Week 9: Serological and Molecular Detection of Bacterial and Viral infections
1/82
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
83 Terms
Host-microbe relationships
symbiotic: host and microbes live tgt for a long time, indigenous microbiota
commensalistic: no benefit or harm to either organism
Mutualistic: both host and microbes benefit
parasitic
Infectivity
Organism’s ability to establish an infection
pathogenicity
ability or an organism to cause disease
virulence
extent of pathology caused by an organism when it infects a host
Bacterial virulence factors
endotoxin
lipid A portion of LPS in gram-negative cell walls
powerful stimulator of cytokine release
Pili
adherence to host cells; R to phagocytosis
Flagella: adherence to host cells; motility
capsule: blocks phagocytosis, antibody attachment, complement
exotoxins
potent toxic proteins released from living bacteria
neurotoxins, cytotoxins, enterotoxins
Lab detection of bacterial infections
Culture of causative agent
microscopic: Gram stain or special stains
detection of bacterial antigens: ELISA, LFA, LA
Molecular detection of bacterial DNA/RNA
Serology: detection of anitbodies generated against bacteria
Serology advantages
to detect and confirm infections for which other lab methods not avail
to diagnose infections clin symptoms nonspecific
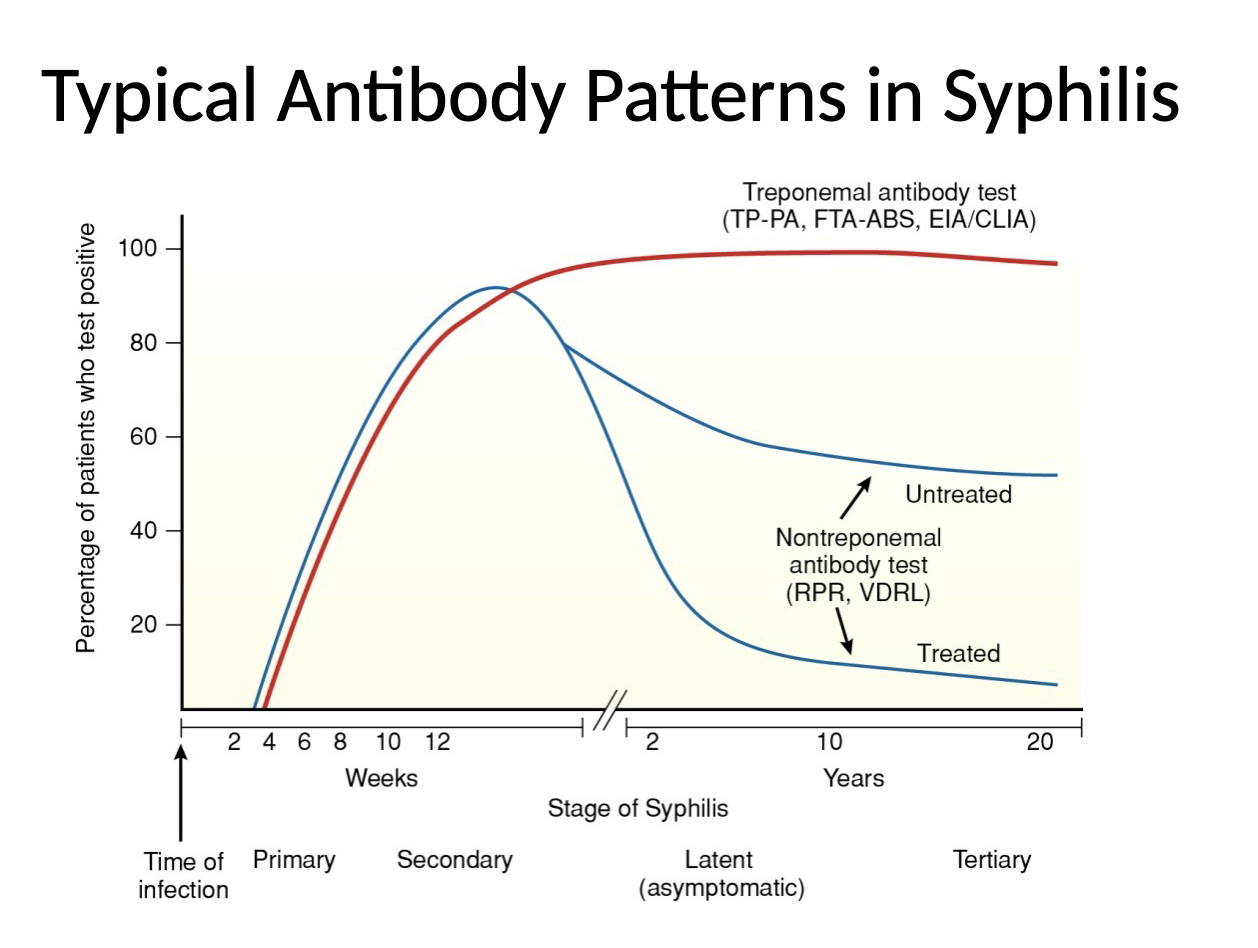
current infection indicated by presence of IgM, high IgG titer, or 4x rise in antibody titer between acute and convalescent samples
determine past exposure to an organism (IgM-, IgG+)
assess reactivation or re-exposure
Clinical manifestations of GAS

GAS sequelae
acute rheumatic fever
Poststreptococcal glomerulonephritis
lab diagnosis of acute GAS infections
culture on sheep blood agar
small translucent colonies surrounded by clear zone beta hemolysis
rapid assays to detect GAS antigens
lateral flow immunochromatographic assay (LFA)
Antistreptolysin O (ASO)
nephelometric methods currently used that measure light scatter produced by immune complexes containing streptolysin antigen
Titer elevated in 85%of pts with acute rheumatic fever
doesn’t increase in pts with skin infection
Anti-DNAse B
produced by both rheumatic fever and impetigo patients
tested by EIA and nephelometric methods
Streptozyme test
detects antibodies to 5 streptococcal products
ASO
anti-hyaluronidase (AHase)
