TRIPLE ONLY: change of state (states of matter + specific heat capacity)
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Describe the arrangement and motion of particles in solids + relative density, compress and KE?
Particles are very closely packed in a fixed and regular arrangement by strong forces
Particles vibrate about fixed positions
They have a definite rigid shape and definite volume
High relative density, hard to compress and low KE
Describe the arrangement and motion of particles in liquids
Particles are closely packed (close together) in random arrangement by strong forces
Particles flow over one another at random
They don’t have a definite shape as they are able to flow and will take the shape of a container
They have a definite volume
High relative density (aunque less than solids), hard to compress and greater KE than solids
Describe the arrangement and motion of particles in gases
Particles are far apart because they have weak forces between them
Particles move randomly
They don’t have a definite shape nor fixed volume
Low relative density, can be compressed and has highest KE
Why are solids usually dense?
Density is a measure of how tightly packed the particles of a substance are. Solids usually have high densities because particles are very closely packed in a regular arrangement
Why do solids keep their definite shape and are strong?
They have strong forces between the particles
Liquids have weaker forces so particles can move more freely than particles in solids
Gases have very weak forces so their particles are very far apart
Even though liquids and gases share the characteristic of having no definite shape, explain what you would see when you put liquid vs gas in a container
Both will take the shape of the container
But liquids will occupy the lowest part of any container, while gases expand to fill the container
This is because liquid particles tend to stick together while gases will expand to fill the container
Why can gases be compressed while it is hard to compress solids and liquids?
Gases have large gaps between the particles so it is easier to push the particles closer together
It is difficult to compress solids and liquids because their particles are almost as close together as they can be.
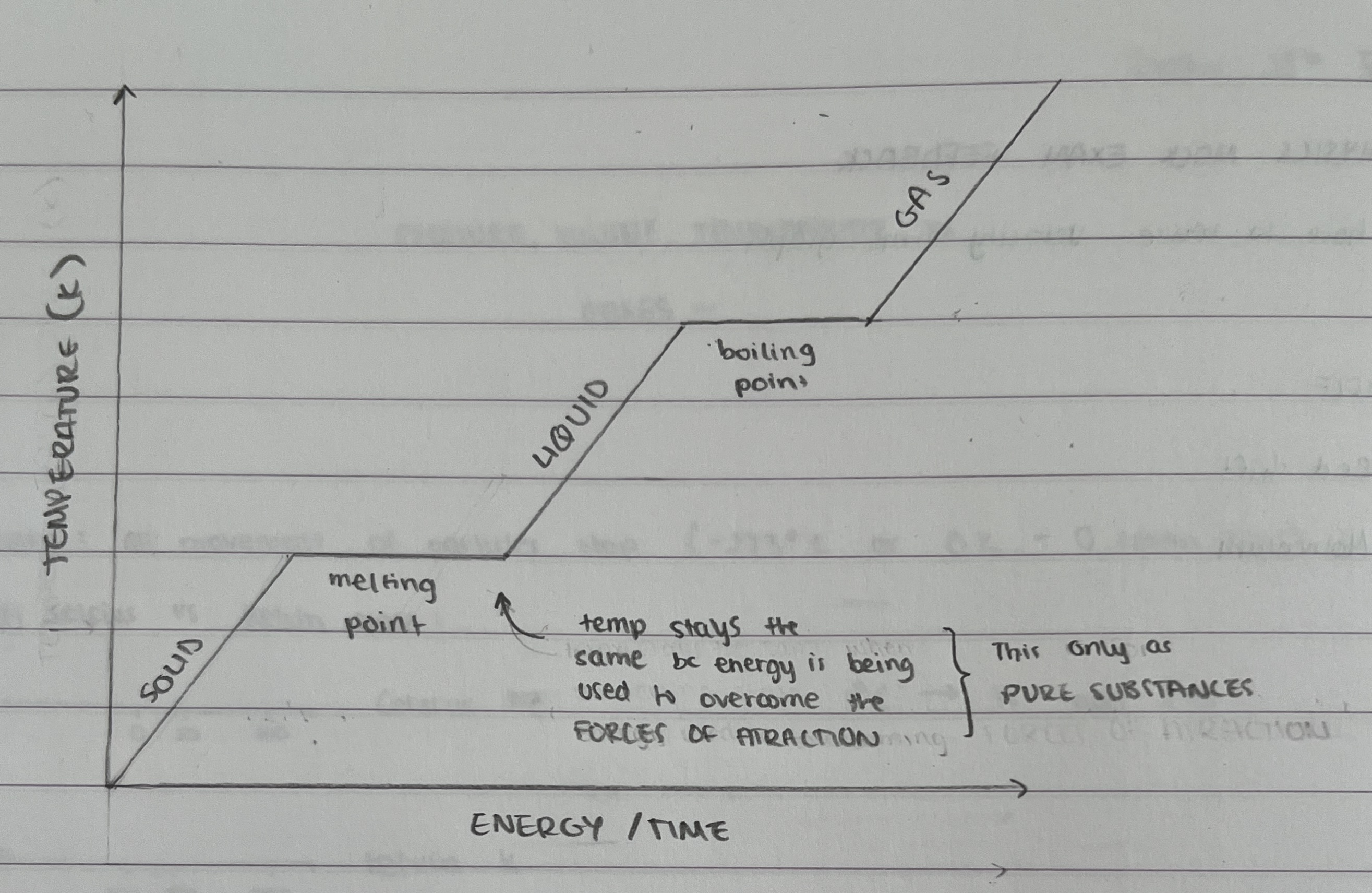
Name the energy conversions between the states of matter
solid ←melting/freezing→ liquid ←boiling/condensing→ gas
