BIOC 4331 Lecture 22
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
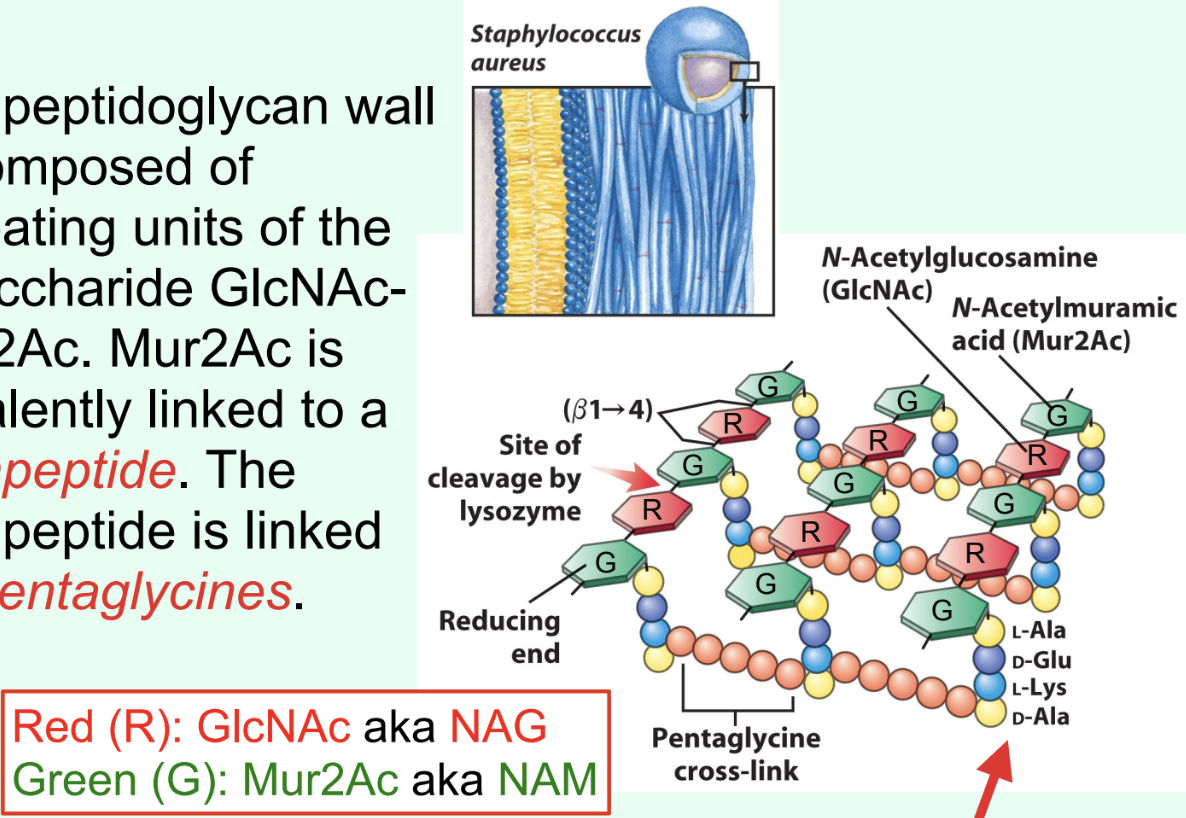
What is the Gram-positive bacterial cell wall primarily composed of?
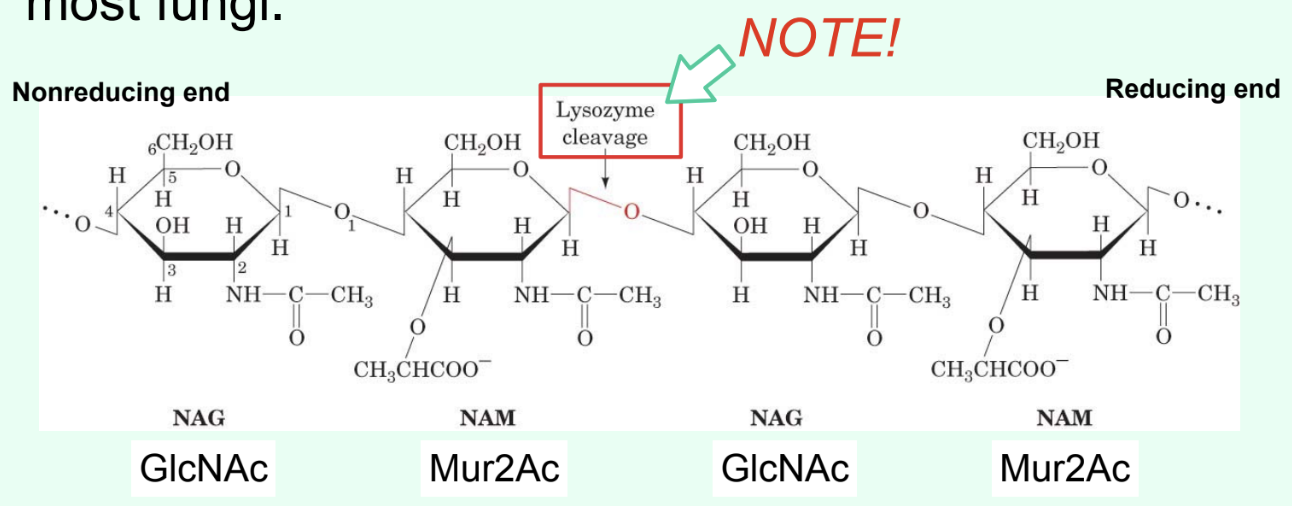
Peptidoglycan made of repeating disaccharide units: GlcNAc (NAG) and Mur2Ac (NAM).
