BIOC 4331 Lecture 3
1/44
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
What are stereoisomers?
Molecules with the same chemical bonds but different configurations.
What two features give rise to stereoisomers?
Double bonds (no free rotation) and chiral centers
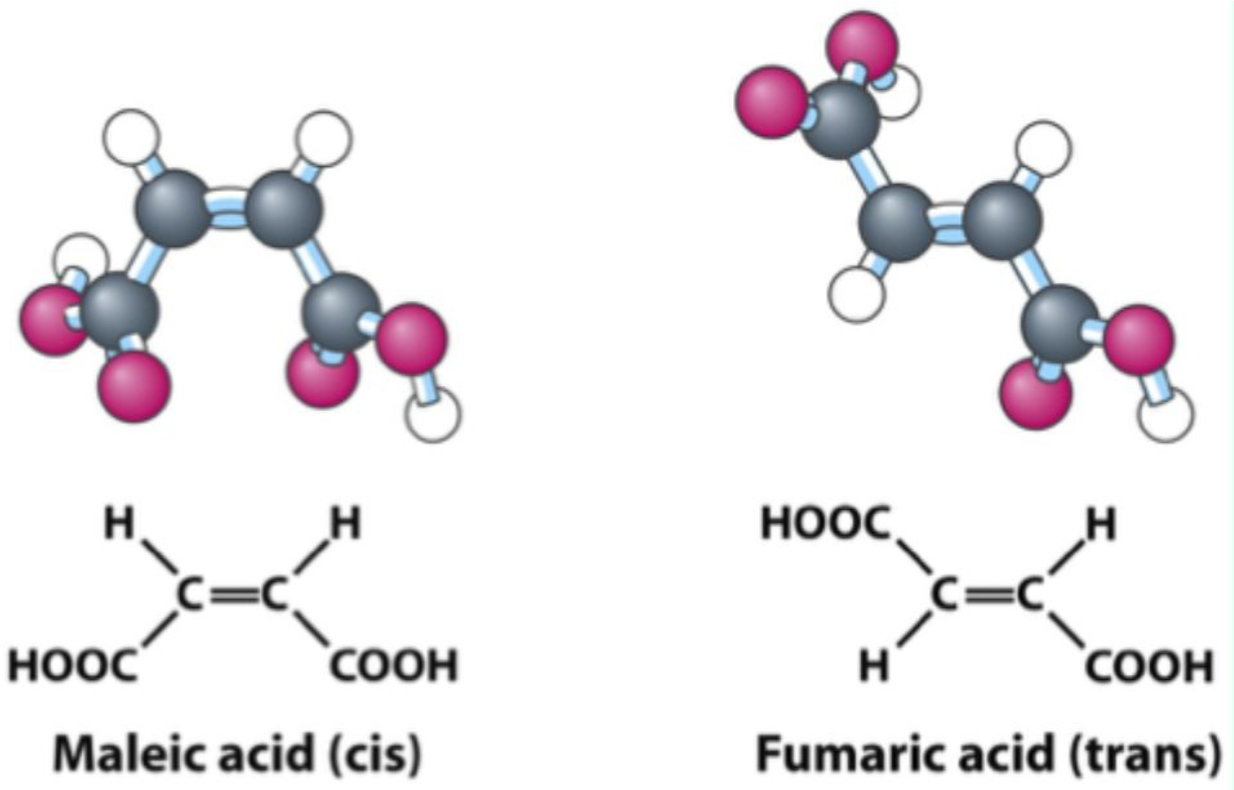
What are geometric (cis-trans) isomers?
Stereoisomers that differ in substituent arrangement around a non-rotating double bond.
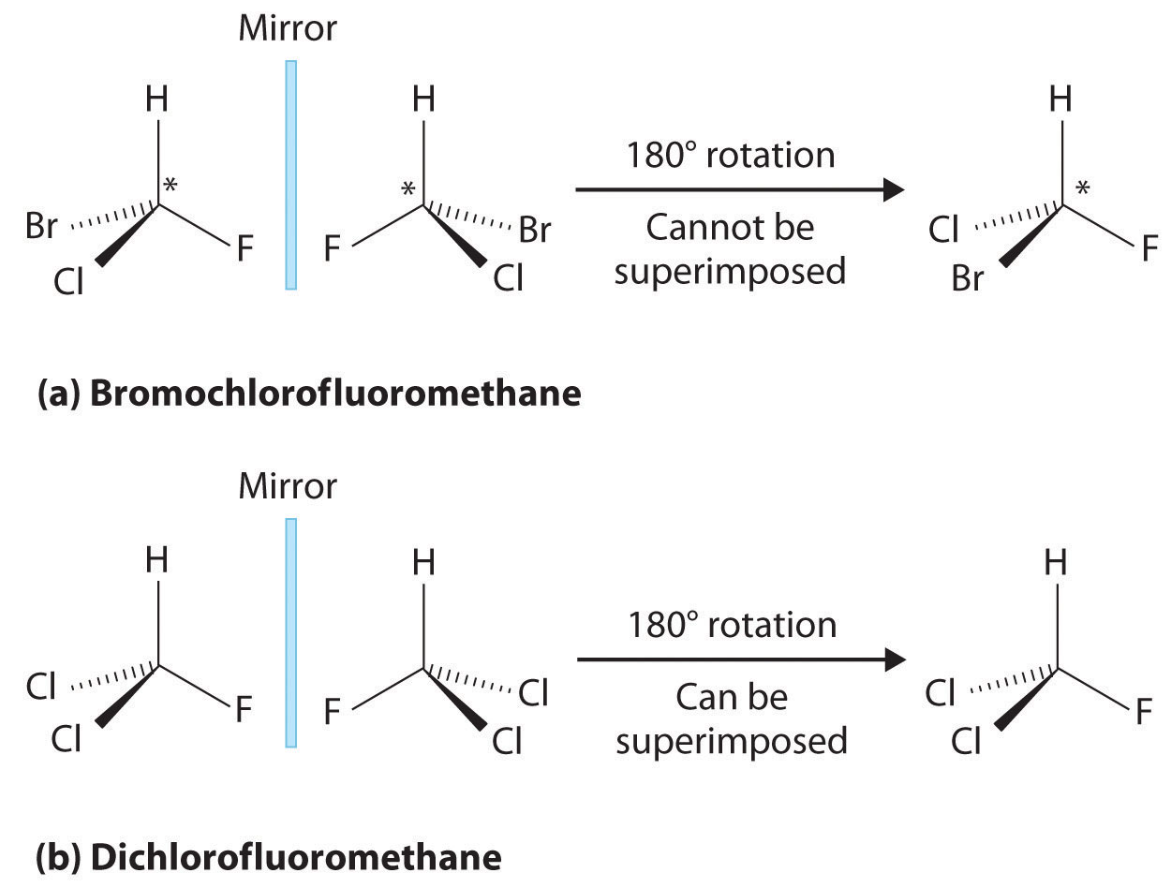
What is a chiral center and what makes a molecule chiral?
A carbon with four different substituents and it cannot be superimposed on its mirror image.
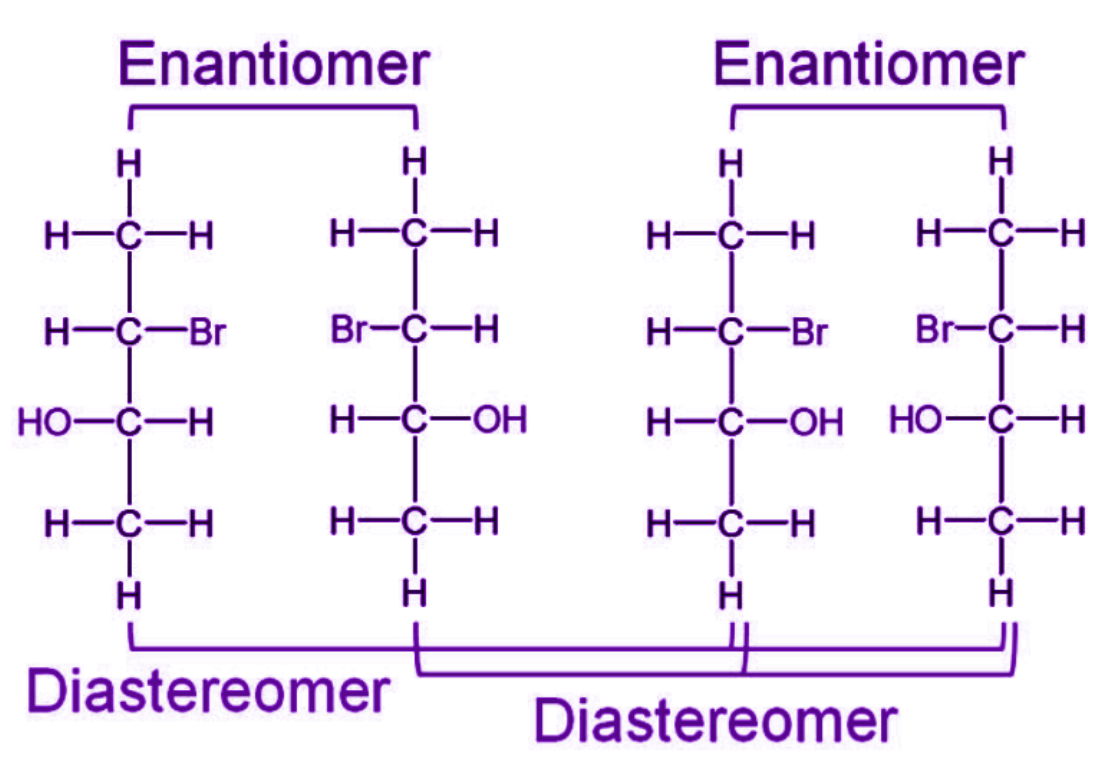
What are the differences between enantiomers and diastereoisomers?
