Cerebral Blood Flow and Metabolism
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
[STATs] Cerebral energy metabolism
STATs
High rate of energy utilization
ATP is generated through oxidative phosphorylation
Uses 20% of resting blood oxygen
Main fuel is blood glucose, very limited beta-oxidation
Uses 50% of resting blood glucose
Very little glycogen storage
Describe the relationship between CBF (cerebral blood flow) and CPP
What are the 3 mechanisms could contribute to CBF regulation
CBF regulations:
Cerebral pressure autoregulation
Flow-metabolism coupling
Neurogenic regulation

Describe Cerebral pressure autoregulation
Maintains?
Mech
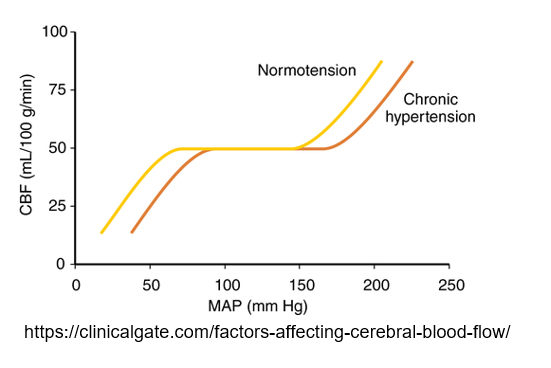
Cerebral pressure autoregulation
maintains constant CBF in the ~70-150 mm Hg MAP range.
Mech:
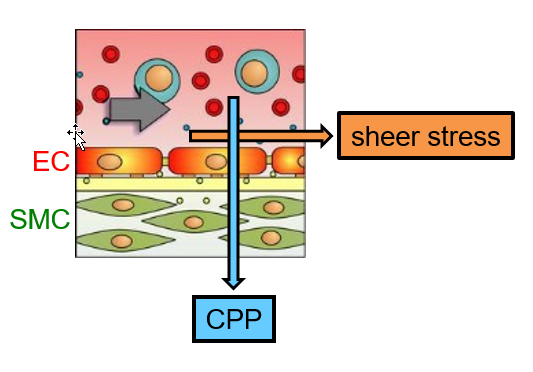
SMC + EC → "mechanical sensors" for CPP + Sheer Stress Sensor
Increases = Vasoconstriction → Increases CVR → Reduces CBF
Draw out the graph depicting MAP vs CBF
List the Flow-Metabolism Coupling
How does Neuronal Activity lead to increasing CBF
Increased neuronal Metabolism?
What is the Result of these things on Vasculature:
Low Blood/Tissue pO2:
Respiratory Acidosis/Alkalosis:
Flow-Metabolism Coupling
Increased Neuronal Activity:
→ increased metabolism → Increases CBF
→ increases Extracellular K+ + adenosine → Vasodilation
Increased Neuronal Metabolism:
→ Increases CO2 → H+ → Vasodilation (increases CBF)
Low Blood/Tissue pO2:
→ Vasodialtion
B/C release of K+, adenosine and NO
Respiratory Acidosis/Alkalosis:
Acidosis: → vasodilation
REMINDER: hypoventillation, high pCO2
Alkalosis: → Vasoconstriction
REMINDER: hyperventillation, low pCO2
Describe Neurogenic regulation of CBF
Extrinsic vs Intrinsic
Mech of intrinsic?
Extrinsic innervation
Para/sympathetic regulation of CBF in vessels outside of the brain parenchyma.
Intrinsic Innervation:
CBF Regulation in vessels inside brain parenchyma.
via astrocyte end-feet (most likely)
Mech: K+ release/prostaglandins /NO synthesis
(due to increased [Ca2+]i) by the astrocyte.
Describe the Main utilization of ATP in the nervous system
Compare/Contrast Kinesins/Dyneins
ATP Utilization in NS:
Maintaining ion gradients (e.g. Na-K ATPase).
Biosynthetic pathways
Axonal transport or organelles, proteins, mRNA
Kinesins/Dyneins:
Both: motor proteins that hydrolyze ATP to move cargo along the microtubules of axons.
Kinesin:
Anterograde motility
Supply of new components by fast/slow axonal transport
Dynein:
Retrograde:
Retrograde survival signaling and degradative traffic
Describe ATP generation from glucose in the brain
Glut1 vs 3
Additional source of energy?