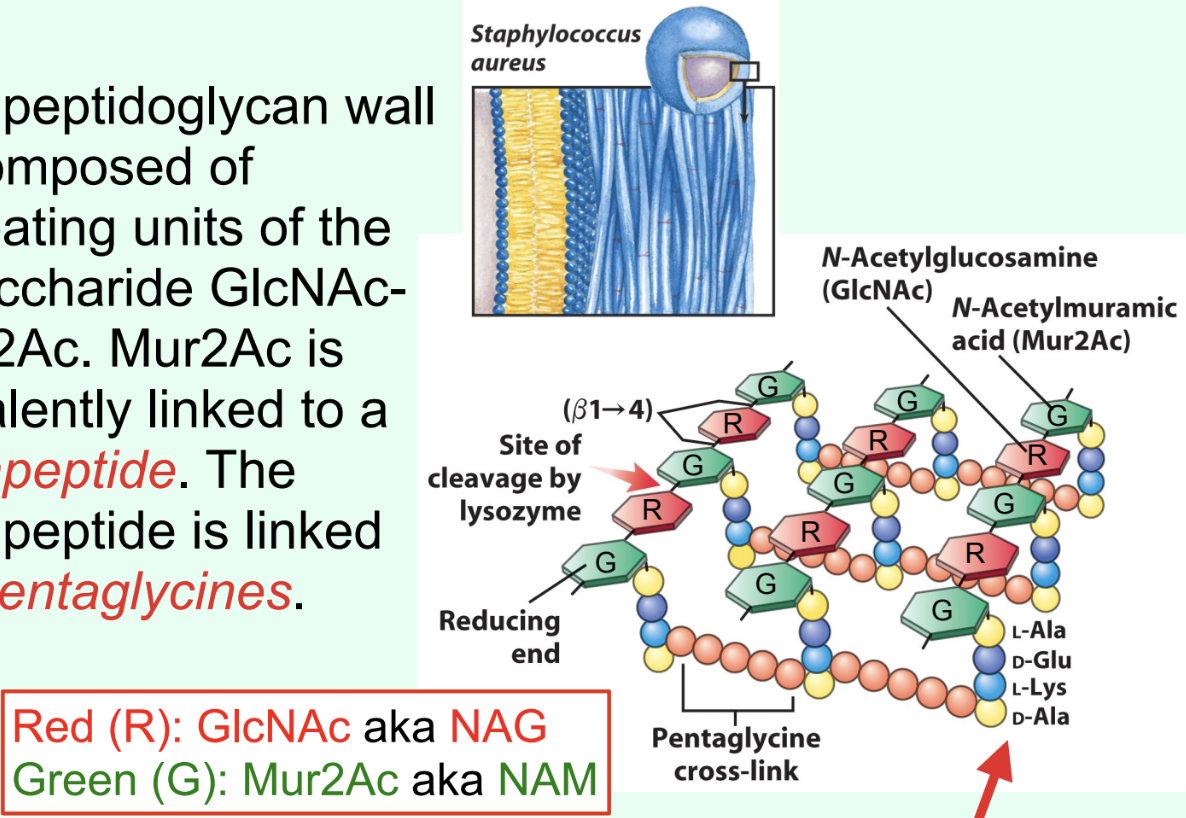
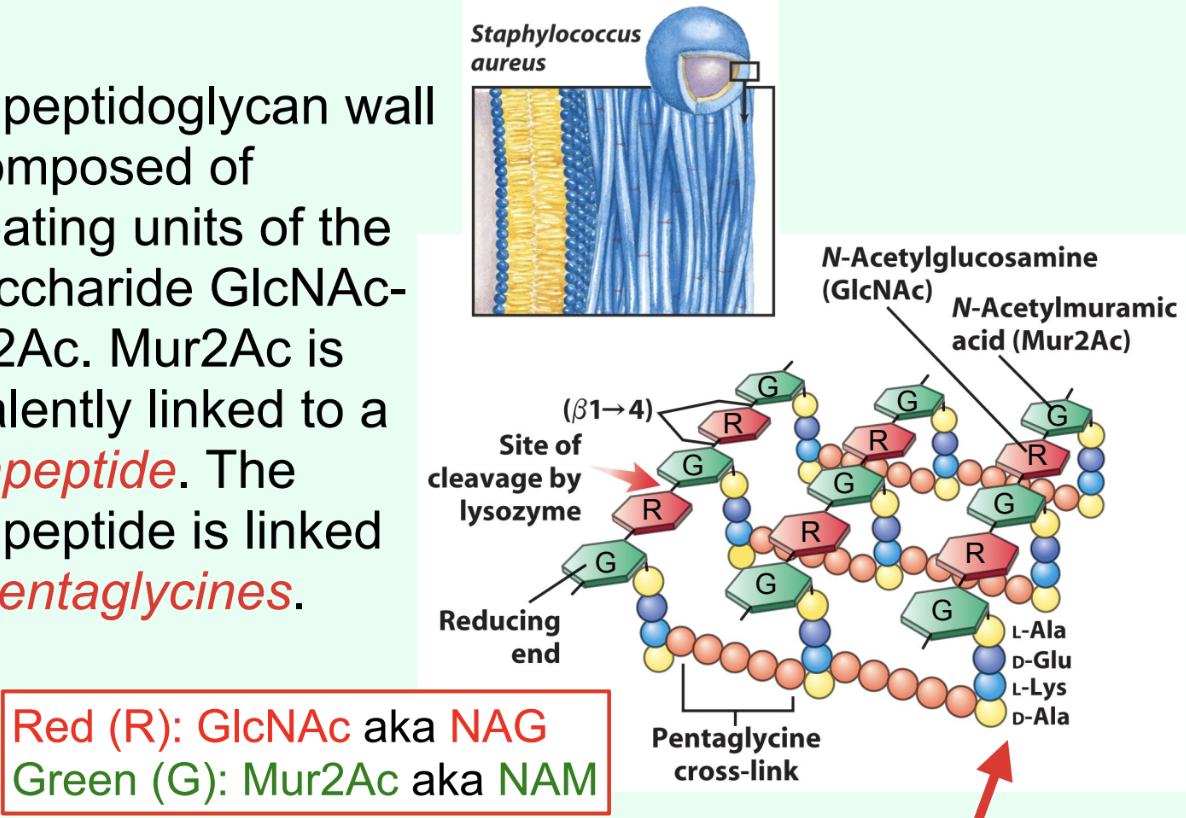
What is attached to Mur2Ac (NAM) in peptidoglycan?
A tetrapeptide covalently linked through the lactyl group.
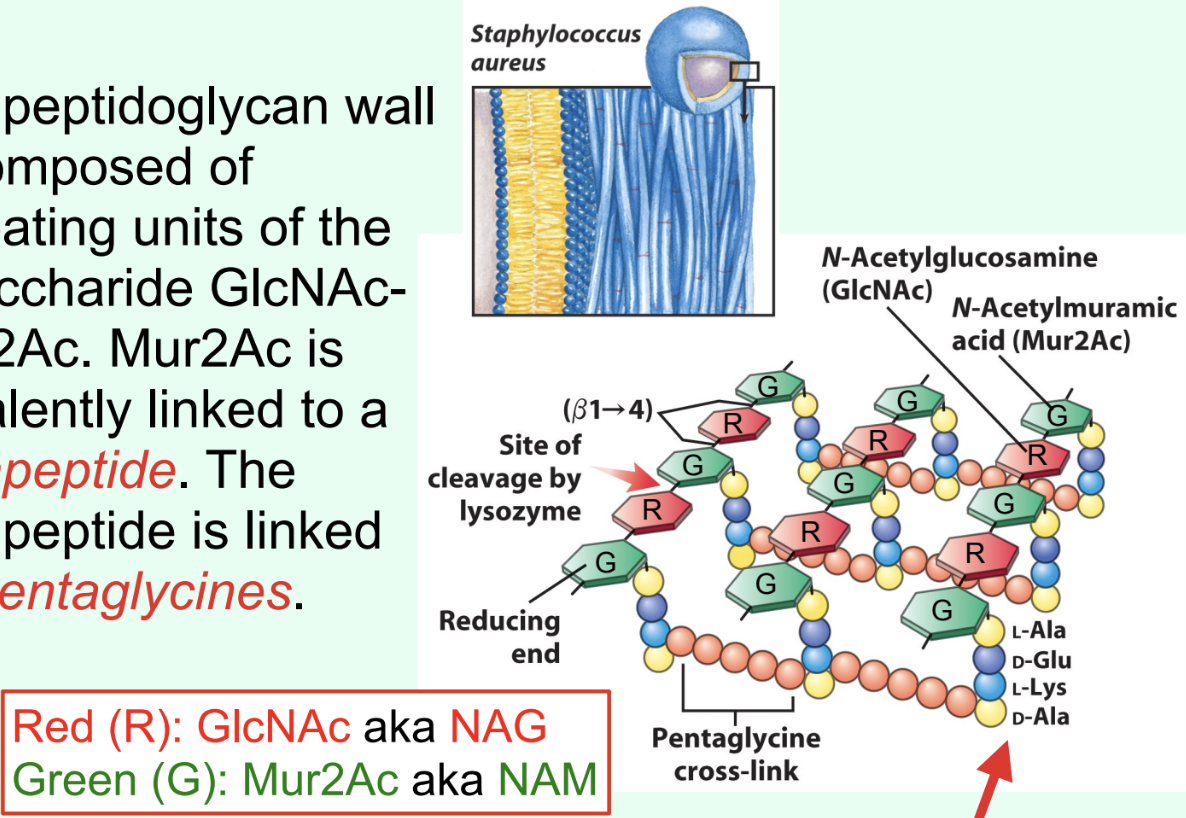
How are tetrapeptides cross-linked in Gram-positive bacteria?
