enthalpies of solution and entropy
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
what is the enthalpy of solution (ΔHsol)? give an example eqn and state whether it is exo or endo:
ΔH when 1 mole of an ionic substance dissolves in enough solvent to form an infinitely dilute soln
NaCl (s) → Na+(aq) + Cl-(aq)
varies :(
give the general cycle for the calculation of an enthalpy of solution:
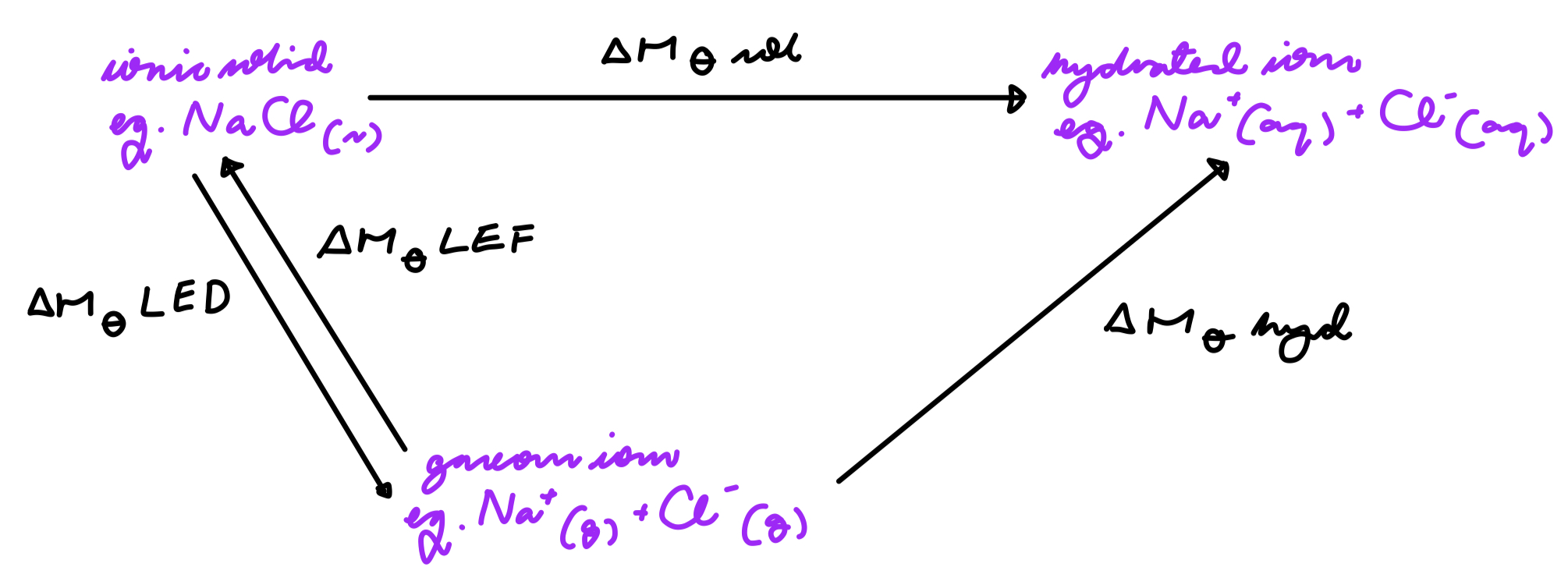
give the eqn for the calculation of the enthalpy of solution:
enthalpy of solution = enthalpy of lattice dissociation + enthalpy of hydration
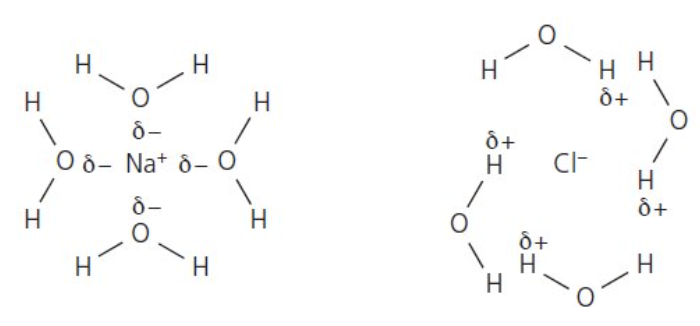
describe what happens when ionic compounds dissolve in water:
polar water molecules attracted to charged ions
δ- O atoms in H2O molecules are attracted to the +ve ions and the δ+ H atoms in H2O molecules are attracted to the -ve ions
why are enthalpy of hydration values exothermic?