Enantiomers are mirror-image stereoisomers, while diastereoisomers are not mirror images of each other.
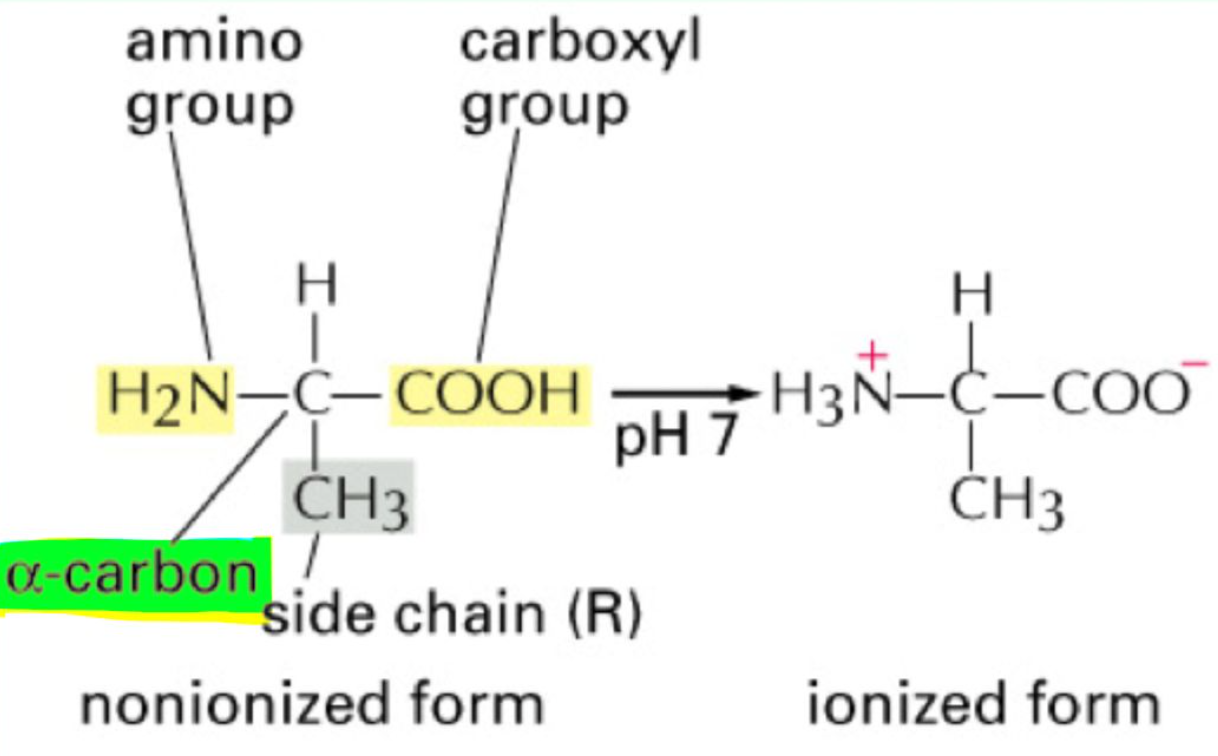
What four groups are attached to the α-carbon of amino acids?
Amino group, carboxyl group, hydrogen, R group (side chain).
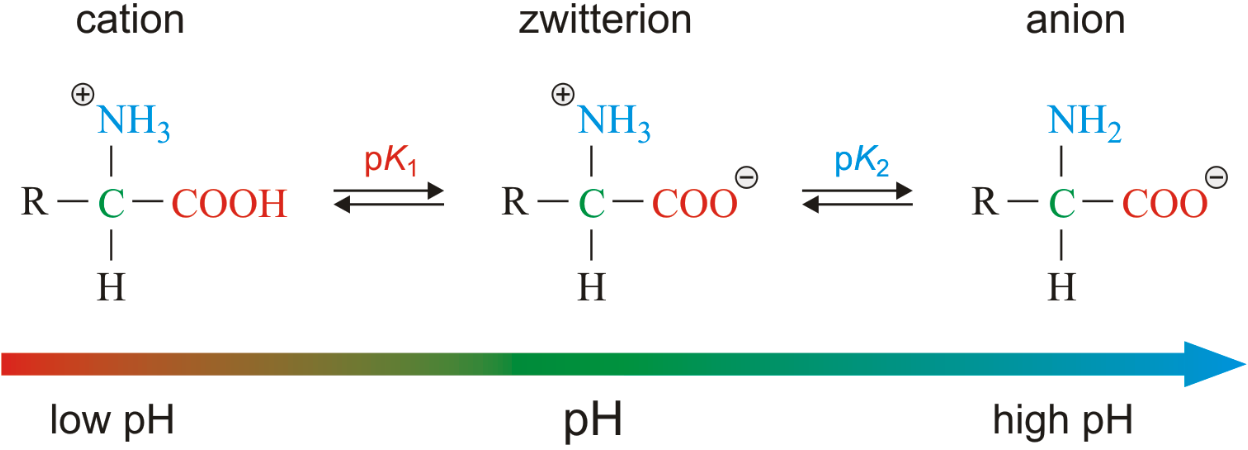
At neutral pH (~7), what form do amino acids exist in?
Zwitterion (both amino and carboxyl groups are ionized).
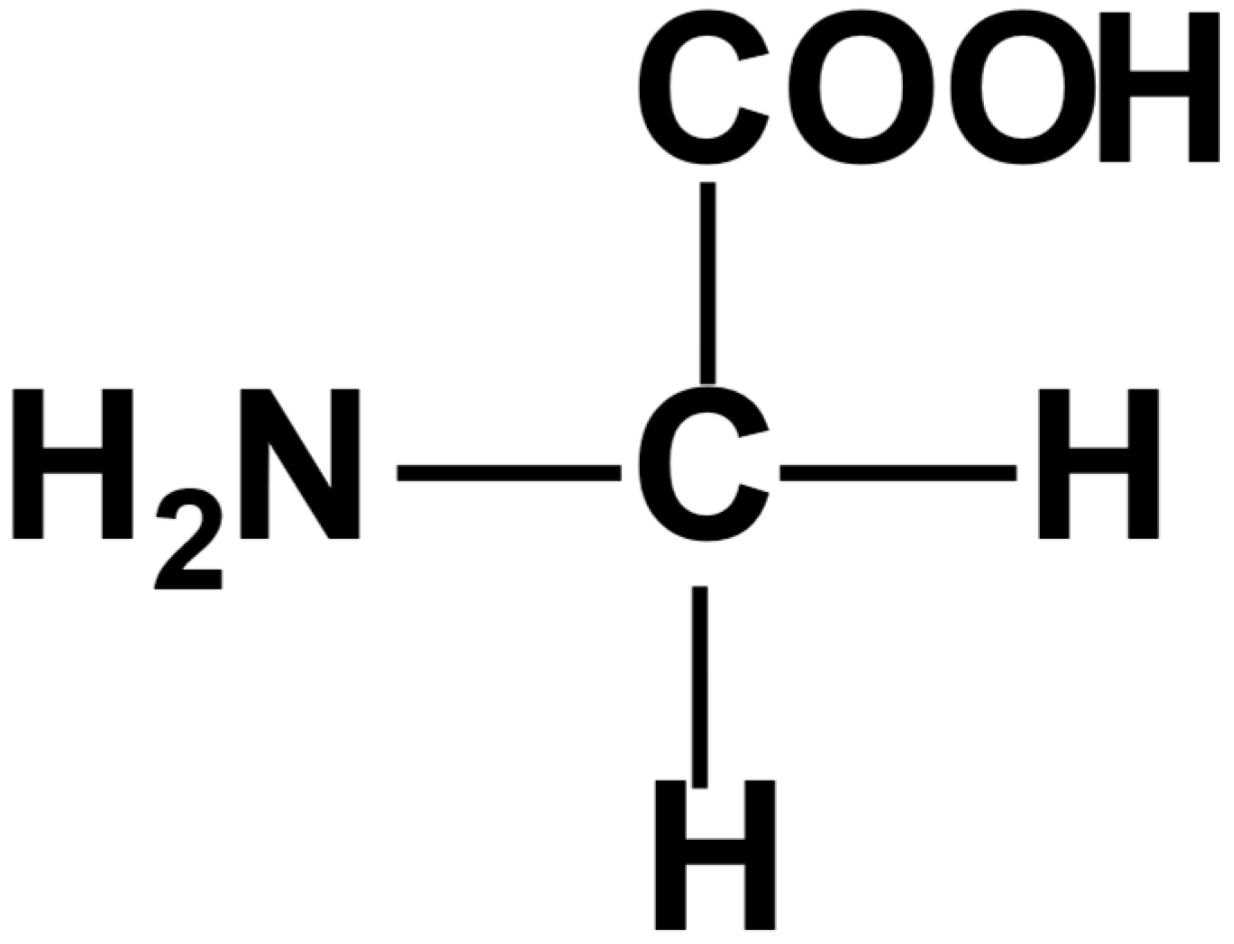
Which amino acid is achiral?
Glycine
Which amino acid configuration is used in proteins?
