Biochem practice problems
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
1. | Which of the following is the most abundant element in the human body? | |
A) | nitrogen | |
B) | carbon | |
C) | oxygen | |
D) | phosphorous | |
E) | none of the above | |
C) oxygen
proteins
structure, enzyme
nucleic acids
information
lipids
membrane, energy
polysaccharides
energy storage
Which of the following molecules contains the most oxidized form of carbon? |
| ||
| A) | acetaldehyde | |
| B) | ethanol | |
| C) | acetic acid | |
| D) | ethylene | |
| E) | carbon dioxide | |
E) carbon dioxide
Which of the following correctly identifies the progression from individual molecules to a functioning multi-cellular organism? |
| |
| molecules, cell, organelle, organ, organism | |
| molecules, organelle, organ, cell, organism | |
| molecules, organelle, cell, organ, organism | |
| molecules, organ, organelle, cell, organism | |
| molecules, cell, organ, organelle, organism | |
Molecule → organelle → cell → organ → organism
Hydrogen bonds are approximately _____% of the bond strength of covalent C-C or C-H bonds.
A. 1
B. 5
C. 20
D. 50
E. 95
B) 5
Hydrogen bonds within liquid water are _________.
A. attractions between the protons of the oxygen nuclei
B. ion-induced dipole interactions
C. dipole-dipole interactions
D. attractions between two oxygen atoms
E. attractions between the H+ and -OH ions of the liquid
C. dipole-dipole interactions
Considering the energetics of transferring nonpolar molecules from water to a nonpolar solvent, the factor TDS is generally _____, causing DG to be _____.
A. positive: negative
B. negative; negative
C. positive; positive
D. negative; positive
E. ngligible; either positive or negative
A. positive: negative
What would be the resulting pH if one ml of 1.0 M NaOH was added to one liter of pure water (assume pH 7.0)?
A. 1
B. 3
C. 7.3
D. 11
E. 13
D. 11
What is the conjugate acid of H2PO4-?
add a proton
H3PO4
At a pH above its pKa, the phenolic group of tyrosine is ___.
A. protonated and neutral
B. protonated and positively charged
C. deprotonated and neutral
D. deprotonated and negatively charged
E. none of these choices
D. deprotonated and negatively charged
Which of the following structures shows the prevalent form of glutamic acid at pH 3?
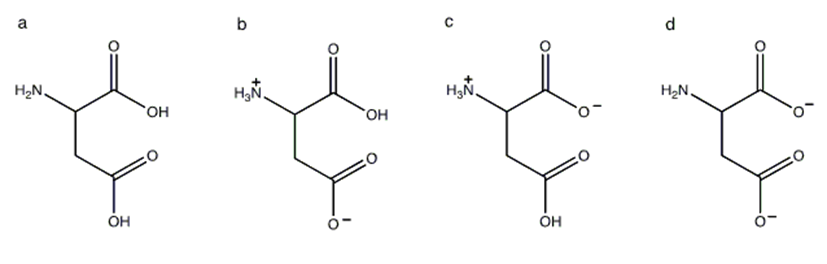
A.
B.
C.
D.
E none of these
C.
What is the N-terminal residue of the peptide AYSDG?
A. glutamic acid
B. glycine
C. aspartic acid
D. alanine
E. none of these choices
D. alanine
peptide sequence is written N-terminal → C-terminal
The following is an example of ___.
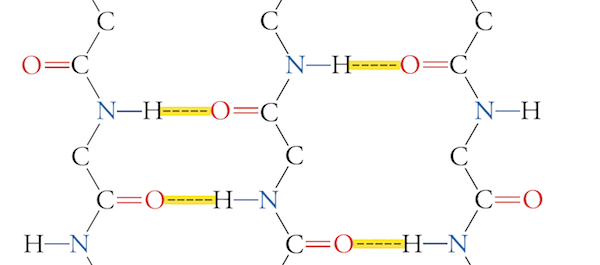
A. parallel alpha helix
B. parallel B-sheet
C. antiparallel alpha helix
D. antiparallel B-sheet
E. none of these choices
D. antiparallel B-sheet
Given pKa values of 2.3 and 9.7, what is the pI of alanine?
A. 12
B. 7.4
C. 6
D. 4.2
E. none of these
C. 6
A technique for determining protein structure that requires crystals of the protein is ___.
A. NMR spectroscopy
B. X-ray crystallography
C. Electron crystallography
D. mass spectroscopy
E. none of these
B. X-ray crystallography