Solid —sublimation→ gas
Gas —deposition→ gas
Describe and explain what happens when you melt a solid
When a solid is heated it melts to form a liquid
Thermal energy transfer takes place and supplies the particles in the solid with energy in their kinetic store
This breaks the rigid bonds between the particles meaning they can now flow over each other
Describe and explain what happens when you boil a liquid
When a liquid boils to form a gas thermal energy is transferred to the particles in the liquid so they have more energy in their KE store.
Until they are able to overcome the forces of attraction so bubbles are produced within the whole body of the liquid, allowing gas to form
Particles do not evaporate from the surface
Describe and explain what happens when a liquid evaporates to form a gas
When a liquid is heated it evaporates to form a gas
Thermal energy transfer takes place and supplies the particles on the surface of the liquid with energy in their kinetic store
This removes the bonds between the particles meaning they can move about randomly and spread far apart
Explain evaporation vs boiling
Evaporation
occurs at the surface of a liquid when particles at the surface have enough KE to overcome the forces of attraction and are able to become a gas
It can occur at any temp
Particles have to be at the surface + they need to have enough KE
Boiling
occurs in the body of the liquid (bubbles appear)
occurs at a specific temp (boiling poitn)
Draw a diagram on conversions of states of matter (name the axis + explain the shape)
Practical: investigate temperature during a change of state
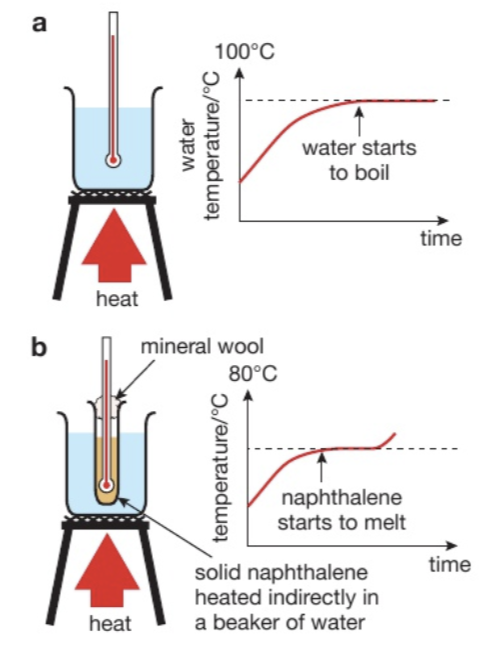
Draw the apparatus
Naphthalene is a solid that can be used in these experiments because it has a low MP. Suggest why a water bath is used for this substance. What precaution should students use?
It is flammable so it must be heated in a water bath WITH CARE
Avoid skin contact with naphthalene and avoid inhaling its vapour
Explain why there are flat lines in the graph showing change of state
The thermal energy supplied does not contribute to the average kinetic energy of the particles in the ice - rather, it is used to weaken the bonds between the particles so they become freer to slide around each other (i.e. a liquid!). Once the ice is fully melted, the temperature of the liquid water begins rising again.