anti-streptokinase (ASKase)
Anti-nicotinamide-adenine dinucleotide (anti-NAD)
Anti-DNAse B
Helicobacter pylori
GN microaerophilic spiral bacterium
transmission likely by fecal-oral route
major cause of gastric and duodenal ulcers
can survive in acid environment bc of production of urease, which provides buffering zone around bacteria
treated with antibiotics and anti-ulcer meds
if untreated, can lead to gastric carcinoma or mucosa-associated lymphoid tumors
Detection of H. pylori infection
detect urease in stomach biopsy (CLOtest)
urea breath test
antigens/antibodies
ELISA is method of choice
IgG in serum indicates active infection
titers decrease after successful treatment
Mycoplasma pneumoniae
tiny bacteria that lack cell wall
leading cause of resp infections
fever, headache, malaise, cough
walking pneunomia
Raynaud syndrome
causes Steven-John syndrome in minority of cases
spread by resp droplets
Lab diagnosis of M. pneumoniae infection
culture: produces mulberry colonies with ‘fried egg’ appearance on specialized media
gold standard
ABs to M. pneumoniae
most useful diagnostic assay
IgM antibodies = recent infection
IgG antibodies = possible reinfection
cold agglutinins
present in about 50% of pts but not specific for infection
molecular methods: film array resp panel
Rickettsial infections
obligate intracellular GN bacteria
spotted fever group (rocky mountain spotted fever)
typhus group (epidemic typhus)
organisms transmitted by arthropods through biting on an infection animal
Rocky mountain spotted fever (RMSF)
caused by R. rickettsii
transmitted by 3 species of ticks
headache, nausea, vomiting, diarrhea, skin rash; death
diagnosis by clinical presentation, serology by IFA
Syphilis
sexually transmitted diseases caused by spirochete Treponema pallidum
rapidly destroyed by heat, cold, and drying
direct contact with open lesion needed
transmission to fetus during pregnancy
bloodborne transmission rare
Clinical manifestations of syphilis
primary stage: development of chancre
secondary: generalized lymphadenopathy, malaise, fever, pharyngitis, rash
latent stage: asymptomatic
tertiary stage: Gummatous, cardiovascular, neurosyphilis
treated with penicillin when detected in early stages
Congential syphilis
transmission of treponemes to fetus occurs when pregnant woman has early-stage or latent syphilis
