Antibacterial Agents
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
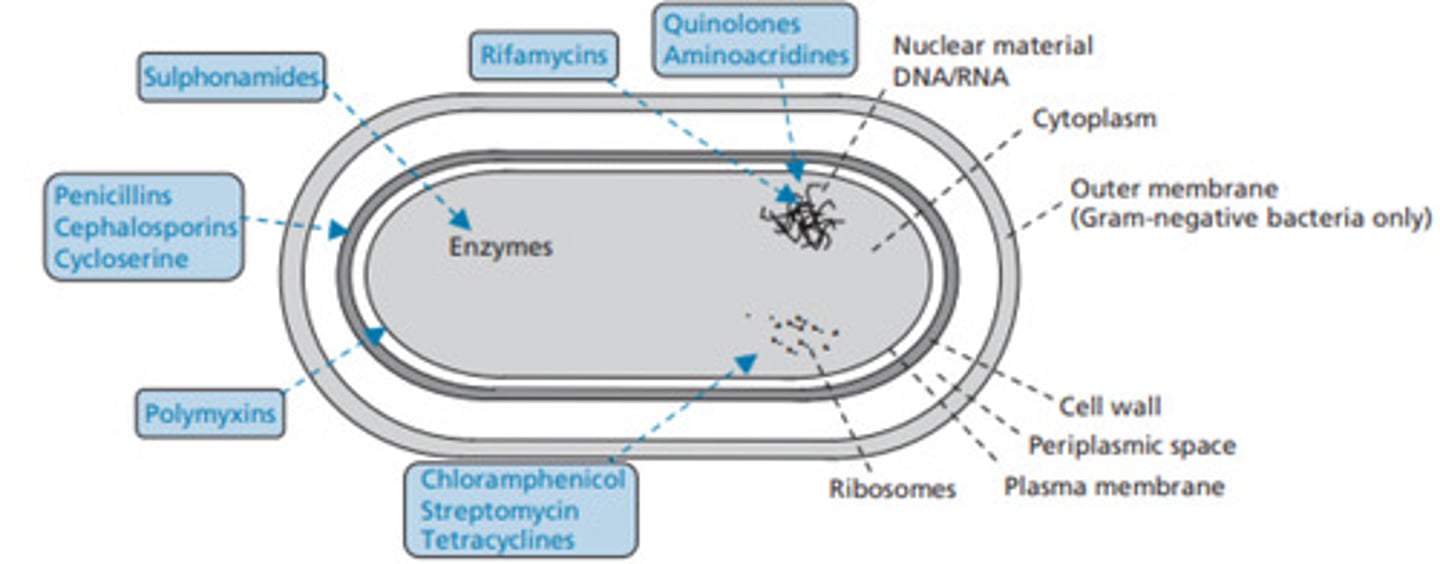
What are the mechanisms behind how antibacterial work
Depends what type:
-Inhibition of bacterial cell wall synthesis: Faulty wall, which is unable to control the flow of nutrient, leads cell lysis and death, e.g. β-lactam antibiotics
- Interaction with the plasma membrane: Affect membrane permeability, e.g. polymyxins
- Inhibition of cell metabolism (Antimetabolites): Target bacterial enzyme catalysis impending bacterial growth, e.g. Sulphonamides
-Disruption of protein synthesis: Block the synthesis of essential proteins and enzymes, e.g. Chloramphenicol and tetracyclines
-Inhibition of nucleic acid transcription and replication: Target transcription and replication, which impedes cell division e.g. Nalidixic acid and proflavine
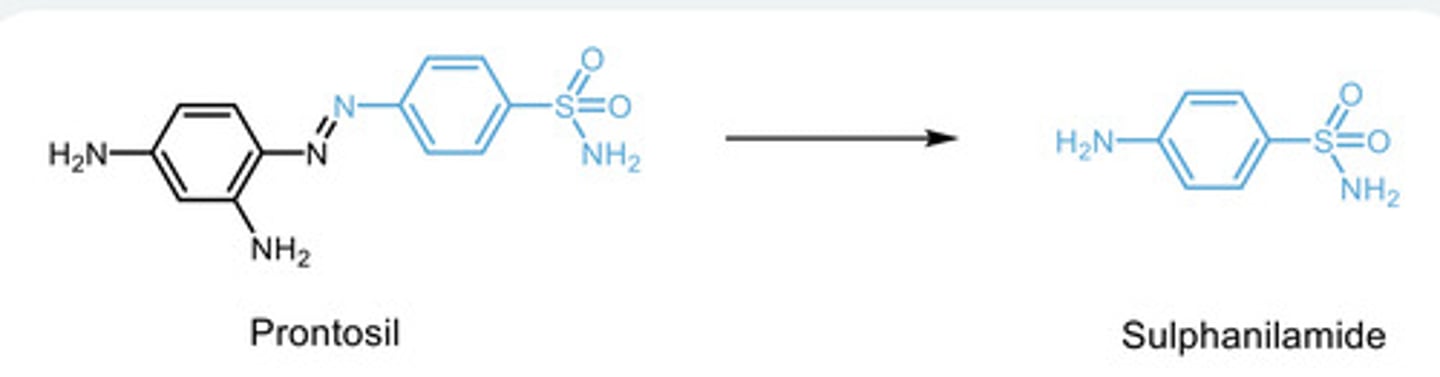
What is prontosil metabolised to form?