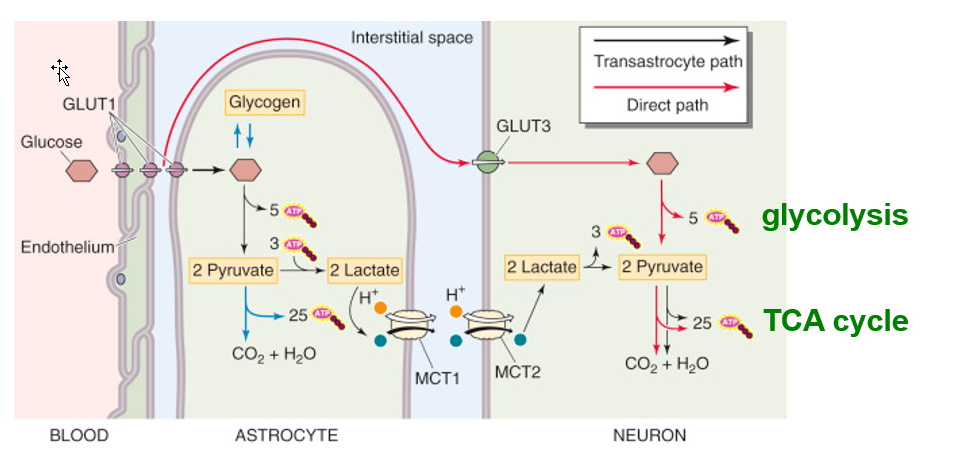
ATP generation from glucose in the brain
GLUT1: glucose → BBB
GLUT3: Glucose → Neurons
Additional Source:
From Lactate produced by astrocytes from blood glucose or glycogen.
Explain how fats can be used to fuel the brain
Nonessential FA cannot pass BBB:
Nonessential FA → oxidized in liver → acetyl-CoA converted to ketone bodies → circulation.
MCT1: Ketone →BBB
MCT2: Ketone → Neurons
NOTE: MCT = Monocarboxylate transporter
Explain how these can occur → Energy deficiency in brain:
Insufficient oxygen delivery
Lack of energy sources
Suboptimal energy generating pathways
Insufficient oxygen delivery
Vasovagal syncope,
head trauma
stroke
myocardial infarction
pulmonary embolism
high altitudes
Lack of energy sources
Hypoketotic hypoglycemia – b-oxidation deficiencies
GLUT1 deficiency – low glucose uptake by the brain
Suboptimal energy generating pathways
Vitamin B1 deficiency
Wernicke-Korsakoff syndrome in chronic alcoholics (ATP generation from glucose is inefficient).
Describe Cerebral Lipid Metabolism:
What cannot pass BBB
What does the brain synthesize?
How do essential FAs taken up?
What does the brain uses Fatty acids for? Describe this thing
Describe Cerebral cholesterol metabolism
STATs about ChL and Brain
What cells can synthesize ChL?
What can provide ChL?
What does ChL synthesis Drive?
Cerebral Lipid Metabolism
Cannot pass BBB: Nonessential fatty acids and ChL
brain synthesizes most of its own fatty acids and cholesterol.
Essential fatty acids (provided in diet) are taken up by specific transporters in the brain.
Fatty acids → membrane lipids.
very long chain (C>22) and branched chain fatty acids in membrane lipids.
Cerebral cholesterol metabolism
~20% of the body's cholesterol are in the brain.
~70-80% in myelin.
Synthesis of ChL:
Neurons and glial cells
What can provide ChL:
Astrocytes via ApoE-cholesterol-phospholipid complexes
What ChL synthesis drive:
ChL synthesis in oligodendrocytes drives myelination in early life.
Describe Niemann-Pick disease type C
Normal Physiology?
Pathophysiology?
Consequence?
Niemann-Pick disease type C
Normal:
Astrocytes generates ApoE-cholesterol lipoproteins
→ Neurons Endocytosis → ChL removed in lysosomes → ER/Plasma Membrane
Pathophysiology:
Defect in removal process → ChL accumulation in lysosomes.
Consequence:
inhibits ganglioside degradation in neuron
Leads to secondary ganglioside accumulation
Leads to Neurodegen.
Describe Myelin Formation
describe the myelin Sheet
Differentaite between Oligodendrocytes/Schwann
The myelin sheet is a multilayer cell membrane around the axon
Oligo vs Schwann:
Oligodendrocytes:
myelinate multiple axons
in CNS.
Schwann cells:
myelinate only one axon
PNS.
