Immunology exam 3 Cancer Immunology
1/55
Earn XP
Description and Tags
all done!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Necessary characteristics of cancer cells (7)
They stimulate their own growth, they ignore growth-inhibiting signals, they avoid death by apoptosis, they develop a blood supply (angiogenesis), they leave their site of origin to invade other tissue (metastasis), they replicated constantly to expand their numbers, they evade and outrun the immune response
angiogenesis definition
the physiological process through which new blood vessels form from pre-existing vessels, cancer cells form their own tissue essentially
4 steps of cancer growth + descriptions (1, 3, 2, 2)
initiation (mutational event), cancer progression (mutation and genome destabilization, dysregulation of growth control pathways), evasion of cancer cell elimination (apoptosis and killing by T cells blocked), tumor growth and dispersal (angiogenesis, metastasis)
how many mutations do you need for cancer
many!! just one usually won’t lead to cancer yet, usually 4-6 needed
how do you explain the statement: cancer is a genetic disease
DNA damage causes cancer
what cells are at greatest risk for cancer?
Epithelial cells: they are exposed to the most radiation (eg uv radiation from the sun), also cells that divide a lot
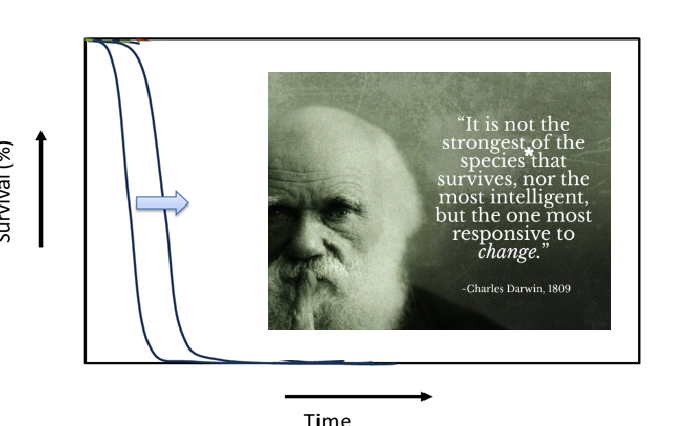
What does this image show?
Typical response to cancer therapy with metastatic cancer, at most life is prolonged a little bit but survival rate doesn't go up overall, graph just shifts to the right and not up
how has the BCG vaccine been used in cancer treatment
already in 1970s, found to help prognosis with melanoma and bladder cancer
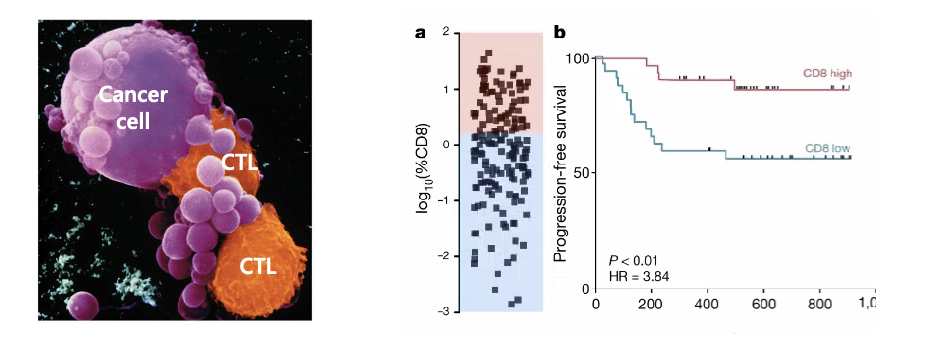
What does the image show?
CD8 T cell infiltration led to a more favorable prognosis
explain the statement “all cancer therapy is immunotherapy”
immune system always needed in fighting cancer, eg chemo causes cell death which alerts the immune system → helps fight the cancer off
three features that make the immune system suited to fight cancer
Adaptive, access (can be anywhere in your blood), memory (will remember cancer over time)
what makes CD8 T cells special with fighting cancer
they can see inside cells (with MHC-I) (bc initially meant to fight viruses), where cancer peptide most common
how do you get neo-antigens
Mutated genes → mutated proteins → neo-antigens
in what way do T cells kill?
with great precision - granzymes directed directly at cell it recognizes
requirements of effective T cell immunity against cancer (4)
Recognition of tumor antigen, activation, migration, effector function
3 examples of viral tumor antigens
papillomavirus - associated with uterine cancer, Hep B - liver cancer, EBV - some lymphomas
what different tumor antigens are there?
neo-antigens (tumor specific antigens)- presentation of mutant peptide from mutant cellular protein,
tumor associated antigens: reactivation of embyonic genes not normally expressed in the differentiated cell; overexpression of self-protein by a tumor cell increases self-peptide presentation and recognition by T cells
what does initiation of an immune response require?