Sulphanilamide (metabolised by intestinal bacteria)
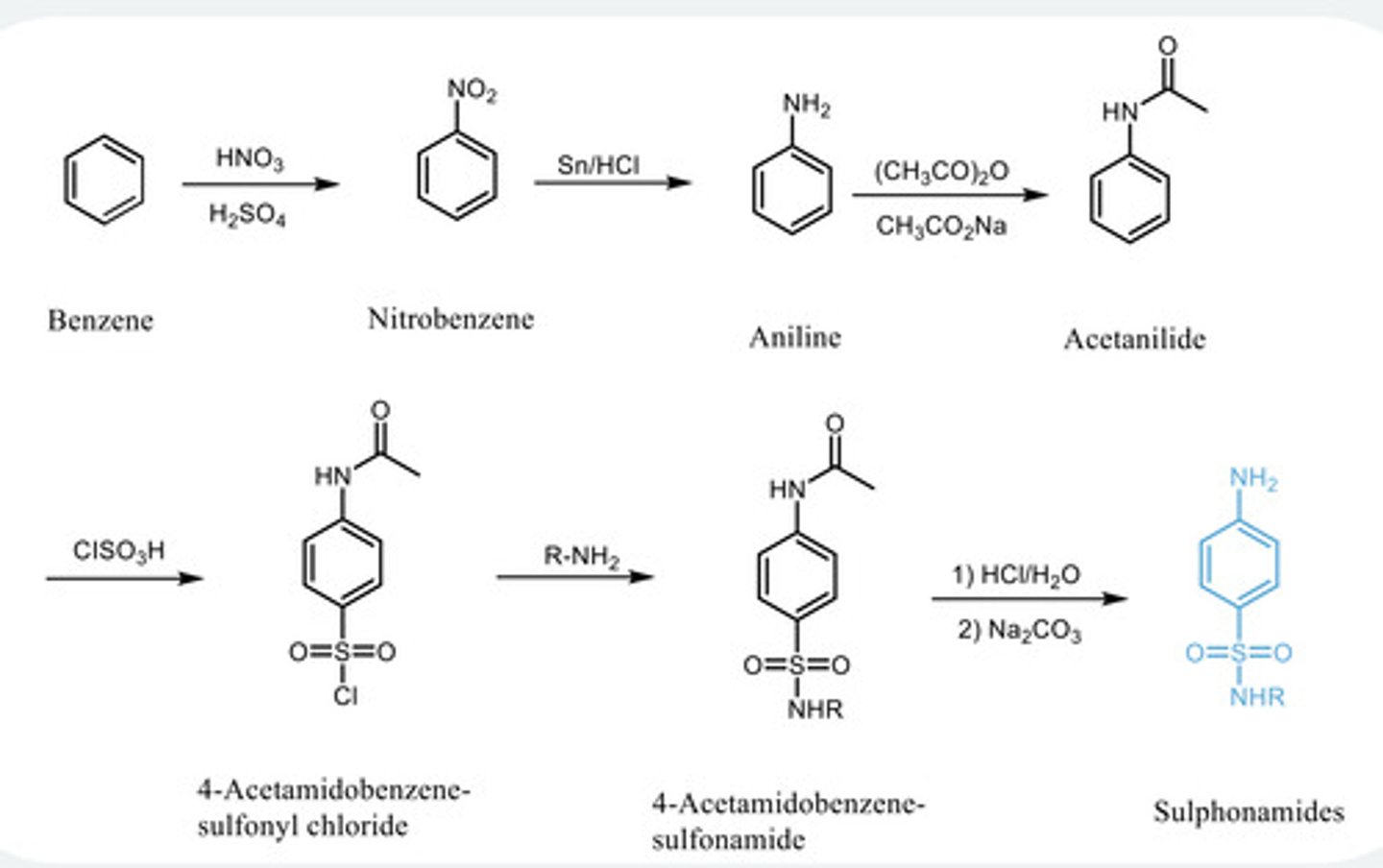
Sulfa drug synthesis:
Benzene is nitrated to form Nitrobenzene -> reduced to amide to form aniline -> Acetylation (with acetic acide) to form Acetanilide -> Chlorosulfonation (using chlorosulfonic acid to fomr 4-acetamidobenzenesulfonyl chloride -> Amine reacts with this product to form 4-acetamidobenzenesulfonamide -> This is then deprotected using acid and Sulphonamides are formed
sulphonamide structure
- All need a para amino group N2H attached to a benzene ring and the opposite
side has a sulphonamide with an essential primary/secondary N
-The R group can be varied
-N2H pKa much less than 4 (~2)
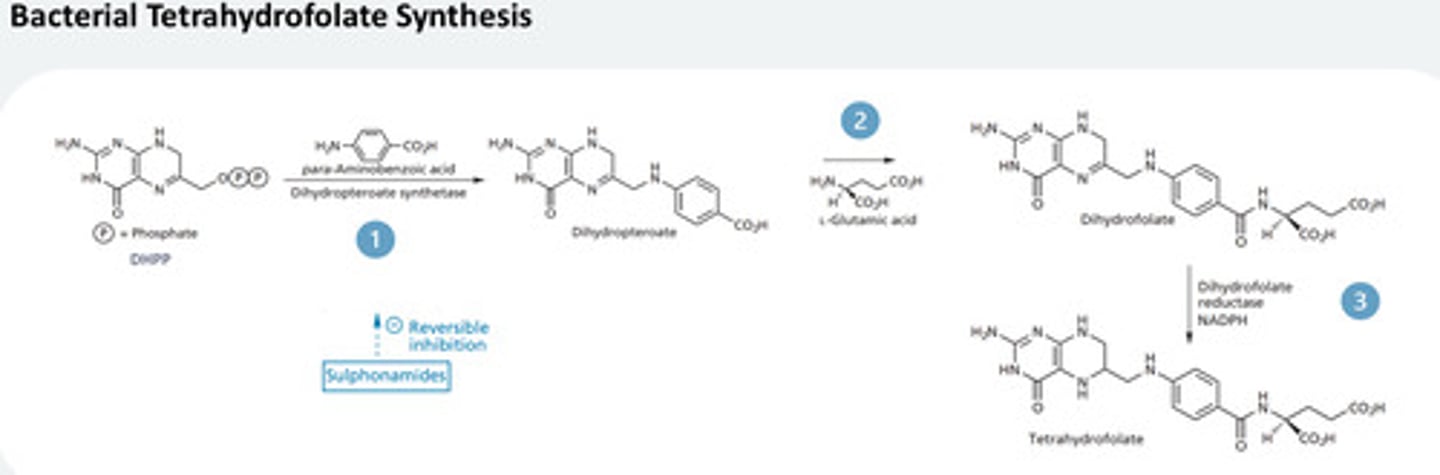
Describe bacterial tetrahydrofolate synthesis and how this mechanism is important for how we understand sulfa drugs to be selective
-Tetrahydrofolate (THF) is an essential cofactor in bacterial cells, required for DNA, RNA, and amino acid synthesis - bacteria synthesise this on their own
-It is key in One-carbon metabolism - a biochemical network that transfers single carbon units ( in this case -CH₂ on pyrimidines ) between molecules, playing a crucial role in DNA, RNA, and amino acid synthesis
-Sulfa drugs selectively target bacteria without harming human cells (as humans do not synthesise folate because we lack the enzyme dihydropteroate synthetase, we get if from diet - Folic acid is carried across animal cell membrane by transport proteins!!)
Key steps in bacterial tetrahydrofolate synthesis:
1- DHPP synthesis: DHPP reacts with PABA and enzyme is
Dihydropteroate synthetase = product incorporates PABA displaces the