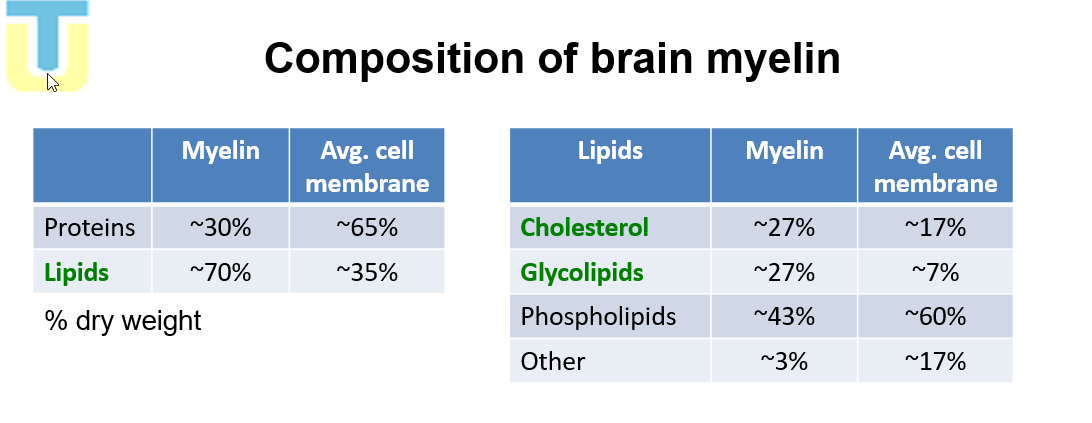
Describe the composition of Brain Myelin compared to Avg. cell membrane
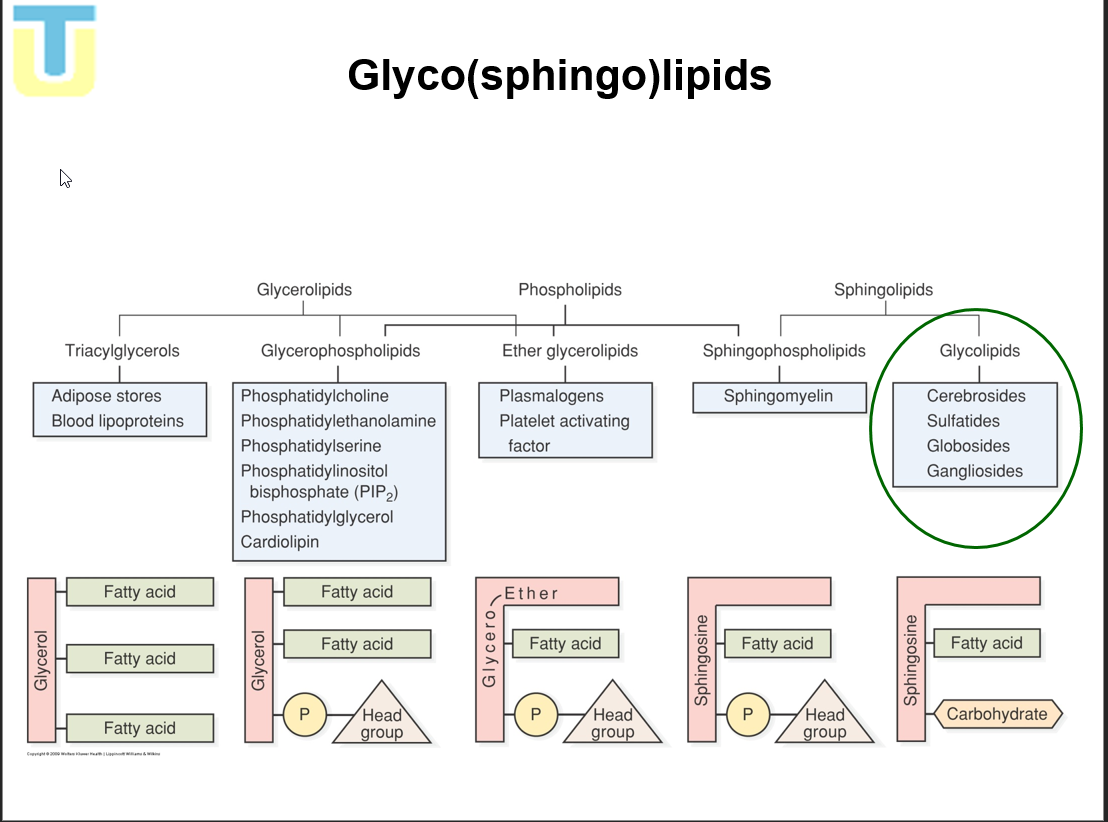
[REVIEW] different types of Lipids
What are the 3 structural components of glycolipids?