inflammation (danger)
how do you get a T cell response against cancer? (3, 3, 2, 3) steps
Inflammatory inducers (bacterial lipopolysaccharides, ATP, urate crystals) → sensor cells (macrophages, neutrophils, dendritic cells) → mediators (cytokines, cytotoxicity) → target tissue (production of antimicrobial proteins, induction of intracellular antiviral proteins, killing of infected cells)
PAMP long, function, issue with cancer
Pathogen associated molecular pattern, trigger inflammation by inducing co-stimulation on DC, usually non-existent in cancer
DAMP long
Damage associated molecular pattern, when tissue homeostasis is lost, often due to a lot of necrotic cell death
what triggers IFN-I
DAMPs and PAMPs
what does the initiation of the T cell response require?
Professional APCs-dendritic cells or macrophages
steps in induction T cell response against cancer (3)
Tumor with dendritic cells, DCs activate T cells in lymph node, T cells migrate to the tumor and kill the tumor cells
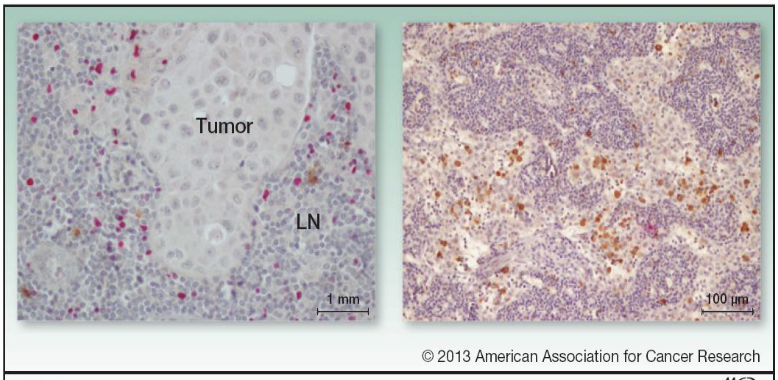
What are these, explain
left: cold tumor- very little inflammation, difficult for immune cells to penetrate
right: hot tumor- inflamed
3 options for immune penetration in cancer + descr.
immune inflamed- many CD8 T cells in tumor
immune excluded- CD8 T cells only at border, not inside- very bad for prognosis
immune dessert- no CD8 T cells at all
reasons why there is immune exclusion in a tumor (5)
Stromal barriers, aberrant vasculature, lack of chemokines (often genes producing them supressed), oncogenic pathways, hypoxia (lack of oxygen)
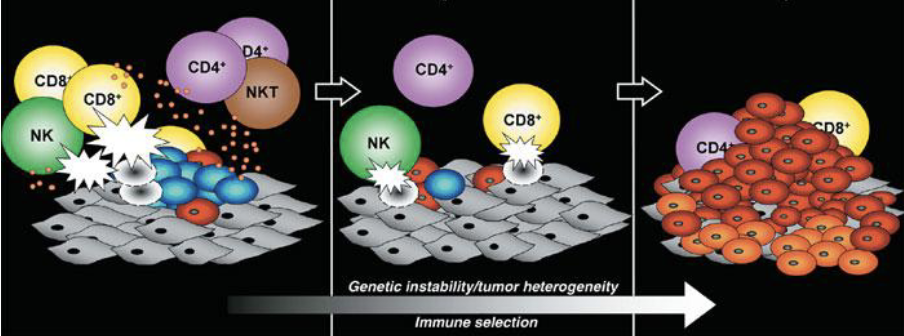
What does this image show?
The 3 E’s of immunoediting: elimination, equilibrium, escape
two methods of escape + example
Escape by loss of tumor antigen expression, eg loss of MART-1 which is normally expressed in melanocytes
escape by HLA loss in cancer - mutations in TAP common, then can be undetected by T cells but still detectable by NK cells
why do T cells need inflammation?
