(CHE120) Chapter 1 Slides
1/169
Earn XP
Description and Tags
Status: Incomplete
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
170 Terms
Chemistry
Study of composition, properties, and interactions of matter
Attempts to understand the behaviour of matter
the “Central Science”
Science based on observation & experimentation
Matter
Anything that takes up space & has mass.
What were the four elements believed by the ancient Greeks to constitute all matter?
Earth, air, fire, and water.
What was the primary goal of alchemists in early chemical history?
To transform base metals into noble metals.
What is one reason alchemy is not considered scientific by modern standards?
It lacked systematic experimentation and empirical validation.
Despite its limitations, what did alchemy contribute to the development of chemistry?
Useful ideas about manipulating matter.
This history of chemistry extends back over _____ years.
This history of chemistry extends back over 2500 years.
Provide 2 examples of how chemistry is used by humans in everday life.
Digesting food
Health and Medicine, e.g. vaccines, sanitation
Synthesizing polymers for clothing, cookware, and credit cards
Refining crude oil into gasoline and other products
Energy and the environment, e.g. solar energy
Organic Chemistry
Define
Name 1 application
Def: The structure, properties, and reactions of carbon-containing compounds
Application: Drug synthesis
Inorganic Chemistry
Define
Name 1 application
Def: The structure, properties, and reactions of inorganic and organometallic compounds.
Application: Catalyst chemistry
Analytical Chemistry
Define
Name 1 application
Def: Quantitative measurements of composition and structure of matter.
Applications:
Forensics
Quality control
Biochemistry
Define
Name 1 application
Def: The chemistry of living systems
Examples of Subfields:
Molecular genetics
Protein science
Physical Chemistry
Define
Name 1 subfield
Def: Law of physics applied to chemical systems
Examples of Subfields:
Kinetics
Quantum chemistry
Theoretical chemistry
What 2 things is chemistry fundamentally based on?
Observation & experimentation
Scientific Law
Concise statement
Summarizes several experimental observations & describes or predicts aspects of the natural world
Hypothesis
A tentative explanation for observations made during scientific research.
Theory v.s. Hypothesis
Theory
Well-supported & tested explanation of a natural phenomenon
Based on extensive evidence
Hypothesis
Tentative explanation for observations made
Testable, falsifiable statement
Predicts an outcome
Theory v.s. Scientific Law
Law = Statement about observed phenomenon
E.g: Law of Conservation of Mass summarizes the observation that mass can not be destroyed nor created.
Theory = Explanation of hypothesis that’s been tested & well-supported
E.g: Dalton’s Atomic Theory explains the nature of atoms and their behavior.
Substance
Form of matter that has a definite composition & distinct properties.
Mass v.s. Weight
Mass = Measure of amount of matter (particles) in an object
Weight = Force exerted by gravity on an object
For example:
on earth → your mass is 150lbs, your weight is 150lbs
on moon → your mass is 150lbs, but your weight will change as gravity changes
T or F: The mass of an object/being changes depending on their location in the universe.
False.
The mass of all objects/beings remains constant regardless of their location
What does change w/ location is weight → bc weight depends on the gravitational force of the celestial body the object/being is on
The mass of an object/being only changes if matter is _______ or _______.
The mass of an object/being only changes if matter is added or removed.
Astronaut Paul weighs 180 lbs on the Moon. What will happen to his weight when he comes back to Earth?
His weight will increase since Earth has higher gravity than the Moon.
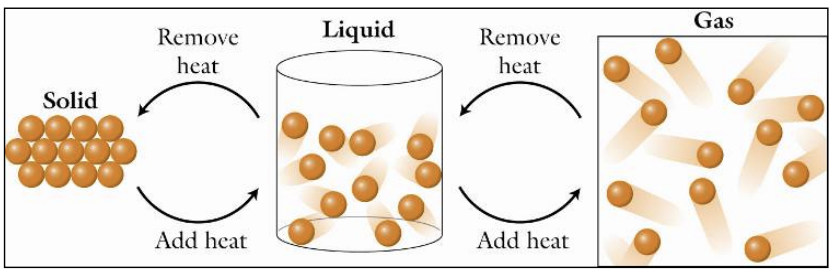
The 3 Phases of Water
Phase of Matter
Arrangement of Molecules
Example
1) Solid (ice)
Molecules → Close together & organized
E.g. Icebergs
2) Liquid (liquid water)
Molecules → Close together & disorganized
E.g. Lakes, oceans
3) Gas (water vapour)
Molecules → Far apart & very disorganized
E.g. Moisture in air
What do the symbols (g), (s), and (l) represent?