For the practical to investigate temp during a change of state, why was the solid substance naphthalene used? Suggest why it was heated indirectly in a water bath with care.
Has a lower melting point than many solids.
It is flammable
Explain how heating a system will change the energy stored within the system.
Heating a system will change the energy stored in a system by increasing the kinetic energy of its particles
What does the temperature of a material tell us?
Heating a system will change the energy stored in a system by increasing the kinetic energy of its particles
The temperature of the material, therefore, is related to the average kinetic energy of the molecules (the internal energy)
An increase in KE (and therefore energy stored in the system) can:
Increasing the internal energy will have TWO effects:
Either it can cause the temp of the system to increase
Or produce a change of state
What is the specific heat capacity, c of a substance? What is its unit?
The amount of energy required to increase the temperature of 1 kilogram of that substance by 1°C.
Unit: J/kg/°C
Remember that in physics we use kilograms (not grams like in chemistry)
What is the equation to work out how much energy is needed to change the temperature of an object by a given amount?
change in thermal energy (joules) = mass (kg) x specific heat capacity (J/kg/°C) x change in temperature (°C)
ΔQ = m x c x ΔT
What is the specific heat capacity of water?
4200 J/kg/°C
Practical: investigate the specific heat capacity of materials including water and some solids
Method to find c of water/solid block of aluminium? Include equipment used
Measure the mass in kg of the substance using a digital balance/electronic scales (plural)
Place the immersion heater and thermometer in the beaker of water (or solid block of aluminium)
The block would be drilled to allow an electric heater/immersion heater and a thermometer to be inserted
Connect the immersion heater with the power supply (ammeter in series and voltmeter in parallel)
Record initial temp of water using thermometer at time 0s
Turn on power supply and set it at 10V
Start the stopwatch
Measure the voltage in volts supplied to the heater, the current drawn in amps and the length of time in sec the heater is on
Record the highest temperature reached and calculate the rise in temp ΔT
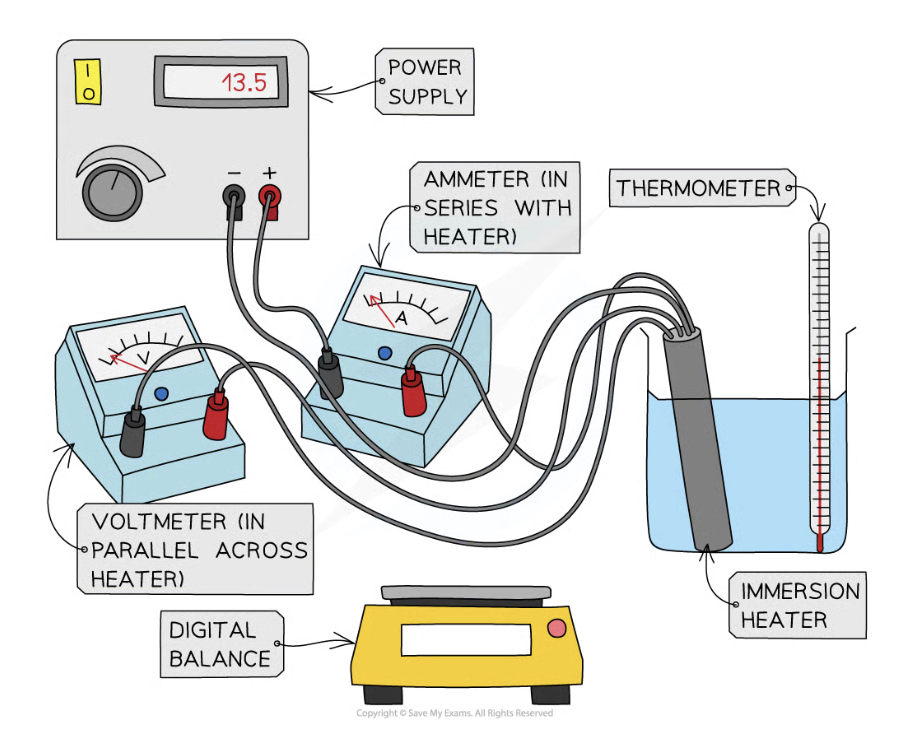
How can you improve the experiment?
Use insulation to reduce heat loss
How can you calculate the specific heat capacity using the results from the experiment?