L-amino acids only
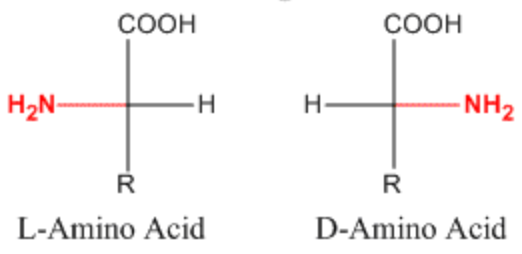
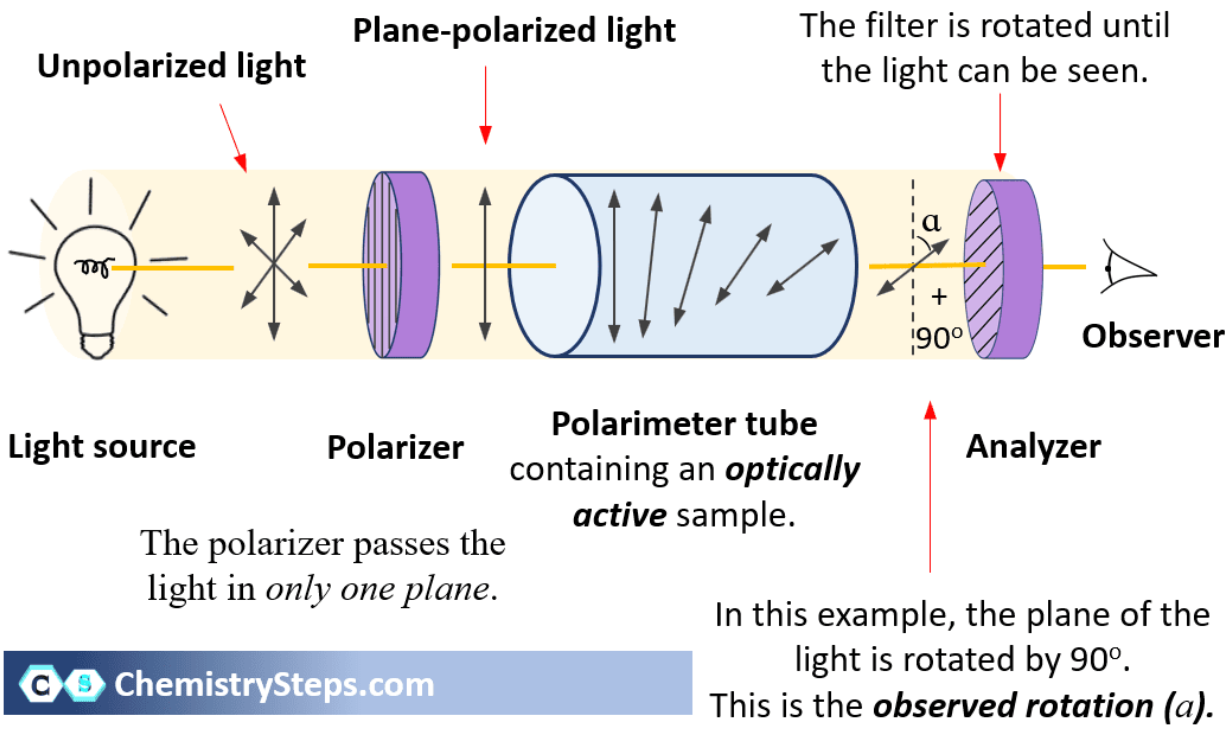
How can enantiomers usually be distinguished experimentally?
By interaction with plane-polarized light (optically active).
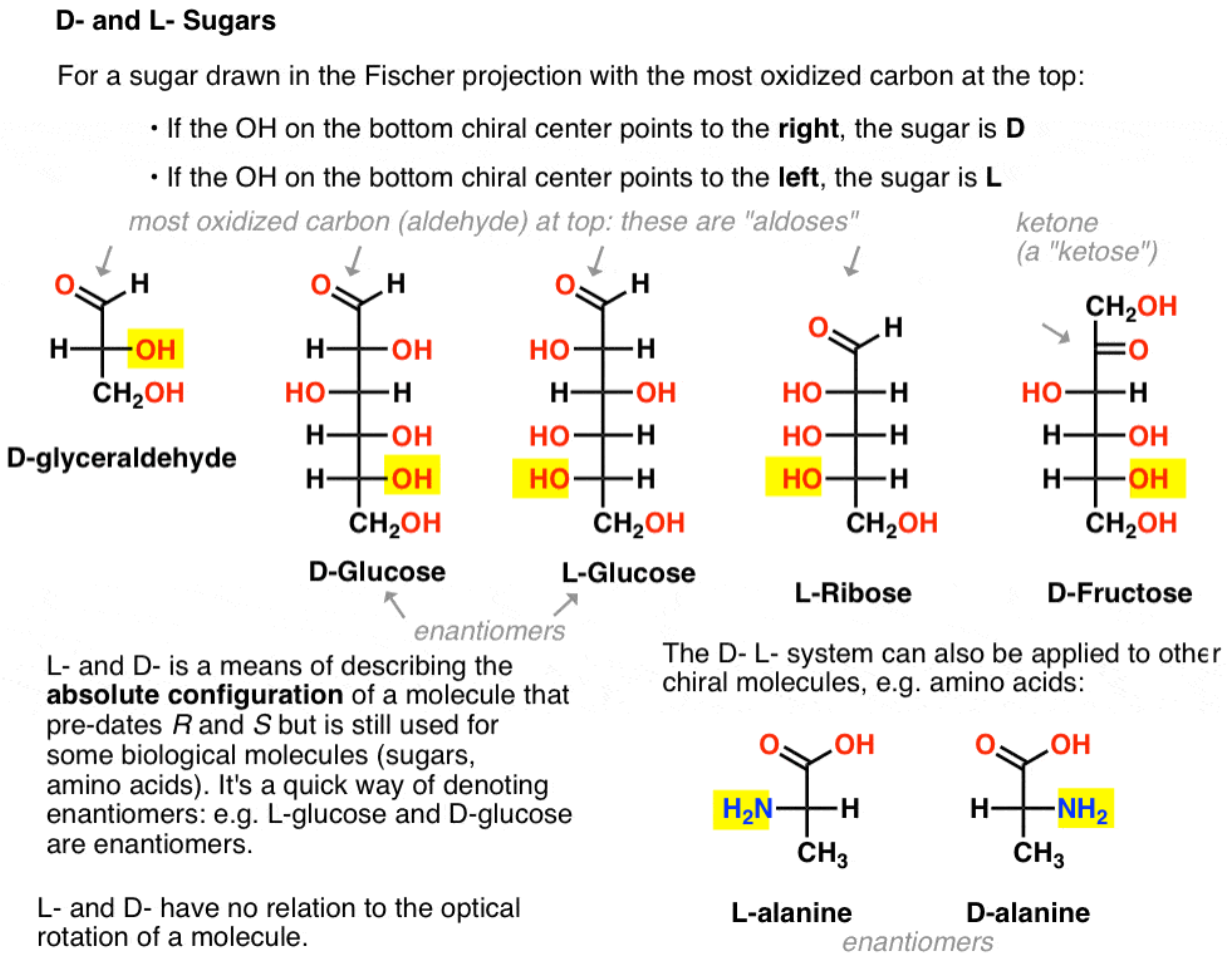
What convention is used to define L and D amino acids?
Comparison to glyceraldehyde.
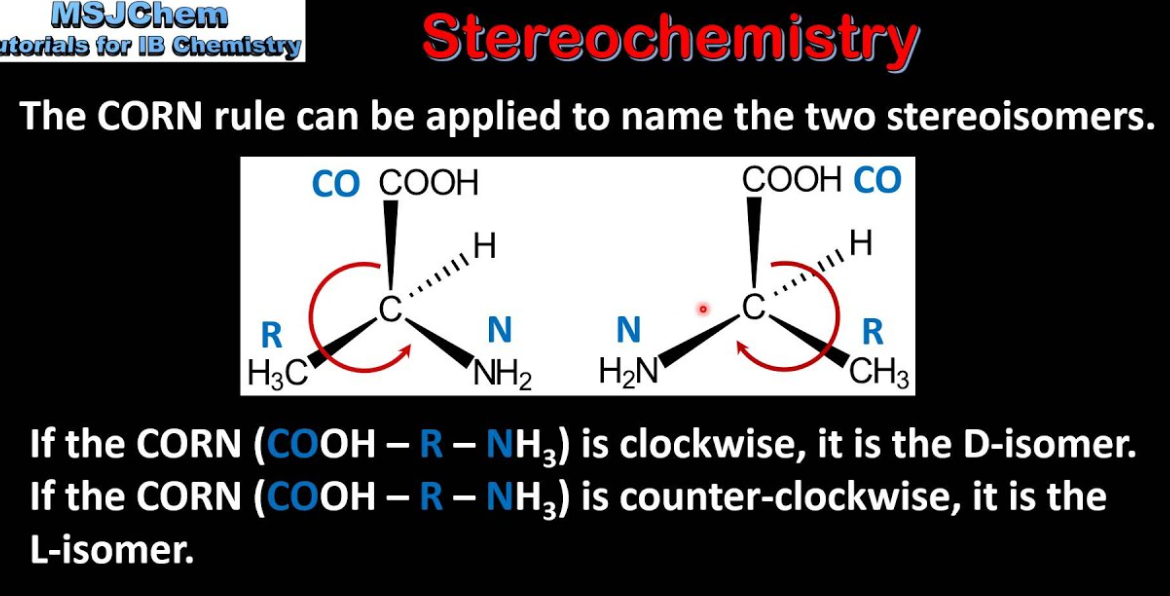
How do you determine L vs. D configuration using the CORN rule?
Look down the H-Cα bond
If CO -> R -> N (CORN) goes clockwise, it's L configuration.
In a Fischer projection, which substituents point behind the page?