They do not inherently know if a peptide is a self antigen or a viral peptide so they need inflammation to know to be activated
HARD MHC-1 lesion
Genetic alterations leading to permanent loss
SOFT MHC-I lesions
Are reversible, eg by IFNγ stimulation
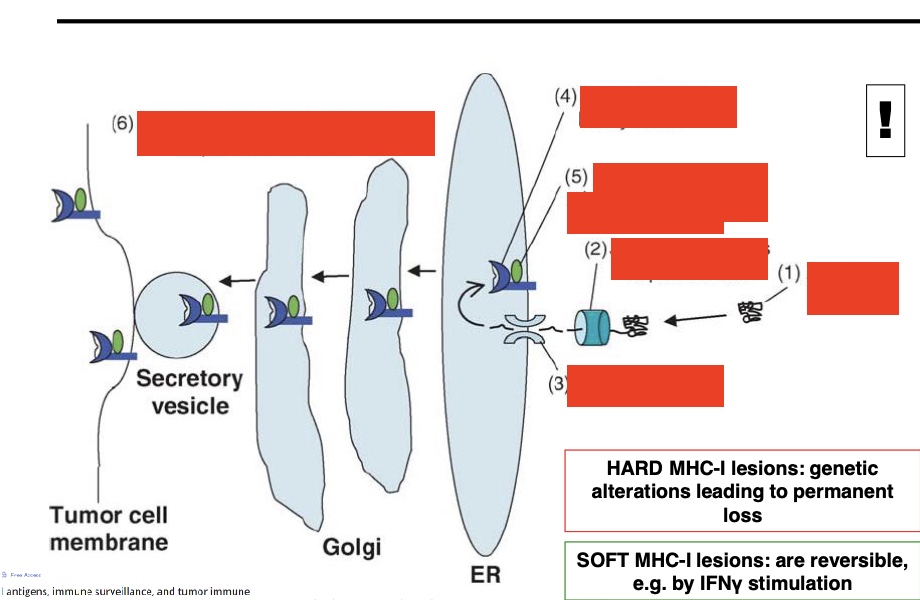
label- how HLA-I loss happens in cancer
antigen loss, decrease of a subunit of proteosome, decreased TAP, loss of MHC-I heavy chain, loss of β2 microglobulin, diminished transport of MHC-I-pep complexes to cell surface
forms of T cell tolerance (4)
Regulatory T cells, exhaustion, TAMs, MDSC
Tregs function
Inhibit responses to self/innocuous antigens
what is Treg abundance correlated with?
Poor cancer prognosis in many different cancers
when naive T cells are activated by DCs to be differentiated in cancer what is good news and what is bad news
Good news: Th1
Bad news: Th2, Treg (!!), Th17
what is an important cause of therapy resistance
Subverted myeloid cells (MDSC, M2 macrophages) release pro-angiogenic and tumor invasion promoting factors
how to tumors create an inhospitable metabolic tumor environment
Hypoxia (low O2), increased lactate and acidification, etc
what does exhaustion mean for T cells?
Loss of effector function/proliferation, sustained expression of inhibitory receptors
What is another way of looking at T cell exhaustion?
Tolerance mechanism, if an antigen doesn't go away for significant periods of time is beneficial for the host to tolerate it to prevent chronic inflammation, this is very beneficial for cancers to keep growing
conclusions of cancer immunity (6)
Many cancers or immunogenic, tumor antigens/neo-antigens, adaptibility is an immune response asset, immune editing (loss of antigen/HLA), tolerance mechanisms (Tregs, MDSC, TAM; T cell exhaustion), inhospitable tumor environment
what are PD-1 and CTLA4?
inhibitory receptors- immune checkpoints, if they are inhibited then T cells can be activated more
what is on the intracellular part of PD1
ITIM- Immunoreceptor tyrosine-based inhibitory motif, it is the intracellular tail
what do immune checkpoint inhibitors do to the survival curve of cancer
they bring it up finally!! people survive and stay cured rather than just living for a few more months
why does immunotherapy not always work?
there are many inhibitor receptors, only a few are blocked at a time, other still exist
role of antigens in immune inhibition + example
responsiveness to checkpoint inhibition correlates with mutational load- eg in lung cancer cause by smoking where many more mutations, easier to treat than not cause more different to healthy body cells
negative side effects of immune checkpoint inhibition
autoimmunity- can cause severe side effects like retina can become inflamed and patient loses sight
TIL long, description
Tumor infiltrating lymphocytes- objective clinal response (70%), long term remission (20%), take T cells out of tumors, replicated them in vitro, put them back in patient to overwhelm the tumor, helps even if checkpoint therapy has failed
disadvantages of TIL
patient specific- has to be patients own T cells- laborious + expensive
BITE description + advantages
more universal therapy approach, activates all T cells in tumors, make recombinant protein that attaches on target monoclonal antibody and then connects to TCR- makes target cell activate T cell, cheaper than TIL, don’t need antigen specific T cells, HLA loss doesnt matter
BITE disadvantages
T regs can still inhibit them, doesnt work on T cell excluded tumors
CAR T cells long
chimeric antigen receptor
CAR T cell description
antibodies bind diff types of antigens than TCR & can be generated against any protein- then you can fuse an Ab and TCR to create the CAR against a target protein
where has CAR t cell therapy had success?
with B cell leukemia- does kill all B cells but you can live without them with just having IGIVs, was with patients on their death bed
new therapies against cancer (4)
checkpoint inhibition, TIL therapy, BITES, CAR T cells