Reactions, Energy Changes, and Bond Energies in Chemistry
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What accompanies all chemical reactions?
Energy changes
What is an example of a combustion reaction?
The oxidation of hydrocarbons to heat houses and drive cars.
What is the reaction formula for the combustion of hydrogen?
2H2 + O2 → 2H2O
What is the ΔH for the combustion of hydrogen?
ΔH = -286 kJ/mol
What processes require energy during a reaction?
Breaking bonds
What processes produce energy during a reaction?
Forming bonds
What is a bond dissociation energy (BDE)?
The energy required to break a bond in the gas phase.
What is the BDE for breaking an H-H bond?
486 kJ/mol
How is ΔH for a reaction calculated?
ΔH = ΣBDE bonds broken - ΣBDE bonds formed
What is the significance of a negative ΔH value?
It indicates an exothermic reaction that releases energy.
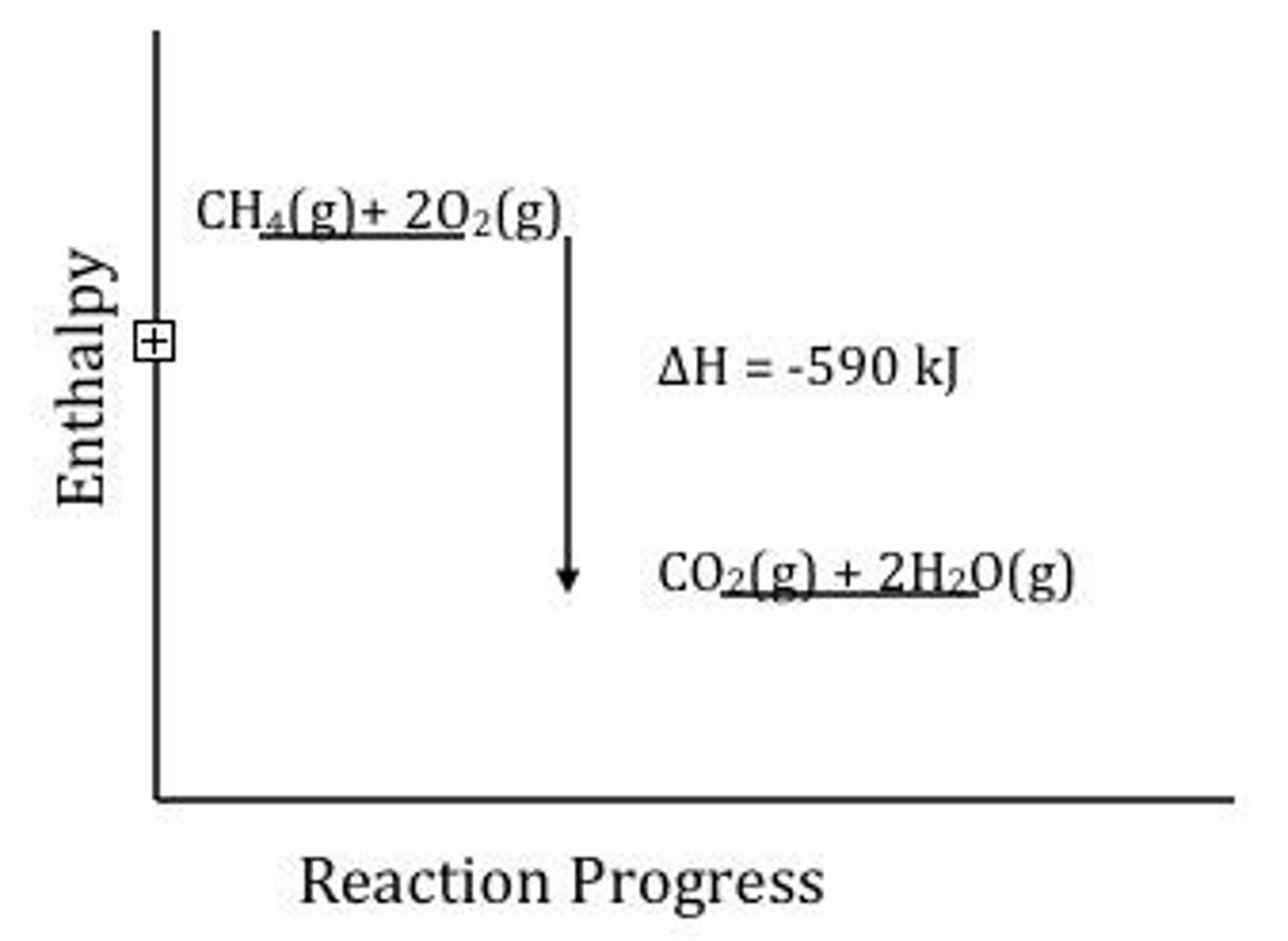
What is the ΔH for the reaction CH4(g) + 2O2(g) ⇌ CO2(g) + 2H2O(g)?