Vertical substituents
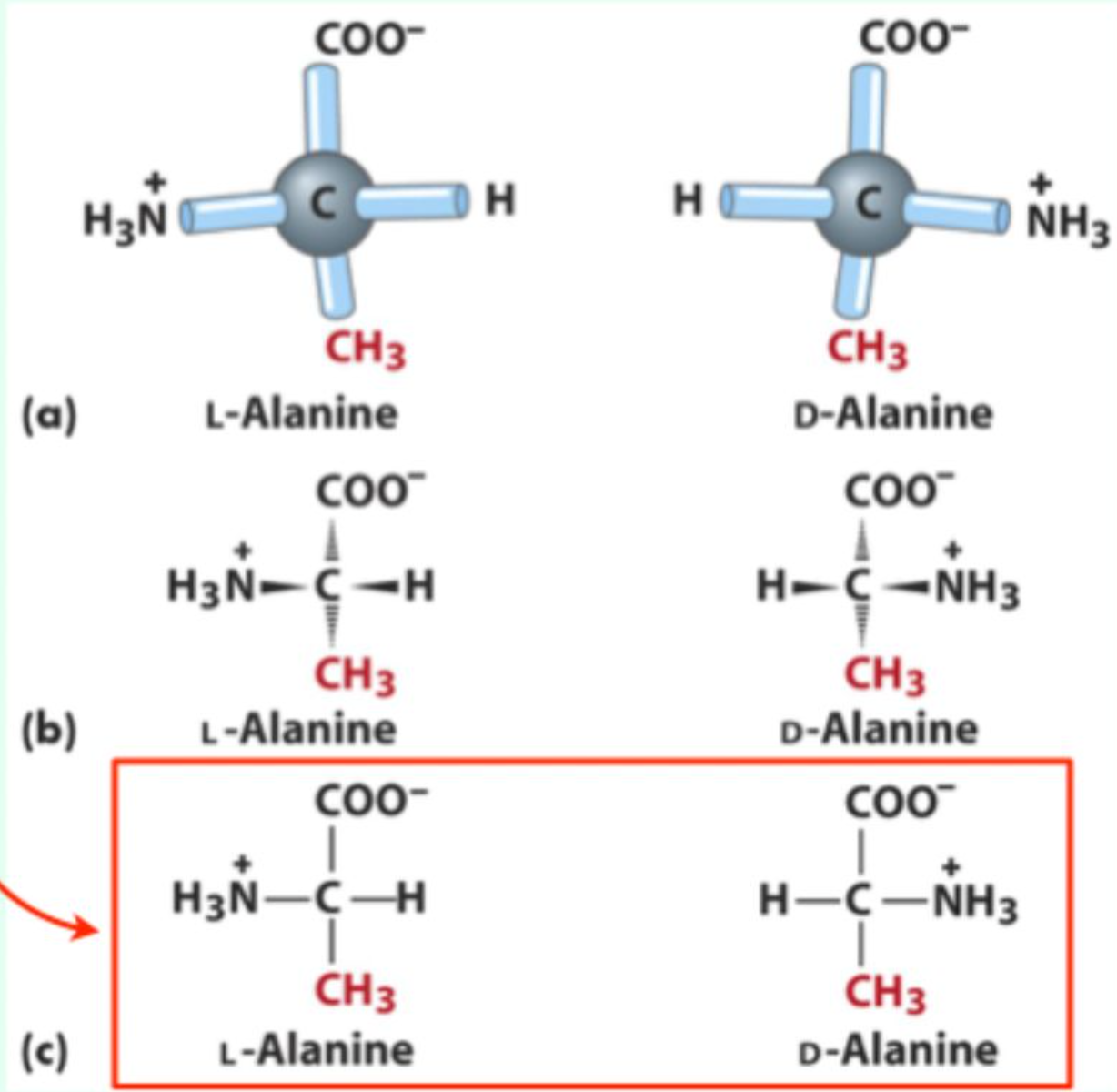
In a Fischer projection, which substituents point out of the page?
Horizontal substituents
Where can D-amino acids be found?
Bacterial cell walls and some peptide antibiotics.
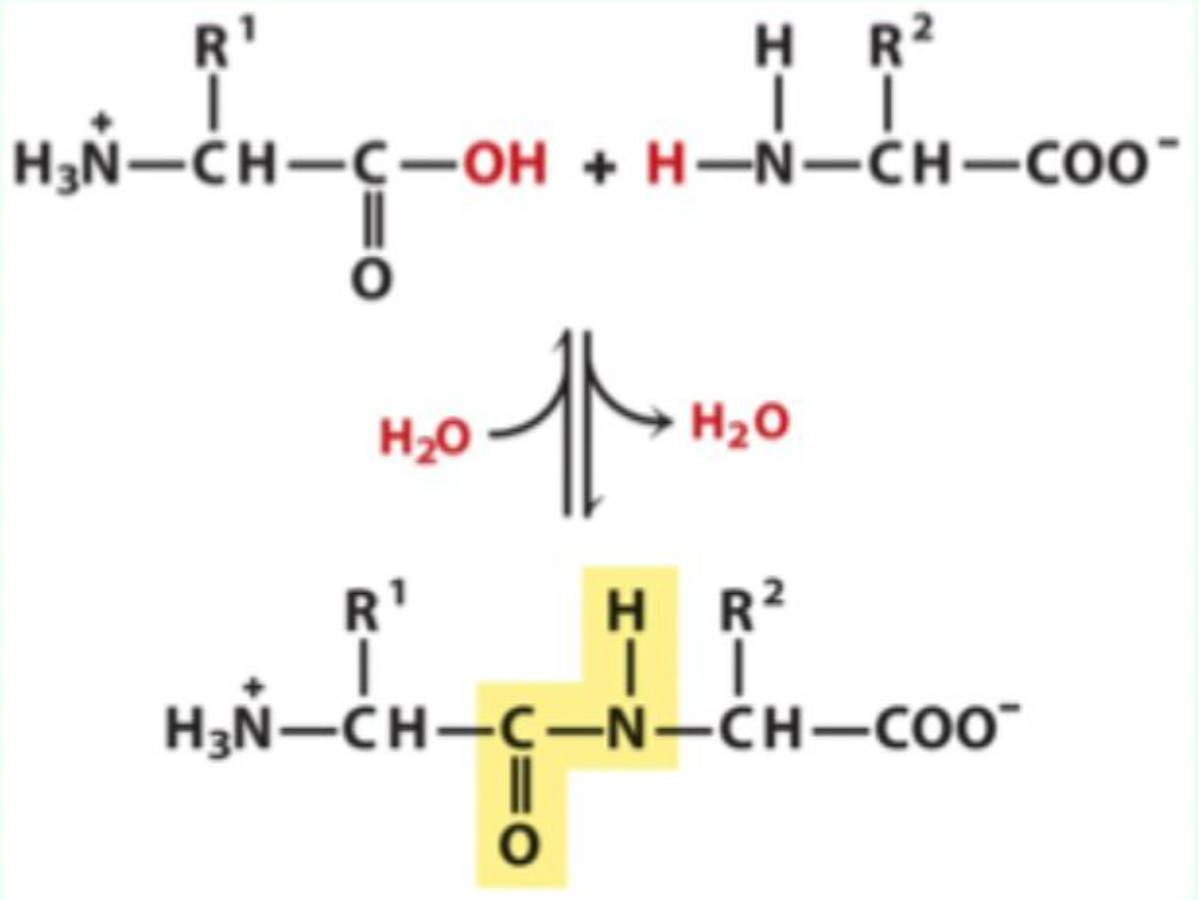
What bond links amino acids together?
Peptide bonds
What is the protein backbone?
The repeating main chain, excluding side chains.
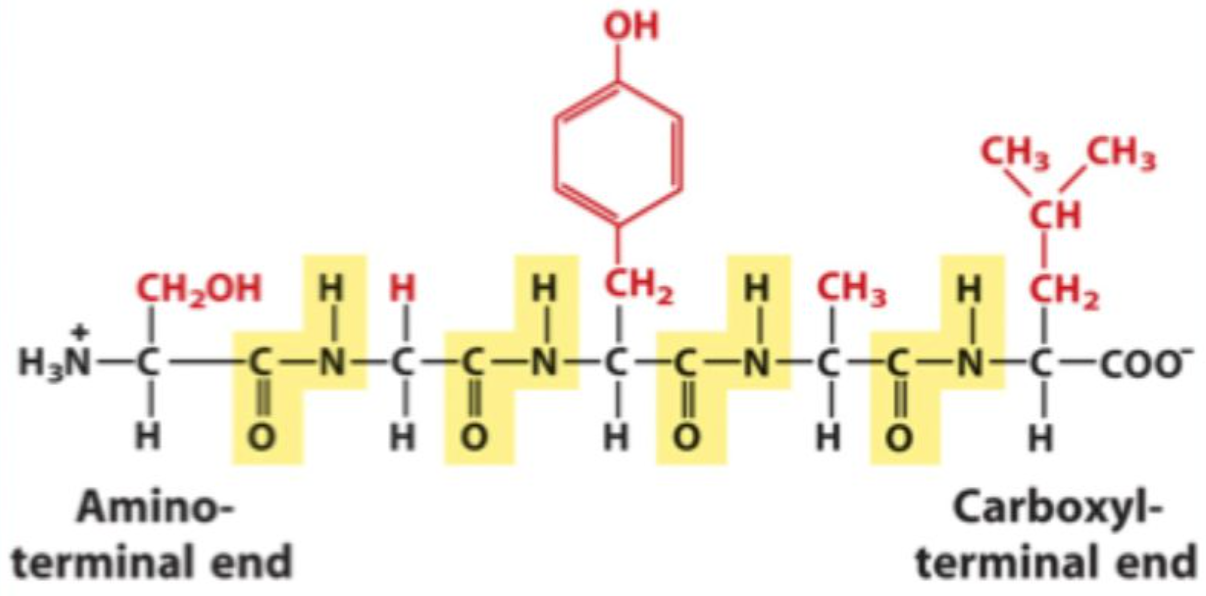
What are residues?
