BIOC 4331 Lecture 14
1/56
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
For myoglobin (Mb), what is the ligand?
O2.
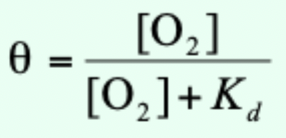
Why do we often use pO2 instead of dissolved [O2] in solution?
Dissolved O2 concentration is difficult to measure; pO2 in gas phase above solution is varied/measured.
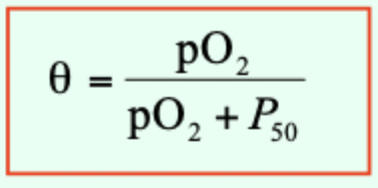
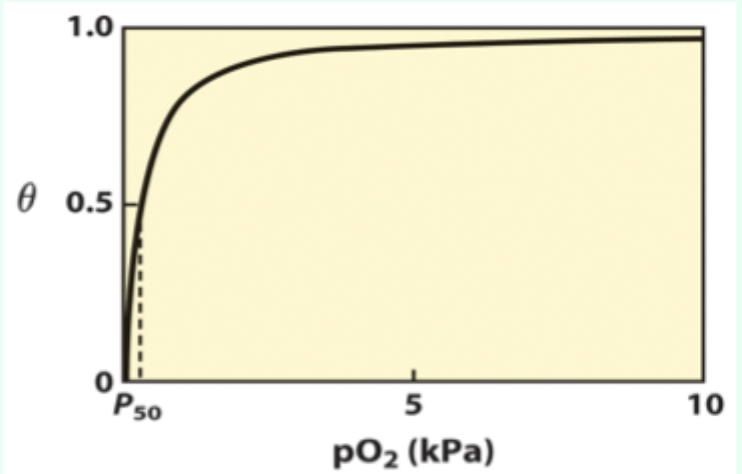
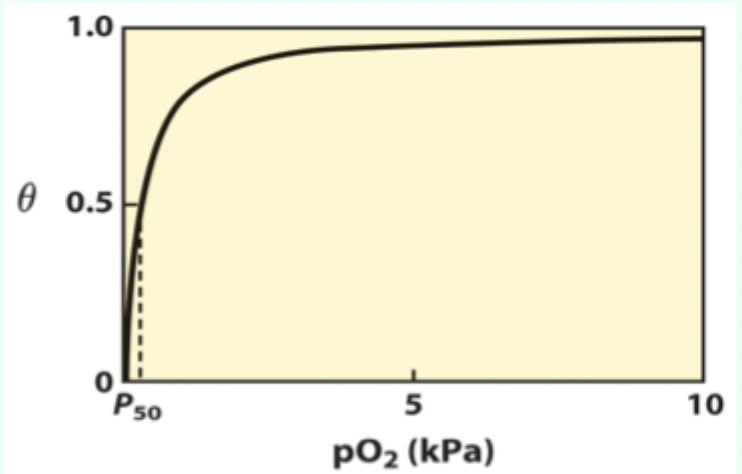
What is P50?
Partial pressure of O2 at which half of binding sites are occupied (θ = 0.5 (50% saturation)).