pyrophosphate to give dihydropteroate
- 2- Dihydropteroate reacts with glutamic acid forming an amide with -COOH
giving Dihydrofolate which is reduced to become tetrahydrofolate
Note: - Inhibiting Dihydrofolate reductase = cannot make tetrahydrofolate = inhibits
protein synthesis
Sulphonamide MOA
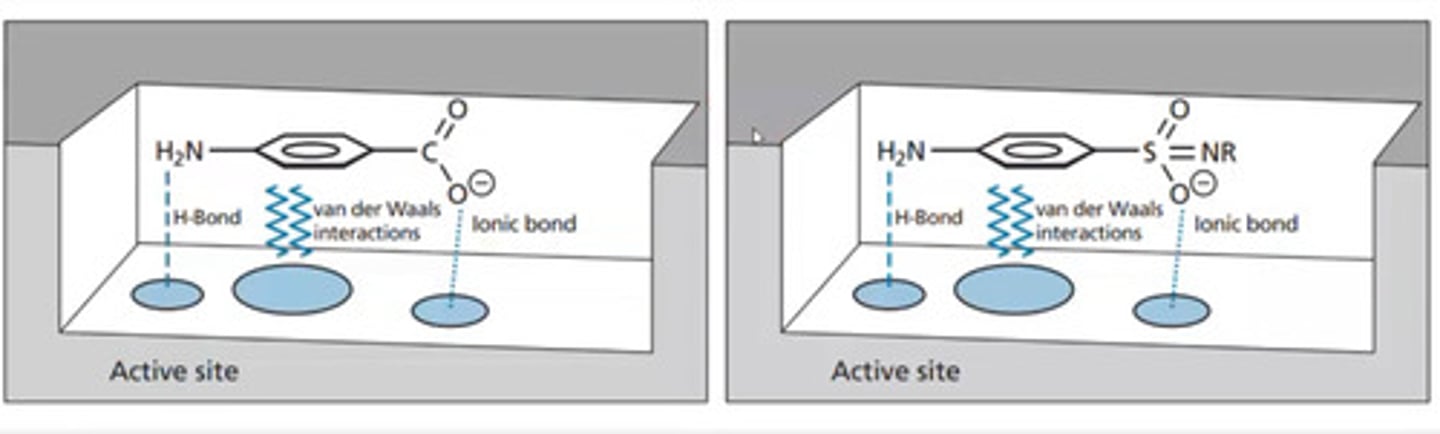
Act as competitive enzyme inhibitors (preventing PABA from binding) of dihydropteroate synthetase and block the biosynthesis of tetrahydrofolate in bacterial cells
Mimics PABA binding with active site using H, Van der waals and ionic bonding
Note: Doesn't kill bacteria just stops its growth ! - Bacteriostatic
-Body defence hunter-killer CD8 cells eliminate the invaders
Common clinical uses for sulphonamides
treating infections of the eyes, mucosal membranes, GIT and urinary tract
How can bacteria develop resistance to sulphonamide drugs (hint 3 ways)
- Bacteria synthesise more PABA
- Higher concentrations of PABA can compete with sulphonamides for the
target enzyme
- Some bacteria have become resistant- mutations of the enzyme whereby it
has low affinity for the sulphonamides
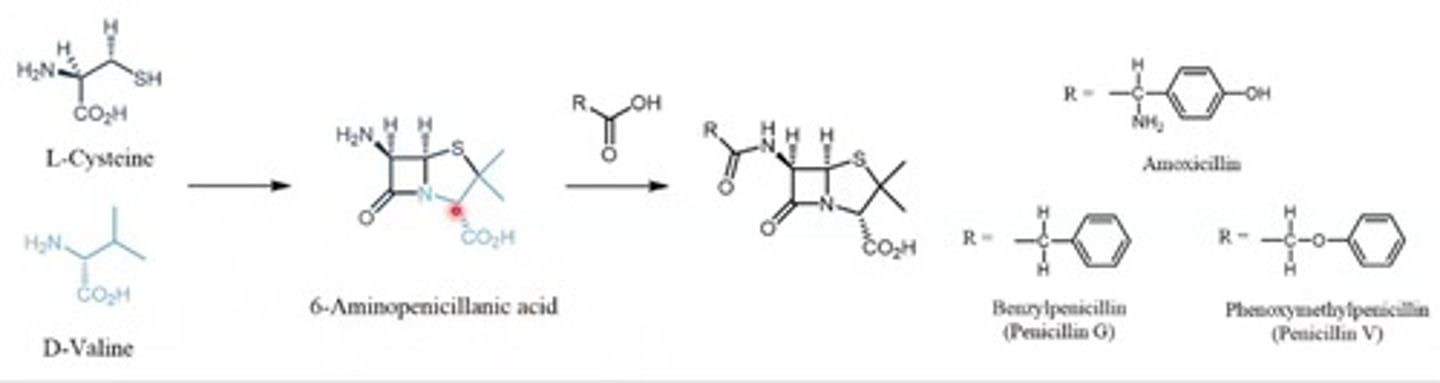
How is Penicillin biosynthesised?
-By a fermentation process
- L-Cysteine and D-Valine react to product 6-Aminopenicillinic acid, 6-APA
- Different carboxylic acids (which form different penicillins) are added in the fermentation into the penicillin that
is produced
What is the structure of Penicillin?
How does its structure affect the stability of the beta lactam ring?
Has a 4-membered Beta-lactam ring which is fused to a five membered