Glycolipids have 3 structural components
FA chain
Carbohydrate
sphingosine (an amino alcohol)


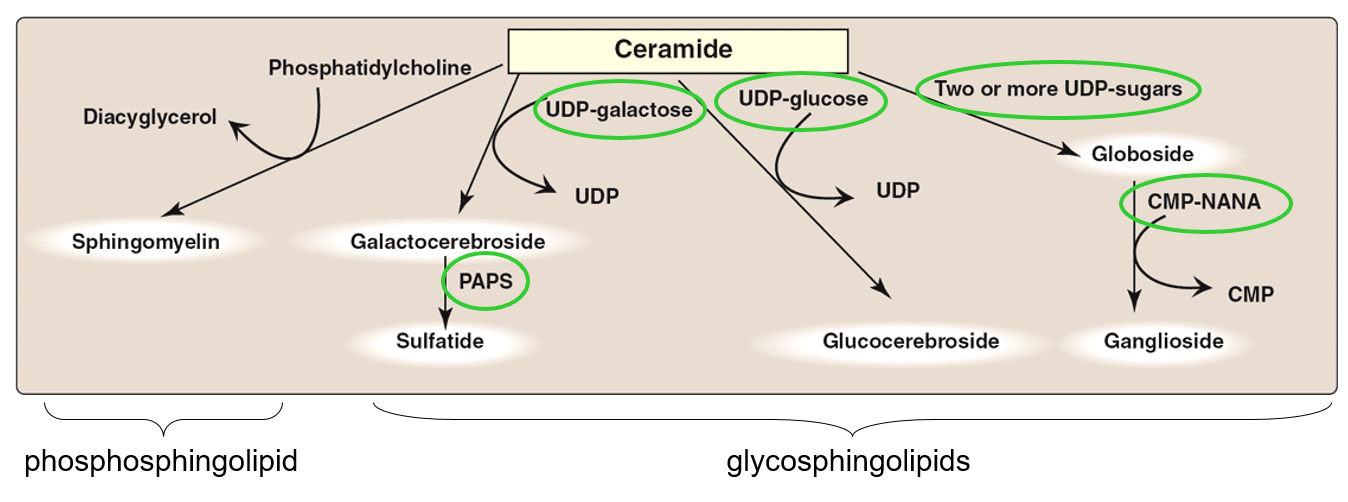
Draw out the synthesis of glycosphingolipids
Where does sugar add. start/completed?
What form does the sugar have to be in?
What is the sulfate donor?
Sugar addition starts in the ER and completed in the Golgi.
Sugars have to be in a nucleotide-activated form to be added to the molecules.
PAPS is the sulfate donor.
Describe Galactocerebrosides and sulfatides
Function?
Metabolic Deficiencies →?
Describe Gangliosides
Enriched?
Deficiencies →?
Clinical Importance?
Galactocerebrosides and sulfatides
Function:
main glycolipids in myelin sheet
Help stabilize paranodal junctions
(the regions adjacent to the Nodes of Ranvier)
Metabolic deficiencies → suboptimal myelination or demyelination.
Gangliosides
Enriched in the neuron plasma membranes (10-12% of lipids)
Metabolic deficiencies → neurodegeneration
(usually without myelination problems).
Clinical Importance:
Autoantigens in certain autoimmune neuropathies.