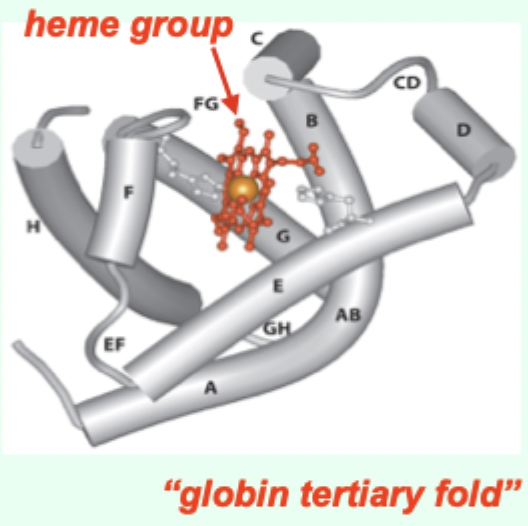
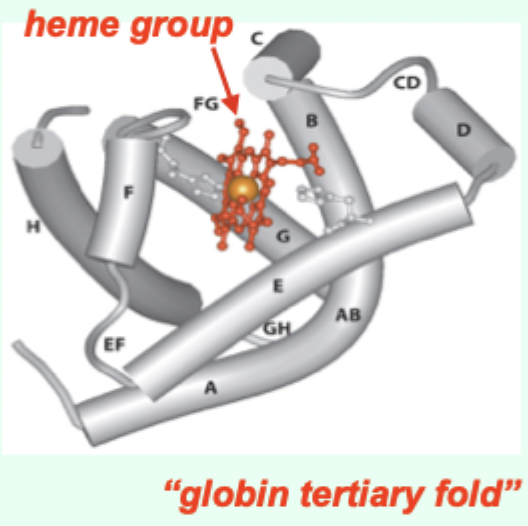
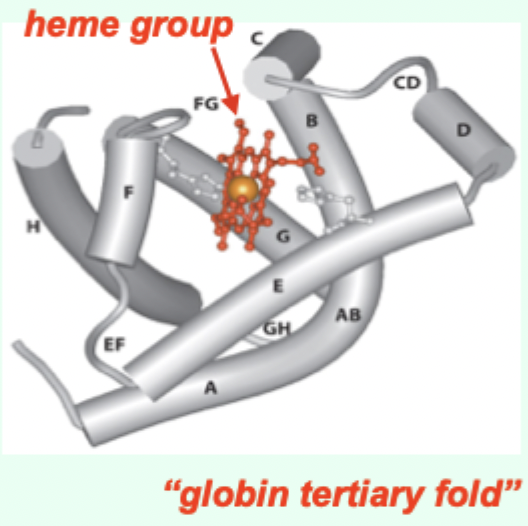
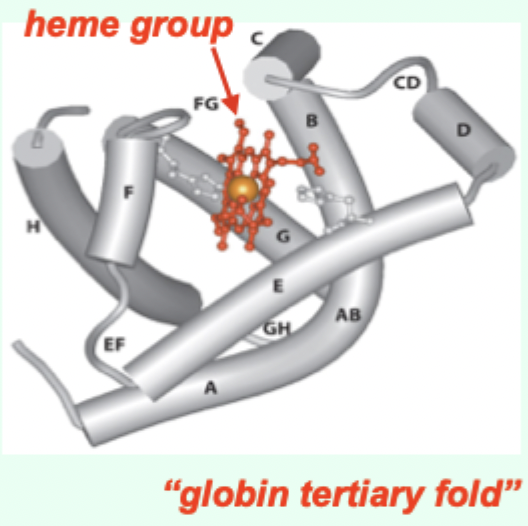
Myoglobin (Mb) is what kind of globin structurally?
A single-chain globin.
Where is myoglobin most abundant?
Muscle
Primary physiological role of myoglobin (Mb)?
Aids O2 transport through tissue.
Why do diving mammals have very high levels of myoglobin?
For O2 storage during dives.
What key prosthetic group does myoglobin contain?
A heme (porphyrin ring).
