Lecture 16 – Glycolysis: Pathway Analysis
1/39
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
Energy Investment vs. Energy Return
glycolysis requires a small input of energy to initially break down glucose
two phases ◦ Energy investment vs. Energy return
Energy investment requires 2 ATP → 2ADP
Energy return gives back 4 ATP, and 2 reduced NADH molecules to be used later Therefore, though glycolysis requires energy, it gives more than is initially required, and many organisms (and many times, cancer) almost entirely use glycolysis because of its quick return on investment
Energy investment phase
Glucose → 2ATP into 2 ADP → 2 triose phosphates
Energy Generation Phase
2 triose phosphates → 4 ADP into 4 ATP→ 2 NAD+ into 2 NADH → 2 pyruvate
Net gain of glycolysis
Glucose → 2 pyruvate
2 ADP→ 2ATP
2 NAD+ → 2 NADH
Anaerobic vs. Aerobic Energy Production
Because this pathway is used by many different organisms (bacteria, archaea, the eukaryotes), it is likely very old
◦ The oxygenation of our atmosphere did not occur until roughly 2 billion years ago, which necessitated this pathway be used without the use of oxygen which allows an increased amount of energy we can derive from glucose
◦ We now use the end products of glycolysis in the other steps down the line, which requires O2
So, how was this pathway (Anaerobic vs. Aerobic Energy Production) useful without oxygen as a final electron acceptor?
◦ Needed to include other organic compounds as electron acceptors to be reduced, so that we can reuse the cycled electron carrier NAD+, and get it back from its reduced form, NADH