By pentaglycines (bridges).
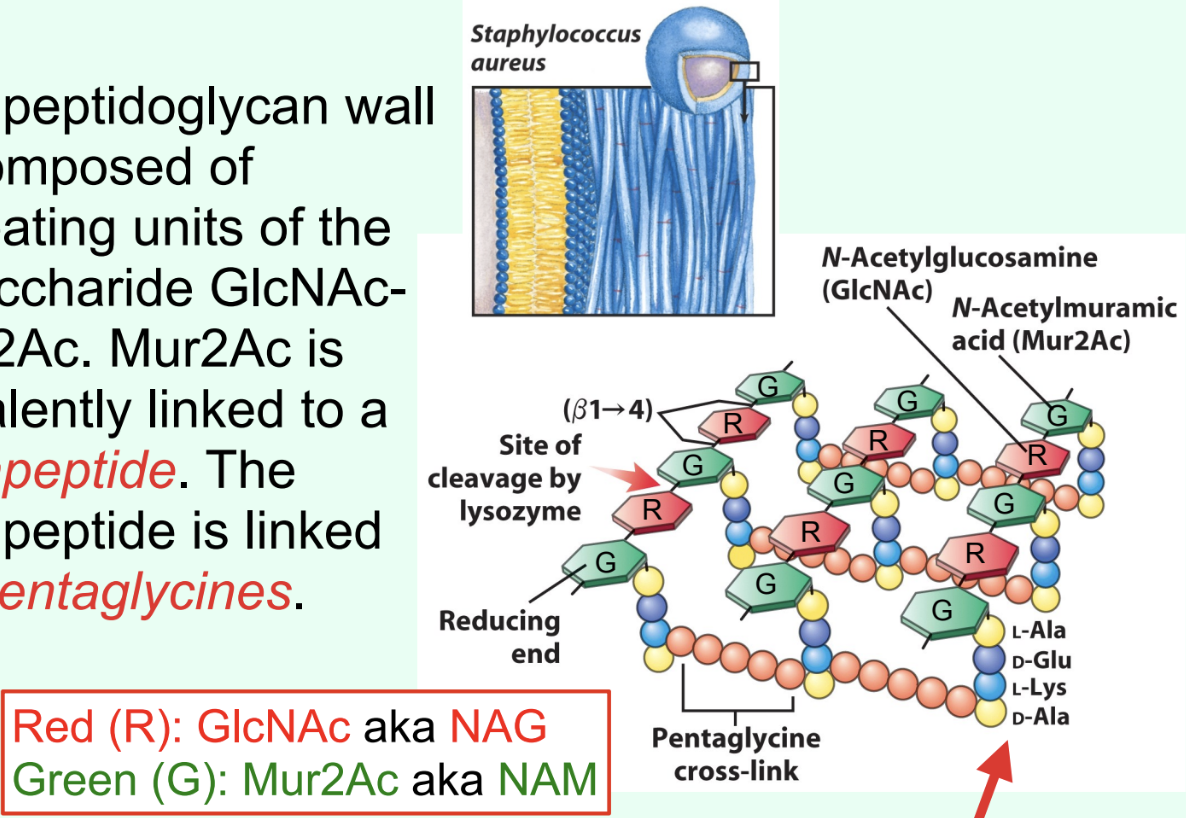
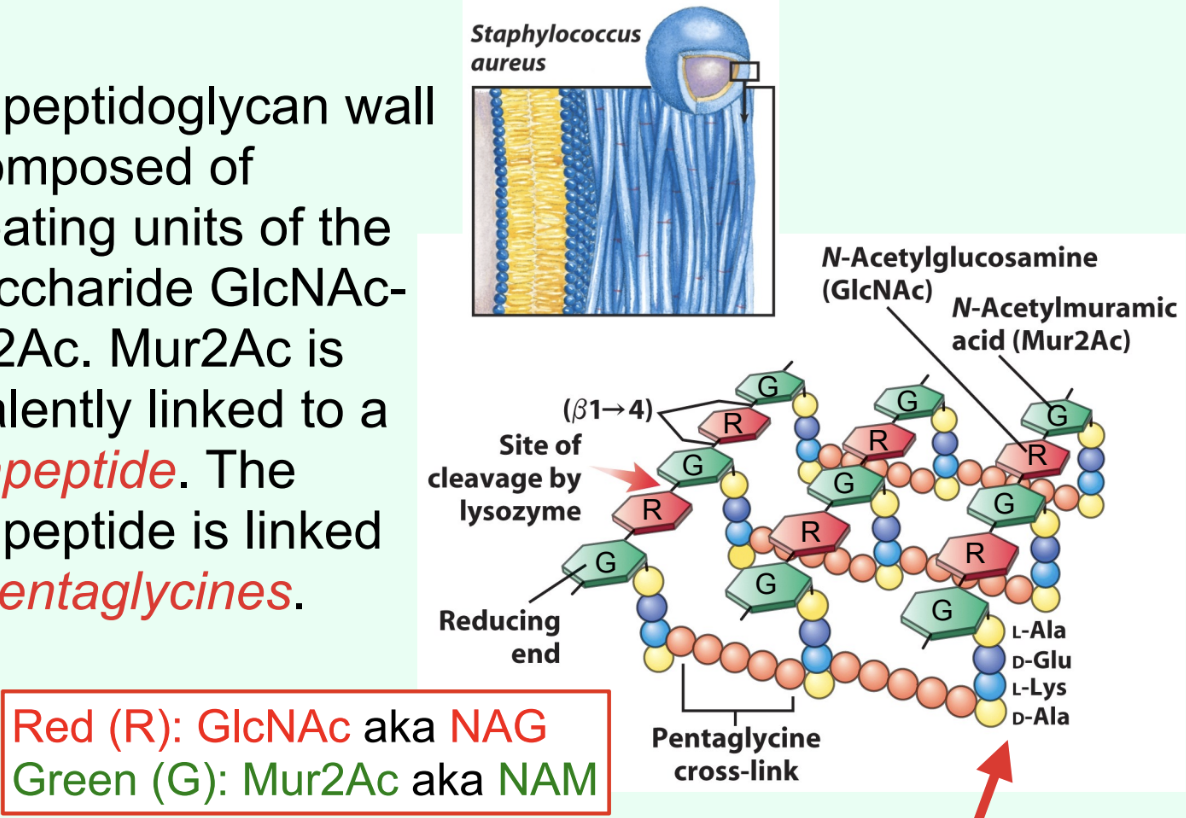
What is unusual about the tetrapeptide in peptidoglycan?