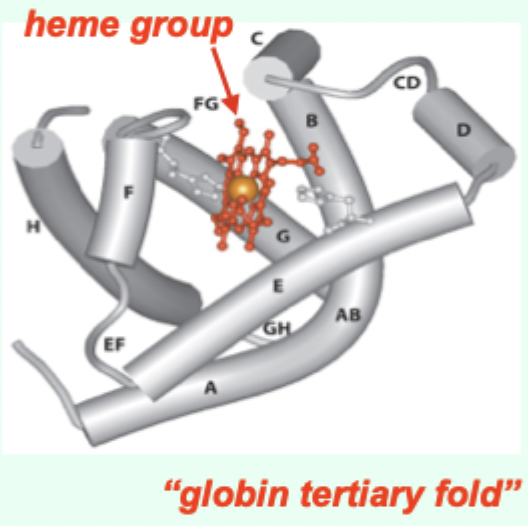
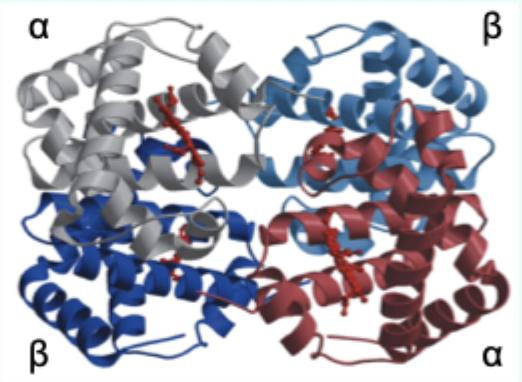
Hemoglobin (Hb) is composed of how many subunits (HbA)?
4 subunits: α2β2 (HbA = adult).
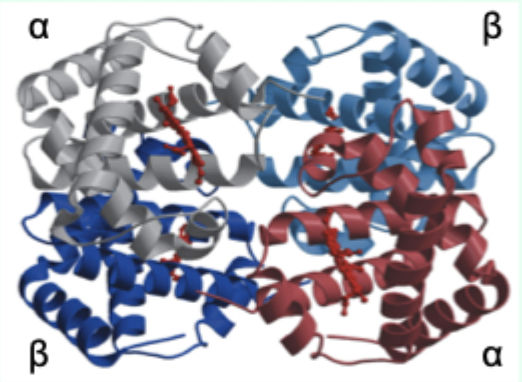
Each Hb subunit contains what two key structural features?
A globin tertiary fold + a heme prosthetic group.
Maximum number of O2 molecules Hb can bind?
4 (one per subunit/heme).
Hb is the major carrier of what in blood?
O2
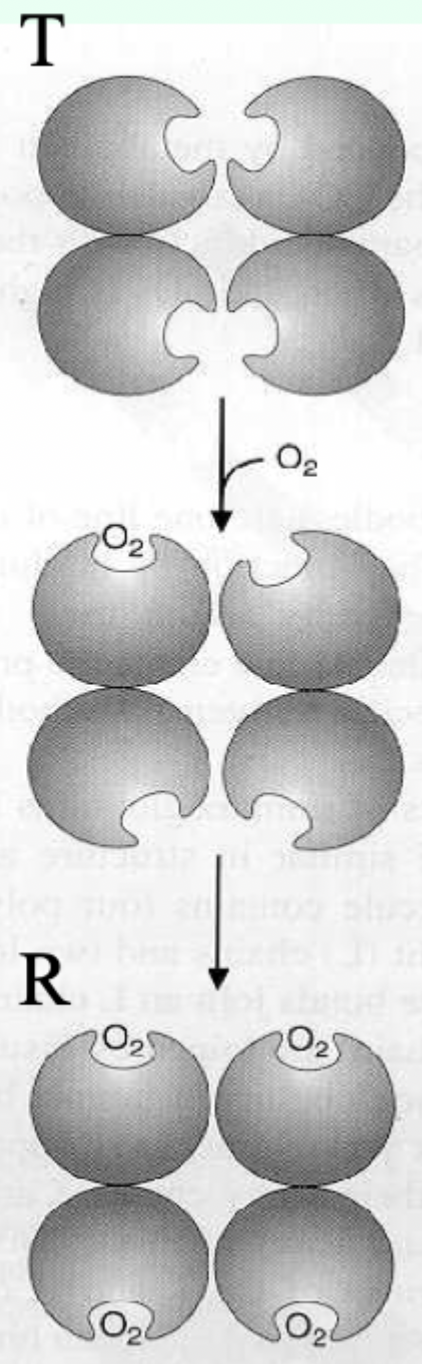
Hb can undergo a conformational change between which two states?
T state and R state.
During the T→R transition, do individual subunits change a lot?
No—little change in individual subunits; major changes occur at αβ interfaces.