E released when ions are attracted to polar water molecules
why is the enthalpy of hydration of F- more exo than the enthalpy of hydration for Cl-?
smaller ion w/ same charge has a higher charge density
so -ve charge on ion attracts δ+ H in H2O more strongly
what is entropy change? give its symbol and units:
measure of disorder - the greater the disorder, the greater the entropy
ΔS
J K-1 mol-1
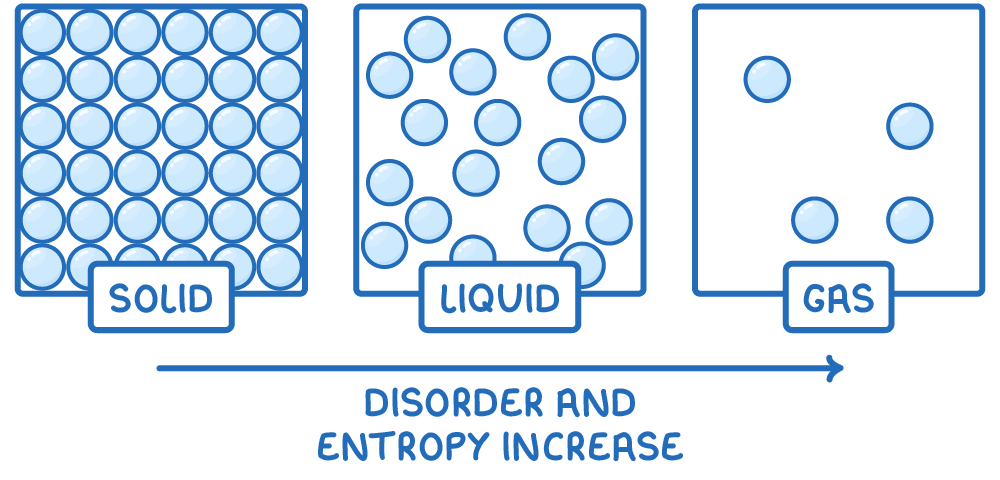
generally, how does the entropy of a solid compare to that of a liquid or gas?
S of solid > S of liquid > S of gas
is the entropy value for ionic, metallic and macromolecular structures typically high or low? why?
low
have regular structures
is the entropy value for simple molecular substances high or low? why?
high - gases and liquids less ordered
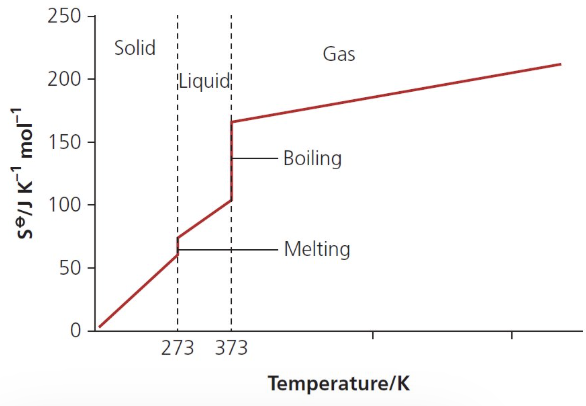
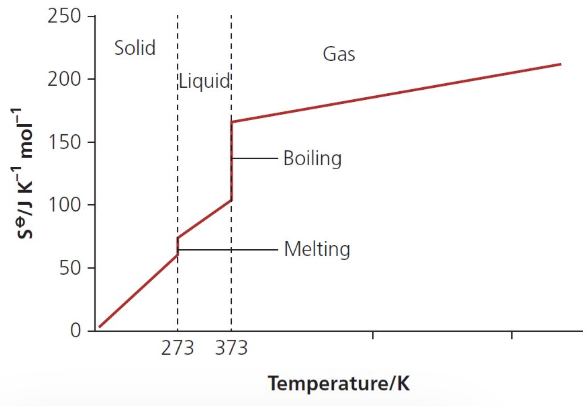
describe and explain the shape of this graph:
at 0K, the S of a perfect crystalline substance is 0
graph shows a general increase in S as temperature increases
w/in a solid, S increases w/ temp as particles gain E and vibrate more
as solid melts and forms a liquid, there is a large increase in S at the mpt
there is a very large increase in S at the bpt as gases are very disordered
give the eqn used to calculate the entropy change of a reaction:
ΔSθ = ΣSθproducts - ΣSθreactants - take molar ratios into account
how can we predict the entropy change of a system?
increase (+ve):
decomposition reactions
increase in moles of gas
overall no. of particles increases
(very situationist)
describe the chelate effect:
bidentate/multidentate ligands replace monodentate ligands to form a more stable complex
because there is an increase in entropy or more particles formed