lecture 14 and 15 - cancer
1/93
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
94 Terms
regulatory genes
regulatory genes control
➢ Genome maintenance and repair
➢ Cell division (mitosis)
➢ Cell fate (differentiation)
➢ Cell survival (metabolism and apoptosis)
Cells have a limited lifespan.
➢ arise from stem cell population
➢ perform a specific job
➢ undergo a tidy packing-up process when they wear out
apoptosis
CANCER CELLS ARISE FROM
DNA damage
cancers are
sporadic
sporadic
the most common form of cancer—comprising 75% to 90% of cases—developing by chance, not through inherited genes, as a result of somatic mutations that accumulate over time.
epigenetic
how your behaviors and environment, such as diet and stress, cause changes that affect the way your genes work without altering the underlying DNA sequence.
Once enough
mutations effect
one cell,
it begins
to change.
Mutations
accumulate
faster and faster.
disrupted pathways
cell fate
cell survival
genome maintenance
cell fate
cell survival
genome maintenance
alterations
point mutations
copy number variants
chromosome rearrangements anupliody
changes in gene expression
point mutations
copy number variants
chromosome rearrangements anupliody
changes in gene expression
symptoms
biomarkers
imaging
mutation detection
Standard somatic cells
Only expresses a standard set of genes
related to the cell’s job
Differentiated to perform a certain job,
takes a standard shape
Anchored, with contact inhibition
(cells don’t divide unless connected)
Damaged cells undergo programmed
cell death (apoptosis)
Cell division tightly regulated or inhibited
cancerous cells
Gene expression patterns change, proteins are mutated, new
proteins appear on the surface
Shape changes, fewer connections to surrounding cells, irregular
growth patterns
Loss of contact inhibition, pressure on surrounding cells, mobility
(cells lose anchors and start moving around– metastasis
Apoptosis mechanisms disabled
Unregulated cell division
What’s the problem?
Cells become less effective at doing their job.
Tissues deform and become less effective at
doing their job.
Rogue cells can start new tumors elsewhere,
disrupting other tissue
CANCEROUS CELLS UNDERGO
UNREGULATED MITOSIS
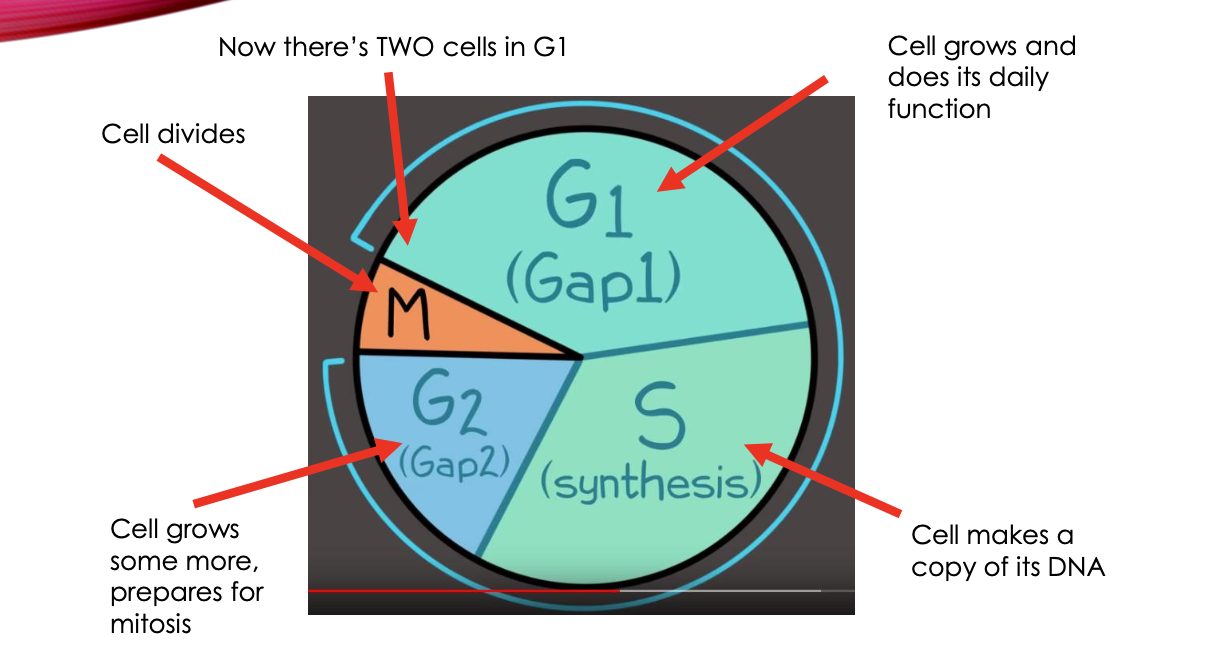
Mitosis
cell cloning
Unlike meiosis, mitosis just duplicates a cell exactly.