It contains D-amino acids (proteins are almost exclusively made of L-amino acids).
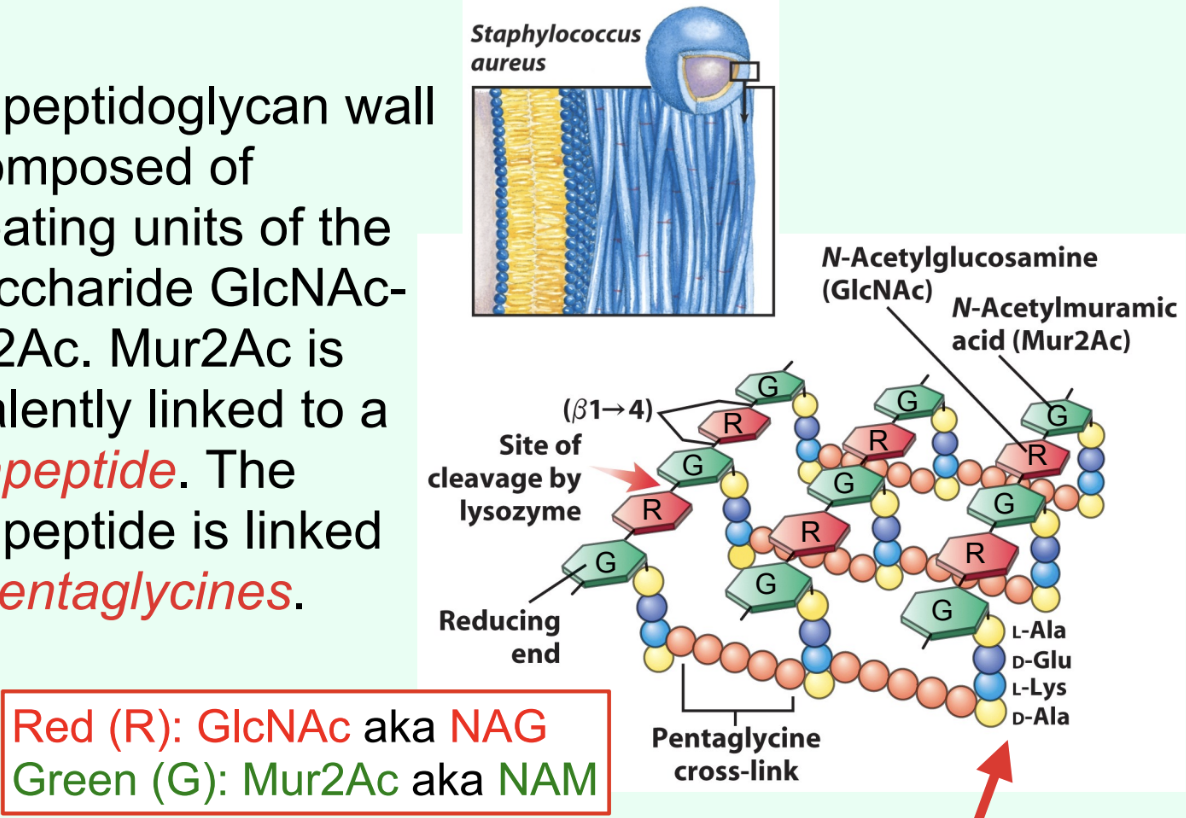
What type of ring do GlcNAc and Mur2Ac contain?
A 6-membered pyranose ring.
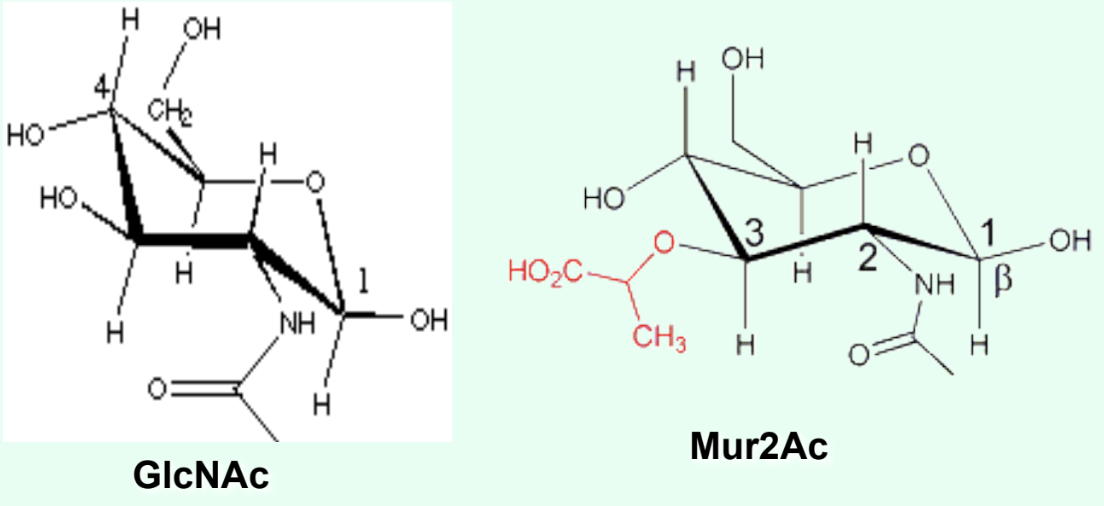
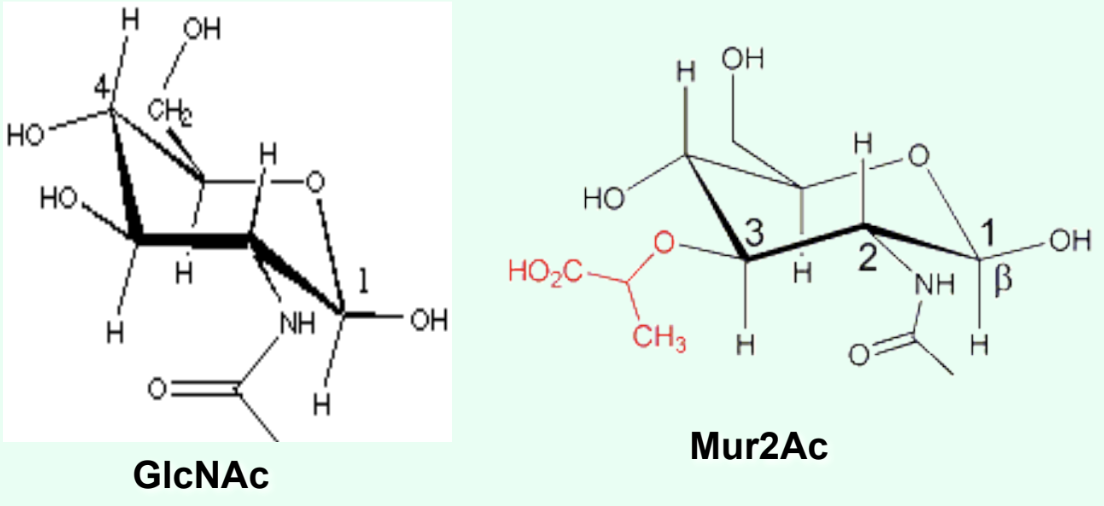
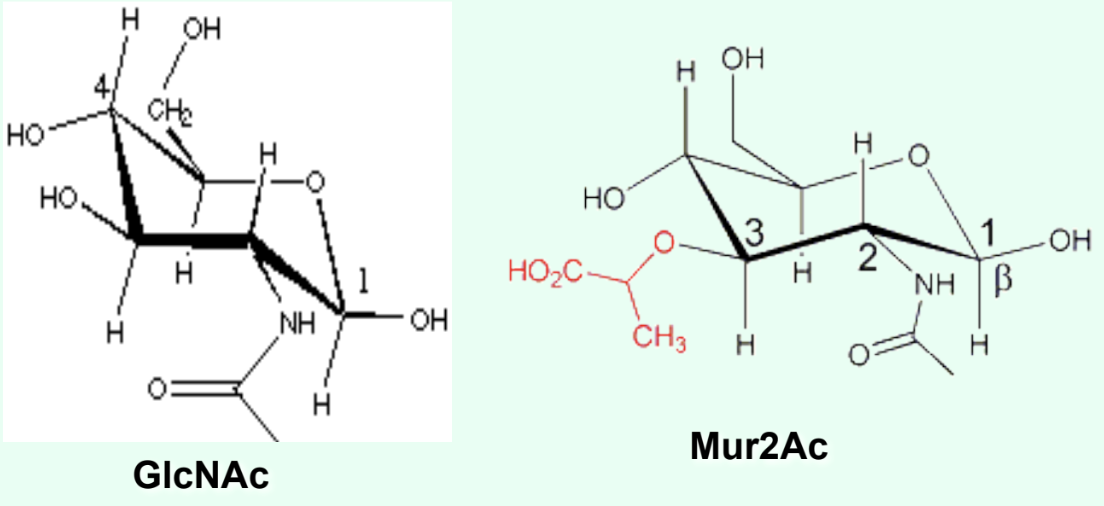
What is the ONLY structural difference between GlcNAc and Mur2Ac?
GlcNAc has an –OH at C3