1- Calculate the thermal energy supplied by the heater
ΔQ = voltage x current x time
Energy = VIt (la Itv)
2- Substitute measured values for mass, ΔT and ΔQ into equation
c = ΔQ/ m x ΔT
Variables?
Independent variable = Time, t
Dependent variable = Temperature, T
Control variables:
Potential difference from the power supply, V
Why is an immersion heater used?
To determine the amount of thermal/electrical energy supplied
What errors might there be? How to improve this experiment?
Some water may be lost to the surroundings by evaporation. Calculate an average mass of water (using the mass before the experiment and the mass after) to account for this
Heat loss to surroundings. Reduce (not prevent) heat loss by using insulation + putting a lid
Stir the water constantly while heating it to ensure the temp measured is the temp throughout the liquid
What should you do when the current or voltage values appear to be changing between two values?
Always be consistent in choosing the higher value
How can we obtain more accurate value for the specific heat capacity? How should we modify our method during the experiment for this?
Plot a graph of the energy supplied (y-axis) against the temperature change multiplied by the average mass (x-axis)
Calculate the gradient of this graph in the straight line region to obtain the specific heat capacity of the water or solid block
To do this, you should record the values of the voltmeter and ammeter every 60s for 10min and then find the electrical/thermal energy supplied (E = VIt)
What should the graph look like? Assuming a perfectly efficient immersion heater
Assuming a perfectly efficient immersion heater, the gradient of the graph = specific heat capacity of the substance
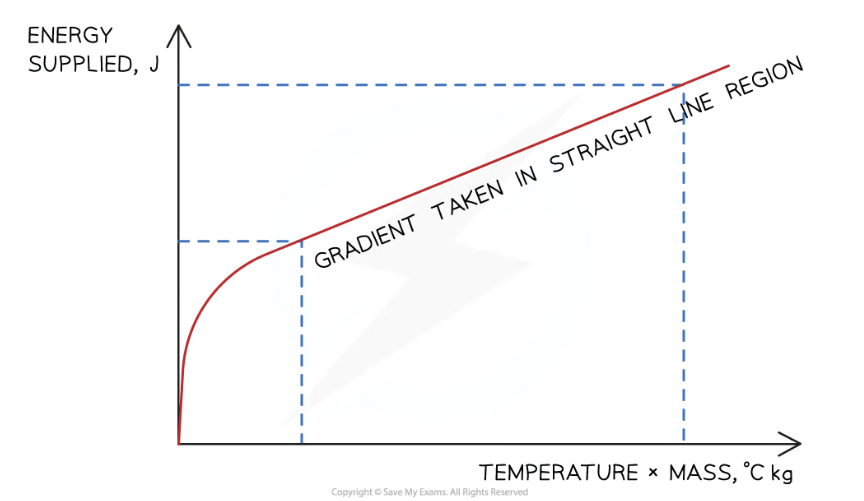
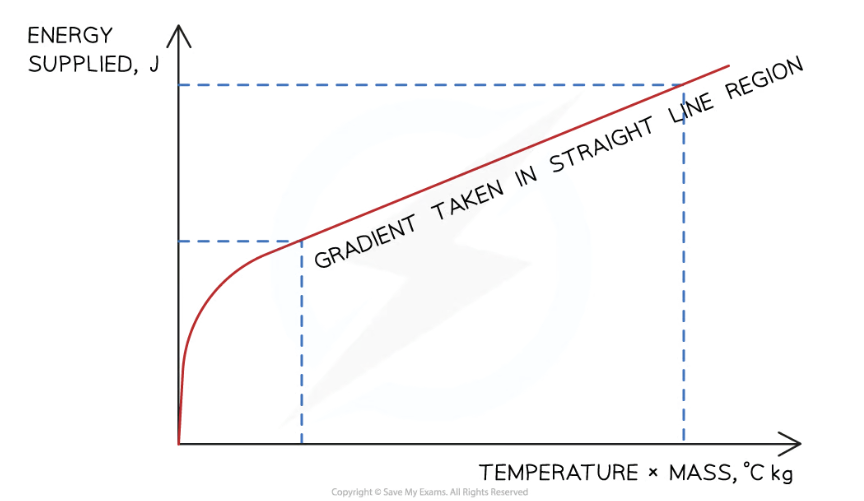
Why should you only calculate the gradient of the straight line and not the curved part?
Before the straight line, the energy supplied is being used to heat the immersion heater itself