(g) → gas
(s) → solid
(l) → liquid
Describe the 3 phases of matter.
(1) Solids
Definite shape
Definite volume
Not compressible
(2) Liquids
No definite shape
Definite volume
Able to flow
(3) Gases
No definite shape
No definite volume
Molecules are very far apart
Highly compressible
__________ take the shape of the container they’re in while maintaining a constant volume.
Liquids take the shape of the container they’re in while maintaining a constant volume.
In molecular models, what do red spheres represent?
Oxygen atoms
In molecular models, what do white spheres represent?
Hydrogen atoms
Briefly explain how substances can be changed from one phase of matter to another.
Substances can be changed from one phase of matter to another by adding or removing energy — usually heat.
Adding energy
particles move faster
substance will eventually melt or evaporate
melt = solid → liquid
evaporate = liquid → gas
Removing energy
particles slow down
substance will eventually freeze or condensate
freeze = liquid → solid
condensate = gas → liquid
Describe the Classifications of Matter
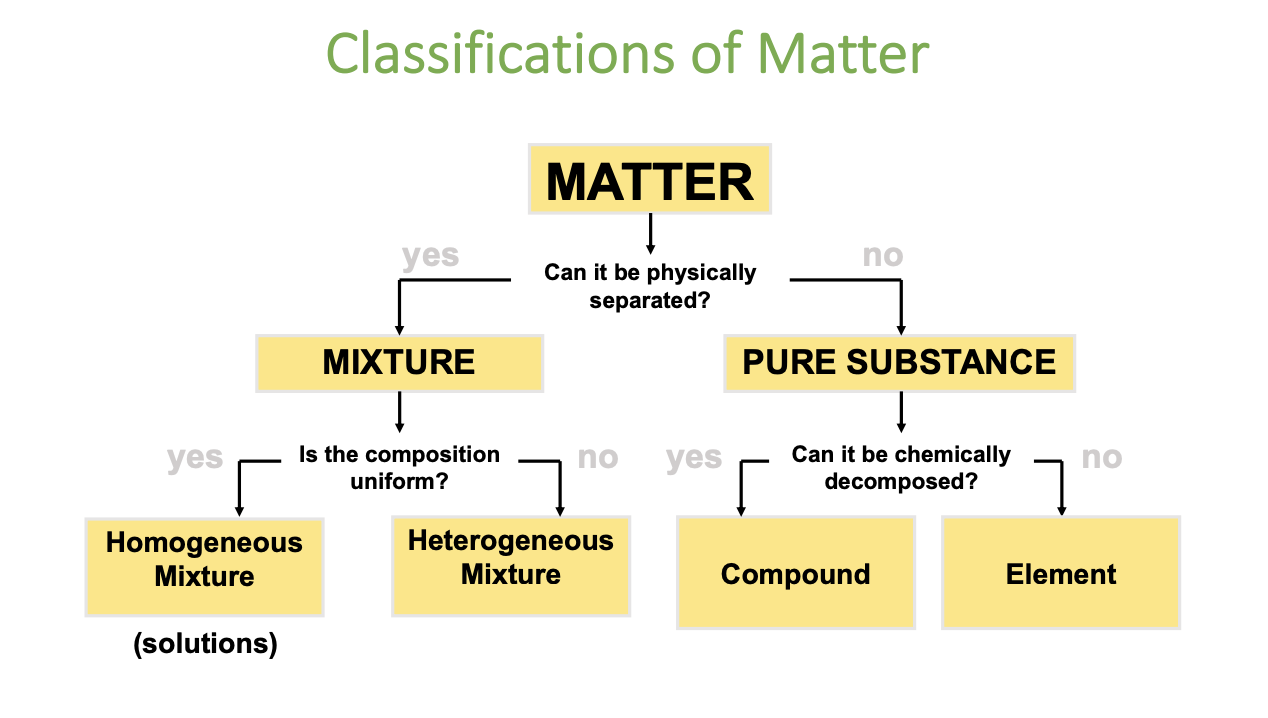
The Two Major Classifications of Matter
(1) Mixture = 2 or more substances that CAN be physically separated
(2) Pure Substance = 2 or more substances that CAN’T be physically separated
Two Types of Mixtures
i) Homogeneous
AKA a solution
Uniform composition throughout (which is also visible)
E.g. sprite
ii) Heterogenous
Non-uniform composition
The different phases of matter are visible
E.g. vinaigrette dressing (oil + non-oil substance; they are visibly separate)
Two Types of Pure Substances
i) Compounds
Pure substance composed of 2+ elements that CAN be chemically decomposed
E.g. Mercury (II) Oxide decomposes into mercury & oxygen when heated
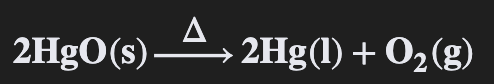
ii) Element
Pure substance that CANNOT be chemically physically decomposed
Examples:
Oxygen (O)
Gold (Au)
Helium (He)
What is the smallest particle that retains the properties of an element?
An atom
T or F: Atoms can be chemically broken down further while retaining their elemental identity.
False.
An ____ is the most basic unit of chemical matter.
An atom is the most basic unit of chemical matter.
What are atoms composed of?
Protons
Neutrons
Electrons
T or F: Molecules can be separated into individual atoms.