One method is still used in (most) eukaryotes, the other is used primarily in single celled organisms…
Homolactic fermentation (Glycolsyis then 2 pyruvate → 2 Lactate
Alcoholic fermentation (Glycolsis then 2 Acetylaldehyde + 2CO2 → 2 Ethanol
The three main processes that need to occur for glycolysis:
1. Priming I. The addition or isomerization of a phosphoryl group to glucose (or fructose-6-phosphate, or other carbon molecule) This step also has the added benefit of trapping glucose in the cell II. These phosphate groups are not lost easily from these two molecules, so therefore we need to…
2. …convert these molecules with low phosphate group transfer potentials to high P group transfer potentials I. Essentially, making the phosphate groups easier to cleave off and reattach to…
3. ADP phosphorylation back to ATP from these high phosphate group transfer potentials
The Reactions of Glycolysis
Overall, there are 9-10 reactions that occur between glucose and its eventual degradation to pyruvate
The first are reactions 1 → 5 of glycolysis
The energy investment phase
◦ This transforms glucose into two molecules of glyceraldehyde-3-phosphate
◦ A six carbon molecule into two phosphorylated three carbon molecules
reactions 6 → 10 of glycolysis
The energy generation phase
◦ This regains, and doubles, the energy invested in the initial phase
◦ It also gives two reduced NADH molecules, and 2 molecules of pyruvate (no longer phosphorylated)
Enzymes used for energy investment, ignoring isomeration:
Hexokinase (Phosphorylation, Glucose → Glucose-6-phosphate G6P)
Glucose-6-phosphate isomerase ( Isomerization, Glucose-6-phosphate G6P → Fructose-6-phosphate (F6P) )
Phosphofructokinase (Phosphorylation, Fructose-6-phosphate → Fructose 1,6-bisphosphate FBP)
Aldoase (Cleavage, Fructose 1,6-bisphosphate → Glyceraldehyde-3-phosphate and Dihydroxyacetone DHAP) (not equal molecules)
Reactions 1-3
Activation by phosphorylation
2 ATPS invested
Reactions 4 and 5
Cleavage of 1 six carbon sugar phosphate to 2 three-sugar phosphates
The first reaction
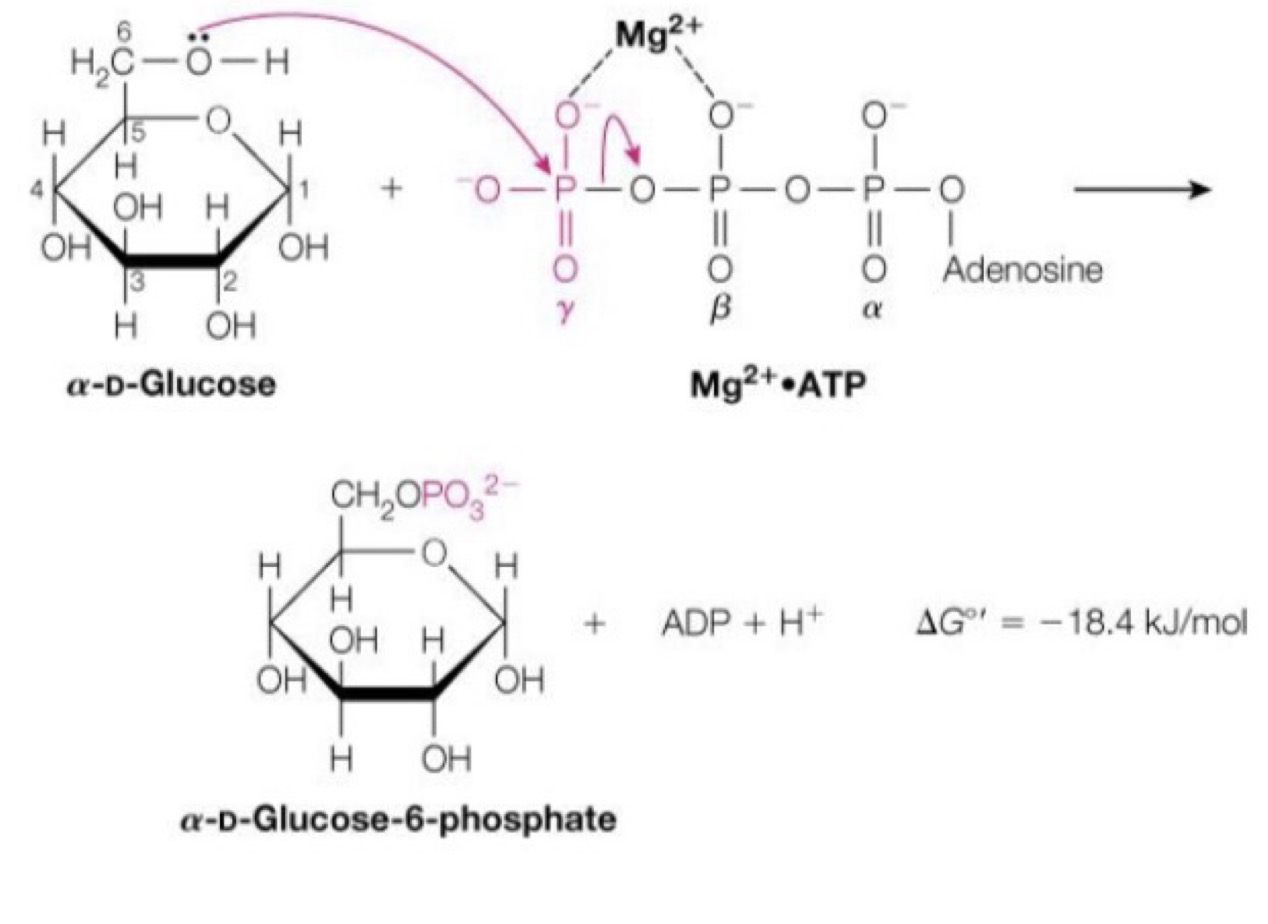
Glucose to glucose 6 phosphate:
Accomplished by hexokinase, a transferase enzyme, requiring a nucleophilic substitution
◦ Takes the gamma (third) phosphate group from ATP
Why is magnesium involved? ◦ It needs to attach to the two oxygen atoms to allow a site of nucleophilic (electron dense molecule) attack. ◦ This is our first reaction, of two, that requires ATP
Hexokinase enzymes
There exist multiple different types of hexokinases, and their concentrations depend on where they’re located and the desired outcomes in that location
For example: Hexokinase I, II, III, vs. Hexokinase IV
Each of the hexokinases 1-3 readily convert glucose to G6P, and have very low Km values
Remember Km? It’s the [substrate] at which V max reaches 50% in an enzymatic reaction In some tissues, it is useful to have an enzyme that readily converts glucose, but in the liver, small intestine, etc. we may want to keep a more consist amount of glucose flowing
Reaction 2
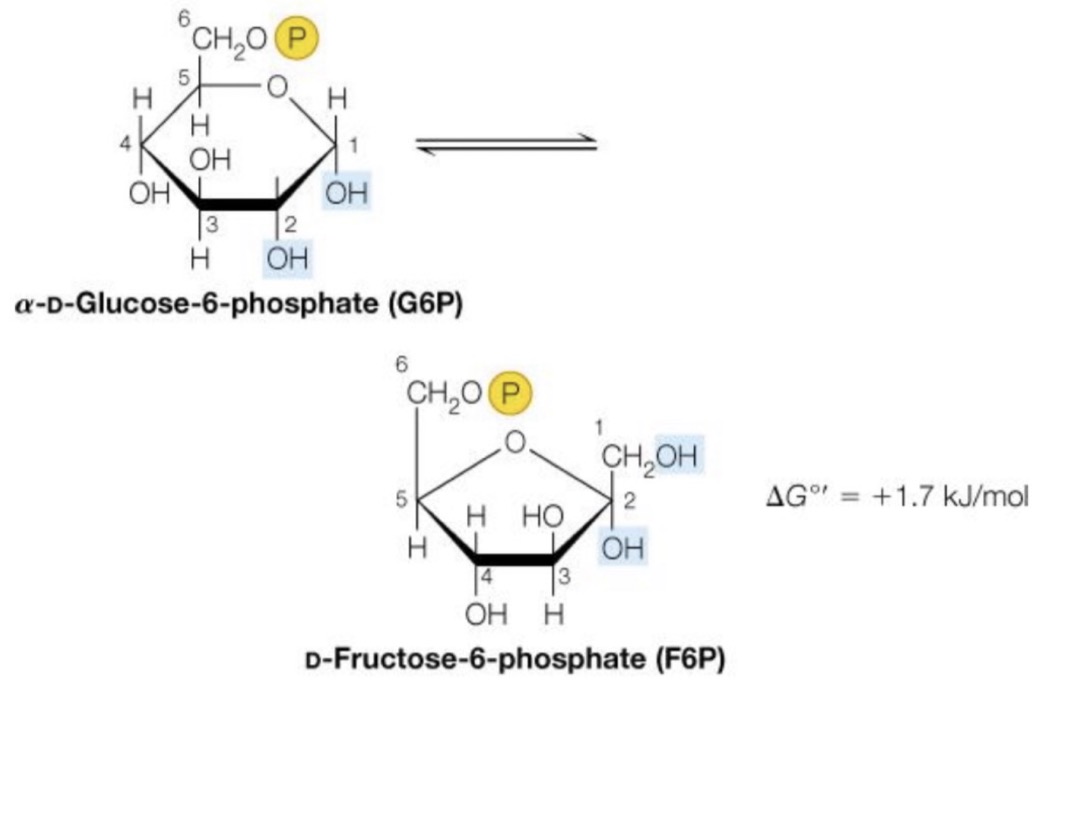
Glucose to fructose
