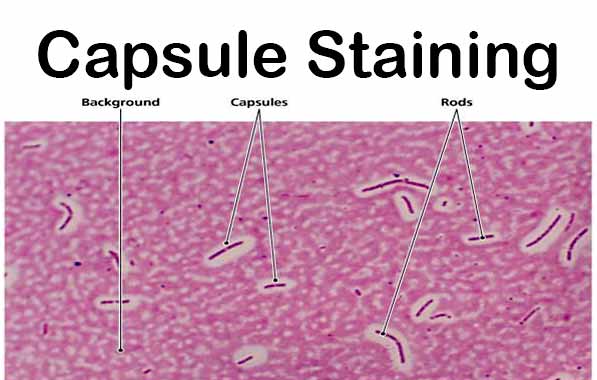
23
New cards
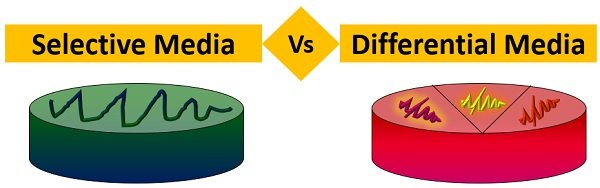
Selective vs. Differential Media
Selective- allows certain bacteria to grow but will inhibit others from growing
Differential- contains substances that cause some bacteria to take on an appearance that distinguish them from other bacteria
Differential- contains substances that cause some bacteria to take on an appearance that distinguish them from other bacteria
24
New cards
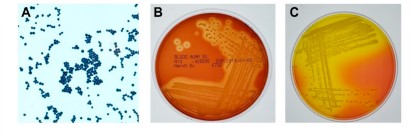
Staphylococcus aureus
- grows in mannitol-salt agar, appears yellow
25
New cards
Nutrient Agar
basic media used to grow our stains in our lab. (Low sensitivity and non-differential)
26
New cards
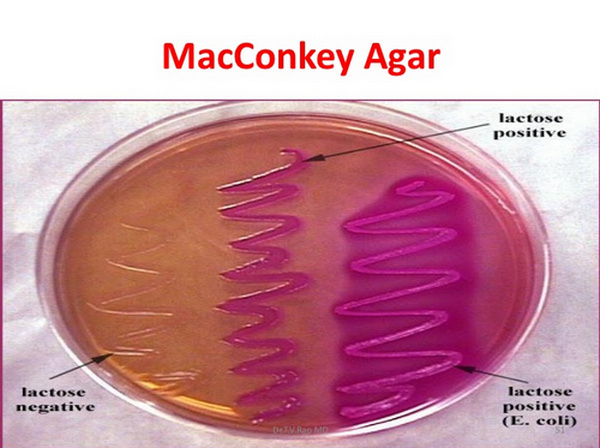
MacConkey's Agar (MAC)
- a differential and selective medium recommended for the use in the detection and isolation of all types of enteric bacteria. (Escherichia coli, Salmonella enterica)
- SELECTIVE: Gram negative bacteria
- DIFFERENTIAL: Lactose fermentation
* Lactose fermentation- red/pink colonies
* Non-Lactose fermentation- white/clear colonies
- SELECTIVE: Gram negative bacteria
- DIFFERENTIAL: Lactose fermentation
* Lactose fermentation- red/pink colonies
* Non-Lactose fermentation- white/clear colonies
27
New cards
Eosin Methylene Blue Agar (EMB)
- a weakly selective and differential plating medium. (Inhibit Gram positive growth)
- SELECTIVE: enteric bacteria; gram positive cannot grow
-DIFFERENTIAL: lactose and or sucrose fermentation
* Sugar fermentation: dark blue/black colonies; E. coli: metallic green
* Non- Fermenters: white/clear/slightly pink colors
- SELECTIVE: enteric bacteria; gram positive cannot grow
-DIFFERENTIAL: lactose and or sucrose fermentation
* Sugar fermentation: dark blue/black colonies; E. coli: metallic green
* Non- Fermenters: white/clear/slightly pink colors

28
New cards
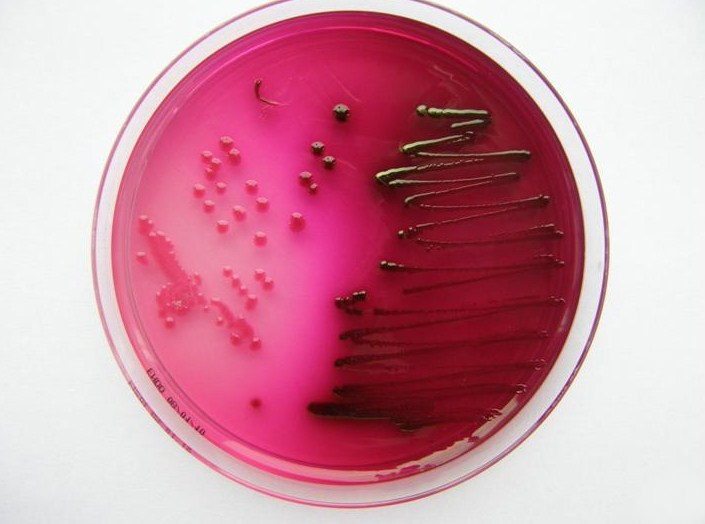
Endo Agar
- is deferential and slightly selective. (Inhibit of gram positive is achieved by the incorporation of sodium sulfite and basic fuchsin)
- SELECTIVE: gram negative
- DIFFERENTIAL: lactose fermenters/non lactose fermenters
*Lactose fermenters: clear colonies
* Non lactose fermenters: red colonies
- SELECTIVE: gram negative
- DIFFERENTIAL: lactose fermenters/non lactose fermenters
*Lactose fermenters: clear colonies