thiazolidine ring = non-planar butterfly-like structure = adds a lot of strain to
the ring so:
-The beta-lactam ring is unstable and is primarily responsible for antibiotic
potency
- Angle in penicillin = two H are pointing up = cis-configuration contributes to
instability of Beta-lactam ring = contributes to good potency as it reacts quickly

What is the major contributor to amide resonance?
The C=N-O⁻ resonance structure, which gives amides partial double-bond character (The C=N-O orbitals overlap = sharing of electrons)
What are the three key conclusions from amide resonance?
1️⃣ Nitrogen is not basic (lone pair is delocalised).
2️⃣ Carbonyl is less reactive (less electrophilic).
3️⃣ N-C-O bond is planar due to resonance.
Why is the carbonyl carbon in amides less electrophilic than in aldehydes or ketones?
The lone pair on nitrogen overlaps with the carbonyl, reducing its partial positive charge and making it less susceptible to nucleophilic attack.
How does the presence of a beta-lactam ring change amide reactivity?
The bicyclic structure prevents resonance stabilisation, making the carbonyl more electrophilic and reactive.
What is a bicyclic beta-lactam ring?
A 4-membered lactam ring fused to a 5- or 6-membered ring, forming a non-planar structure.
Why can’t resonance stabilisation occur in a beta-lactam ring?
The bond angles in the 4-membered ring are 90° instead of 120°, creating strain that prevents nitrogen’s lone pair from overlapping with the carbonyl = nitrogen uses the lone pair of electrons and makes carbonate group more electrophillic
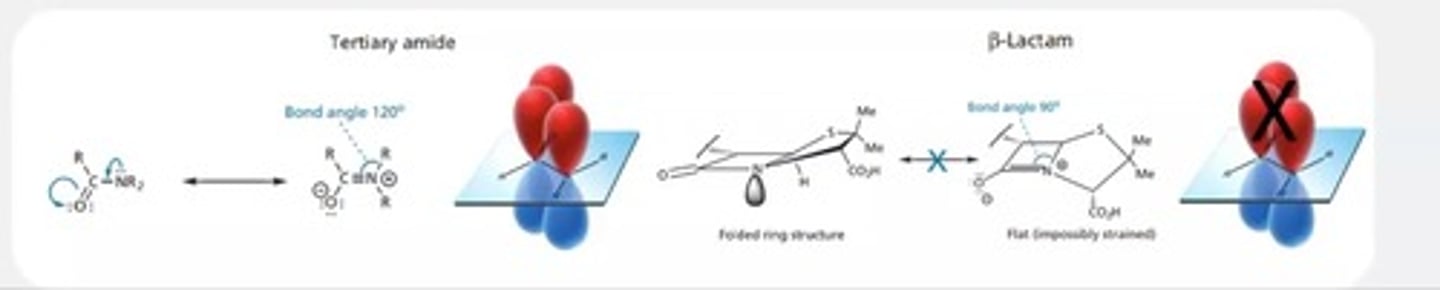
How does bond angle strain in beta-lactams affect reactivity?