We have our glucose-6-phosphate, which needs to be isomerized to fructose-6-phosphate
◦ Still a hexose, but has two carbon atoms out of the ring instead of one
◦ Turns a 6-ring into a 5 ring What happens molecularly?
◦ A transfer of the oxygen from the number 1 carbon to the number 2 carbon Transforms the molecule from an aldose to a ketose
◦ i.e. if it wasn’t a ring structure, there would be a double bond on the 2nd carbon, rather than the first
Number three – More phosphate
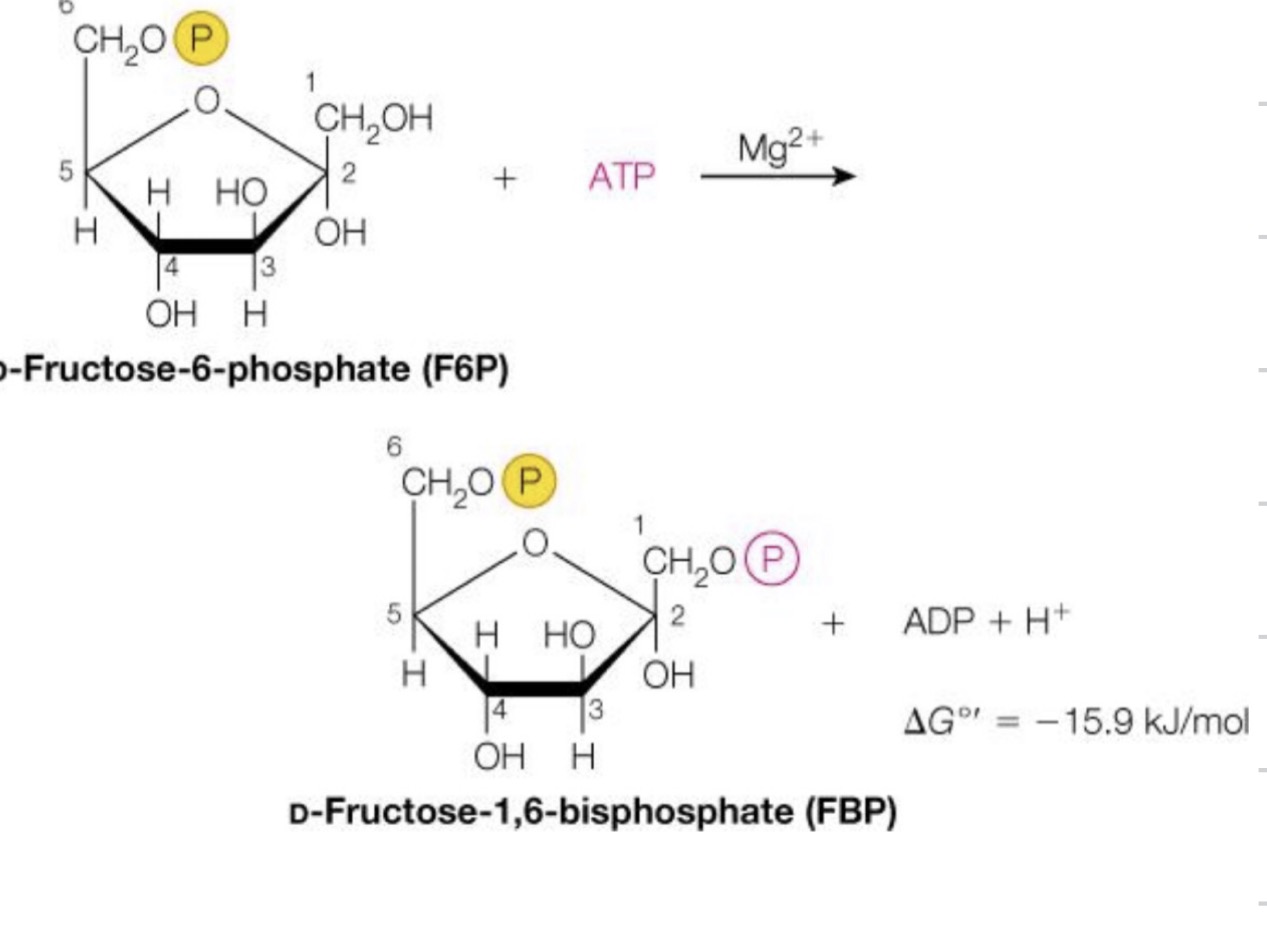
Fructose-6-phosphate to fructose-1,6-bisphosphate
Another addition of a phosphate group, so we know that this enzyme is a transferase
◦ And once again, this requires a magnesium ion to allow for the nucleophilic attack of the phosphorus atom on the gamma phosphate
This is our second, of two, enzyme reactions that require ATP.
From here on out, energy is either not expended (the enzymes are catalyzed/moved forward without the use of phosphate) or is generated from the metabolic process.
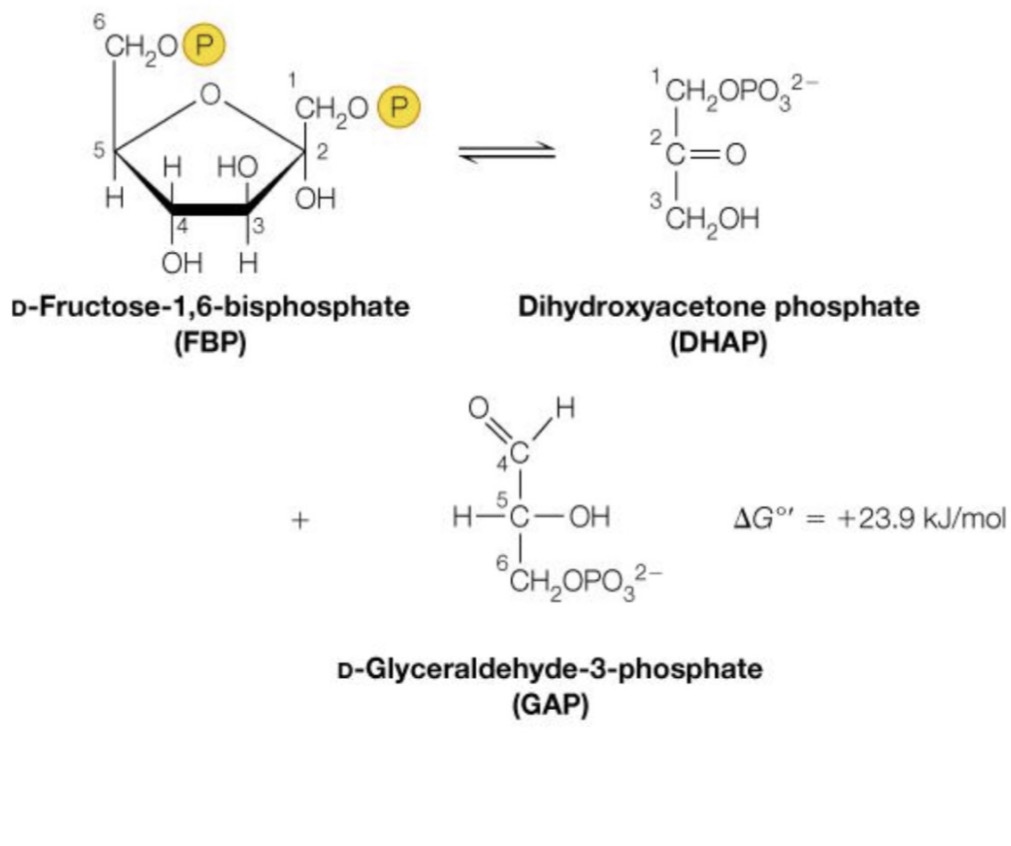
Reaction Number 4:
The cleavage of our fructose-1,6-bisphosphate into two, threecarbon molecules
◦ Glyceraldehyde-3-phosphate and Dihydroxyacetone phosphate
This is accomplished by the enzyme aldolase, a lyase
◦ Why lyase? It cleaves a molecule without the use of water… …this isn’t entirely true, water is used at one point, but it is also generated, so the net water intake / use is zero
◦ just know the aldolase converts the 6 carbon double phosphorylated ring into these two molecules
What reaction is this?