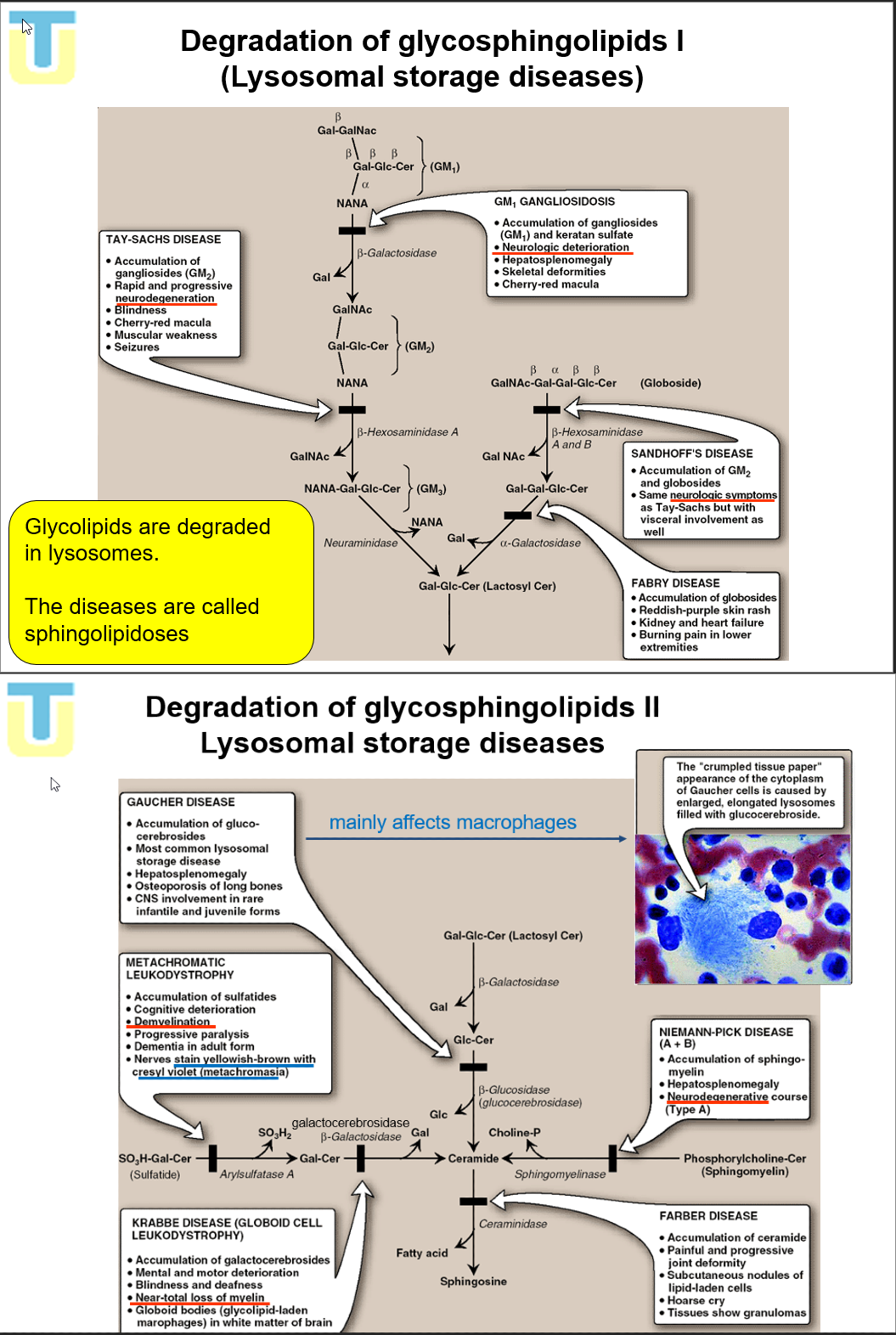
Draw out Degradation of glycosphingolipids II and associated disease
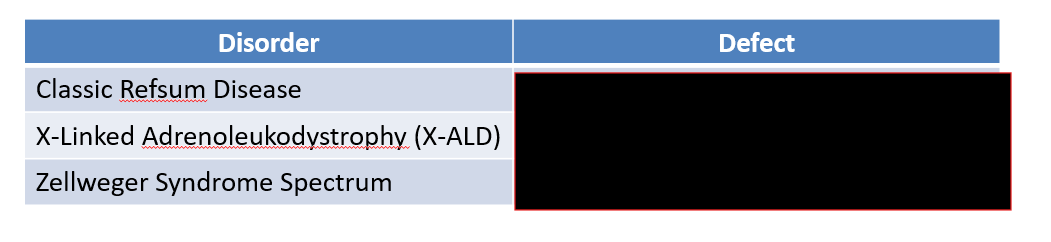
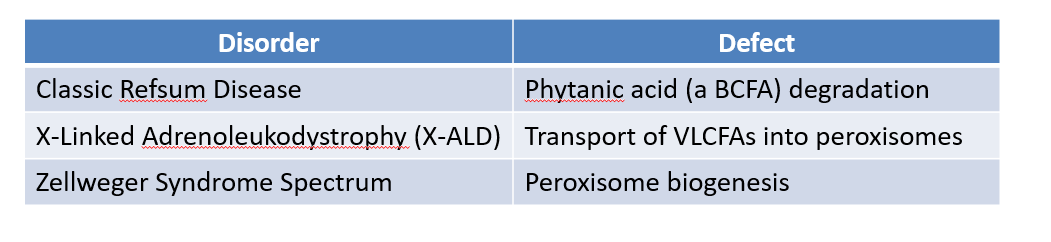
Describe the Peroxisomes:
Function?