causes death in 10% of cases
live-born infants may be asymptomatic at birth but develop symptoms later
Lab diagnosis of syphilis
direct detection
demonstration of treponemes in active lesions
dark-field microscopy
fluorescent antibody staining
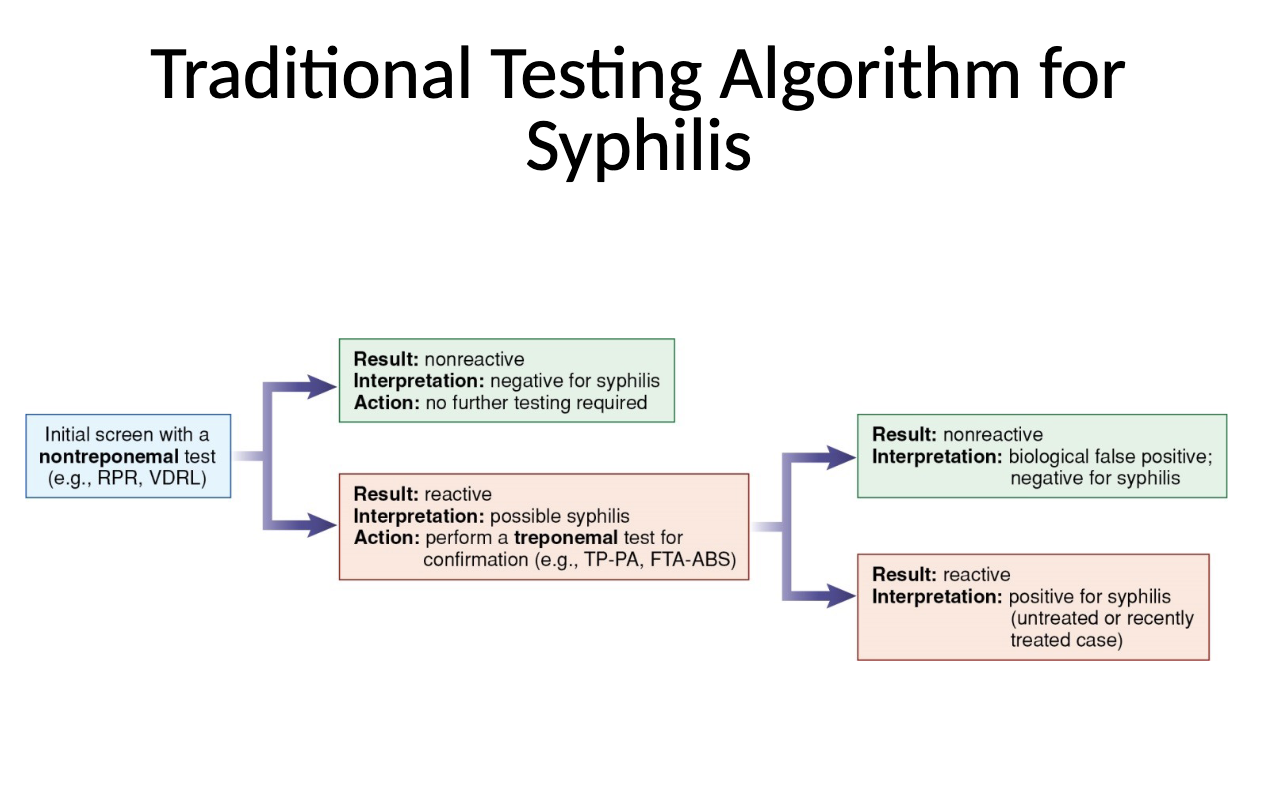
serological tests: nontreponemal/treponemal
Nontreponemal tests
detect antibody against cardiolipin (reagin), a lipid released from membranes of cells damages as a result of infection
VDRL test
RPR test
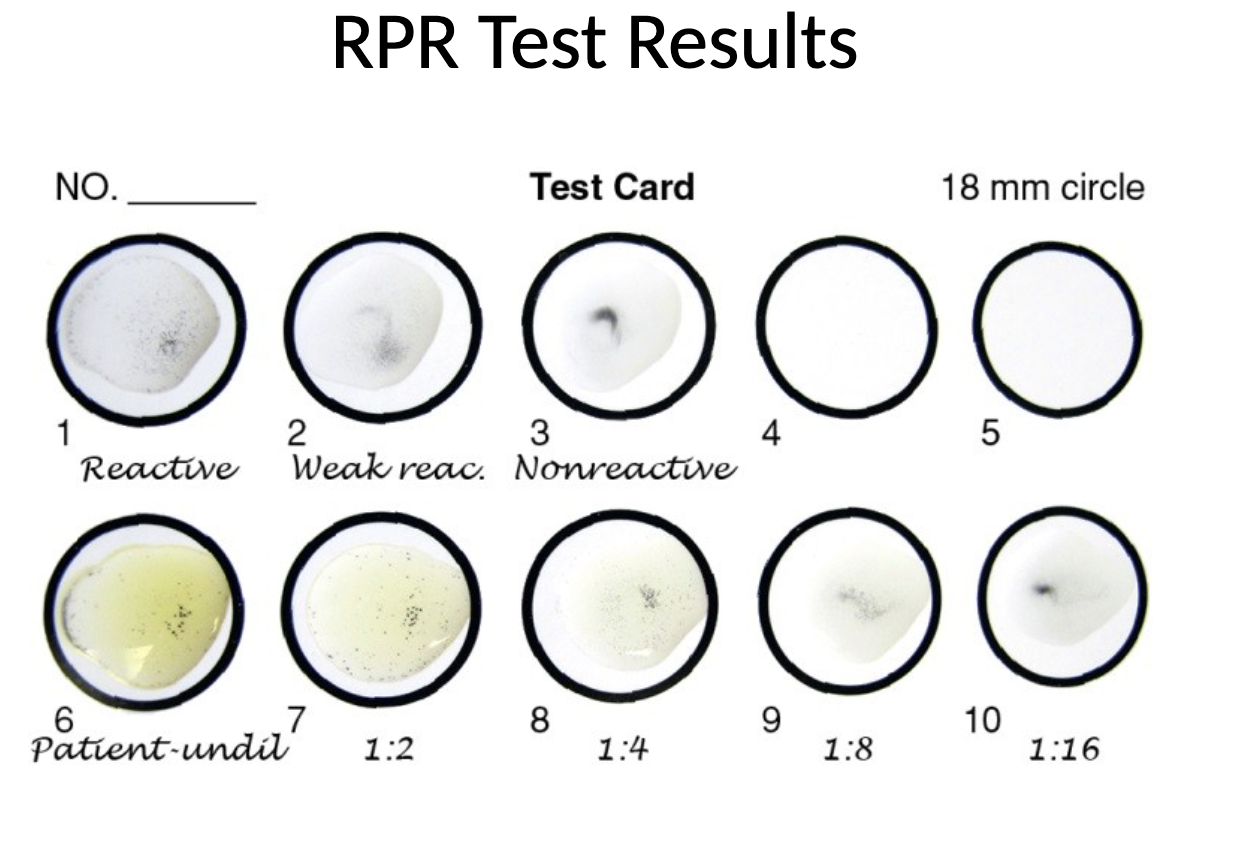
look for flocculation
screen: test undiluted patient serum
titer: test twofold dilutions of patient serum
VDRL test
patient serum mixed on a slide with cardiolipin-lecithin-cholesterol antigen suspension
viewed under light microscope for flocculation
RPR test
patient serum mixed on a card with charcoal particles with cardiolipin antigen
observe for macroscopic flocculation
Treponemal tests
detect antibody to T. pallidum
fluorescent treponemal absorption (FTA-ABS)
T. pallidum particle agglutination (TP-PA)
automated immunoassays
ELISA
CLIA
MFI
FTA-ABS test
an indirect immunoflorescence test for antibody to T. pallidum
patient serum incubated with sorbent to remove cross-reacting anitbodies
absorbed patient serum incubated with microscope slide fixed with T. pallidum
wash → AhG conjugated with fluorescein is added
after 2nd incubation and wash → slide examined under fluorescent microscope
TP-PA test
pt serum and controls diluted and incubated wit unsensitized gel particles or gel particles sensitized with T. pallidum antien
(+) = agglutination
(-) = no agglutination (button)
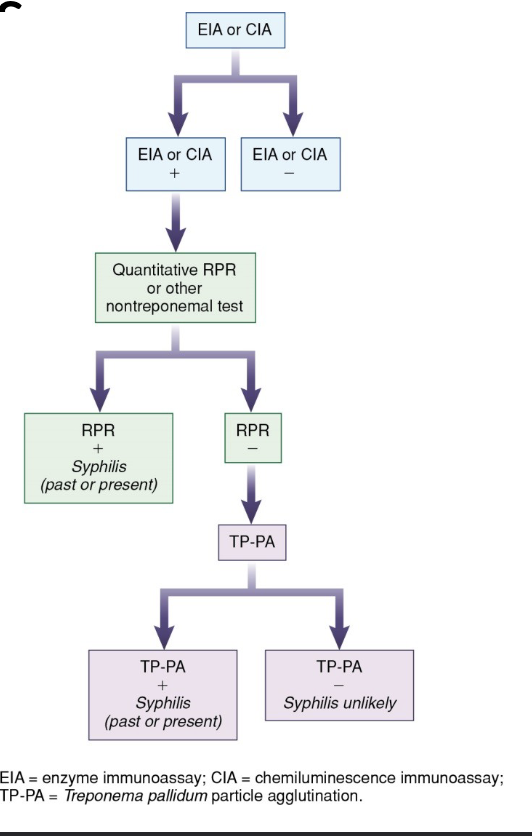
Reverse sequence algorithm for syphilis
screen with an automated immunoassay for T. pallidum antibody
confirm pos results with an RPR
perform TP-PA on samples with discrepant results
Lyme disease
caused by spirochete bacterium Borrelia burgdorferi
transmitted by Ixodes ticks
main resevoir: white-footed mouse
Clinical manifestations of lyme disease
Stage 1: localized rash
Stage 2: early dissemination
Stage 3: late dissemination with arthritis
Two-tiered testing for lyme
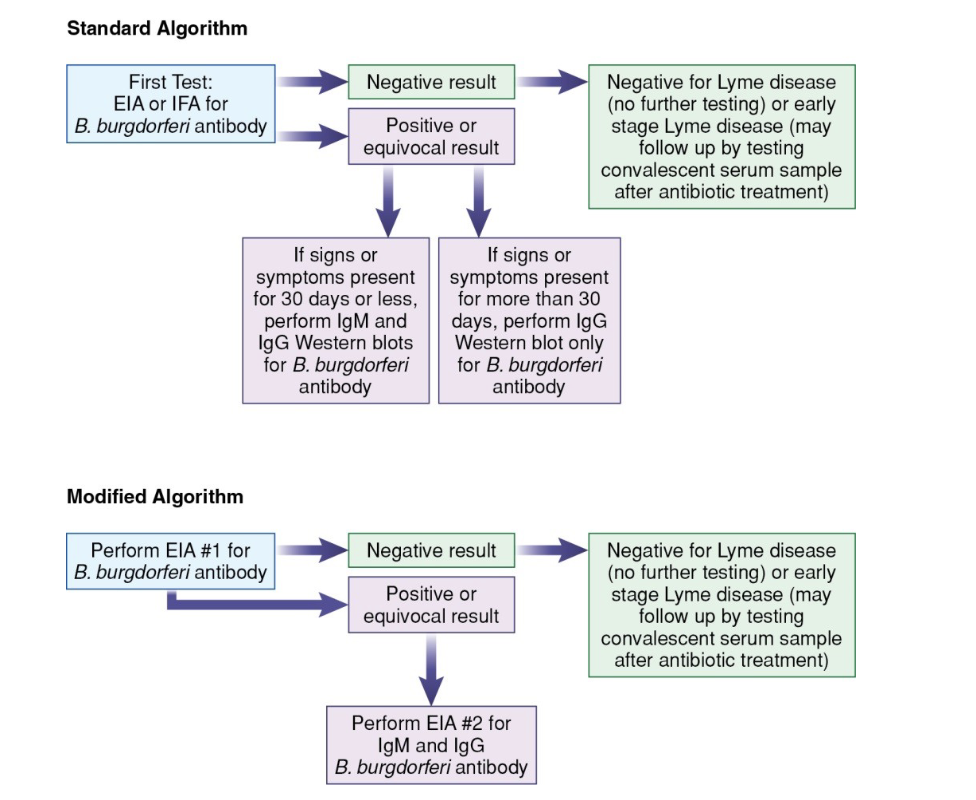
Western blot results for B. burgdorferi antibodies
Patient serum incubated with nitrocellulose membrane containing electrophoresed B. burgdorgeri antibodies