Mur2Ac has a lactyl group at C3
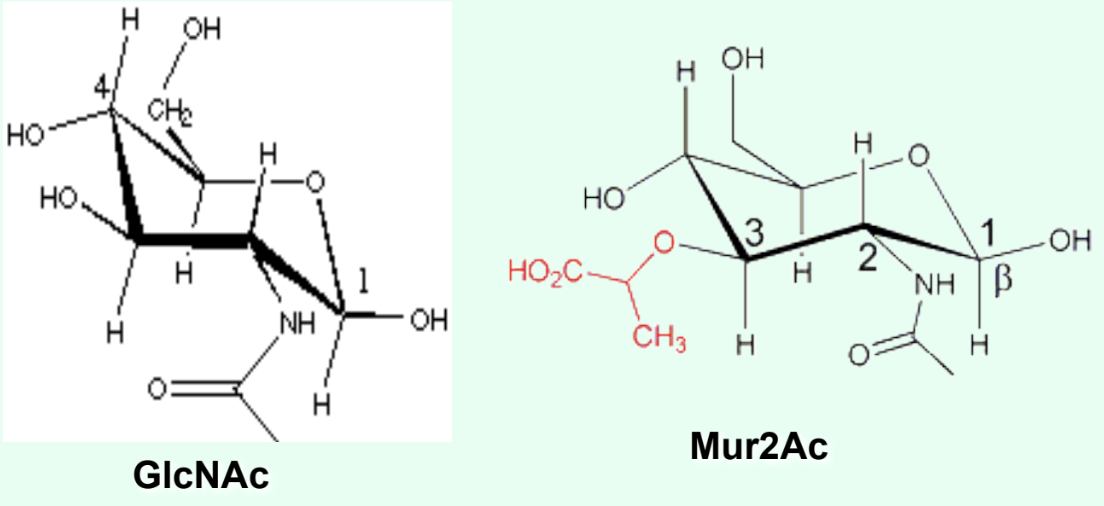
Through what group is Mur2Ac covalently linked to the tetrapeptide?
Through the lactyl group at C3.
What is the most energetically stable conformation of a pyranose?
The chair conformation.
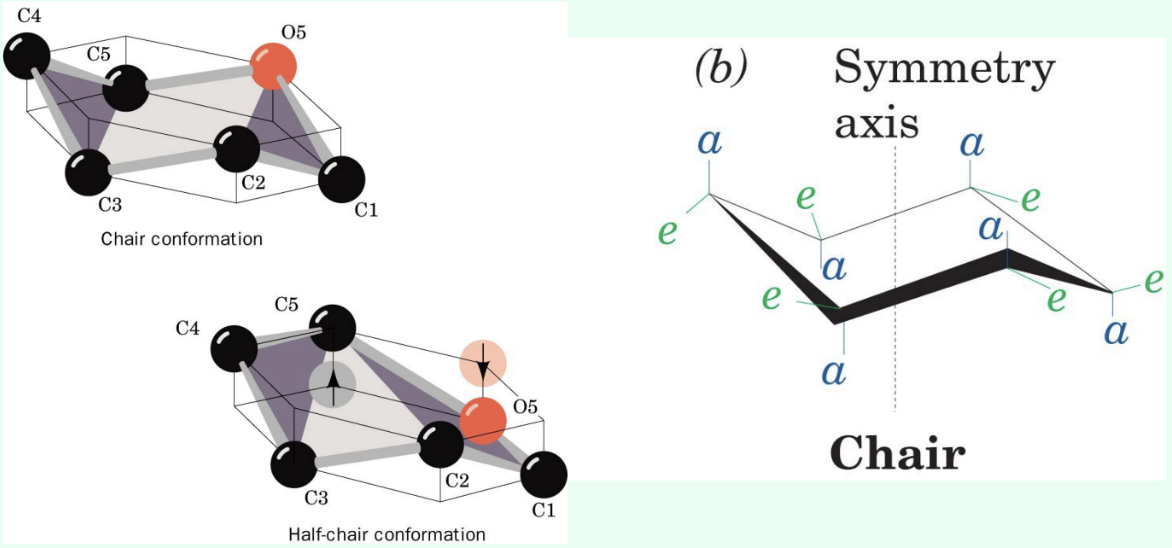
What is the half-chair conformation?
An energetically less favorable distorted conformation where C1, C2, C5, and O5 lie in a plane.