The two daughter cells are genetic copies of the parent.
do ALL cells undergo mitosis?
No, most stay in G1 or G0…
think of this as “daily life” phase for cells.
Multiple proteins
(encoded by genes!) keep mitosis under control.
cell cycle
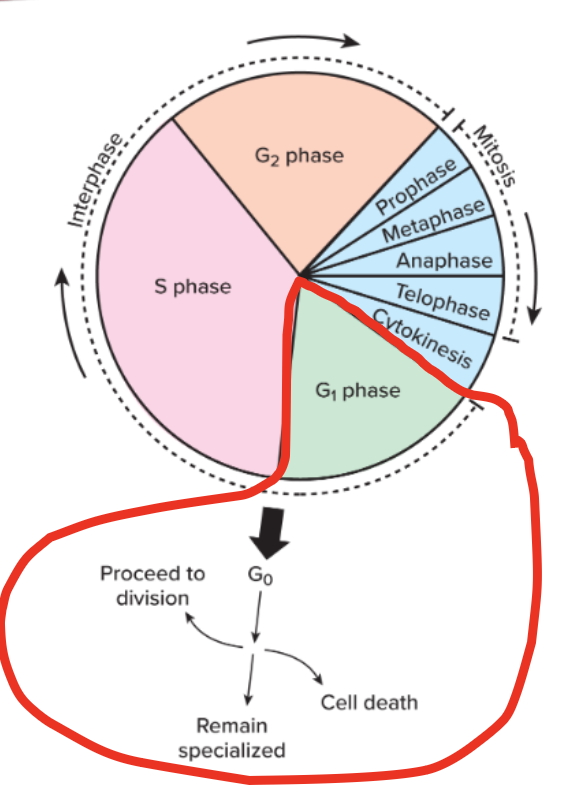
CHECKPOINT PROTEINS PREVENT CELLS
FROM
DIVIDING UNLESS STRICT CONDITIONS ARE MET
Analogy for checkpoints:
gates in a parking lot or at a
train crossing.
❖ Prevent moving forward
unless fee is paid (signal is
received).
❖ Prevent moving forward if
conditions are unsafe
❖ Will never open if you
don’t have the right
permissions!
CELL CYCLE STAGES
CELL CYCLE CHECKPOINTS
Checkpoint 1:
Is there DNA
damage?
(Y/N-
Checkpoint 2:
Is new DNA copy
OK? (Y/N)
Are we big enough
for division? (Y/N)
Checkpoint 3
(metaphase):
Are all our
chromosomes lined
up and correctly
attached to fibers?