+ IgM: 2-3 characteristic bands
+IgG: 5-10 bands
Leptospirosis
zoonotic infection associated with occupational and recreational activities
humans are exposed by mucous membrane contact with urine-contaminated water
causes febrile episode that can progress to severe disease involving renal, liver, pulmonary, CNS
lab testing:
IgM screening with ELISA, ImmunoDOT, LFA
MAT is gold standard for confirmation
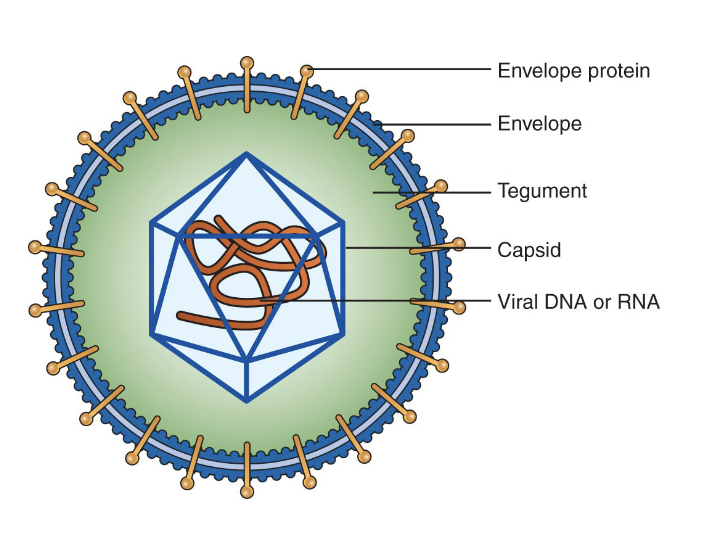
Virus structure
submicroscopic particles (nm)
core of DNA or RNA
protein coat (capsid)
some have outer envelope
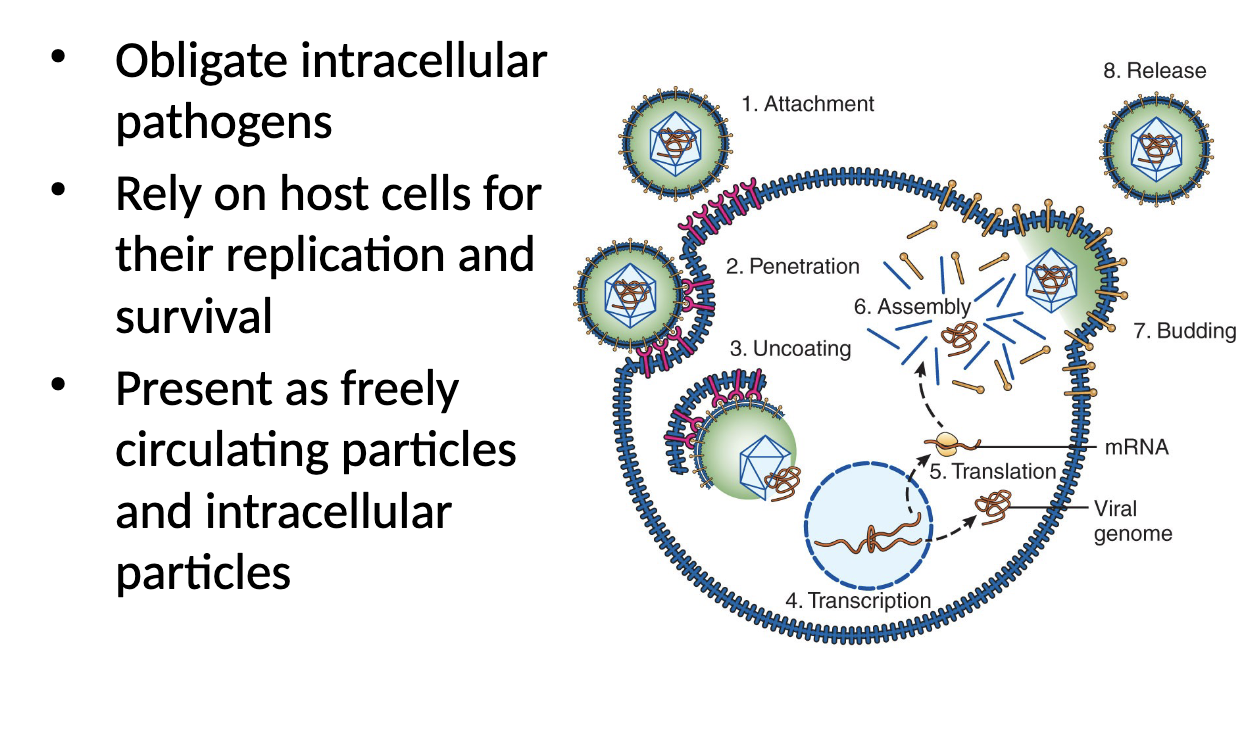
Virus life cycle
Immune defenses against viruses
innate
skin and mucous membrane barriers
recognition of PAMPs on virus-infected host cells
interferons a and B
humoral antibody response
antibodies attack free virus particles
viral neutralization, opsonization, C’ fixation, ADCC
cell-mediated immunity
interferon y and IL-2 produced by Th1 cells
host cells containing intracellular virus destroyed by CTLs
Viral escape mechanisms
mutations result in production of new viral antigens (influenza)
Viruses block action of immune system components (HCV binding C3b)
Suppression of immune response (CMV reducing MHC1 expression)
Immune function altered (EBV stimulating polyclonal B-cell activation)
latent state established
Lab testing for viral infections
serologic tests
distinguishes between current and past infection
IgM (+), IgG(±): current or recent infection; congenital infection
IgM (-), IgG (+): past infection
antibody titers used to monitor course of infection
assess immune status
virus-specific IgG indicates immunity
molecular tests
detect active infection
quantitative tests - guide antiviral therapy
Hepatitis viruses
transmitted fecal-oral route: Hep A/E
transmitted via parenteral route: Hep B/D/C
hepatitis clin and lab findings
general flu-like symptoms early in infection
pain in upper right quadrant of abdomen
hepatomegaly and liver tenderness with progression
jaundice, dark urine, light feces
elevated bilirubin and liver enzymes (ALT)
HAV
RNA virus transmitted by
fecal-oral route
close person-to-person contact
ingestion of contaminated food or water
produces acute hepatitis in majority of adults
infections in children usually asymptomatic
Formalin-inactivated HAV vaccine
HAV immune globulin recommended if unimmunized persons exposed