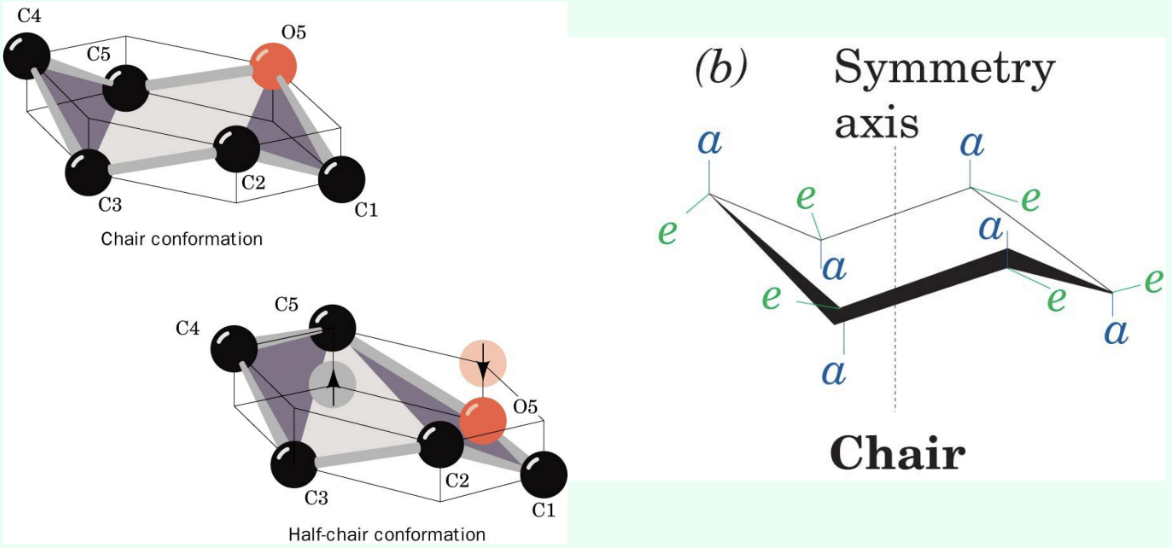
Why is the half-chair conformation important in lysozyme catalysis?
It resembles the transition state and increases the reaction rate.
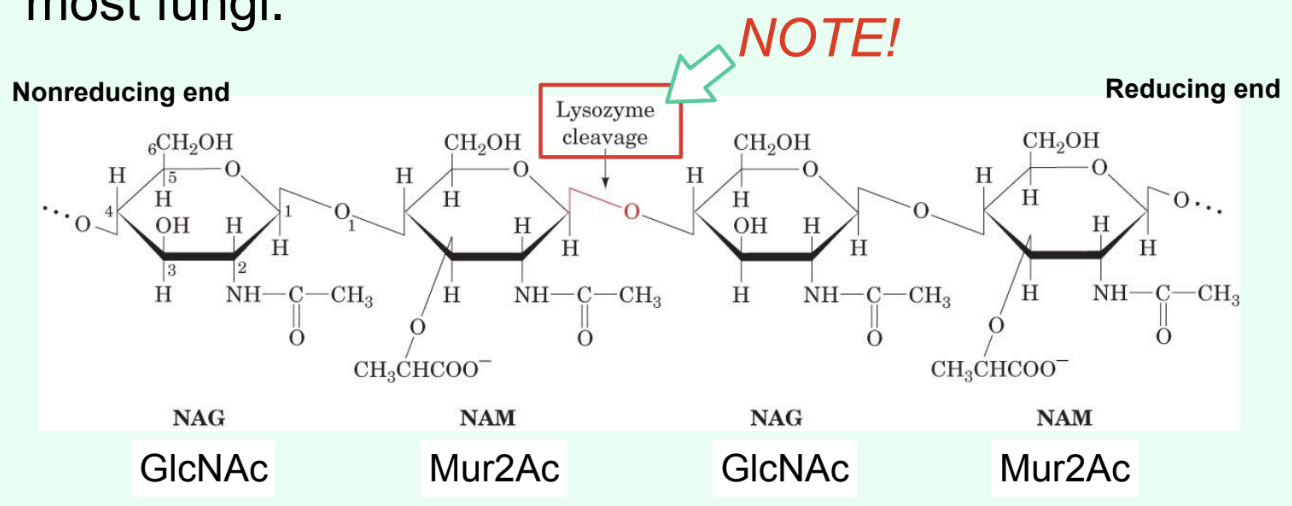
What does lysozyme do?
Breaks down Gram-positive bacterial cell walls.
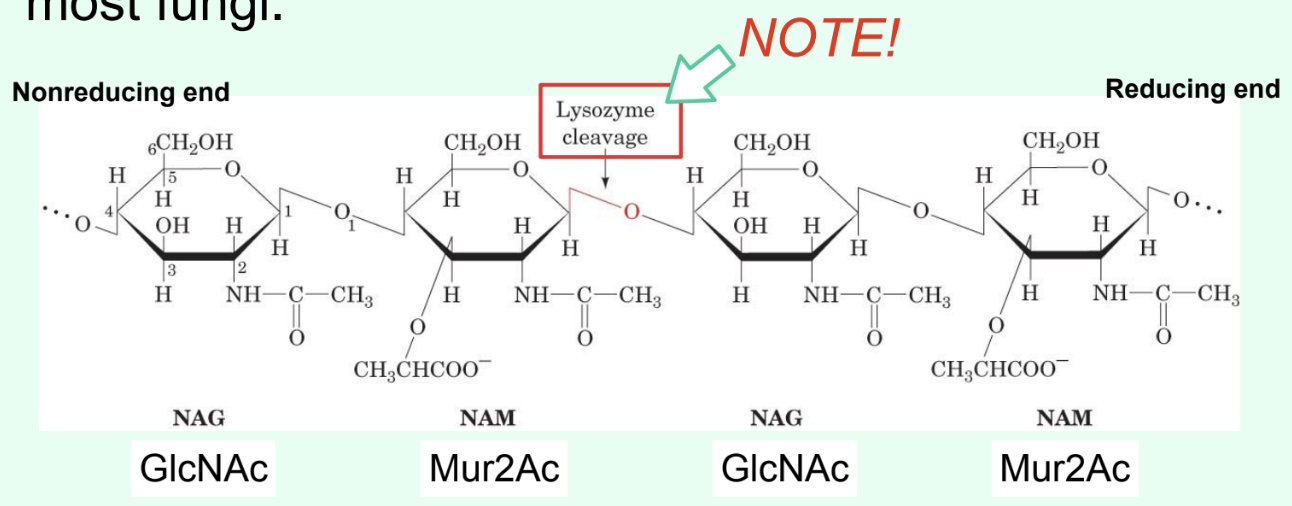
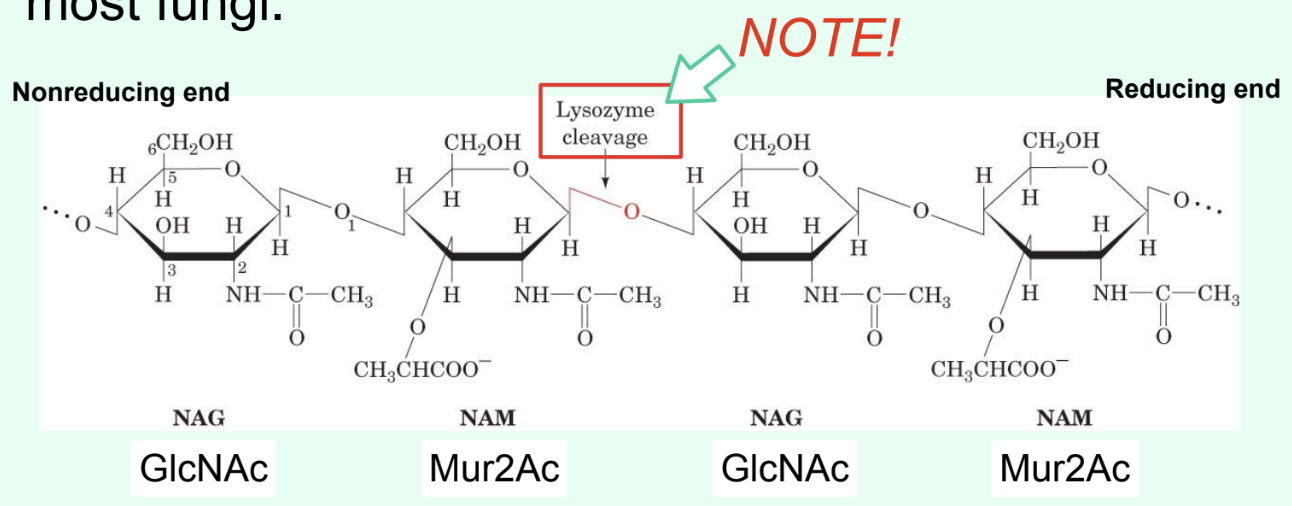
What specific bond does lysozyme catalyze?