The 30° strain increases instability, making the carbonyl highly electrophilic and susceptible to nucleophilic attack.
Why is the beta-lactam ring easily hydrolysed?
✔️ Nitrogen's lone pair is readily protonated, making the ring unstable.
'✔️ Ring strain increases reactivity, leading to rapid ring opening in acidic or basic conditions
How does the increased reactivity of beta-lactams relate to their antibiotic function?
The electrophilic beta-lactam carbonyl reacts with penicillin-binding proteins (PBPs), disrupting bacterial cell wall synthesis and leading to bacterial death
How stable is the beta lactam and what can open up its ring
-Very susceptible to attack under acidic and basic conditions due to the lone pair on the nitrogen being readily protonated
- It can be hydrolysed with water to open up, can react with bases and
nucleophiles
Note:
Neighbouring acyl group can actively participate in a ring opening of the lactam ring
Summary: Contributing factors to unstable penicillin structure
-Amide group
-Beta-lactam
-Acyl Side chain
Directly relates to activity
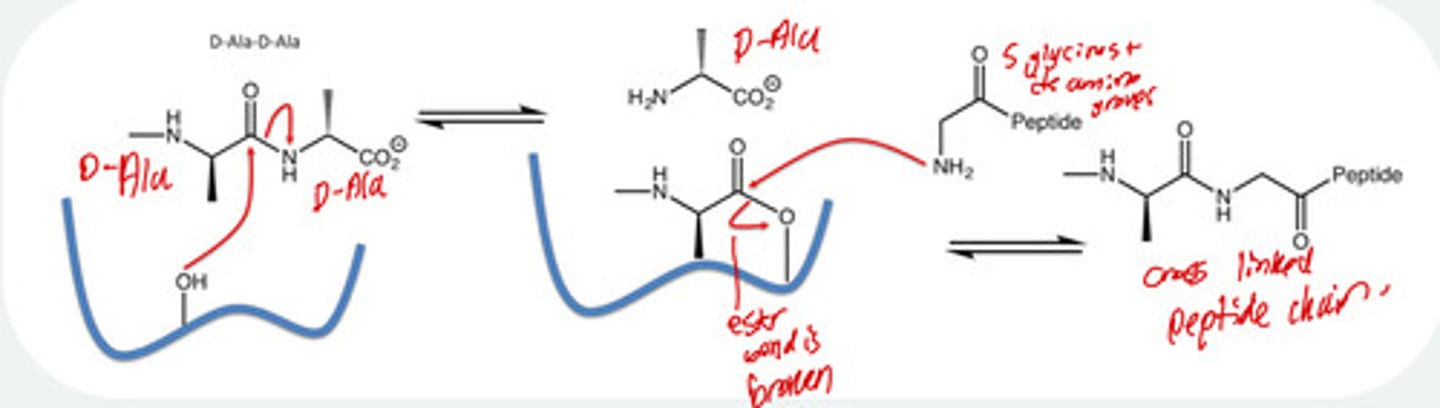
Describe the Bacterial cell wall structure
- Consists of a sugar and peptide backbone- sugars are composed of NAM and
NAG
- In bacterial synthesis we have polypeptide chains cross-linked, when this
happens one of the glycine’s attacks a D-Ala-D-Ala (D-Ala is only seen in bacterial cells) to cross-link the cell membrane and is done by a transpeptidase, (Penicillin binding protein)
How does the transpeptidase reaction occur normally and what is its importance ?
-Penicillin’s resemble D-Ala D-Ala structure to forms a stable complex with the
transpeptidase so reaction cannot proceed and bacterial cell wall cannot be
formed
Summary:
- OH group attacks the amide = hydrolyses amide = reactive transpeptidase
complex
- Another peptide chain with amino group on glycine = attacks the
transpeptidase so now peptides are cross-linked
- Activated Ser-OH group attacks a C of the amide between 2 terminal D-Ala
residues resulting in Nu displacement of 1 D-Ala and the formation of a
peptide chain transpeptidase complex
- Complex is linked as an ester readily attacked by terminal amino group of
5-glycine chain releasing the enzyme and competing cross-link of two
peptide chains
What is penicillins MOA?
- The hydroxyl group from from Transpeptidase attacks the carbon between D-Ala and D-Ala to give a reactive centre and then from another chain the 5-glycines and its amino groups attacks the Serine of
transpeptidase to cross-link breaking the esther bond releasing your enzyme again
- Penicillin’s resemble D-Ala D-Ala structure (which is usually present in the active sites) to forms a stable complex (a covalent penicillin attached to the
transpeptidase) with the
transpeptidase so reaction cannot proceed and bacterial cell wall cannot be
formed (so rather than attacking D-Ala D-Ala the transpeptidase
attacks the penicillin) =This results in incomplete cell walls that are much more fragile and porous and eventually resulting in swelling followed by cell lysis and death.