Relation to CNS?
Defect?
Peroxisomes:
Function:
Metabolizes very long chain fatty acids (VLCFAs, C>22) and branched chain fatty acids (BCFAs).
CNS membranes uses these
Defect → neurological impairment.
Describe Cerebral nitrogen metabolism
Non vs Essential AA:
Glutamine Metabolism:
Glutamine synthetase
Non vs Essential AA:
Essential: Transported through BBB
Non: LImiged Transport (except arginine).
Glutamine Metabolism:
Brain = net glutamine exporter
due to extensive glutamine synthesis by astrocytes
Glutamine synthetase reduces the levels of toxic ammonia in the brain.
Describe the AA transporters
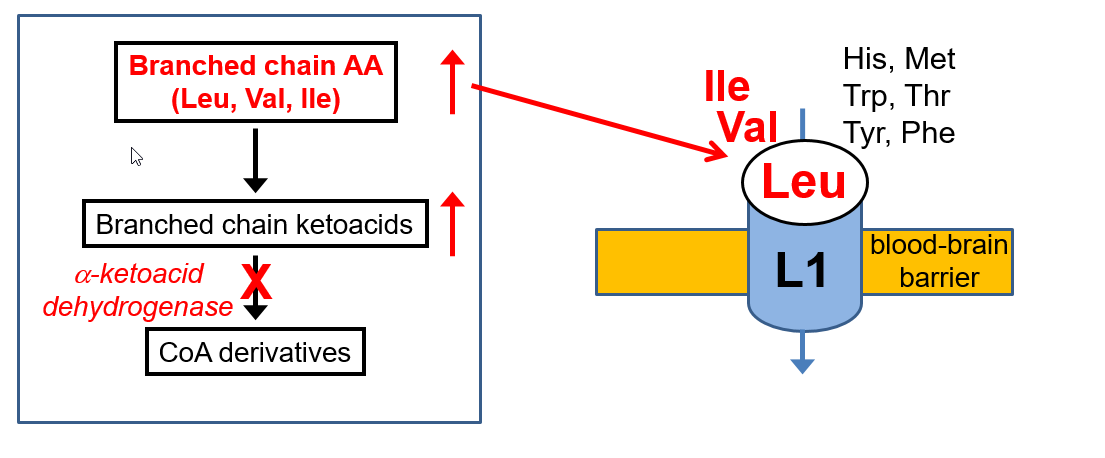
L1 (Leucine-preferred) transporter
y+ transporter:
Na+-dependent transport
L1 (Leucine-preferred) transporter
moves large neutral amino acids (LNAAs) → brain
(Leu, Val, Ile, His, Phe, Tyr, Trp, Met, Thr).
y+ transporter:
moves cationic amino acids → brain
(Lys, Arg)
Na+-dependent transport
Glutamine (Gln) leaves the brain ECF
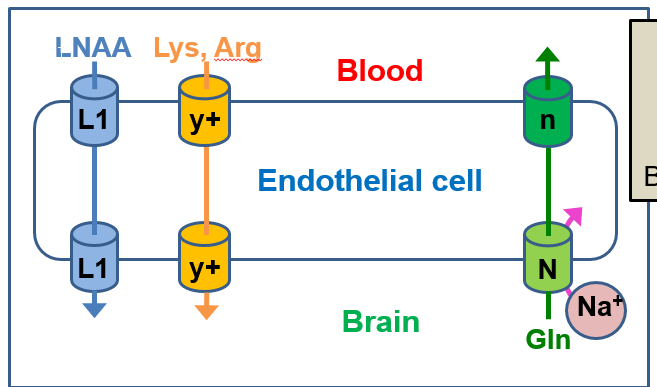
Compare and Contrast BRAIN ECF vs Blood contents

Describe the relationship between L1 and maple syrup urine disease and consequences
High Blood levels of Leu → occupy L1 → other LNAA going in
Low His, Trp and Tyr → compromise NT synthesis → severe CNS dysfunction