Amino acids after incorporation into a peptide.
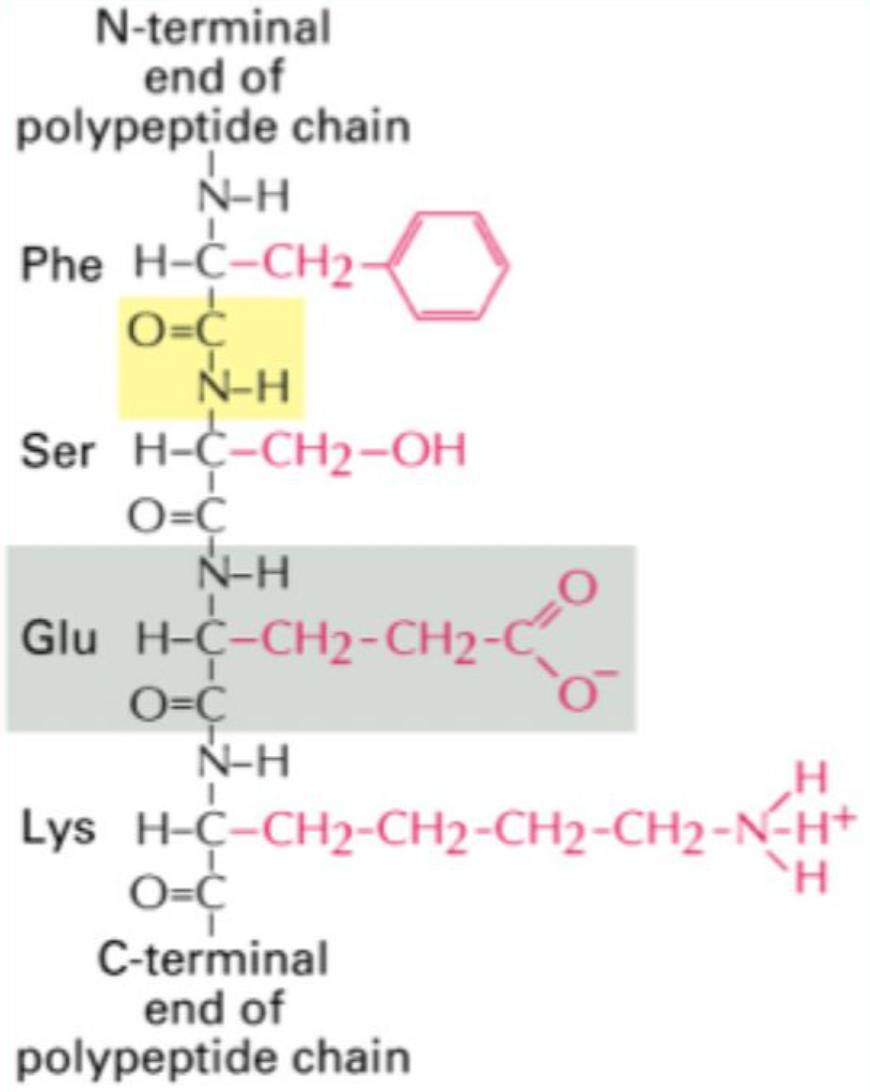
What is the average mass of an amino acid in a protein?
~110 Da
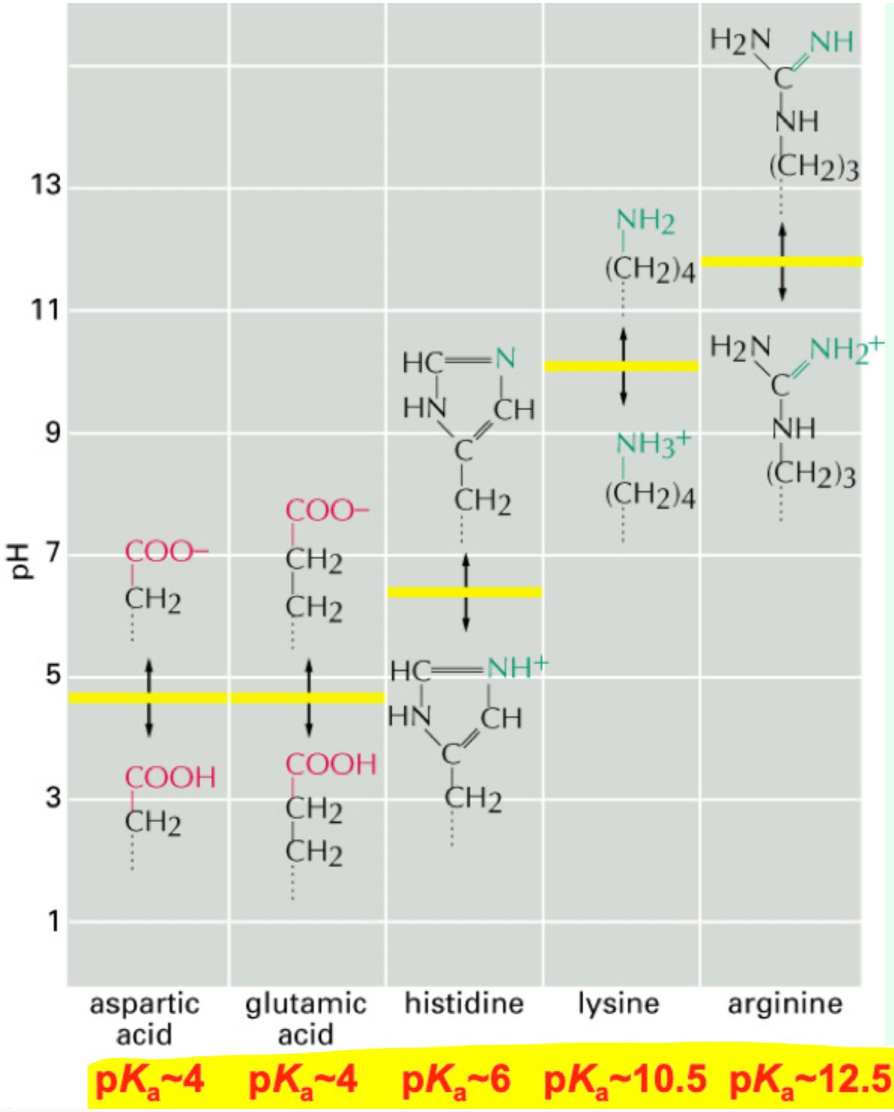
Which amino acids have charged side chains at physiological pH? And what are their pKas?
Arg = 12.5
Lys = 10.5
His = 6.0
Asp/Glu = 4.0
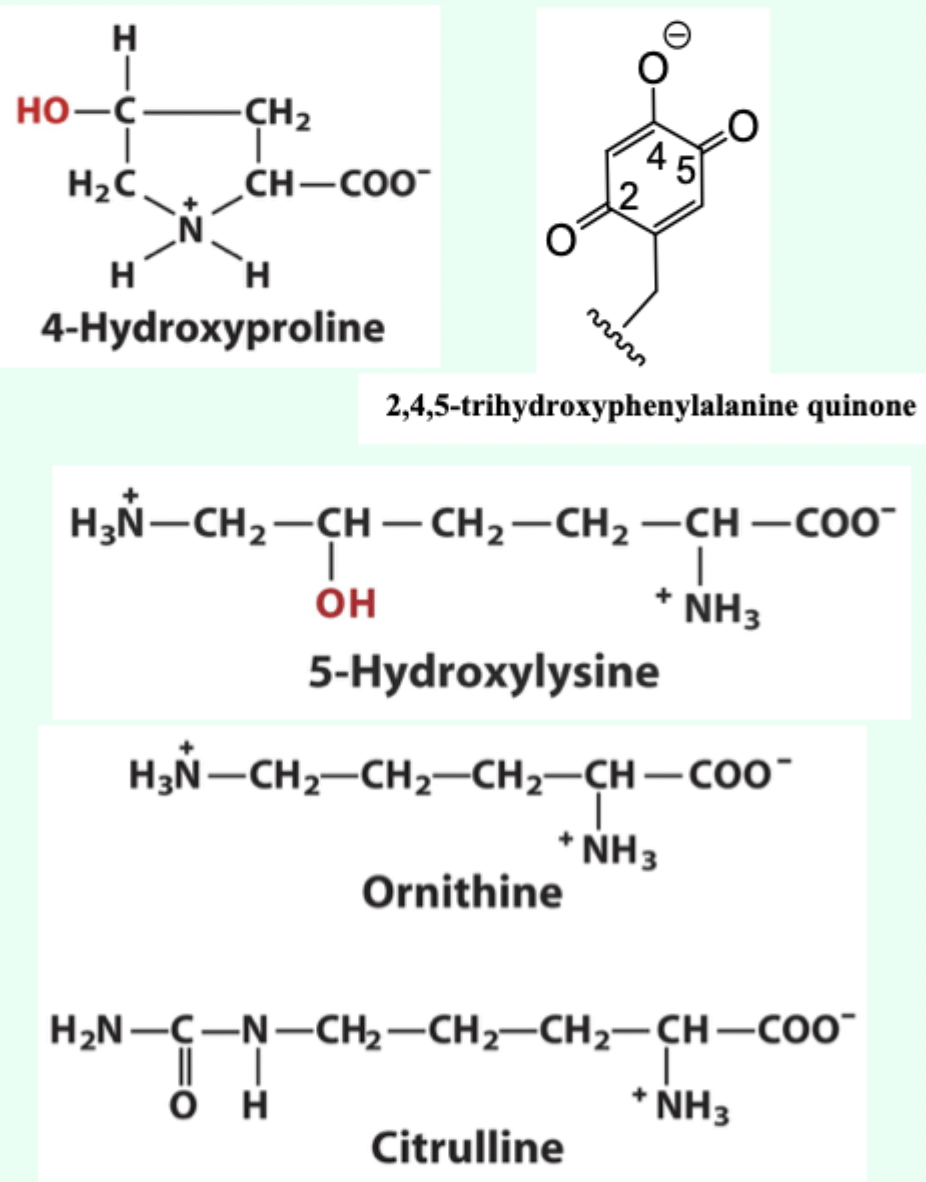
How do uncommon amino acids usually arise in proteins?