acute infection indicated by (+) IgM and anti-HAV
immunity indicated by (+) total anti-HAV along with (-) IgM anti-HAV
HEV
RNA virus with 4 genotypes
HEV-1/2 transmitted primarily through ingestion of feces-contaminated drinking water
HEV-3/4 transmitted mainly by consumption of infected pork
mostly asymptomatic or self-limiting infections
severe infections possible (in immunocompromised or pregnant)
acute infection indicated by IgM anti-HEV
IgG anti-HEV indicates past exposure
can detect HEV RNA in blood or stool samples during acute infection
HBV
DNA virus transmitted through parenteral or perinatal routes
sexual contact
IV drug use
occupational needlestick injury
during birth process
acute infection: symptoms increase with age
chronic infection
persists for 6 months or more
occur sin 90% of infected infants, 10% of infected adults
increases in risk of liver cirrhosis or hepatocellular carcinoma
infection preventable by immunization
HBIG recommended for unimmunized persons exposed to HBV
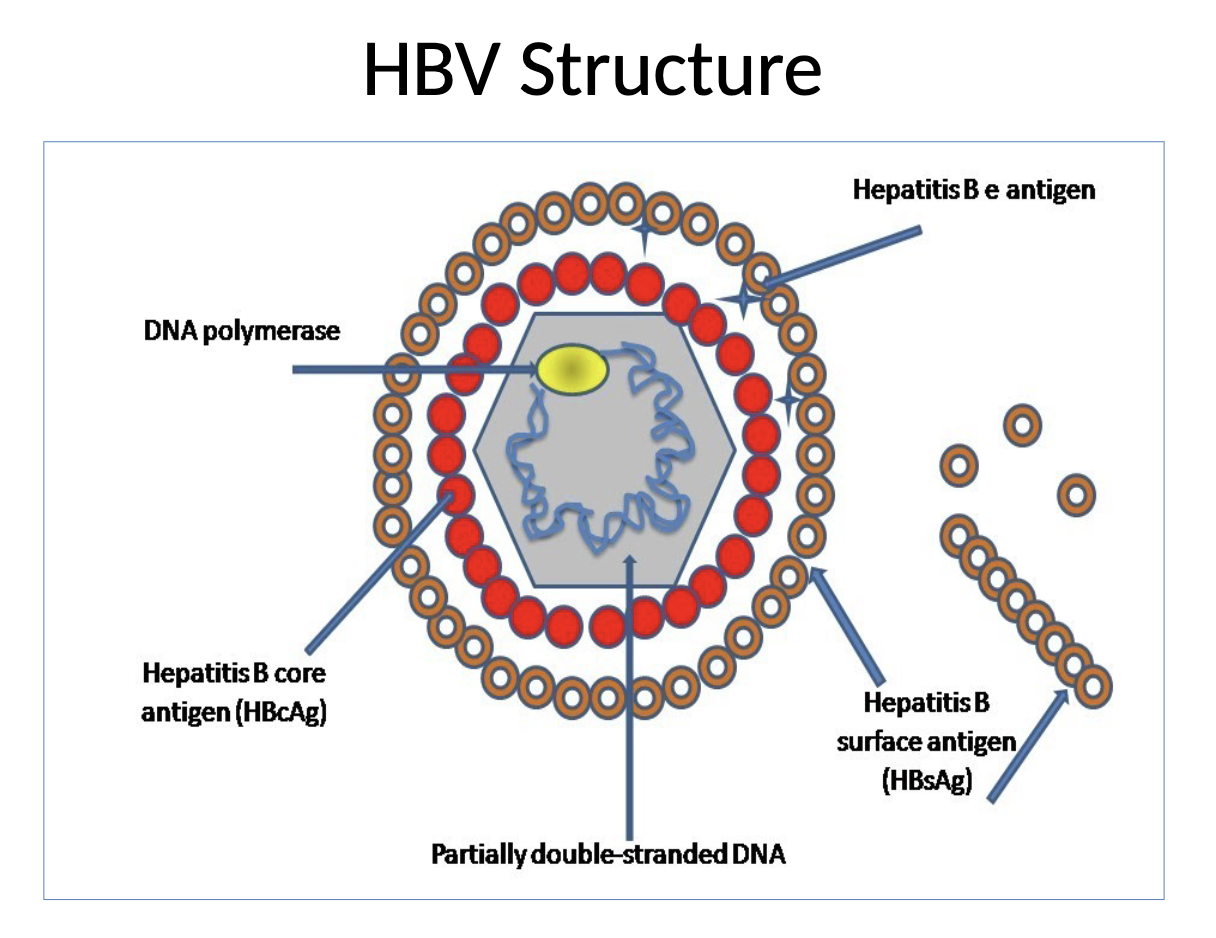
Hepatitis B surface antigen (HBsAg)
protein on outer envelope of virus
excess circulates in virus-like particles in blood
marker for active HBV infection
component of hepatitis B vaccine
Hepatitis Be antigen (HBeg)
protein in core of HBV
marker of active viral replication
indicates high degree of infectivity
Anti-HBc
directed against hepatitis B core antigen
IgM anti-HBc indicates current or recent acute infection; detects “core window”
total anti-HBc consists mainly of IgG and can indicate a current or past infection
Anti-HBe
directed against HBeAg
indicates recovery from hep B
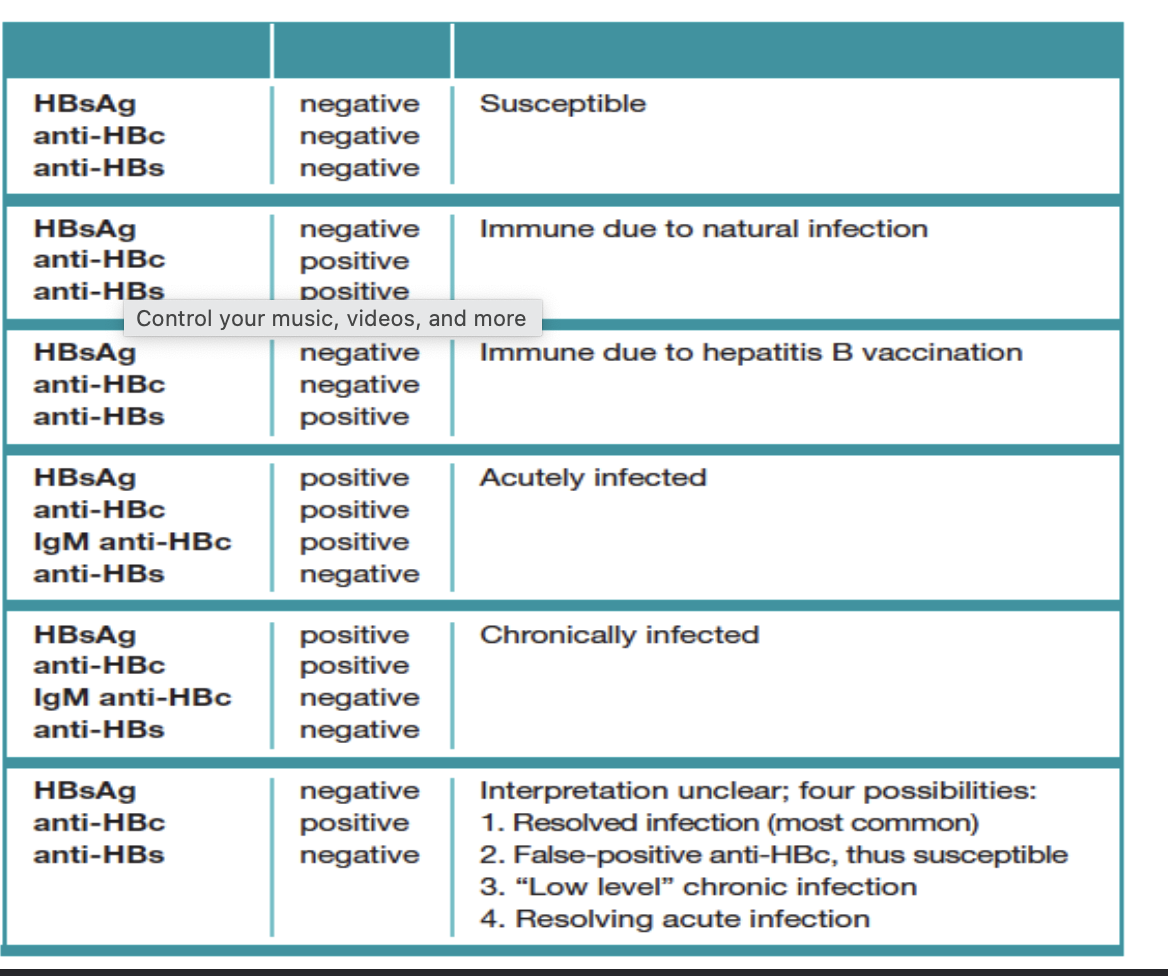
Anti-HBs
directed against HBsAg
indicates immunity to hep B$
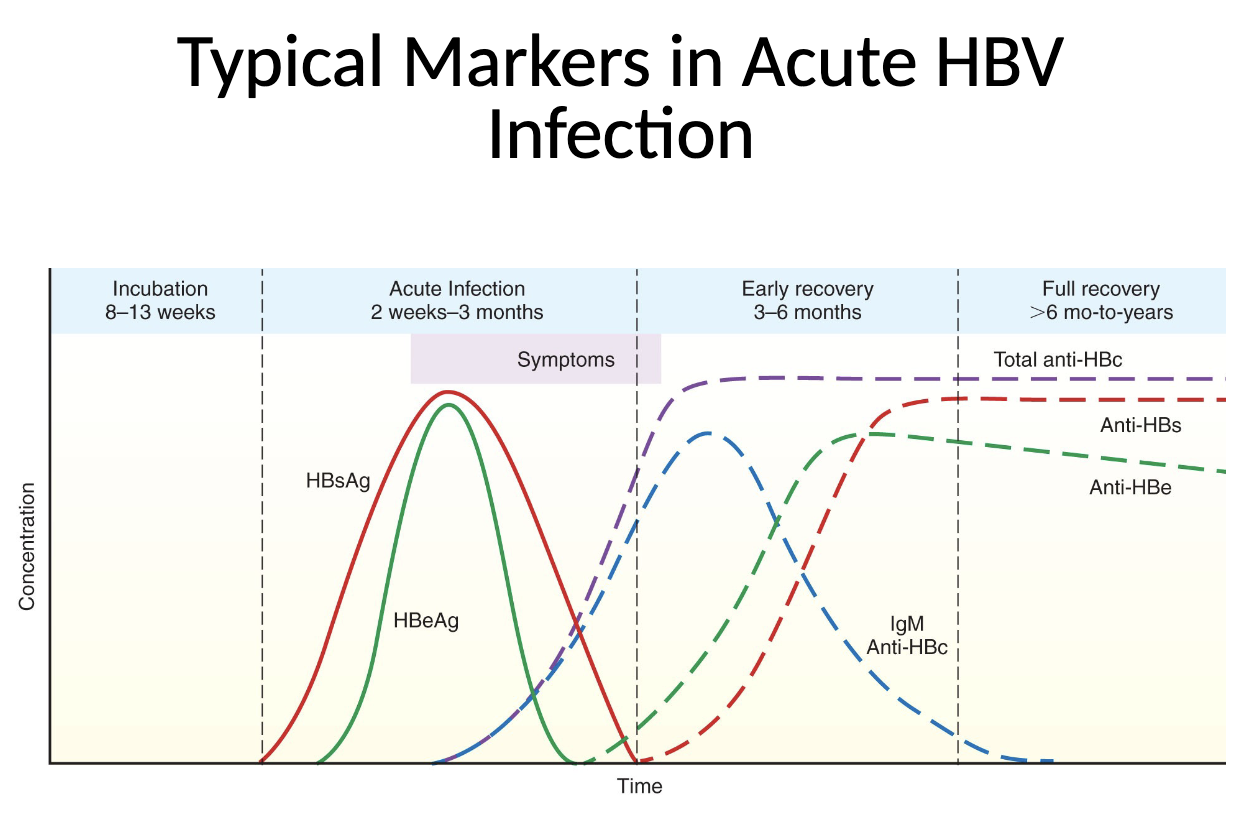
Typical markers in Acute HBV infection
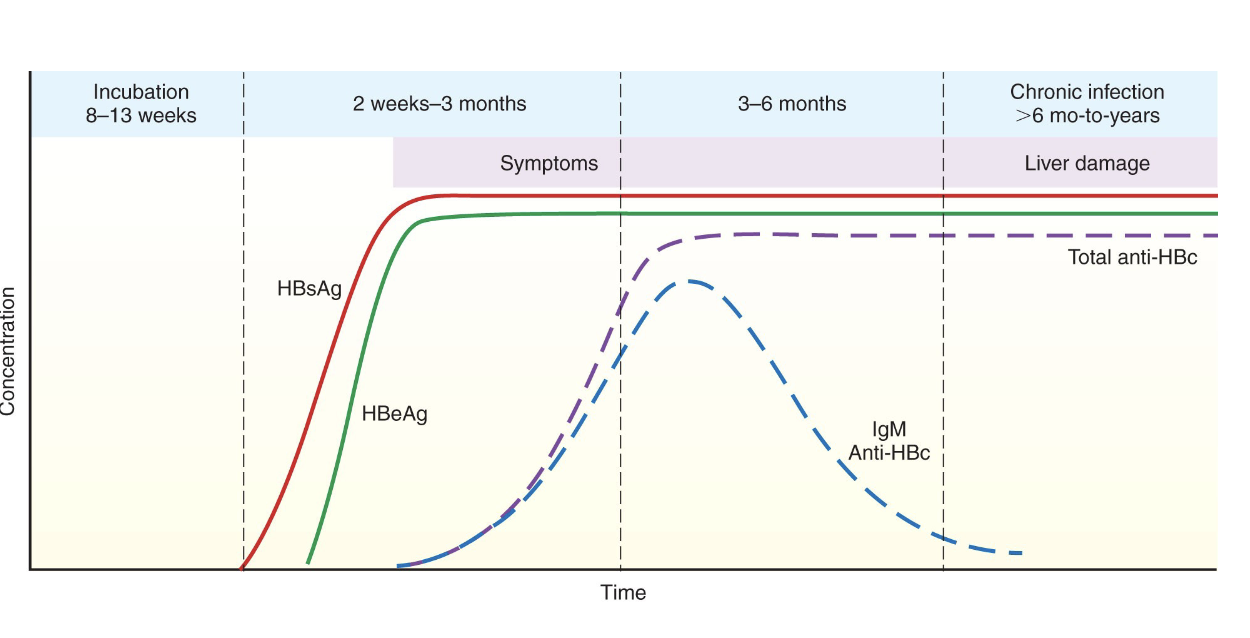
Typical markers in chronic HBV infection
HDV
RNA virus that requires presence of HBV
transmitted through parenteral or perinatal routes
coinfection with HBV
usually results in acute, self-limited hepatitis
(+) for anti-HDV, IgM anti-HBc
Superinfection of chronic HBV carriers
chronic liver disease with accelerated progression to cirrhosis and liver failure
(+) for anti-HDV, IG anti-HBc
HDV RNA= marker of active viral replication
HCV
RNA virus with 7 genotypes