(Y/N)
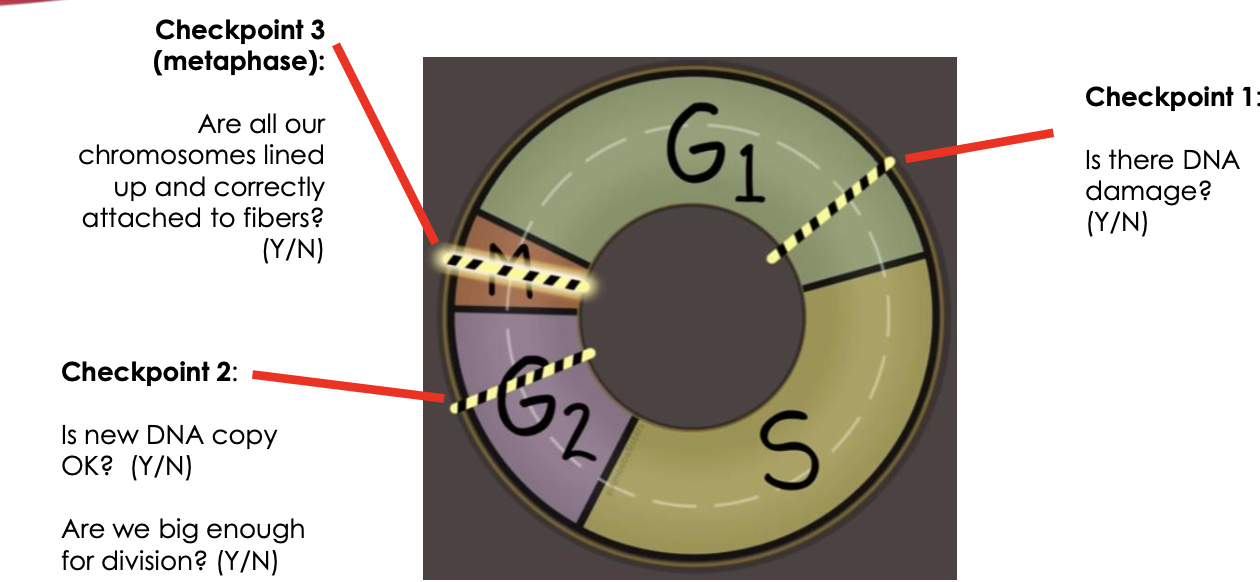
Tumor suppressor proteins at each
checkpoint
prevent the cell from going any
further in the cycle until…
WHAT IF THE ANSWER IS “NO?”
tumor or whatever
…the problem is fixed.
Then the tumor suppressors turn
off, and cell cycle continues
…the problem is
irreparable
Then the tumor suppressors cause
the cell to commit apoptosis
WHAT WOULD HAPPEN IF MITOSIS OCCURRED WITHOUT FIRST
ENSURING DNA WAS PROPERLY COPIED?
A. The cell would divide unevenly
B. The cell couldn’t divide at all
C. The nuclear membrane would prevent chromosomes from moving to
the center
D. Some chromosomes could get missorted (nondisjunction)
E. DNA could be mutated due to mistakes in the DNA synthesis stage (DNA
replication)
Remember how mutations
occur:
❖ DNA template has
chemical damage
❖ DNA polymerase
makes a new strand
with mistakes
❖ Daughter cells now
have new mutations
WHAT WOULD HAPPEN IF MITOSIS KEPT GOING WITHOUT
FIRST ENSURING ALL CHROMOSOMES ARE LINED UP AND
ATTACHED TO SPINDLE FIBERS?
A. The cell would divide unevenly
B. The cell couldn’t divide at all
C. The nuclear membrane would prevent chromosomes from moving to
the center
D. Some chromosomes could get missorted (nondisjunction)
E. DNA would be mutated
GENE EXPRESSION PATTERNS ALSO
CHANGE
Leukemia
affects white blood cells. In leukemia,
useless cancer cells flood the bloodstream and
crowd out the vital red and white blood cells
microarray
measures
the expression of
genes. Each square
represents one gene.
measures if genes are
turned on MORE or LESS
in cancer cells than in
normal cells.
genes in microarray\
This gene is expressed at higher levels in this
cancerous cell than in normal white blood cells.
This gene is expressed at lower levels in this
cancerous cell than in normal white blood cells.
Leukemia affects white blood cells. In leukemia,
useless cancer cells flood the bloodstream and
crowd out the vital red and white blood cells.
If there’s no change, it’s light
pastel
We’re interested in the overall expression
pattern of the chosen genes.
This could indicate dedifferentiation.
It means the cells aren’t making the correct
set of proteins for their specific duties.
dedifferentiation
a transient process by which cells become less specialized and return to an earlier cell state within the same lineage.
Cancer results from
multiple mutations, mostly spontaneous (not inherited),
accumulated over a lifetime
Mutations in cell cycle genes allow unregulated growth
• Mutations in DNA repair genes allow
mutations to accumulate more rapidly
• Mutations in cell-death genes
make the cancer cells harder to kill
• Mutations in cell-cell adhesion genes allow
allow cancer cells to break away and stat new tumors
(metastasis)
• Mutations in metabolism genes allow
cancer cells to steal more resources from the body
Exposure to anything that damages DNA (mutagens)
increases cancer risk
• DNA damage causes mutations
• DNA repair proteins can become overwhelmed and can also become mutated
Untreated cancer is ultimately fatal because
the tumor cells take up resources and do not contribute to keeping the body alive.