By post-translational modification
What are the special properties of proteins made from post-translational modification?
Structural (4-hydroxyproline and 5-hydroxylysine (found in collagen))
Involved in catalysis (2,4,5-trihydroxyphenylalanine quinone (in copper amine oxidase))
Metabolites (Ornithine and citrulline)
What does delocalize mean and what causes electrons to delocalize?
π-electrons are not confined to one bond or atom but are spread out over several atoms in a molecule. Electrons delocalize when double bonds are separated by only a single bond, allowing π-electrons to spread over the entire conjugated system.
What is a conjugated system?
A chain of atoms with alternating single and double bonds that allows π-electrons to delocalize across the system.
What structural feature do conjugated systems have?
They are planar and cannot freely rotate because of electron delocalization.
What are the four levels of protein structure?
Primary, secondary, tertiary, quaternary.
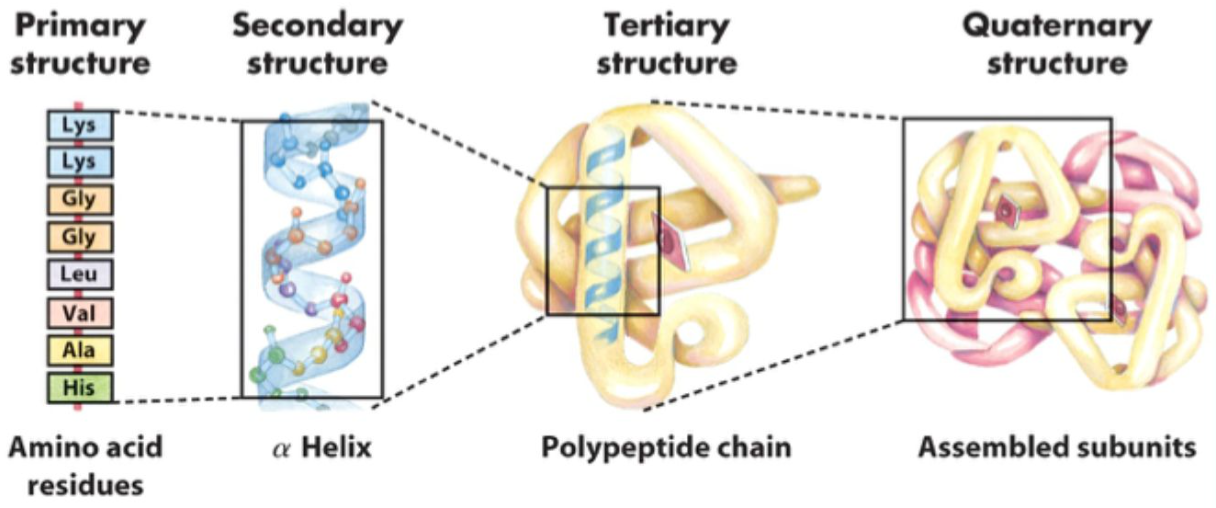
What is primary protein structure?
The covalent structure of the protein.

What does primary structure include ?
The amino acid sequence and all chemical modifications, including disulfide bonds.

What is the difference between an interchain disulfide bond and an intrachain disulfide chain?
Inter: Linking two different polypeptide chains.
Intra: Within the same polypeptide chain.

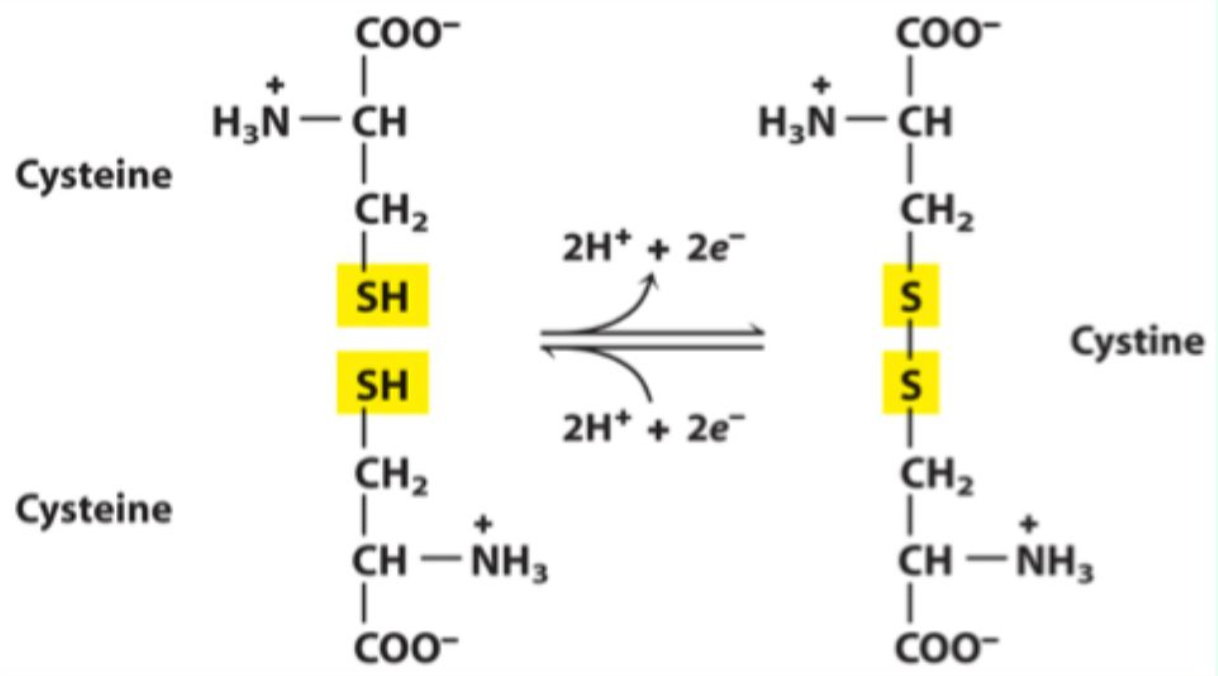
Which functional group of cysteine forms disulfide bonds?
-SH (thiol/sulfhydryl) group
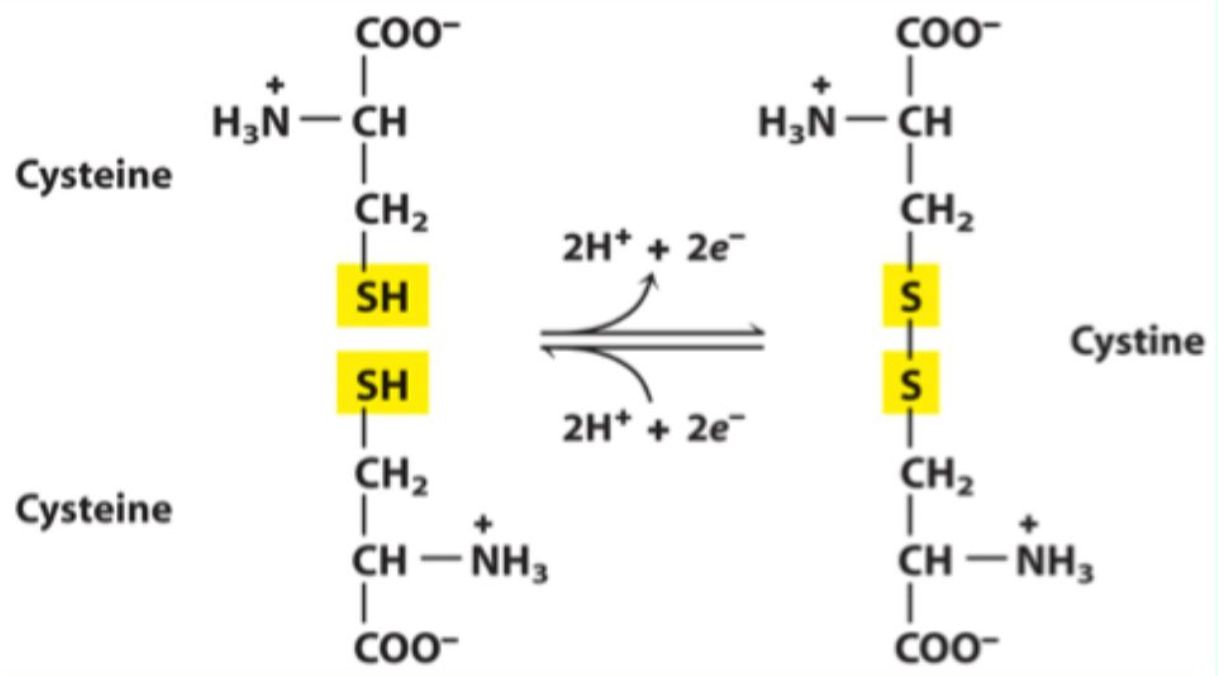
What reaction forms a disulfide bond?
Oxidation
What is oxidation?
Loss of electrons

What is reduction?
Gain of electrons

What is an oxidant?
The species that causes oxidation and is itself reduced.

What is a reductant?