* Non lactose fermenters: red colonies
29
New cards
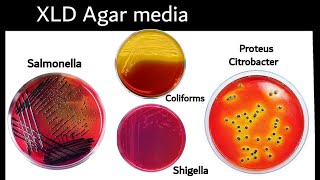
Xylose lysine deoxycholate Agar (XLD)
- a highly selective medium recommended for the isolation of Salmonella enterica and Shigella
- SELECTIVE: gram negative
-DIFFERENTIAL: hydrogen production
*Sugar fermentation: media changes to a yellow color
*No Xylose fermentation: colonies appear res (shigella)
*Hydrogen sulfide production: black precipitate in colonies (Salmonella enterica)
- SELECTIVE: gram negative
-DIFFERENTIAL: hydrogen production
*Sugar fermentation: media changes to a yellow color
*No Xylose fermentation: colonies appear res (shigella)
*Hydrogen sulfide production: black precipitate in colonies (Salmonella enterica)
30
New cards
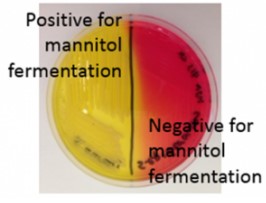
Mannitol Salt Agar (MSA)
- selective for staphylococcus and is differential for staphylococcus aureus from other staphylococci.
- SELECTIVE: salt tolerant organisms
-DIFFERENTIAL: mannitol fermentation; changing the pH indicator
* Growth on Media- organism is tolerant
* Mannitol fermentation- media changes from yellow to red
* Non-Mannitol fermentation- media remains red
- SELECTIVE: salt tolerant organisms
-DIFFERENTIAL: mannitol fermentation; changing the pH indicator
* Growth on Media- organism is tolerant
* Mannitol fermentation- media changes from yellow to red
* Non-Mannitol fermentation- media remains red
31
New cards
osmotic pressure
involves 6.5% NaCl
Temp: 37 degrees C
Temp: 37 degrees C
32
New cards
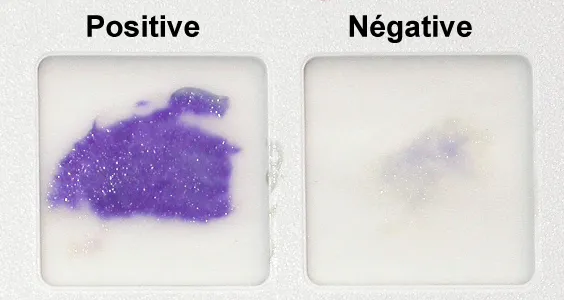
Oxidase Test
- tests for the presence of cytochrome oxidase
* Positive result- formation of a purple color within minutes
* Negative result- no color change
* Positive result- formation of a purple color within minutes
* Negative result- no color change
33
New cards
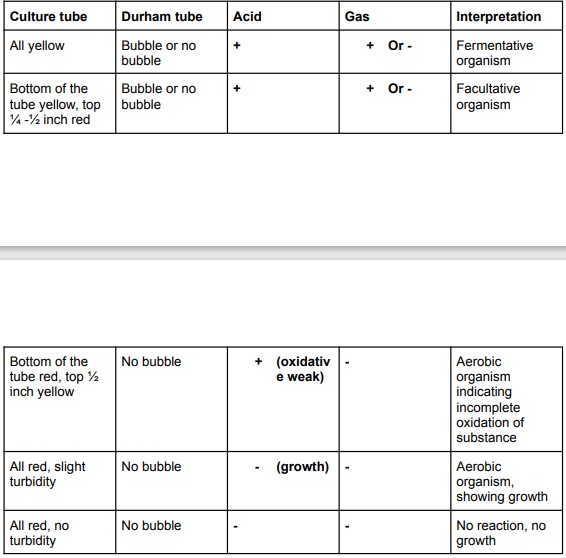
Fermentation of sugars
Reversion: SUGAR-> PROTEINS-> pH CHANGE-> YELLOW-> pH CHANGE-> RED
Temp: 37 degrees C
Temp: 37 degrees C
34
New cards
Carbohydrate Fermentation Test
- objectives
- principles
- procedure
- result and interpretation
- precautions and limitations
- principles
- procedure
- result and interpretation
- precautions and limitations

35
New cards
Extracellular Enzymes
- test for the presence of certain extracellular enzymes: alpha-amylase, and enzyme that catalyzes the breakdown of starch.