• The tumor cells press on and distort surrounding organs– altered structure means they become less
and less able to perform their functions.
• death occurs from organ failure
Everyone’s cancer is unique
Spontaneous mutations are different in each person
• Tumors are heterogenous
• Which means even in the same tumor, different cells have different mutations!
Everyone’s cancer is unique which makes cancer
• hard to detect. We can screen for commonly mutated genes, or other biomarkers, or changes in
overall gene expression.
• Hard to treat with a one-drug-fits-all approach. Treatment uses a variety of approaches:
• Chemotherapy: kill all currently dividing cells (which also kills healthy cells, causing the drastic side effects like
hair loss), encourage apoptosis (cell death)
• Radiation and surgery: remove as many abnormal cells as possible
• New techniques to specifically target tumor cells
MOST INHERITED RISK FACTORS FOR
CANCER ARE
REGULATORY GENES
REGULATORY GENES
control the expression of other genes, acting as "on/off" switches or volume knobs for protein production
You must know the
Example: inheriting mutation X increases risk of
cancer Y by 150% (1.5).
Base rate of getting cancer Y: 0.5% (0.005)
Calculate increased risk:
0.005 x 2.5 (1 + increased risk) = 0.0125
Your new risk of getting cancer Y because of
mutation X: 1.5%
INHERITING ONE BROKEN
ALLELE SPEEDS UP PROCESS OF
CANCER FORMATION
Inherited risk factors are
mutations in tumor
suppressor genes. The second mutation is
much easier to acquire when you already
have one.
Most common: 50% age*
➢ p53 30
➢ BRCA 1 or 2 50s
➢ Rb 1-2
nherited risk factors accoun
for only 5-10% of
cancer cases.
Most do not increase risk this drastically, either.
BOTH NATURAL PROCESSES AND
CARCINOGENS DAMAGE DNA
WHICH CAN
CAUSE NEW MUTATIONS
Natural mutagens:
Oxygen tobacco smoke
Oxygen
X-rays
UV radiation
Chemistry*
Avoidable mutagens
tobacco smoke
industrial chemicals / pollutions
alchohol
OXYGEN CAN CREATE
“FREE RADICALS”
(OR ROS) THAT DAMAGE DNA BASES
If the DNA is DAMAGED
during replication,
the cell
creates a mutation.
The G and C have switched
positions and the code is
changed.
Cells make antioxidants to
help absorb ROS before
they damage DNA.
antioxidants
substances—including vitamins (C, E), minerals (selenium), and enzymes—that protect cells by delaying or preventing damage caused by harmful free radicals (oxidative stress).
These mutations cause point mutations
anywhere in the genome
point mutations
a genetic alteration where a single nucleotide base pair in the DNA sequence is substituted, inserted, or deleted.
UV LIGHT ALTERS DNA STRUCTURE,
CREATING
ABNORMAL CHEMICAL BONDS CALLED
PYRIMIDINE DIMERS
Pyrimidine dimer
Two Ts or two Cs on same strand
form a covalent bond
Sometimes incorrect repair results
in Cs being converted to Ts.
ENZYMES CONSTANTLY SCAN
THE DNA FOR MISTAKES.
MISTAKES ARE FIXED BY CHOPPING OUT A PORTION OF ONE
DNA STRAND AND USING THE OTHER STRAND TO REBUILD IT
These enzymes can fix:
• Mismatches
• Chemically damaged bases
• Thymine dimers
Cannot fix
• Double-stranded backbone
breaks
• Double-stranded backbone
breaks
a severe form of DNA damage occurring when both sugar-phosphate backbones of the DNA helix are severed simultaneously or within close proximity.