The species that causes reduction and is itself oxidized.

What are post-translational modifications (PTMs)?
Covalent modifications added after protein translation.
Name common PTMs
Glycosylation, methylation, phosphorylation, acetylation, membrane anchors (GPI, fatty acids, prenyl groups)
What is glycosylation?
The addition of sugars (carbohydrates) to proteins.
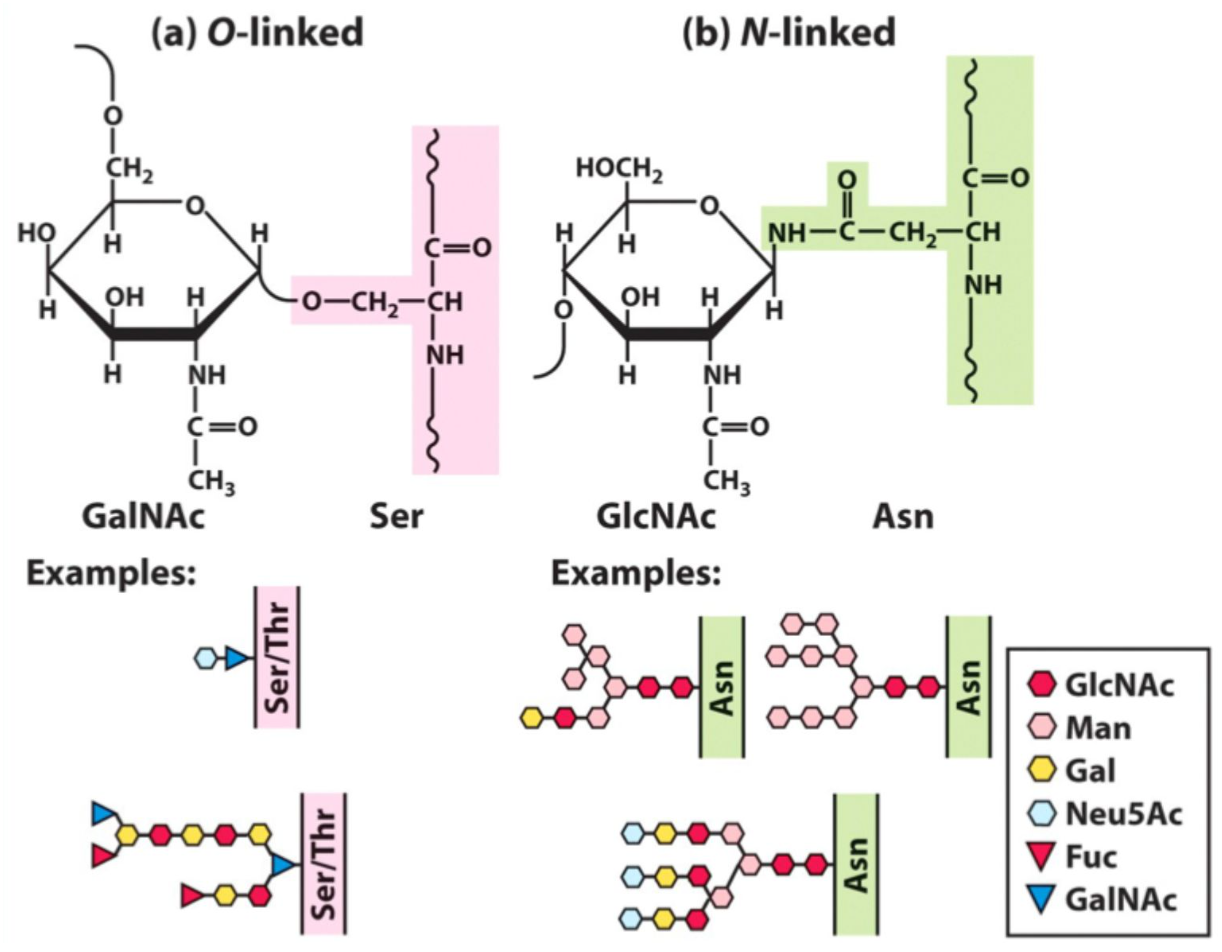
Which amino acids are initially glycosylated?
Ser, Thr, and Asn
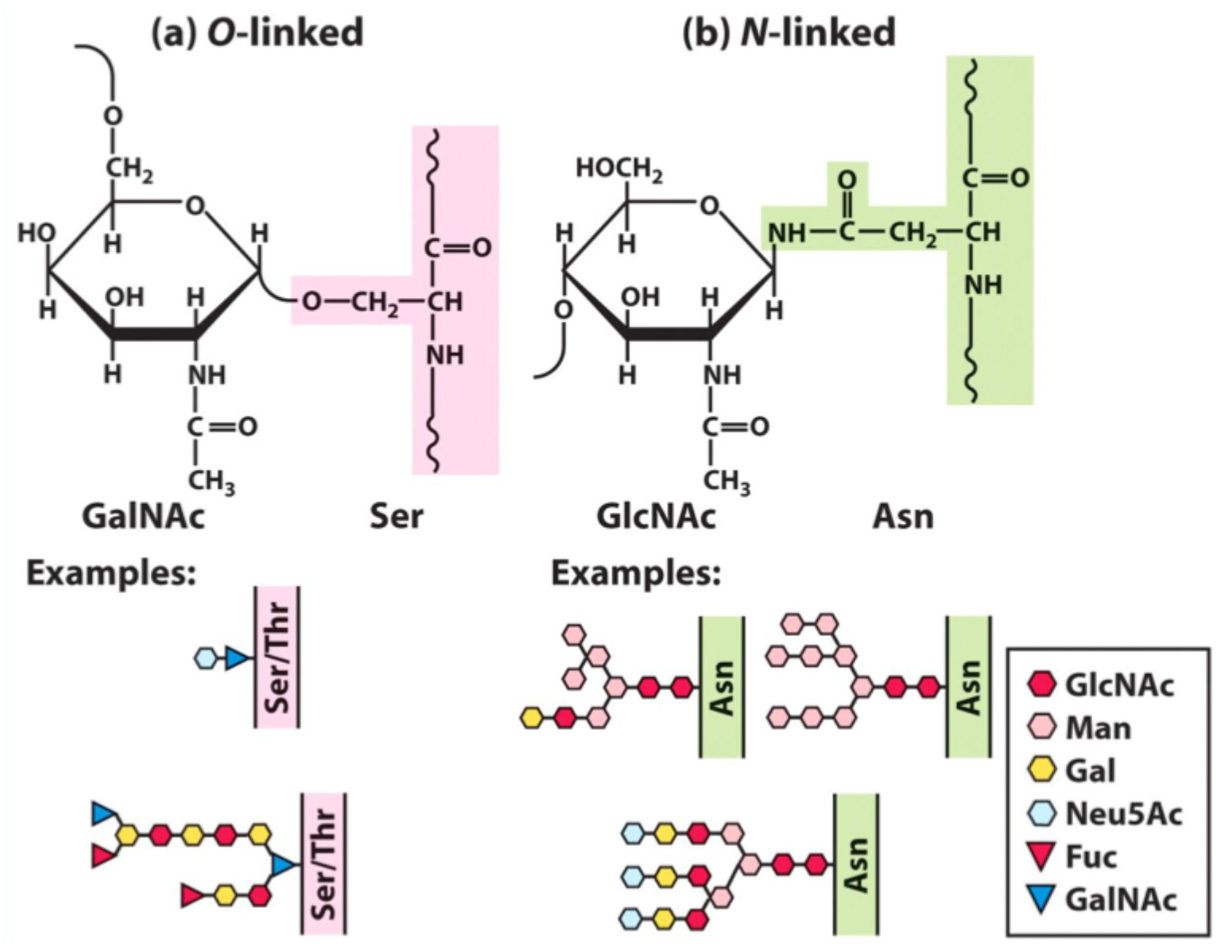
What is O-linked glycosylation?
Glycosylation via Ser or Thr hydroxyl groups.