True. Through chemical reactions
Name 2 examples of molecules made from a single element.
O₂ (oxygen gas)
N₂ (nitrogen gas)
Atoms v.s. Molecules
Atoms (of an element)
Most basic unit of chemical matter
Smallest type of particle that can have properties of an element
Cannot be broken down further while retaining properties of the element
Molecules (of an element)
Combination of 2 or more atoms
Can be broken down into individual atoms
Elements v.s. Compounds
Elements & compounds are the two major categories of pure substances.
Elements
Simplest type of pure substance
Consists of only 1 type of atom
Can’t be broken down further by physical or chemical means
Forms :
Single atoms
e.g. Gold (Au); Helium (He); Iron (Fe)
Molecules of an element — still considered elements because all atoms are of the same type.
e.g., O₂, N₂
Compounds
Pure substances composed of 2+ types of atoms that are chemically bonded
Can be broken down chemically into elements
Compounds have diff properties than the elements that form it
Examples: Water (H₂O), Carbon dioxide (CO₂), Sodium chloride (NaCl).
In one sentence, state the critical difference b/w elements & compounds.
Elements → Cannot be broken down chemically
Compounds → Can be broken down chemically into elements
Compound
Pure substance composed of 2+ elements
How many elements in the periodic table?
118 elements
Each element on the periodic table is identified by its _______________. The atomic number represents the _______________ in the element’s nucleus.
Each element on the periodic table is identified by its atomic number. The atomic number represents the number of protons in the element’s nucleus.
Natural Elements
Define
2 Examples
Def: Elements that naturally occur on Earth
(Elements number 1 through 94 on periodic table)
Examples:
Gold (Au)
Oxygen (O)
Carbon (C)
Sulfur (S)
Synthetic Elements
Define
How can they be identified on periodic table?
2 Examples
Def: Man-made in labs through nuclear reactions
Identify by their atomic number → elements with number 95 through 118 on periodic table
Examples:
Americium (Am)
Seaborgium (Sg)
Discuss the historical context of the periodic table and how its evolved over centuries.
Ancient elements: Known since antiquity (e.g., gold, copper).
Lavoisier’s list (1789): Early classification of known elements.