CHEMICALS CAN CREATE CROSSLINKS,
ABNORMAL COVALENT BONDS BETWEEN
DNA STRANDS
Nitrogen Mustard ICL
Moiyomycin C ICL
Psoralen ICL
Cisplatin ICL
Trying to separate the
strands can
break the backbone
X-RAYS AND RADIATION CAN BREAK
THE
DNA BACKBONE
Highly energetic particles
rip
through cells like mini
wrecking balls!
If both sides of the DNA
backbone are broken, it’s
harder to fix– why?
CRISPR-CAS9 WORKS BY
CREATING
DELIBERATE DNA BACKBONE BREAKS
(WE CAN CONTROL EXACTLY WHERE)
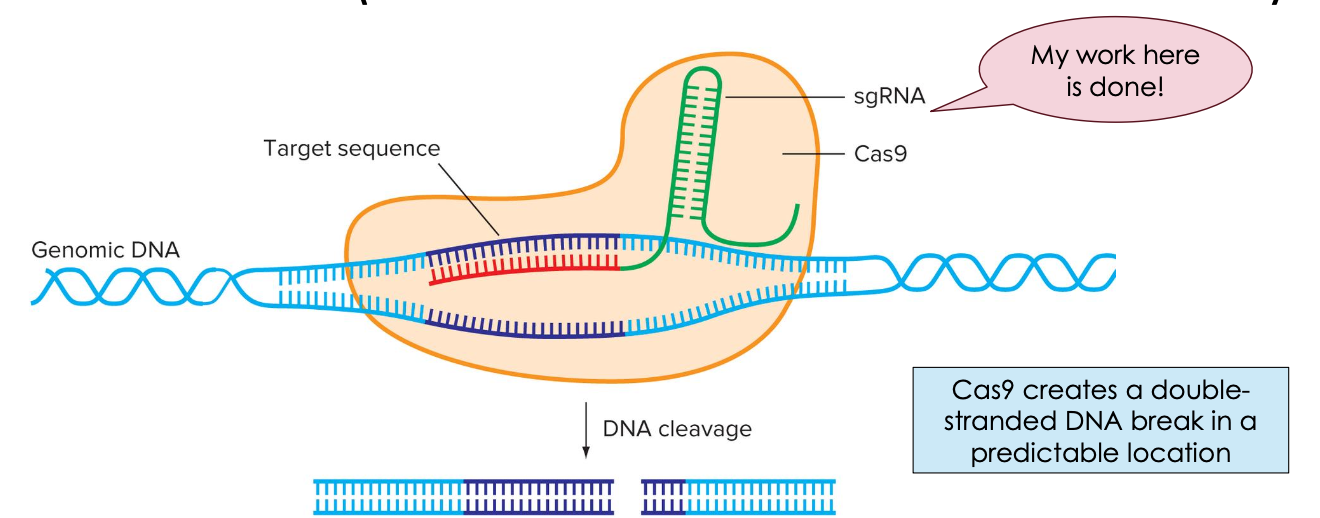
DNA cleavage
the breaking of the covalent sugar-phosphate backbone that links nucleotides in a DNA strand, resulting in smaller fragments
knock in vs knock out in DNA cleavage
Knock-out (KO) and knock-in (KI) are DNA editing techniques that use cleavage to alter genomes. Knock-out uses cleavage to disrupt a gene (loss of function) via error-prone repair, while knock-in uses cleavage to insert specific DNA sequences (gain of function) via precise repair. KO is faster and more efficient, whereas KI is generally more complex.
ZeClinics +3
Provide an intact template with
with the
change we want.
Force cell to use HR to repair to
edit a change into a gene.
example of chemotherapy drugs disrupting the cell
Taxol (paclitaxel)
disrupts spindle
formation.
Anaphase goes
badly wrong!
(some cells “freeze”
in this phase and die)
Cytokinesis in
multiple regions
results in 2+
aneuploid cells
which die
NEWER TECHNIQUES INVOLVE
GENETICALLY
MODIFYING A PATIENT’S CELLS TO HELP THE IMMUNE
SYSTEM DETECT AND FIGHT THE CANCER
Chimeric antigen receptor
CAR cell therapy enables a patients T cells attack cancer cells.
What are environmental factors that can increase your risk of cancer, and why?
Radiation (UV light, X-rays, ionizing radiation): causes DNA strand breaks, pyrimidine dimers, and base modifications that can lead to permanent mutations if not repaired correctly.