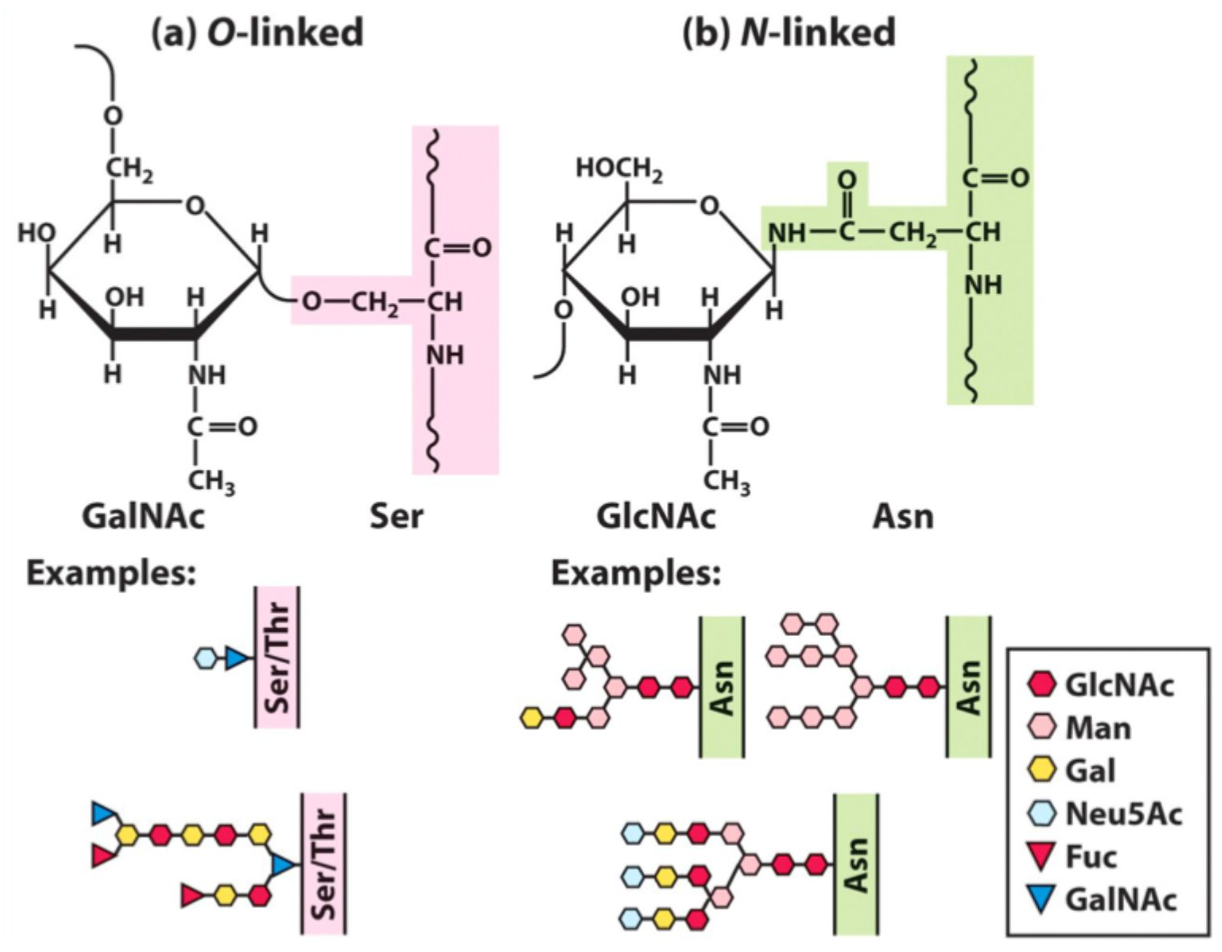
What is N-linked glycosylation?
Glycosylation via Asn amine group.
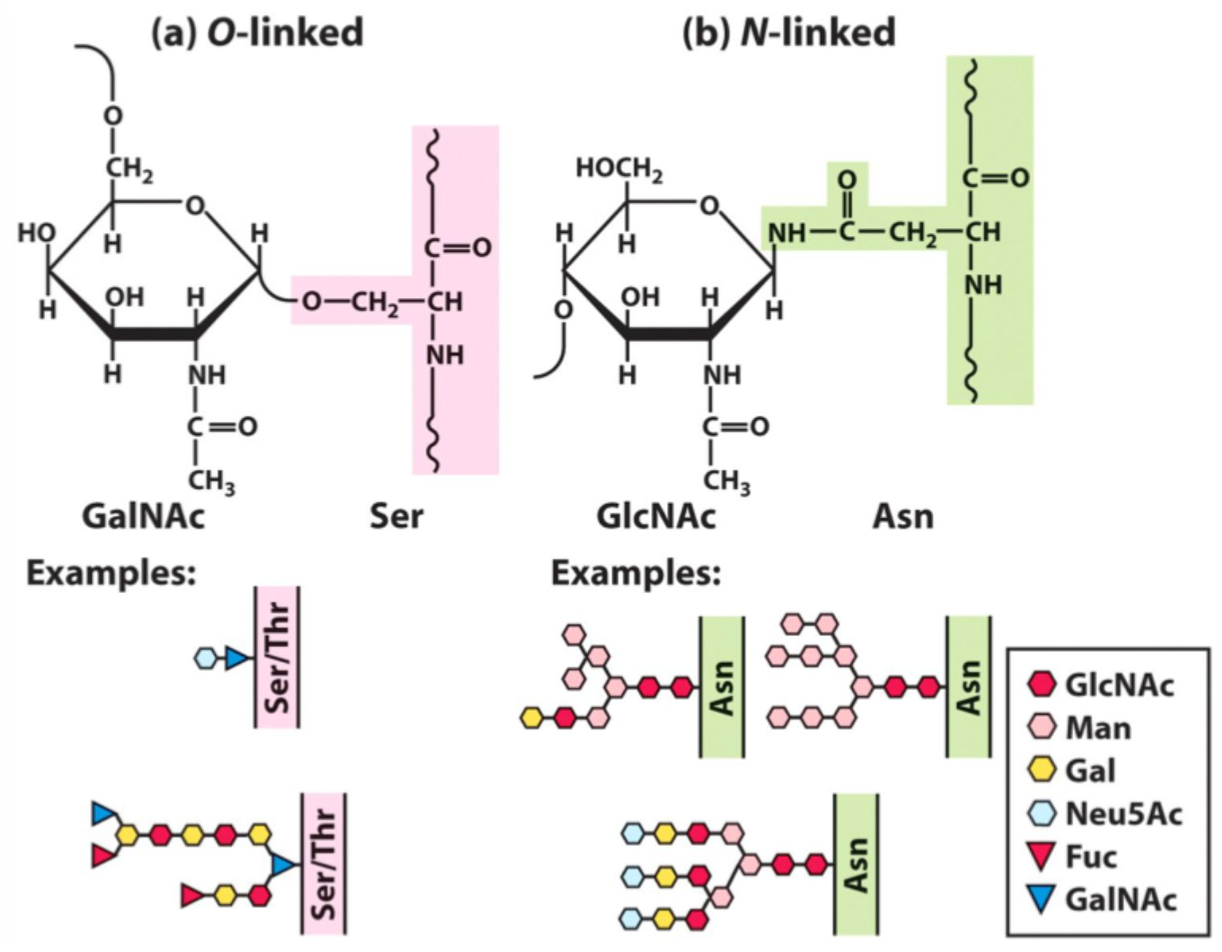
What is the structure of most glycan chains?
Branched oligosaccharides, often charged.
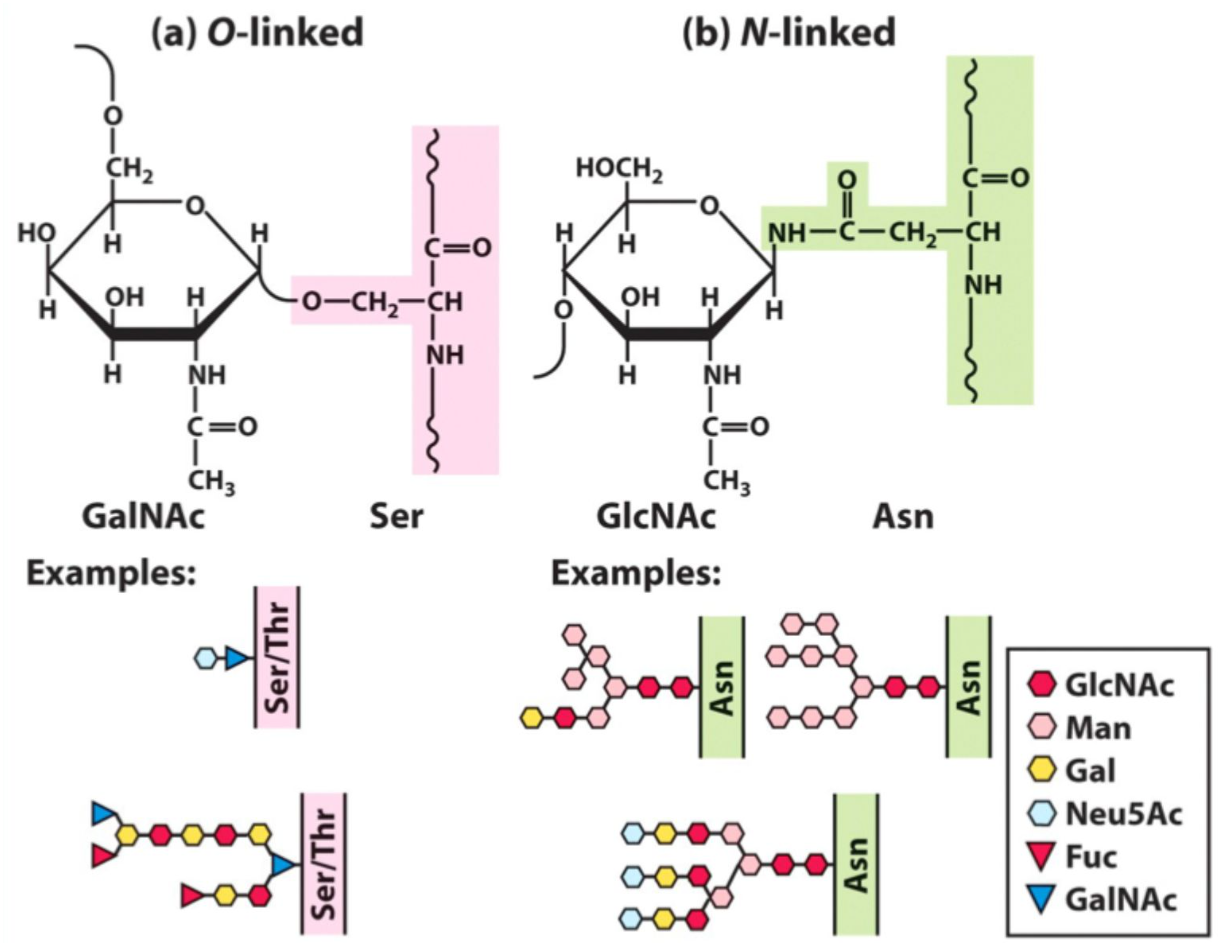
What are glycoproteins?
Proteins with covalently attached carbohydrates.
Most proteins in the blood are what?
Glycoproteins
What is the most important example of acetylation?
Histone acetylation