Number three
What reaction number is this?
Number one
What reaction number is this?
Number four
What reaction number is this?
Number 2
Enzymes used for energy generation:
Glyceraldehyde-3-phosphate dehydrogenase
Phosphogylcerate kinase
Phosphoglycerate mutase
Enolase
Pyruvate kinase
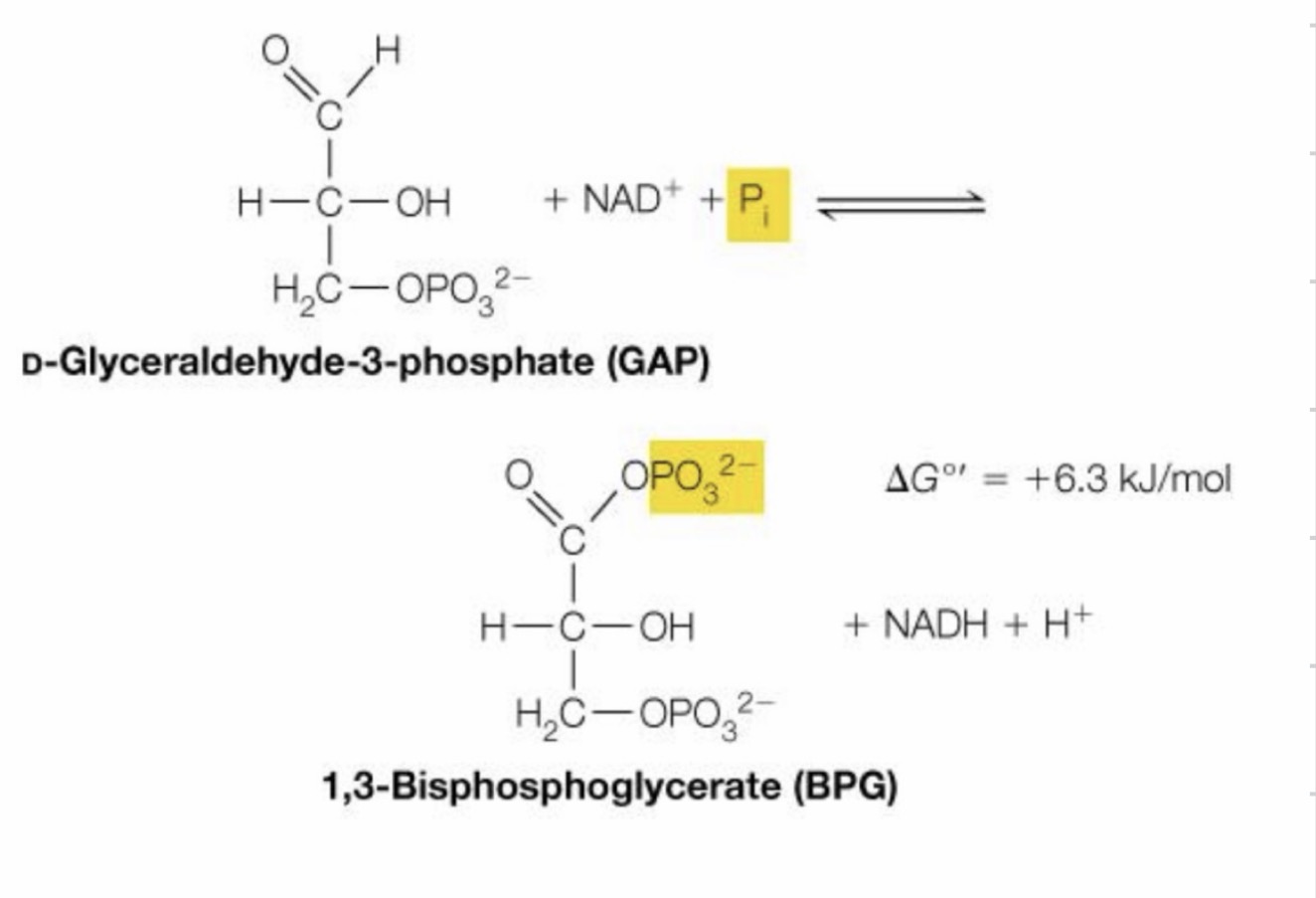
First reaction of energy investment phase
Generation of NADH and more phosphorylation
Group one enzyme (oxidoreductase) ◦ You can assume that any enzymatic activity involving NAD+ and NADH will have oxidation and reduction involved, so look for these molecules
two steps: 1. The aldehyde is converted from its C-O-H bond into a C-O-O (-), through the use of water and NAD+ as an electron acceptor, creating an acid
2. Then, this acid now has an excess of electrons, allowing for the attachment of the phosphate group at the phosphorus site