ΔH = -810 kJ/mol
What is the relationship between bond length and bond strength?
Shorter bonds are typically stronger.
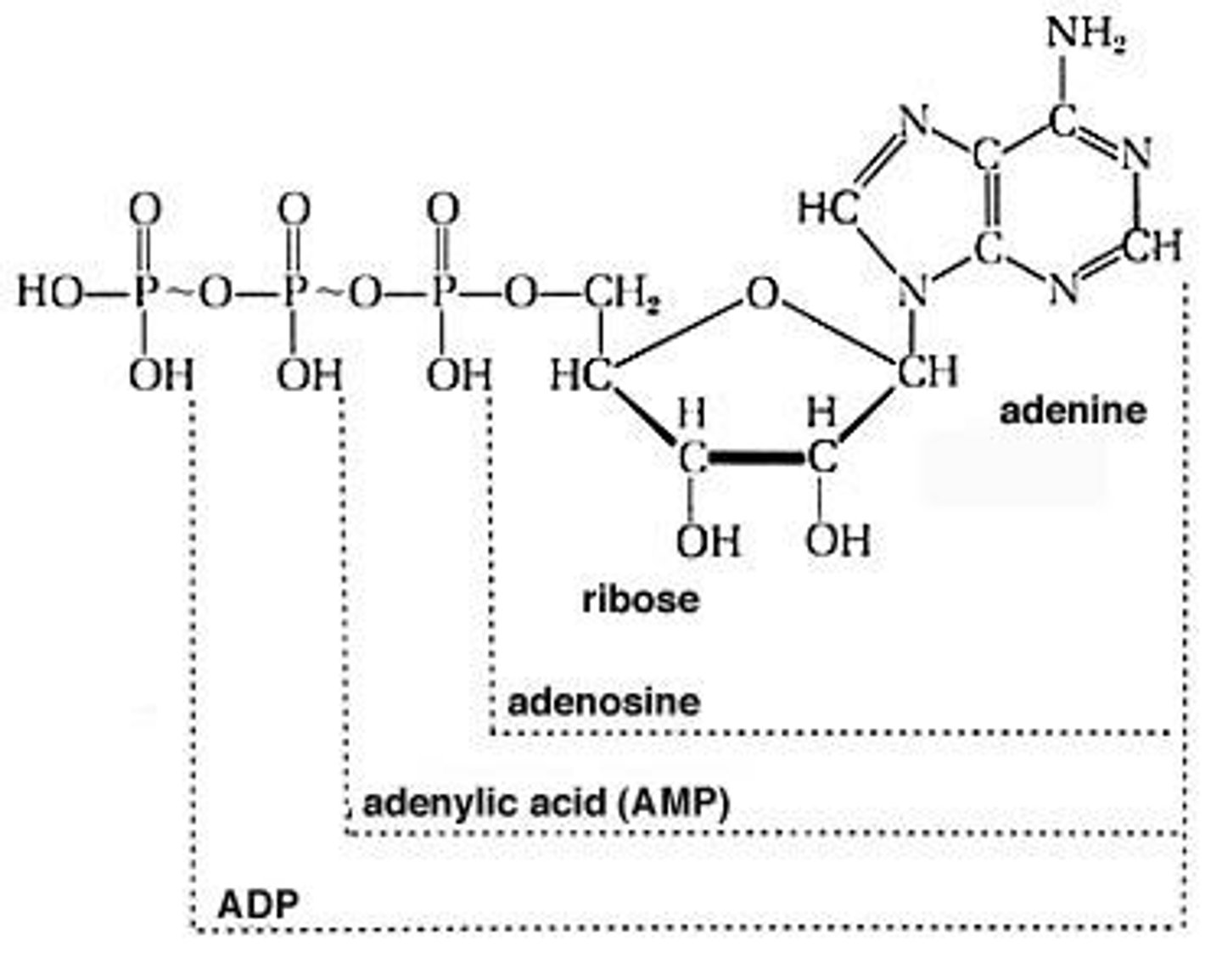
What is the role of ATP hydrolysis in biochemical reactions?
It drives unfavorable reactions due to its negative energy change.
What is the ΔH for ATP hydrolysis?
ΔH° ~ -29 kJ/mol (at pH 7)
What is the consequence of CO2 being a stable product of combustion?
It requires a lot of energy to break its strong bonds.
What is the difference between calculating ΔH from bond energies and calorimetry?
Bond energies are typically measured in gas phase, while calorimetry is usually in solution.
What is the general trend for multiple bonds compared to single bonds?
Multiple bonds are stronger than single bonds.
What is the energy change when bonds are formed?
Energy is released to the surroundings.
What is the energy change when bonds are broken?
Energy input is required from the surroundings.
How can coupled reactions be useful?
They exploit common intermediates to drive unfavorable reactions.
What happens to the potential energy of a system as bonding atoms get closer?
The potential energy drops, forming a stable bond.
What is the significance of bond formation in terms of energy?
Bond formation represents a stable state and releases energy.