36
New cards
Hydrolysis of Starch
Temp: 37 degrees C
Test for amylase using Gram's iodine solution
Positive result: clear zones surrounding bacterial growth indicated the absence of starch.
Negative result: a starch-iodine complex produces a deep purple to black color
Test for amylase using Gram's iodine solution
Positive result: clear zones surrounding bacterial growth indicated the absence of starch.
Negative result: a starch-iodine complex produces a deep purple to black color
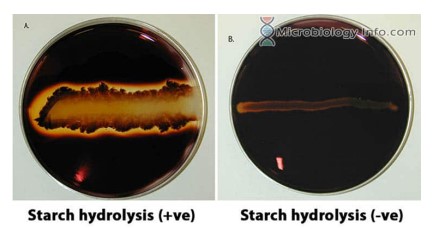
37
New cards
Hydrolysis of Triglycerides
MA: spirit blue agar plate
Temp: 25 degrees C
Positive result: cleaning of the emulsion
Negative result: precipitation of the blue dye
Temp: 25 degrees C
Positive result: cleaning of the emulsion
Negative result: precipitation of the blue dye
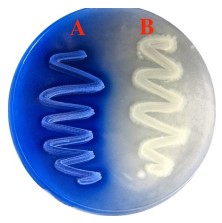
38
New cards
Catalase Production
positive: production of gas
negative: no bubbling
negative: no bubbling

39
New cards
Sulfur Metabolism
- bacteria that are able to use sulfur will produce hydrogen sulfide or H2S.
- uses media allied Klinger's Iron Agar (KIA)
- Temp: 37 degrees C
- uses media allied Klinger's Iron Agar (KIA)
- Temp: 37 degrees C
40
New cards
Protease production
Temp: 37*C
-Positive: If culture remains liquid or liquefied, gelatin hydrolysis occurred by proteases
produced by the inoculums
-Negative: Gelatin solidifies, then no protease was present
-Positive: If culture remains liquid or liquefied, gelatin hydrolysis occurred by proteases
produced by the inoculums
-Negative: Gelatin solidifies, then no protease was present
41
New cards
Ryo or KOH String Test
- The alkaline solution of KOH dissolves the gram-neg. outer envelope, causing the bacteria to
become sticky and tacky, thus generating a slimy emulsion.
● Positive: Stringy bacteria, predicting a gram-neg. Stain result.
● Negative: Non-stringy bacteria predicting a gram-positive stain result.
become sticky and tacky, thus generating a slimy emulsion.
● Positive: Stringy bacteria, predicting a gram-neg. Stain result.
● Negative: Non-stringy bacteria predicting a gram-positive stain result.