transmitted by exposure to contaminated blood, sexual contact, perinatal
most infections asymptomatic at first but develop into chronic liver disease
anti-HCV IgG used for screening and diagnosis
Qualitative HCV RNA used for confirmation
quantitative molecular test used to monitor viral load during antiviral therapy
genotyping used to determine best therapy
EBV
DNA herpes virus most commonly transmitted by intimate contact with saliva secretions
begins in oropharynx in B cells and epithelial cells and spreads through lymphoreticular system
infectious mononucleosis
lymphoproliferative disorders
certain malignancies (Burkitt lymphoma)
Infectious mononucleosis (IM)
absolute lymphocytosis
20% or more atypical lymphocytes
heterophile antibody
reacts with antigens from 2 or more species
monospot
paul-bunnet test
Antibodies to EBV antigens
early antigens: EA-D {acute IM}, EA-R
late antigens: viral capsid antigens, IgM anti-VCA (acute) IgG anti-VCA (acute or past IM)
latent phase antigens: EBV nuclear antigens (EBNA), anti-EBNA appear during convalescent IM
CMV
DNA herpes virus transmitted through oral secretions, genital secretions, congenitally, transfusion/transplantation
may be asymptomatic or induce mononucleosis-like syndrome in health individuals
in sick people, can disseminate to lungs, liver, GI tract, CNS, and eyes and cause life-threatening infections
may cause congenital defects an decreased survival in infants
Lab diagnosis of CMV
direct virus detection
viral culture
ID of CMV antigens
molecular tests for CMV DNA
serology
used to screen blood and organ donors; pregnant women
presence of IgG anti-CMV indicates infection
low avidity antibodies indicative of recent infection
VZV
DNA herpes virus
transmitted by inhalation of infected resp secretions or aerosols from skin lesion
cause of: varicella, Herpes zoster (shingles)
Lab diagnosis of VZV
diagnosis is usually based on characteristic clin findings
real-time PCR
serology is most useful in determining immunity
Rubella
RNA virus transmitted through resp droplets or across placenta
cause of German measles
can cause:
congenital abnormalities
miscarriage
stillbirth in infants born to infected mothers
Lab testing for rubella
serology
presence of IgG used to screen for immunity
congenital infection indicated by IgM antibodies in fetal blood, cord blood, or neonatal serum