Note:
When 5-glycine binds here = penicillin irreversibly bound to transpeptidase =
enzyme no longer functional – no cell wall made
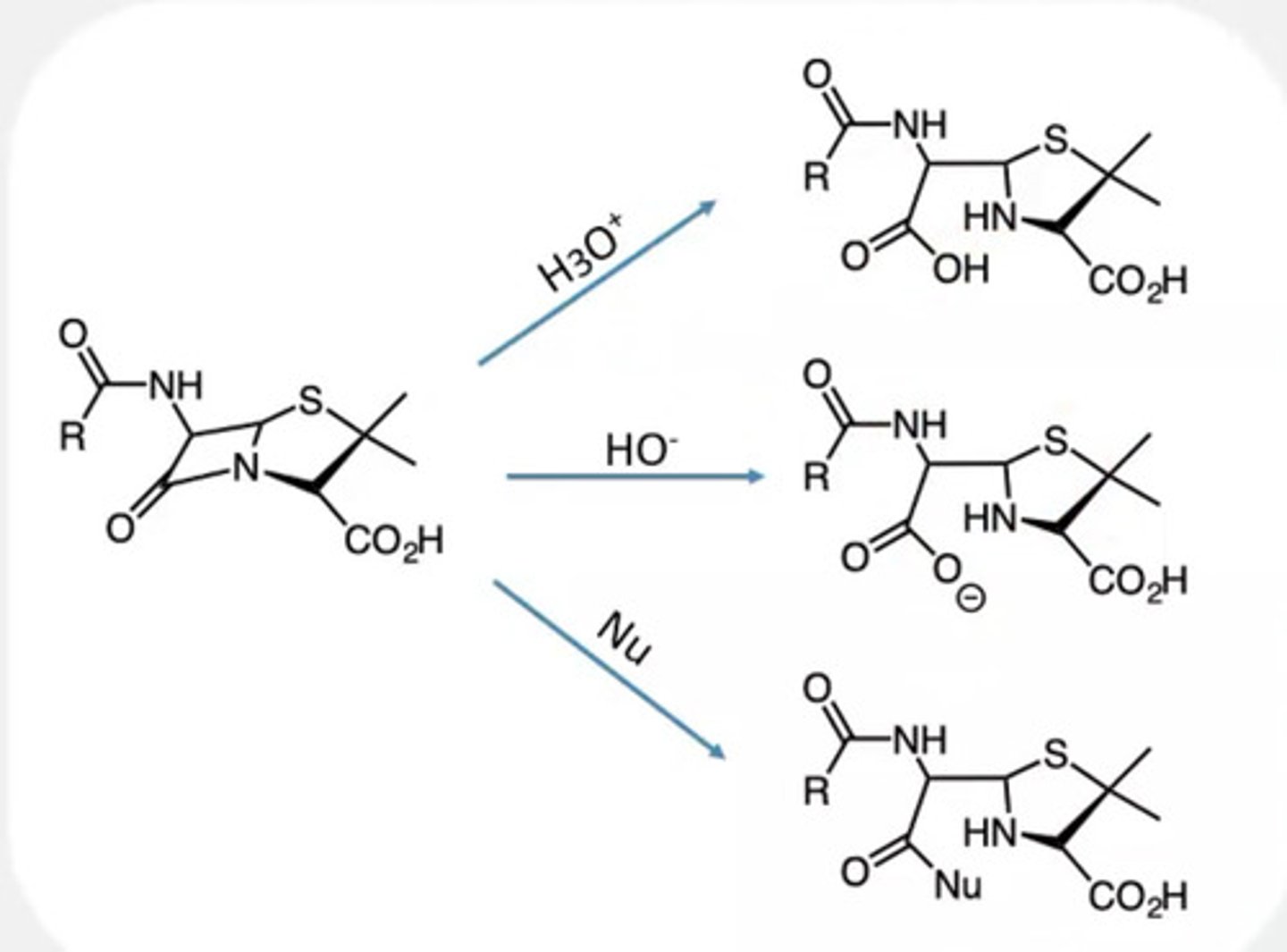
How does bacterial resistance against penicillin's occur?
-Beta-Lactamase catalyses the hydrolysis Beta lactam ring = inactivation of penicillin.
e.g. penicillin G which is sensitive to all Beta -lactamase's
Note:
Only beta lactam of penicillin is reactive as there is no resonance because groups are not on the same plane so NH-O group is stable
How do we tackle this bacterial resistance ?