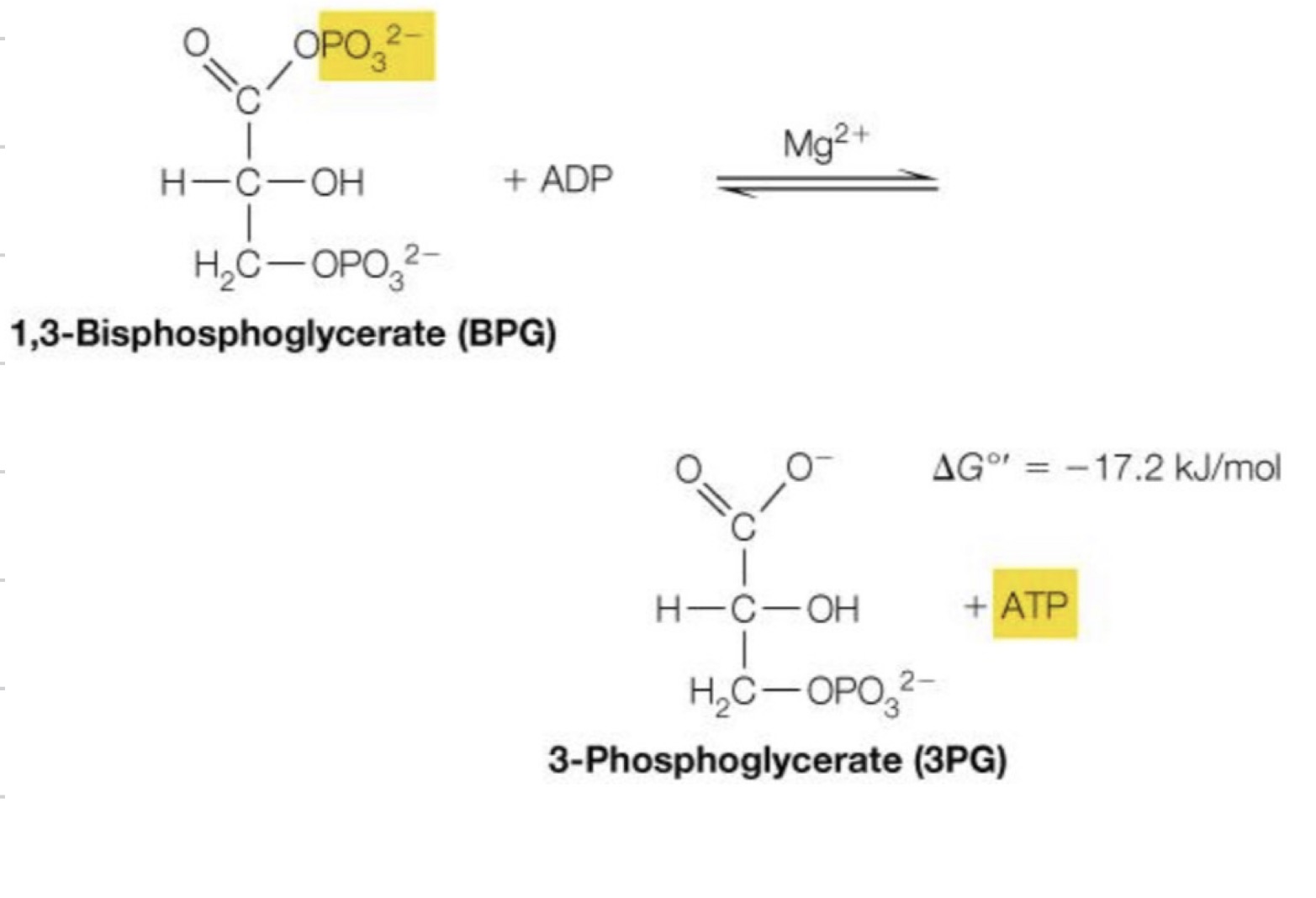
Second reaction of energy investment phase
Much like the addition of ATP molecules, this step also uses a transferase in the likes of phosphosglycerate kinase ◦ Except this time, it’s taking phosphate away
1,3 bisphosphoglycerate → 3-phosphoglycerate
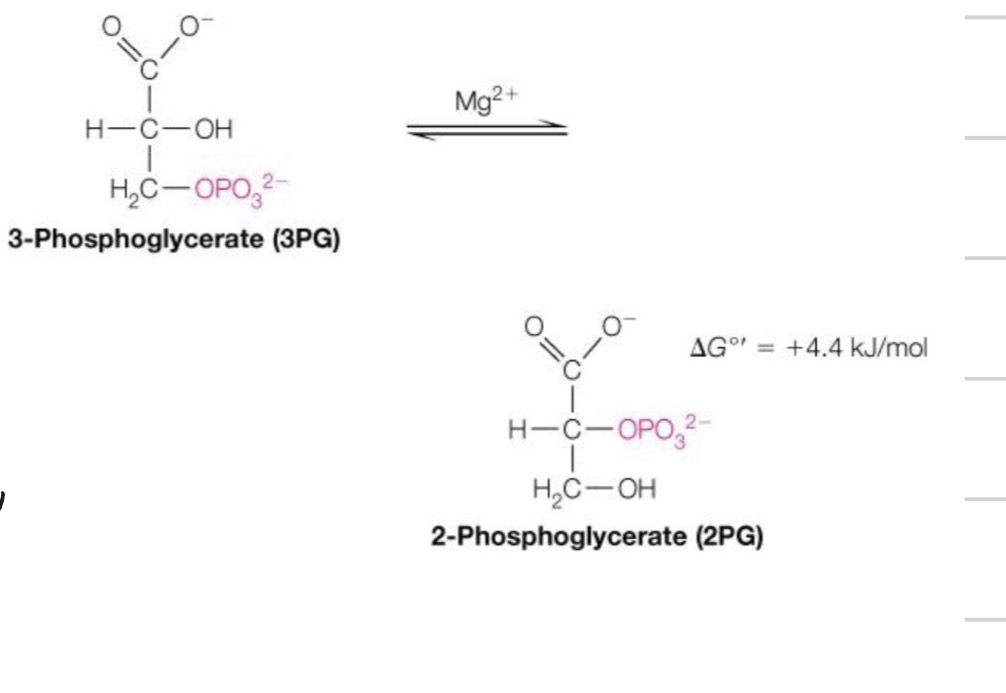
Third reaction of energy investment phase:
Priming the 2nd P group for its eventual departure:
This necessitates the movement of the phosphate group from the third carbon to the second carbon, via phosphoglycerate mutase
◦ This is an isomerase, since it is moving a structure from one site to another on the same molecule