current infection indicated by rubella-specific IgM or fourfold rise IGG
low avidity antibodies indicate recent infection
viral culture
molecular methods
Rubeola virus - measles
RNA virus transmitted through resp droplets
cause of: measures, subacute sclerosing panencephalitis (SSPE)
diagnosis based on clin presentation and confirmed by serology
Rubula virus- mumps
RNA virus transmitted through resp droplets, saliva, fomites
most common clin manifestation: parotitis
diagnosis based on clin presentation
confirmation by culture or RT-PCR
HTLV-I/II
closely related to retroviruses that preferentially infect T lymphs
transmission mainly bloodborne, sexual contact, perinatal (breastfeeding)
HTLV-I cause of adult T-cell leukemia/lymphoma (ATL_ and HAM/TSP
serologic tests for antibodies to HTLV I/II are used to diagnosis infections and screen blood donors
ELISA or CLIA used to screen; Western blot or LIA used for confirmation of + results
Human Immunodeficiency virus (HIV)
cause of AIDS
HIV-1
cause of most HIV infections worldwide
4 groups (M, O, N, P)
9 subtypes in group M (ABCDFGHJK)
HIV-2
originated in West Africa
Modes of HIV transmission
sexual contact involving exchange of body fluids
contact with blood or other body fluids
perinatal: mother to child
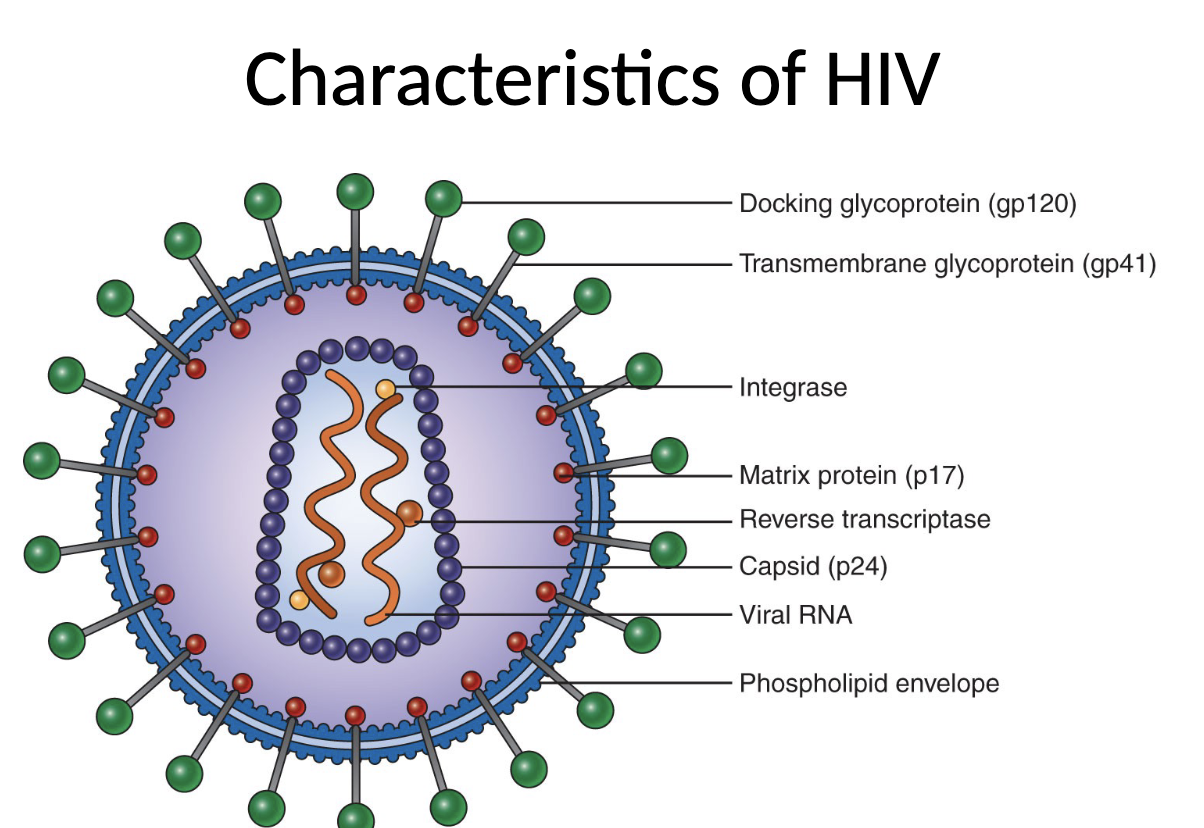
Characteristics of HIV
retrovirus
contains 2 copies of ssRNA
reverse transcriptase transcribes viral RNA into DNA
surrounded by protein coat (capsid)
outer envelope of glycoproteins embedded in lipid bilayer
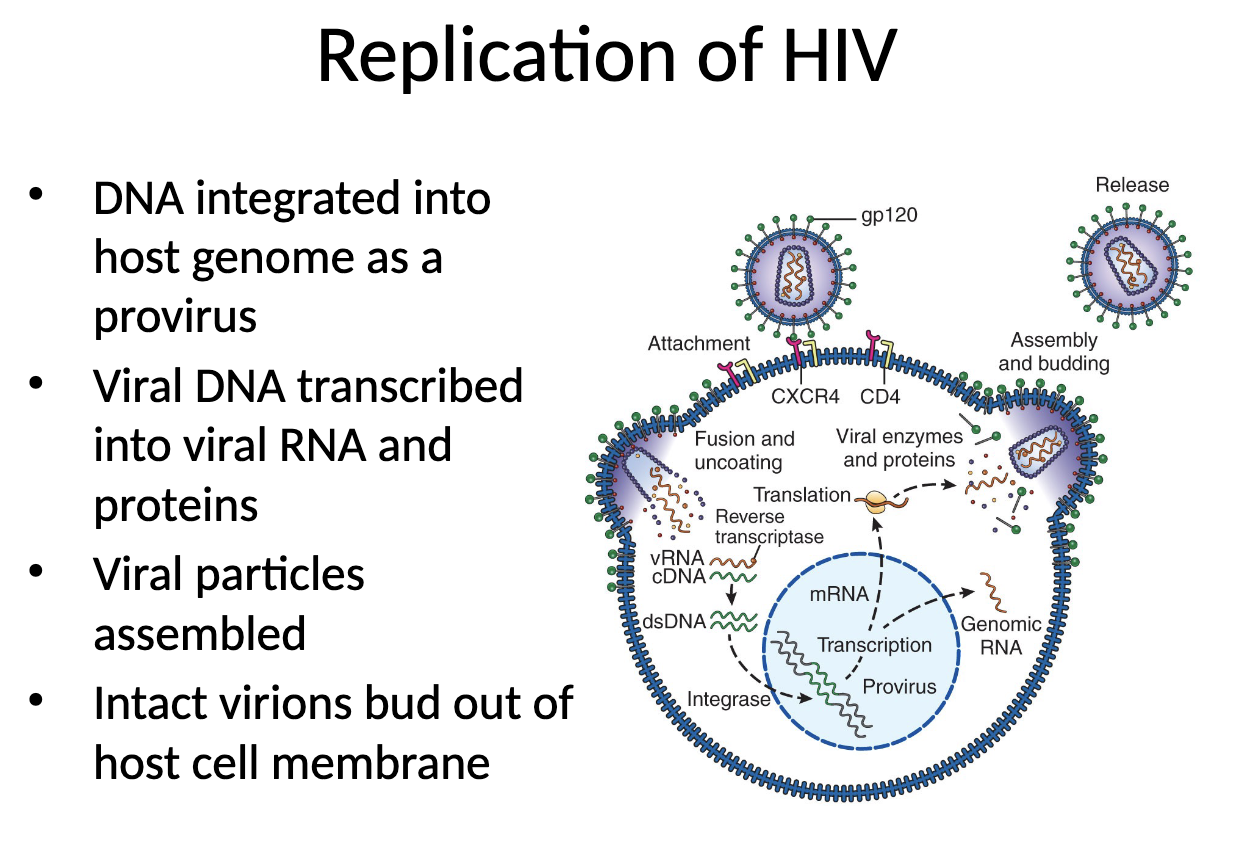
Replication of HIV
attachment of HIV into host cell
main target: CD4 Th
coreceptor required
fusion and undercoating
RT converts viral RNA into complementary DNA
Immune responses to HIV
innate defenses
NK cells mediate cytolysis of HIV-infected cells
dendritic cells stimulate release of cytokines that have antiviral effects
humoral antibody production
antibodies are detected by 6 weeks after infection
antibodies produced later may prevent HIV from infecting host cells and participate in ADCC
cell-mediated immunity
T cells produce cytokines with antiviral activity
CTLs destroy HIV-infected host cells
HIV escape from immune responses
genetic mutations rapidly occur, generating new viral mutants with altered antigens
HIV can down regulate expression of MHC-I molecules on infected host cells