Mendeleev’s table (1869): Organized elements by atomic mass and predicted undiscovered ones.
Seaborg’s table (1945): Included actinides and expanded the table.
Modern updates: Continued discoveries up to 2012 and beyond.
How many elements naturally occur on Earth?
94
How many elements are man-made (synthetic)?
24
________________ are elements that are created through nuclear reactions.
Synthetic elements are elements that are created through nuclear reactions.
Give 2 examples of natural elements.
Iron (Fe)
Oxygen (O)
Nitrogen (N)
Phosphorus (P)
Give 2 examples of synthetic elements.
Americium (Am) and Seaborgium (Sg).
In the periodic table the first ___ elements are _________, whereas the rest are _________.
In the periodic table the first 94 elements are natural, whereas the rest are synthetic.
Who published the first organized periodic table in 1869?
Dmitri Mendeleev.
What defines an element’s position on the periodic table?
It’s atomic number
What was Seaborg’s major contribution to the periodic table?
Inclusion of the actinide series and expansion of synthetic elements.
When the compound Mercury II Oxide is __________ it decomposes into two elements: ___________ and __________.
When the compound Mercury II Oxide is heated it decomposes into two elements: liquid mercury and oxygen gas.
Physical Properties v.s. Chemical Properties
Compare definitions
State the 5 most common examples/scenarios for each
Physical Property
Can be observed w/o changing composition of substance
Most common examples:
State/Phase of Matter — solid v.s. liquid v.s. gas at room temp
Boiling/Melting Point — temp at which phase change occurs
Density — Relationship b/w mass & volume (D = m / V)
Malleability — Ability to be re-shaped w/o breaking
Solubility — How well a substance dissolves in a solvent
Chemical Property
Describes how likely a substance is to undergo a chemical change that transforms it into a new/different substance,
Most common examples:
Flammability — How easily substance ignites/burns
Reactivity — How a substance reacts with water, acids, or oxygen (e.g. rusting)
Toxicity — Potential for substance to damage an organism
Acidity/Alkalinity (pH) — How a substance behaves as an acid or a base
Chemical Property v.s. Chemical Change.
Provide an example for each.
Chemical Property
A characteristic or "personality trait" of a substance.
Think “Ability”
E.g. Wood is flammable
Chemical Change
An event where a substance actually transforms.
Think “Process” or “Event”
E.g. Wood is burning
Physical Change v.s. Chemical Change
Compare definitions
State the 5 most common examples/scenarios for each
Physical Change
Change in substance’s state/phase
E.g. Ice melting
Chemical Change
Chemical reaction
E.g. rusting, burning, baking
Condensation
High-Yield
DEF: Phase change of a water vapour into a liquid
Not from slides (extra info to read for better understanding)
Occurs when warm, moist air cools to below its dew point
As water vapour molecules cool, their kinetic energy decreases
Results in their motion slowing down enough for intermolecular attractions to pull them together close enough to form liquid water droplets on a surface or in the air
Evaporation
High-Yield
DEF: Phase change of a liquid water into water vapour (gas)
Not from slides (extra info to read for better understanding)
Phase change of liquid water into water vapor (gas).
Occurs when surface molecules of a liquid gain enough energy to escape into the air.
Happens below the boiling point, often when air is warm, dry, or moving.
Molecules absorb energy, their kinetic motion increases, and they overcome intermolecular forces.
High‑energy molecules leave first, causing cooling of the remaining liquid.
Name & briefly describe the components of a measurement.
Every measurement includes:
Number (size/magnitude)
Unit (standard of comparison)
Uncertainty (estimated last digit)
Example: 4.7 ± 0.1 cm
Number: 4.7
Unit: cm
Uncertainty: 0.1
Without units, the number in a measurement is ________.
Without units, the number in a measurement is meaningless.
What are SI units? Explain its importance in the world of science.
SI = International System of Units
Most widely used measurement system worldwide
Adopted in 1964
Modernized, globally agreed-upon version of metric system
Compare the 2 systems of measurement used in the USA.