Again, coming back to our main steps for glucose metabolism through glycolysis, this is the transference of the phosphate group to help prime it to transfer to make it a high energy molecule capable of delivering a phosphate group to ADP
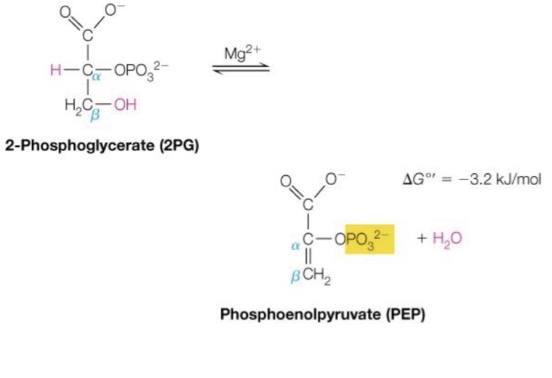
Fourth Reaction of energy investment phase:
Preparing for ATP transfer
To make the phosphate group more accessible, the removal of the hydroxyl group on the third carbon is required ◦ This is done through a dehydration reaction through enolase, a lyase enzyme
2-Phosphoglycerate → Phosphoenolpyruvate
Fifth Reaction of energy investment phase:
Pyruvate kinase (transferase), takes the phosphate group and attaches it to ADP, forming ATP So, magnesium is there, but why potassium? ◦ Helps to stabilize the enolpyruvate form, prior to transformation to pyruvate
2-phosphoglycerate → Phosphoenolypryvate
What reaction is this?