HIV can survive as a latent provirus for prolonged periods
CD4 T cells are prime targets of destruction
HIV primary infection
acute, early infection
may be asymptomatic or have flu-like syndrome that resolves
high level of viremia and decrease in CD4 T-cell #
HIV clinical latency
absence of clinical symptoms
decrease in viremia increase in CD4 T-cell #
AIDS
resurgence of viremia and decrease in CD4 T-cell #
profound immunosuppression, with appearance of life-threatening opportunistic infections and malignancies
Antiretroviral therapy (ART)
drugs that block various steps of HIV replication cycle
most effective when used in combination (CART)
have significantly improved morbidity and mortality of HIV-infected persons and have reduced the rate of perinatal transmission
Previous testing algorithm
screen for HIV1/2 antibodies by ELISA or rapid EIA
confirm + test results by repeating ELISA, followed by Western Blot
CDC algorithm
labs should begin antigen/antibody HIV screening immunoassay, followed (when reactive) by an HIV-1/2 antibody differentiation immunoassay
when differentiation assay interpretation is negative or indeterminate for HIV-1, perform HIV nucleic acid test (NAT)
Disease monitoring
peripheral blood CD4 T-cell counts
quantitative viral load assays
drug resistance testing
CD4 T-cell Enumeration
these counts are best indicator of immune function in HIV-infected individuals
incubate peripheral blood with fluorescent-labeled anti-CD4; analyze results by flow cytometry
in untreated patients, CD4 T-cell # declines progressively, and CD4 T: CD8 T-cell ratio is less than 1:1
CD4 T-cell count of less than 200 cells/uL indicates stage 3 infection (AIDS)
quantitative viral load assays
measure amount of HIV RNA circulating in patient plasma
methods: qPCR, bDNA
HIV RNA detectable about 11 days after infection
Phenotype resistance testing
determine ability of HIV from clinical samples to grow in presence of antiretroviral drugs
involve sophisticated technologies only performed by specialized reference labs
Testing of infants younger than 18 months
maternal antibodies in infant serum can complicate serologic test results
qualitative HIV-1 DNA PCR using infant’s peripheral blood mononuclear cells is preferred method
serologic testing at 12-18 months of age may be used to confirm diagnosis