U.S. Customary System (USCS)
Used for:
Daily life
Commerce
Includes:
Length → inches, feet, yards, miles
Weight → ounces, pounds, tons
Volume → Gallons, quarts, pints, cups
Internal System of Units
Used for:
Science
Medicine
Internal trade
For each of the 7 base SI units, provide:
Property
Name of Unit
Symbol of Unit
What It Represents
Why It's a Base Unit
Property | Name of Unit | Symbol of Unit | What It Represents | Original Definition | Current Definition |
Length | Meter | m | Distance b/w 2 points | 1/10,000,000 of distance from North Pole to equator | Distance light travels in a vacuum in 1/299,792,458 |
Mass | Kilogram | kg | Amount of matter | Mass of a litre of water | Defined in terms of the Planck constant, h, which states |
Time | Second | s | Duration of events | ||
Temperature | Kelvin | K | Average kinetic energy of particles | ||
Electric Current | Ampere | A | Flow of electric charge | ||
Amount of Substance | Mole | mol | Number of particles | ||
Luminous Intensity | Candela | cd | Received brightness |
Each of the 7 base units were selected because they correspond to …
to a fundamental physical property that cannot be broken down into something more basic.
Which 2 other countries besides the USA have not officially adopted SI units?
Liberia
Myanmar
What is the SI base unit for length?
Meter (m)
What is the SI base unit for mass?
Kilogram (kg)
What is the SI base unit for time?
Second (s)
What is the SI base unit for temperature?
Kelvin (K)
What is the SI base unit for electric current?
Ampere (A)
What is the SI base unit for amount of substance?
Mole (mol)
What is the SI base unit for luminous intensity?
Candela (cd)
What is Kelvin (K)?
Absolute temperature scale
Has no degree symbol
What is Celsius (°C)?
Common scientific temperature scale
What is the formula to convert °C to °F?

What is the formula to convert °C to K?
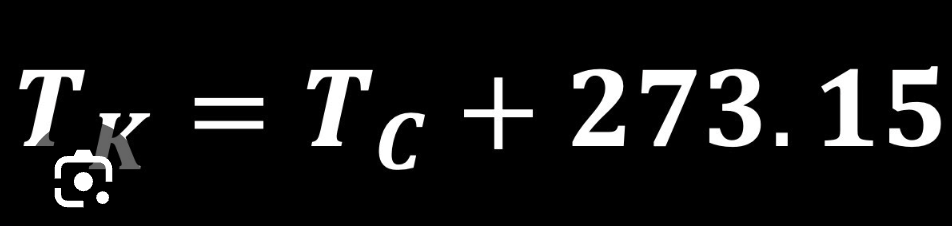
SI unit for volume
Cubic meter (m³)
What is 1 dm³ equal to?
1 L
What is 1 cm³ equal to?
1 mL
Extensive Properties vs. Intensive Properties
Extensive Properties
Dependent on amount of matter
Changes when sample size changes
Examples:
Mass
Volume
Total energy
Intensive Properties
Independent of amount of matter
Stays constant regardless of sample size
Examples:
density
colour
melting point
temperature
T or F: The periodic table organizes elements into groups with similar properties.
True
Elements in the same group (vertical column) share similar chemical properties (bc they all have same number of valence electrons).
What is the formula density?
Density = mass / volume
What are common density units for solids and liquids?
g/cm³
What are common density units for gases?
g/L
How is scientific notation written?
N × 10ⁿ
What must N be between in scientific notation?
What must N be between in scientific notation?
Why is scientific notation used?
For very large or very small numbers.
What is Avogadro’s number in scientific notation?
6.022 × 10²³
What does kilo (k) represent?
10³
What does centi (c) represent?
1A: 10⁻²
0⁻²
What does milli (m) represent?
10⁻³
What is accuracy?
Closeness to the true value.
What is precision?
Reproducibility of measurements.
What are exact numbers?
Counting or defined quantities; infinite significant figures.
What are uncertain numbers?
Measured values that include estimation.
What digits are included in significant figures?
All certain digits plus one uncertain digit.
Which digits are always significant?
Nonzero digits.