First reaction of energy investment phase, 6th reaction total
What reaction is this?
Second reaction of energy investment phase, 7th reaction total
What reaction is this?
Third reaction of energy investment phase, 8th reaction total
What reaction is this?
Fourth reaction of energy investment phase, 9th reaction total
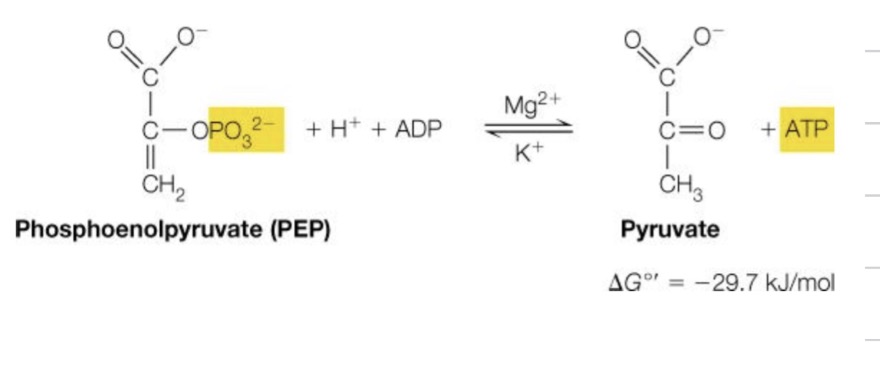
What reaction is this?
Fifth reaction of energy investment phase, 10th reaction total
So we have pyruvate: What next?
It depends on whether it goes through aerobic or anaerobic breakdown
Aerobic: Would continue to electron transport chain, generating ATP -lots of ATP
Anaerobic: Has one of two fates, depending on the organism involved
◦ Most animals, and some bacteria, go the route of lactate production
◦ This is only temporary in animals. This lactate is eventually reconverted to pyruvate once aerobic conditions prevail◦ Many others, including yeast, go a different route and produce ethanol through the conversion of pyruvate to acetaldehyde, then to ethanol
Auto-brewery syndrome or gut fermentation syndrome
The “pathogen”, in this case, is Saccharomyces cerevisiae, otherwise known as yeast.
◦ Therefore, this disease is not from the body directly converting sugars as such, but rather from the ingestion of yeast which lives on in the body to break down sugars into ethanol in the body directly In rare cases, this can result in blood concentrations of over 0.08 g / 100 mL of blood (i.e. over the legal limit in most of Canada)
◦ Once case study, describes a case of a 61 year old man that had a BAC of 0.21+ without having consumed any alcohol.
auto-brewery syndrome or gut fermentation syndrome treatment
The man was eventually treated through a combination of an anti-fungal agent and an oral dose of pro-biotics (lactobacillus bacteria), and followed a strict no-sugar, no carbohydrate diet
auto-brewery syndrome or gut fermentation syndrome treatment in court
Hhas been used as a successful defence for driving under the influence
Keep in mind, this is a very rare condition, and most patients recognize something is amiss.
Gluconeogenesis – In a nutshell
Very similar to glycolysis, but uses slightly different enzymes for some reactions
◦ Three reactions require so much free energy to reverse, if they were using their original enzyme, that different enzymes are required:
Most of the steps that are difficult to reverse are the ones that attached a phosphate group (steps 1 and 3), and their enzymes do not work as well in reverse
The last step, going from PEP to pyruvate, also requires two different enzymes that we’ll get into
Glycolysis and Gluconeogenesis The reactions of these two pathways, and their differences:
◦ Reaction 1: ◦ Glucose-6-phosphotase instead of hexokinase
◦ Reaction 3: ◦ Fructose-1-6-bisphosphatase instead of phosphofructokinase
◦ Reaction 10: ◦ Pyruvate carboxylase instead of pyruvate kinase
Overall energy investment required for gluconeogenesis:
Overall energy investment required for gluconeogenesis
◦ 4 ATP ◦ 2 GTP
◦ 2 NADH → 2 NAD+
To (re)use these compounds for gluconeogenesis, they need to be converted via different enzymes and bypass the enzymes in the glycolysis system.