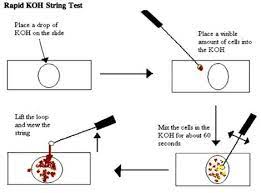
42
New cards
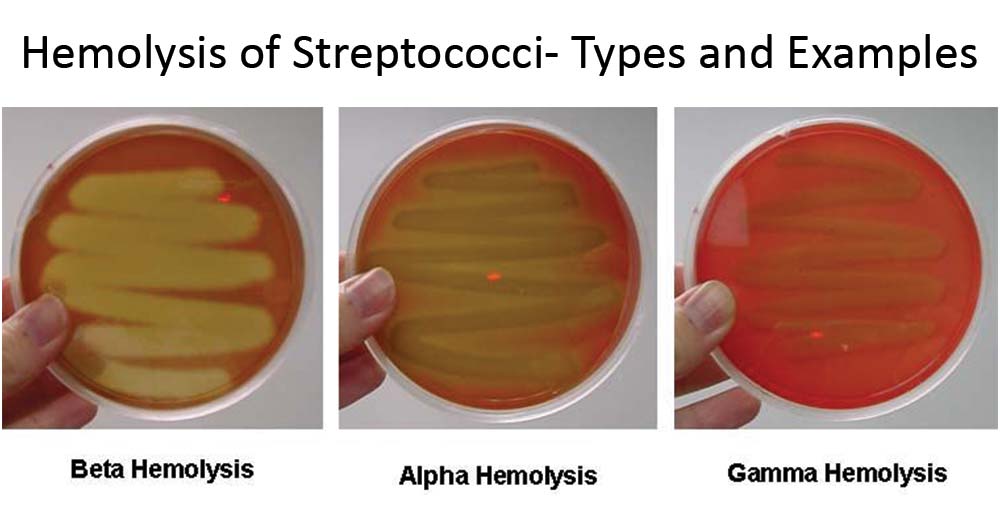
Alpha, Beta, and Gamma Hemolysis
- Alpha hemolysis: green coloring under bacteria colonies; partial lysis of cells
- Beta hemolysis: Clearing around the colonies; complete lysis of cells
- Gamma Hemolysis: Growth on plate without any lytic action (non-hemolytic)
Temp 37*C
- Beta hemolysis: Clearing around the colonies; complete lysis of cells
- Gamma Hemolysis: Growth on plate without any lytic action (non-hemolytic)
Temp 37*C
43
New cards
Microbial Biofilms
Test the ability of bacteria to form biofilm using the dye crystal violet
Temp 37*C
Temp 37*C

44
New cards
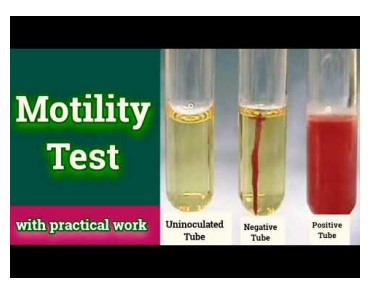
Motility Test Medium (MTM)
Semisolid agar media to detect motility
Temp 37*C
● No growth is an inconclusive result
● Non-motile growth is only visible along the line of inoculation
● Motile growth disperse away from the stab line of inoculation (positive result)
Temp 37*C
● No growth is an inconclusive result
● Non-motile growth is only visible along the line of inoculation
● Motile growth disperse away from the stab line of inoculation (positive result)
45
New cards
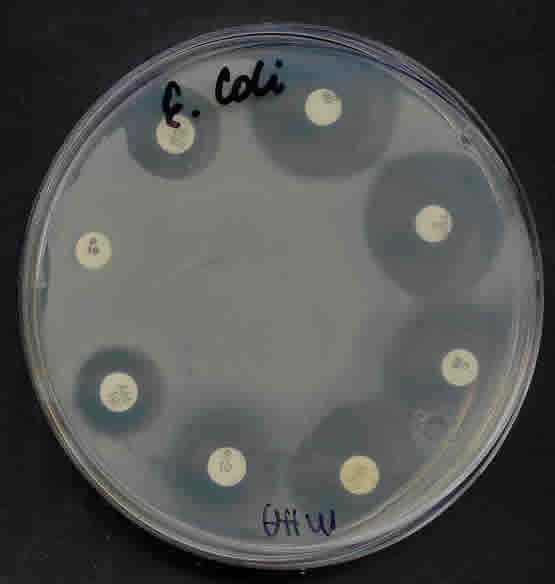
Antimicrobial Susceptibility Testing
- An important technique to determine the effectiveness of antibacterial agents for a partial
target organism is sensitivity disc assay.
- This assay is also called the Kirby-Bauer method after the inventors.
- The MIC is the lowest concentration of a compound that prevents growth of a particular
organism.
-Bactericidal: death to an organism
-Bacteriostatic: block and slow the growth or metabolisms of the organism
- Temp 37 degrees C
target organism is sensitivity disc assay.
- This assay is also called the Kirby-Bauer method after the inventors.
- The MIC is the lowest concentration of a compound that prevents growth of a particular
organism.
-Bactericidal: death to an organism
-Bacteriostatic: block and slow the growth or metabolisms of the organism
- Temp 37 degrees C