What happens at the αβ interfaces during the T→R transition?
Extensive changes; the αβ interfaces slide past each other.
αβ interfaces consist of >30 interactions of what 3 kinds?
Mainly hydrophobic
Some hydrogen bonds
A few key ionic interactions (ion pairs/salt bridges).
What happens to ion pairs during the T→R transition?
Some ion pairs stabilizing T are broken; a few new ones form in R.
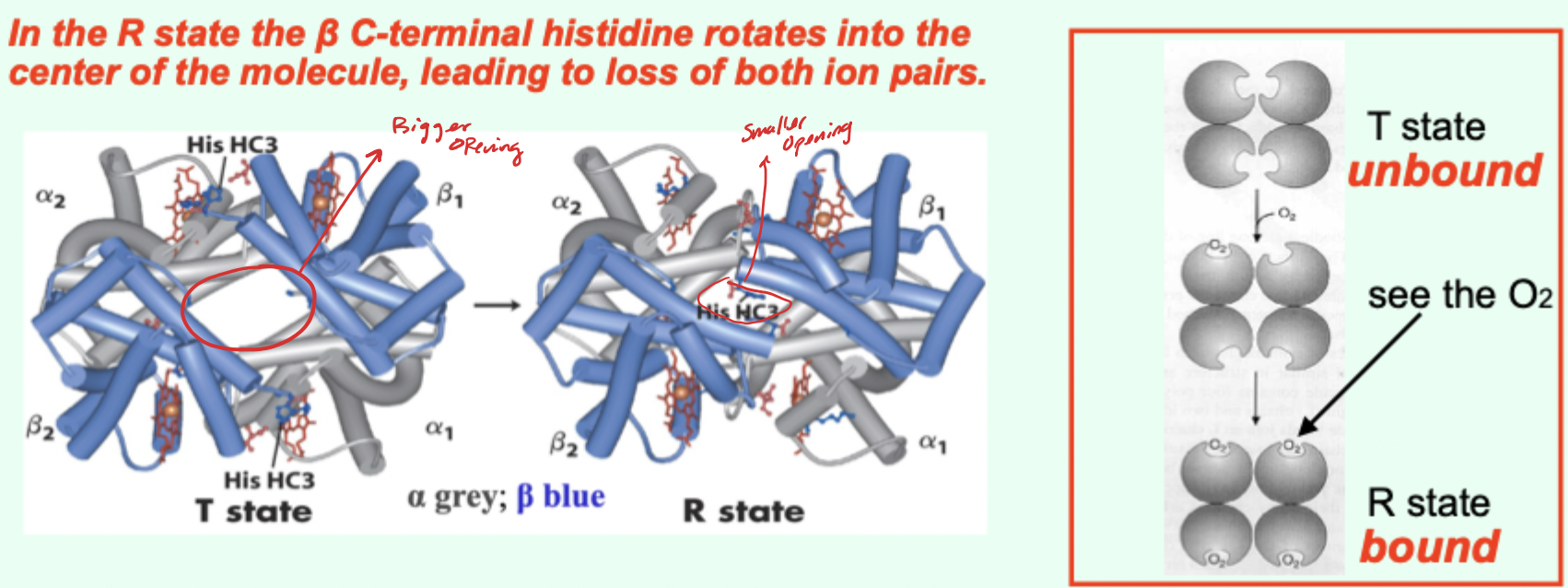
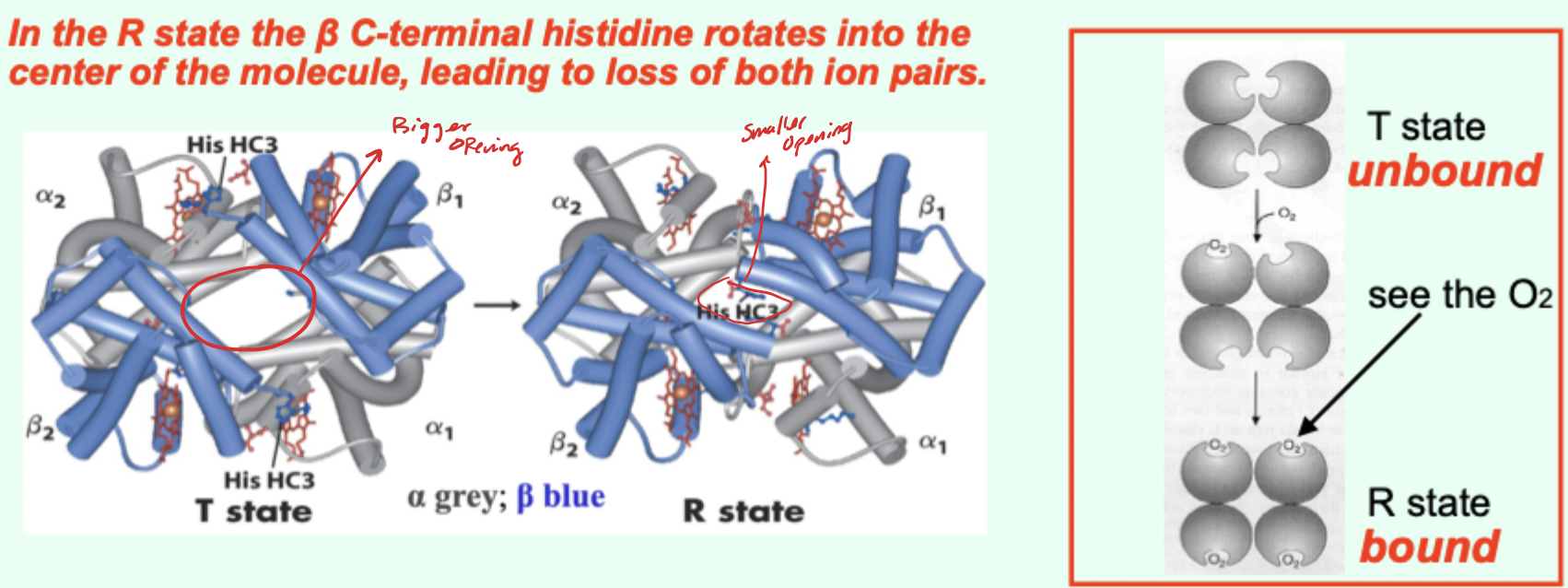
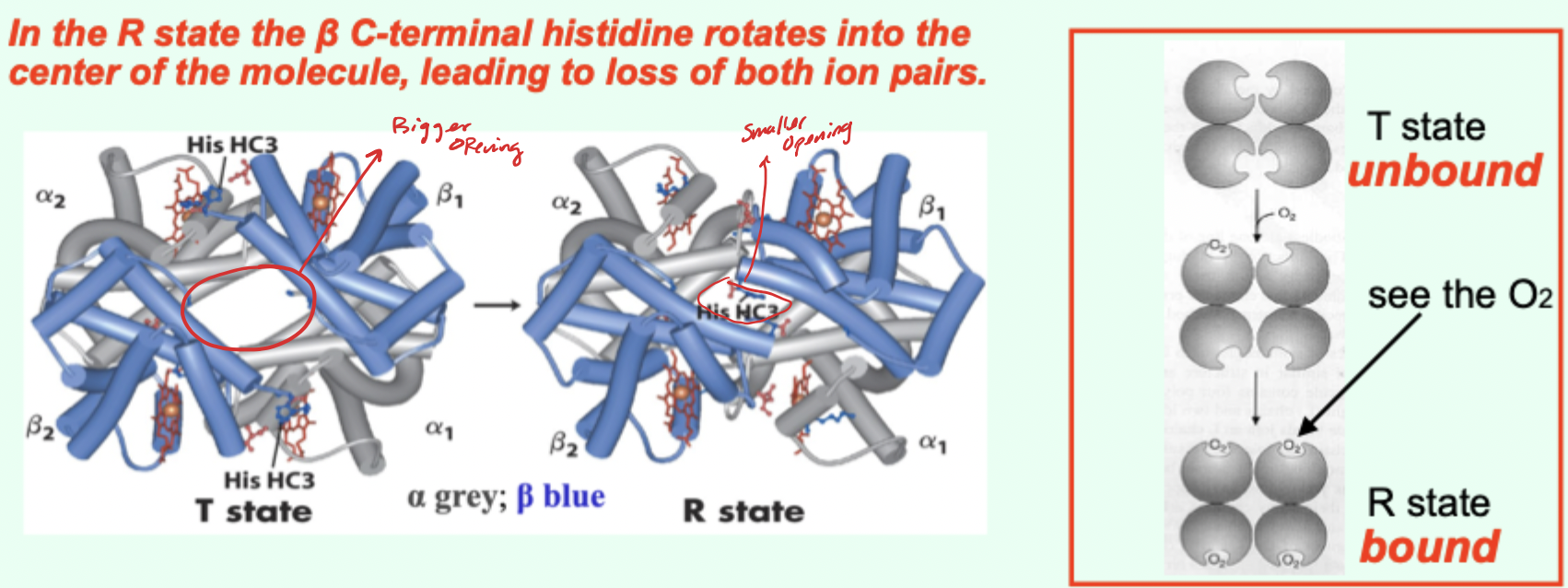
Which Hb state has higher affinity for O2?
R state has higher affinity.
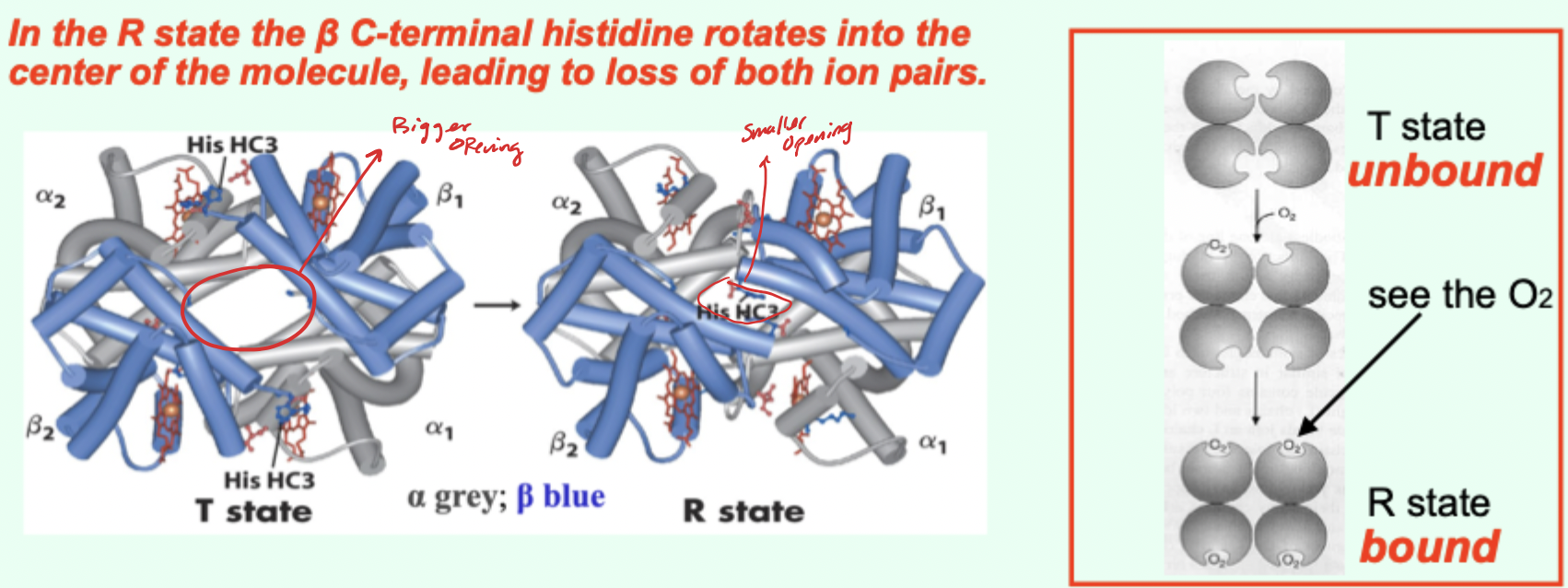