Create a beta lactamase inhibitor
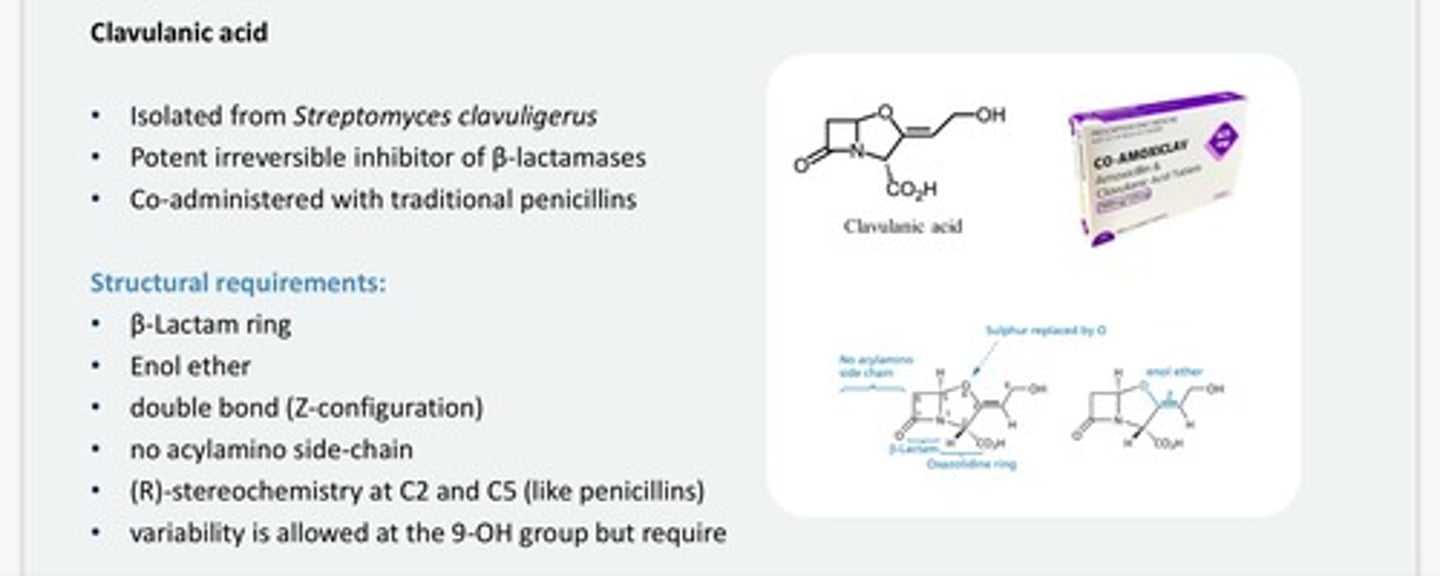
What is Clavulanic acid?
- Clavulanic acid is a potent irreversible inhibitor of Beta-lactamase
- MOA: carbonyl addition followed by elimination is essential
-Administered with penicillins
-Clavulanic acid reacts with beta-lactamase twice which completely traps and
inhibits it
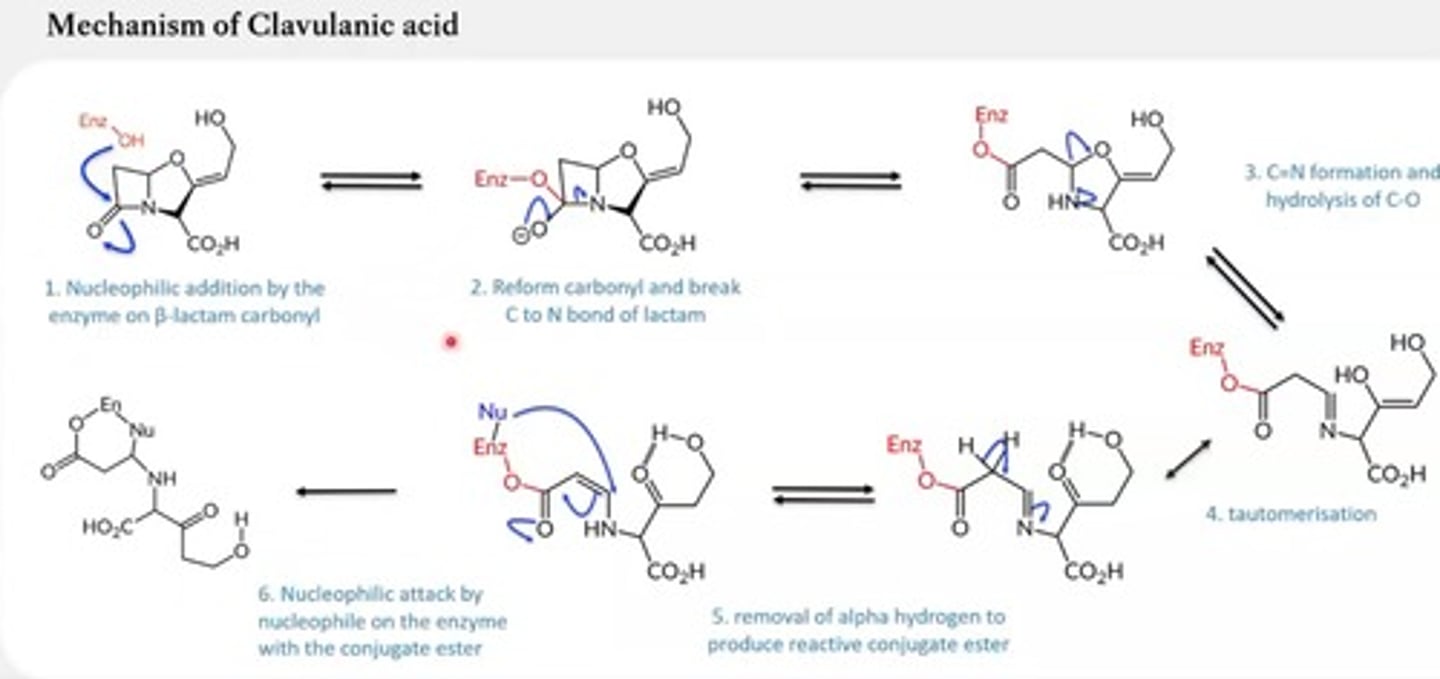
Clavulanic acid MOA?
Clavulanic acid reacts with beta-lactamase twice which completely traps and
inhibits it
What is the benefit of developing an acid resistant penicillin and how is this done?
- Acid resistant due to NH2 group - electron withdrawing group decreases the tendency for the carbonyl carbon to act as a nucleophile (more orally active)
What is the benefit of developing an Beta lactamase resistant penicillin and how is this done?