The hydrolysis of the β(1→4) glycosidic linkage between Mur2Ac and GlcNAc.
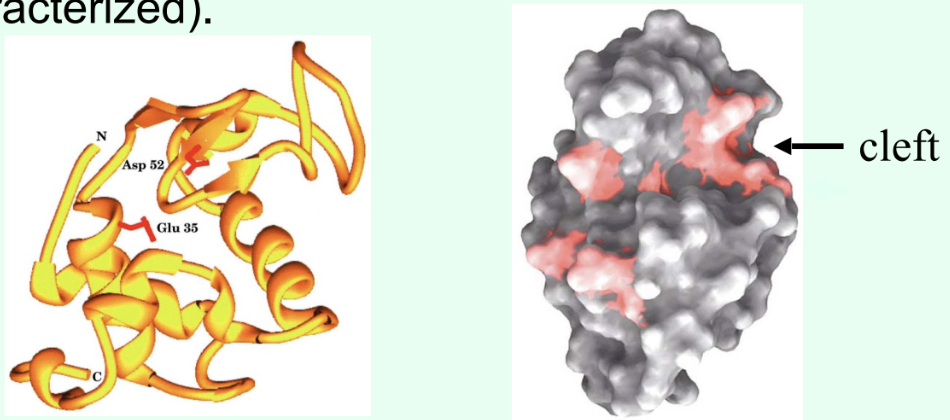
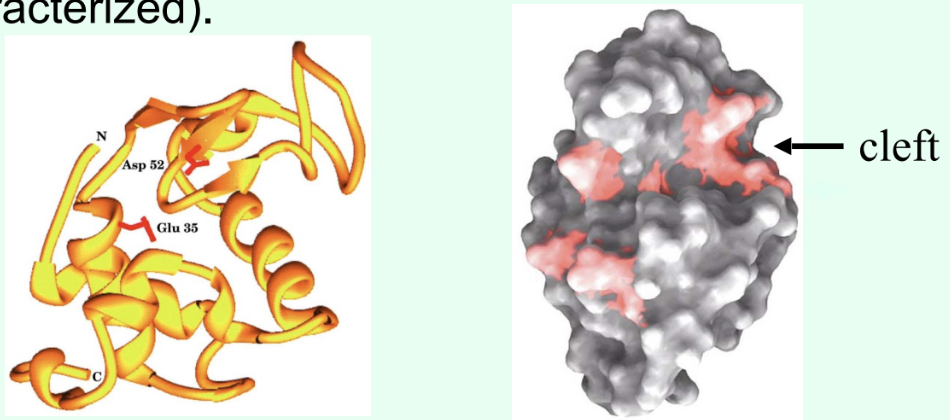
How were catalytic residues in HEWL identified?
By functional testing of specific residues:
Chemical modification of active-site groups → loss of activity
Site-directed mutagenesis (swap one amino acid by changing its codon), then express + assay mutant enzyme
Residues whose alteration dramatically reduces catalysis are catalytic (classically Glu35 & Asp52).
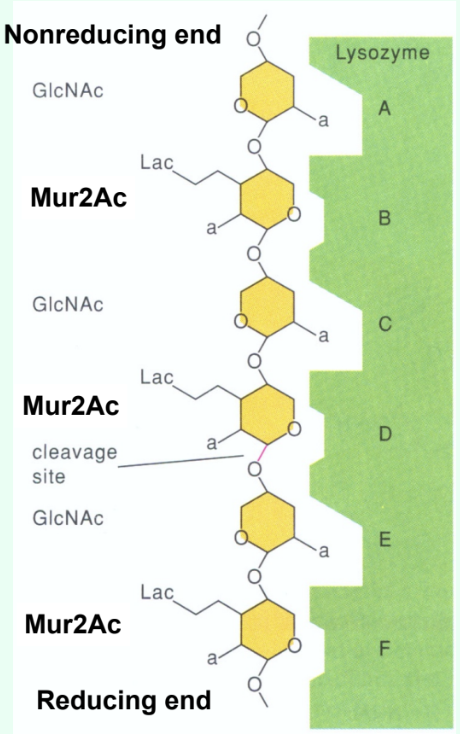
How many sugar-binding subsites does lysozyme have?
Six: A, B, C, D, E, F.
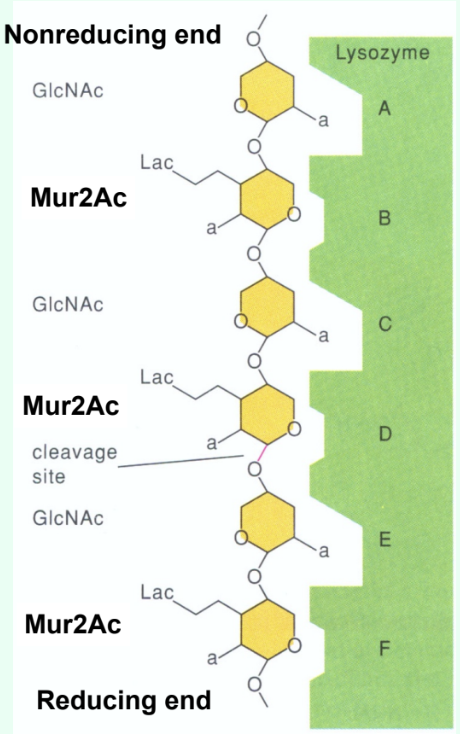
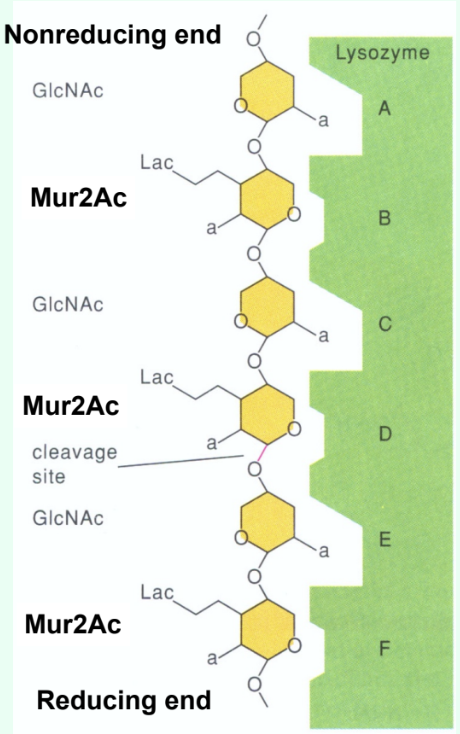
How are short GlcNAc oligosaccharides (<5 residues) hydrolyzed by lysozyme?
