Atomic structure
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
What were atoms believed to be before the theories?
-Democritus:
-Before the discovery of the electron, atoms were thought to be tiny spheres that could not be divided
What was John Dalton’s theory?
-Matter is made up of tiny spheres, ‘atoms’
-Each element is made of a different type of atom
What was JJ Thompsons theory?
-The discovery of the electron led to the plum pudding model of the atom
-The plum pudding model suggested that the atom is a ball of positive charge with negative electrons embedded in it
What was Ernest Rutherford’s first theory?
-Alpha particle scattering experiment
Observations:
-most alpha particles passed straight through
-some particles are deflected
-a few particles are deflected by large angles
Conclusion:
-most of the atom is empty space
-nucleus is positively charged
-most of the mass of the atom is in the nucleus
What was Niels Bohr’s theory?
-He adapted the nuclear model by suggesting that electrons orbit the nucleus at specific distances
-The theoretical calculations of Bohr agreed with experimental observations
What was Ernest Rutherford’s second theory?
-Later experiments led to the idea that the positive charge of any nucleus could be subdivided into a whole number of smaller particles, each particle having the same amount of positive charge
-The name proton was given to these particles
What was James Chadwick’s theory?
-The experimental work of James Chadwick provided the evidence to show the existence of neutrons within the nucleus
-This was about 20 years after the nucleus became an accepted scientific idea
what is the structure of an atom?
-small positively charged nucleus
-Nucleus composed of protons (positive charge) and neutrons (no charge)
-Most of the mass of the atom is in the nucleus
-Surrounded by negatively charged electrons
-Electrons are at differently energy levels
What are the number of protons?
-atomic number
-changing the number of protons changes the element
what are the number of neutrons?
-changing the number of neutrons doesn’t change the element but it does change the mass
-Number of protons and neutrons (nucleons) is the mass number
-Atoms of the same element with different numbers of neutrons are called isotopes
How are electrons arranged in an atom?
into electron shells/energy levels
How can electrons move energy levels?
-To move up an energy level they have to absorb electromagnetic radiation
-To move down they emit electromagnetic radiation
what is the radius of an atom?
1×10-10 m
what is the radius of the nucleus?
less than 1/10000 of the radius of an atom
what is ionising radiation?
Radiation that has enough energy to cause electrons to leave an atom
what is radioactive decay?
-A random process where:
-Some atomic nuclei are unstable
-The nucleus gives out radiation as it changes to become more stable
What is activity?
the rate at which a source of unstable nuclei decays
how is activity measured?
-Becquerel (Bq)
-1 Bq is one decay per second
What is count-rate?
The number of decays recorded each second by a detector (e.g Geiger-Muller tube)
What does an alpha particle consist of?
An unstable nucleus emits 2 protons and 2 neutrons
what are the properties of alpha particles?
-Low penetration (stopped by paper)
-Low range in air (2cm)
-Strongly ionising
What is the charge of alpha particles?
2+
What is another name for an alpha particle?
a helium nucleus
what symbols represent alpha particles?

what does a beta particle consist of?
-a neutron turns into a proton and an electron
-the high speed electron is emitted from to nucleus
What are the properties of a beta particle?
-Mid penetration (stopped by 5mm aluminium)
-Mid range in air (80cm)
-Medium ionising
What is the charge of a beta particle?
1-
what is another name for a beta particle?
an electron
what symbols represent a beta particle?

What is gamma radiation?
-nucleus emits high electromagnetic radiation
-the nucleus doesn’t change
what are the properties of gamma radiation?
-high penetration (only stopped by thick lead)
-very high range in air
-weakly ionising
What is the symbol for gamma radiation?
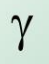
what happens during alpha decay?
The unstable nucleus emits an alpha particle, which is 2 protons and 2 neutrons
How do the mass number and atomic number of an unstable nucleus change during alpha decay?
the mass number decreases by 4 and the atomic number decreases by 2
what happens during beta decay?
A neutron decays into a proton and an electron
The
How do the mass number and atomic number of an unstable nucleus change during beta decay?
The mass number stays the same and the atomic number increases by 1
Do the mass number or atomic number of an unstable nucleus change during gamma decay?
no
when writing nuclear decay equations, how can alpha particles be represented?
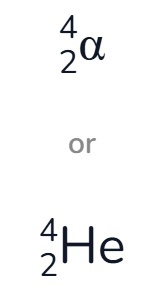
when writing nuclear decay equations, how can beta particles be represented?

when writing nuclear decay equations, how can gamma particles be represented?
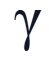
Can you tell when an individual nucleus will decay?
No it is a random event:
-Cannot predict which nuclei will decay
-Cannot predict when the nuclei will decay
What is half-life?
-The half-life of a radioactive isotope is the time it takes for the number of nuclei of the isotope in a sample to halve
-This is also the time it takes for the count rate or activity to fall to half its initial level
How do you find the half life from an activity-time graph?
find the time it takes for activity to fall by half
What device is used to record radioactive decay?
Geiger Muller tube and counter
What is contamination?
-The unwanted presence of materials containing radioactive atoms on other materials
-The contaminated object becomes radioactive
-The hazard is due to exposure to ionising radiation from the decay of the contaminating atoms
-The hazard is highest for alpha emitters as they are the most ionising
what is irradiation?
-The process of exposing an object to nuclear radiation
-The irradiated object doesn’t become radioactive
-The hazard is due to exposure to ionising radiation
-The hazard is highest from gamma emitters as they are the most penetrating
What are the suitable precautions to protect against contamination?
-Don’t touch radioactive sources (use tongs)
-Don’t eat and drink while handling
-Wash hands after handling
-Wear protective equipment (e.g gloves, goggles, respiratory mask)
What are the suitable precautions to protect against irradiation?
-Minimise time of exposure (keep sources in a box until needed)
-Increase distance from source (use long tongs, or robots)
-Use shielding