Principle of steric shields - block penicillin from accessing the β-lactamase active site by placing a bulky group on the side chain = ensures its activity
How does membrane permeability affect penicillin in regards to gram negative bacteria (what it is usually used for), why would we make a pro drug for it?
-G-negative bacteria have a coating on cell wall of fats and sugars which acts
as a barrier
-Penicillin has Free carboxylic acid which if ionised would be repelled by the cell membrane fatty position of the membrane may act as a barrier to polar hydrophilic penicillin molecules
-Also would have poor bioavailability through the gut wall as both the amino group and the carboxylic group are ionized
Solution: pro-drug (that contains an ester instead of the normal carboxylic acid) which is hydrolysed by esterase as it enters cell wall and then is
activated to work on gram-negative bacteria
-Once ester hydrolysed = releases formaldehyde
giving penicillin = quite effective
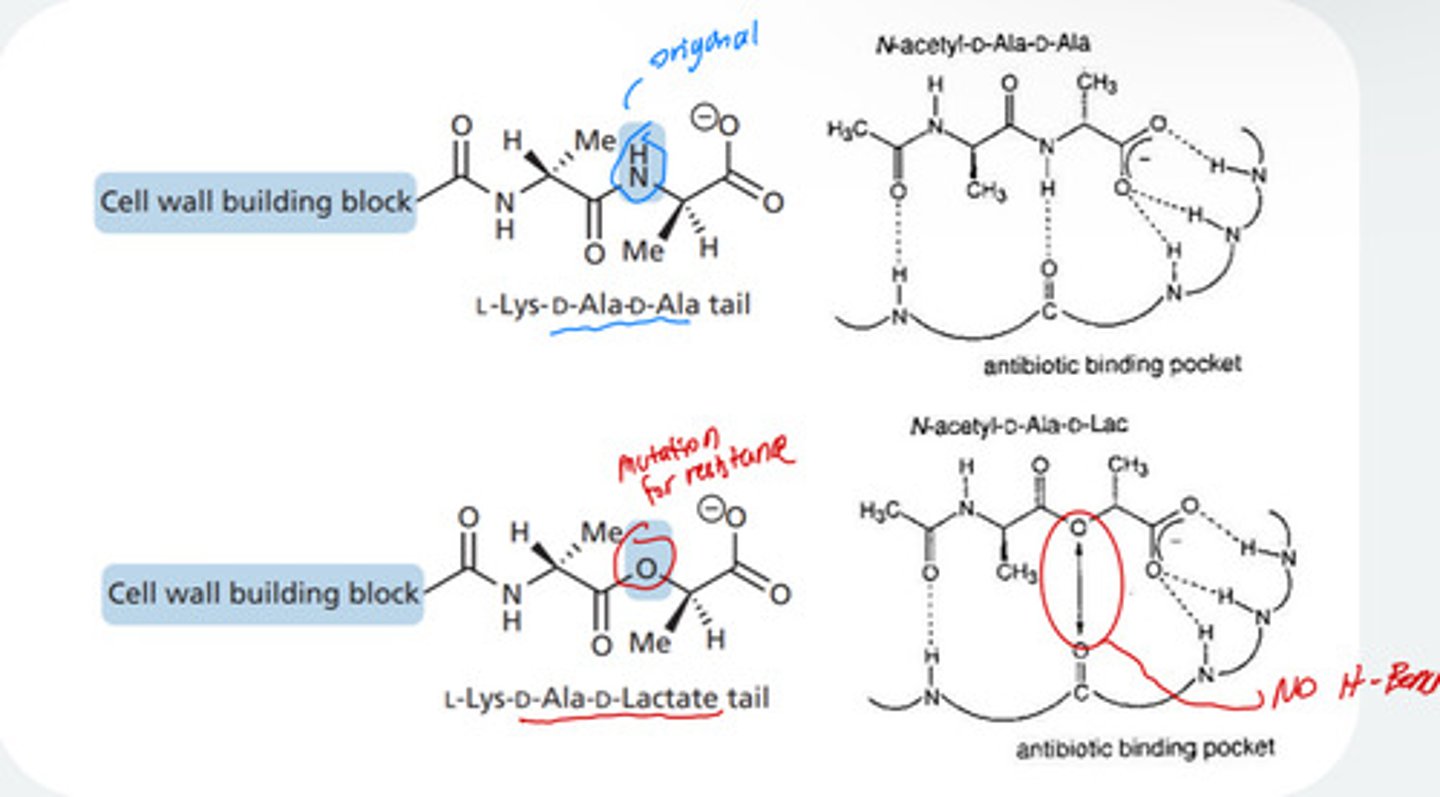
How does glycopeptide vancomycin work?
-Vancomycin molecules are complex to form a number of H-bonds (5) with D-Ala D-Ala, with O-, carbonyl, NH and with O of second which forms a stable complex
-Bind to D-Ala-D-Ala terminal and block peptide incorporation into cell wall
How is bacteria resistant to vancomycin?
-Mutation of NH to O = Vancomycin resistant bacteria have mutated the DAla-D-Ala termination of pentapeptide to D-Ala-DLac (lac means lactate)
-bacteria evolved by having two oxygens which want
to stay away as they cannot H-bond at that position = much lower affinity of vancomycin for this peptide in the resistant bacteria in the antibiotic binding pocket