Chemical carcinogens (tobacco smoke, asbestos, benzene, aflatoxins): these molecules can directly alkylate or intercalate DNA, causing miscoding during replication.
Viral infections (HPV, Hepatitis B/C, EBV): oncogenic viruses can insert viral DNA near proto-oncogenes, produce proteins that inactivate tumor suppressors (e.g., HPV's E6 protein degrades p53), or drive chronic inflammation that increases replication errors.
Chronic inflammation: increases reactive oxygen species (ROS), which oxidize DNA bases (e.g., 8-oxoguanine), leading to transversion mutations.
Poor diet / obesity: excess calories increase cell proliferation rates and hormonal signaling (e.g., insulin/IGF-1), raising the chance of replication errors.
➢ Why does inheriting mutations in DNA repair genes give you a strong predisposition to developing cancer?
DNA repair genes encode the cellular machinery that fixes replication errors and damage from environmental mutagens. If you inherit a defective copy of a repair gene (e.g., BRCA1/2, MSH2/MLH1 in Lynch syndrome), every cell in your body starts life with already-compromised repair capacity. This means:
Errors that would normally be corrected now persist as permanent mutations.
Mutations accumulate far faster than in someone with normal repair.
The threshold for hitting the critical set of driver mutations needed for cancer is reached much sooner and more easily.
You don't need an environmental "first hit" to disable repair — it's already impaired from birth.
This is why BRCA1/2 carriers have dramatically elevated lifetime risks of breast and ovarian cancer.
➢ Why would inheriting a mutation in a tumor suppressor gene like p53 or Rb give
you a strong predisposition to developing cancer?
Tumor suppressor genes act as brakes on cell division. Most cells carry two functional copies (two alleles). Under Knudson's "two-hit hypothesis", both copies must be inactivated to lose suppressor function.
If you inherit one mutant copy (one hit already present in every cell):
You only need one somatic mutation in any susceptible cell to completely lose function, versus two in the general population.
The probability of that second hit occurring in at least one cell over a lifetime is very high.
p53 ("guardian of the genome") normally halts the cell cycle and triggers apoptosis in response to DNA damage — lose it, and damaged cells divide unchecked.
Rb normally prevents the cell cycle from advancing past G1 — lose it, and cells proliferate even without proper growth signals.
This is why Li-Fraumeni syndrome (inherited p53 mutation) and familial retinoblastoma (inherited Rb mutation) cause very early-onset, multiple cancers.
➢ How many cancers (%) are caused by mutations that are NOT inherited, but accumulate over the course of a lifetime?
Approximately ~90–95% of all cancers are caused by somatic mutations — mutations that are not inherited, but accumulate over a lifetime in individual cells due to replication errors, environmental exposures, and random stochastic events. Only ~5–10% of cancers are strongly linked to inherited germline mutations.
➢ Why are all cancers genetically different?
Each cancer arises from a unique cell that accumulated a unique sequence of mutations over time, influenced by:
The cell type of origin (a lung epithelial cell vs. a B-lymphocyte have different baseline gene expression and vulnerabilities).
The specific mutagens the individual was exposed to (e.g., tobacco causes C→A transversions; UV causes C→T transitions).
Random replication errors — DNA polymerase makes ~1 error per 10⁹ bp, and which specific bases are hit is stochastic.
The order in which mutations accumulate alters which subsequent mutations are selected for.
Because the path to cancer involves hitting multiple driver genes through random processes in a unique individual environment, no two cancers will ever have exactly the same mutational profile.
➢ Why are cancer cells in the same tumor genetically different?
The founding tumor cell continues to divide and accumulate new mutations after transformation.
Cancer cells have increased genomic instability (often due to defective repair or checkpoint genes), so daughter cells mutate at higher-than-normal rates.
Different subclones within the tumor evolve independently through clonal evolution and natural selection — some subclones may acquire resistance to therapy, faster proliferation, or invasive capacity.
Spatial differences in the tumor microenvironment (oxygen, nutrients, immune pressure) apply different selective pressures on different regions of the tumor.
This heterogeneity is clinically important because it means a biopsy from one region may not represent the whole tumor, and it underlies the development of drug resistance.