They are very slowly (several weeks) hydrolyzed by lysozyme.
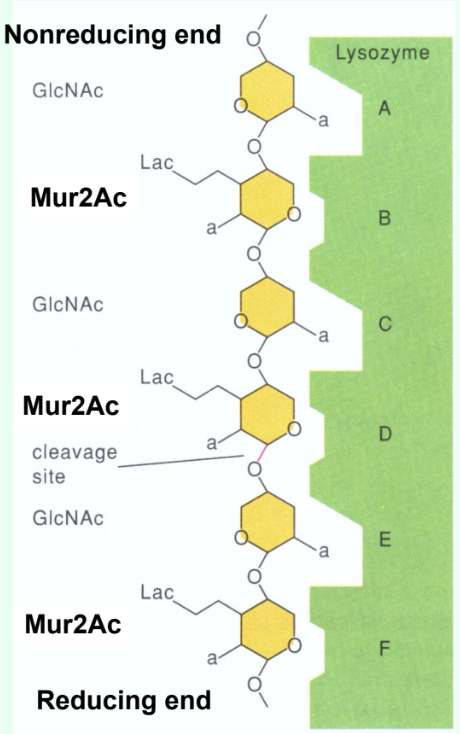
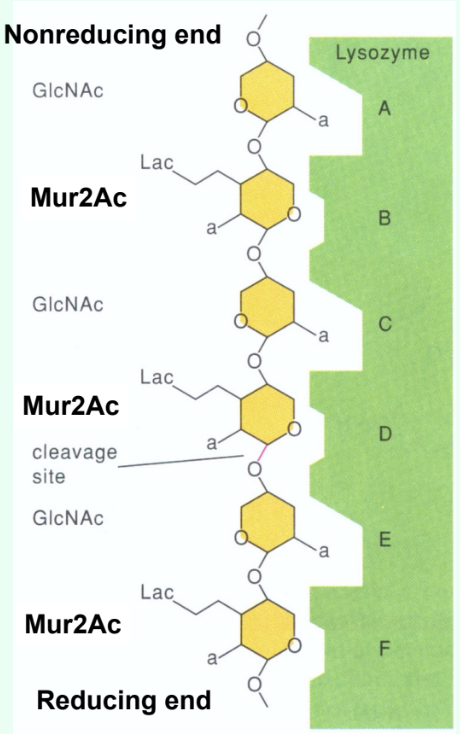
Why do short GlcNAc oligosaccharides inhibit lysozyme?
They bind the active site but react slowly, acting as competitive inhibitors.
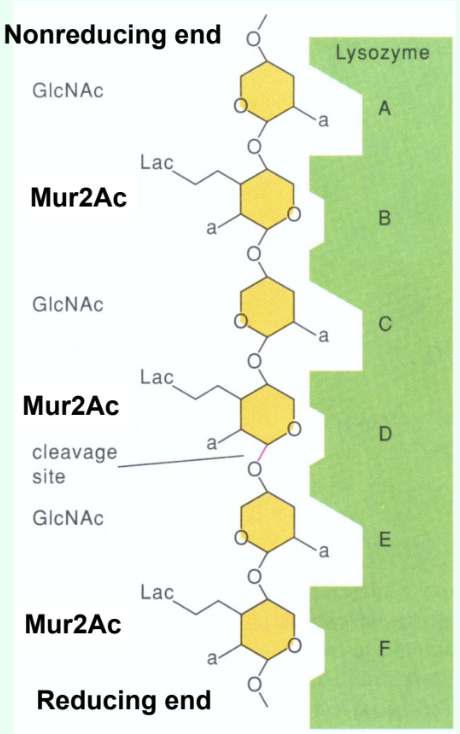
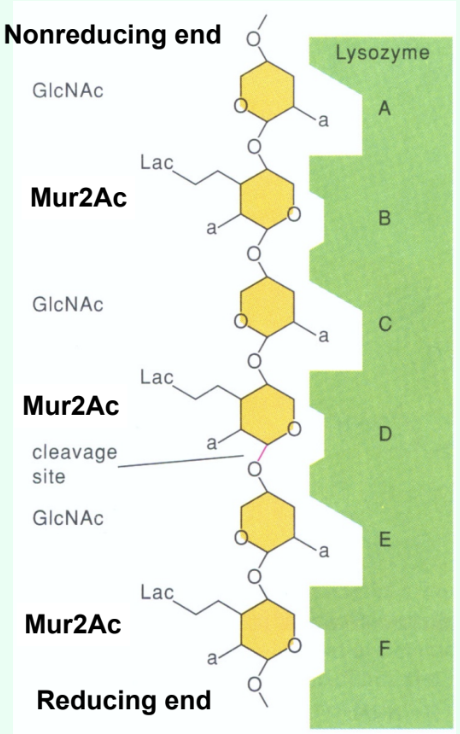
What type of inhibitors do short substrate analogs act as when they bind the lysozyme active site?
Competitive inhibitors.
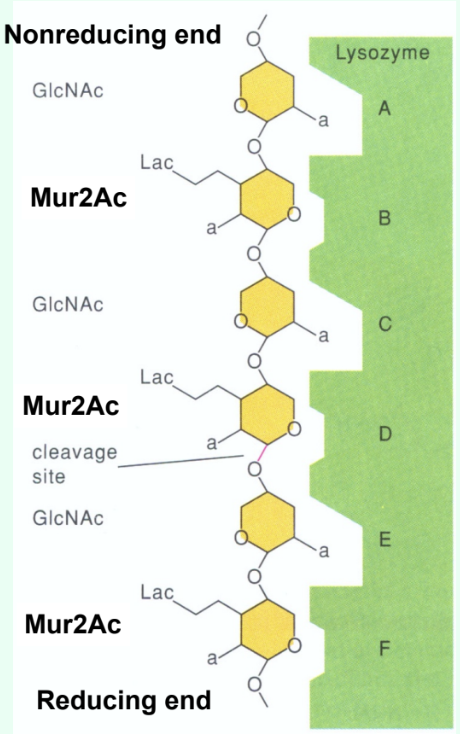

Why is there a large jump in kcat (s-1) between (GlcNAc)4 and (GlcNAc)5?
Due to binding across the D site allows transition-state distortion (half-chair) and proper positioning for cleavage.

What important concept is illustrated by the huge jump in kcat between (GlcNAc)₄ and (GlcNAc)₅?
Lysozyme becomes much more catalytically efficient once the substrate length reaches ~5 sugars.