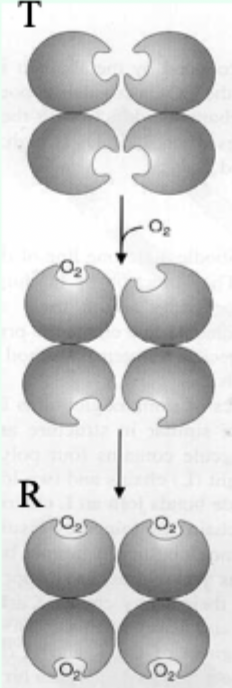
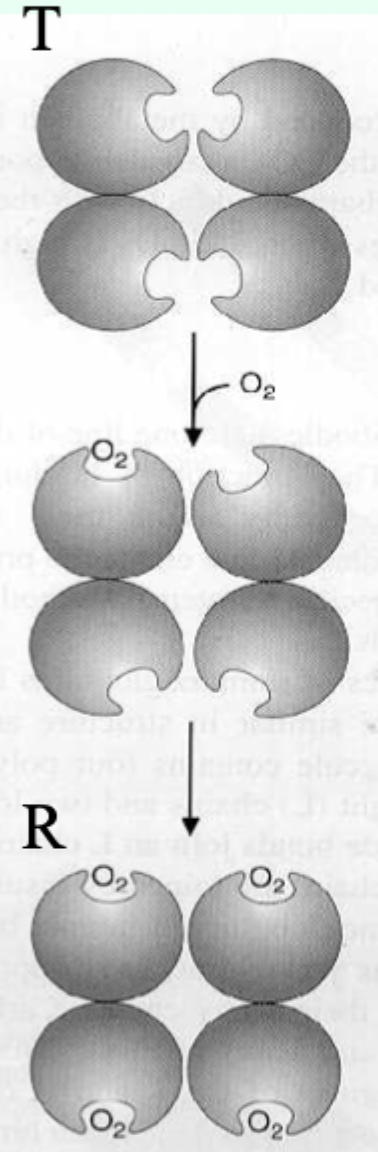
Hb vs Symmetry (MWC) model: can O2 bind T and R?
Yes.
O2 can bind both T and R, but affinity is higher in R.

Which state is favored when O2 is absent and what does that imply?
T state predominates (more stable) when O2 is absent → deoxy/low-affinity form is favored.
O2 binding in which pocket stabilizes the R state?
The heme distal pocket.
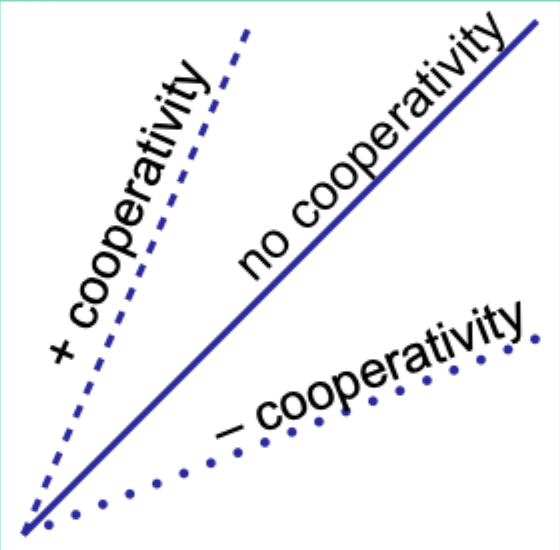
Positive cooperativity (Hb): what does it mean?
When a ligand binds to one binding site/subunit on hemoglobin, it increases the affinity of the remaining sites—so subsequent ligand binding becomes easier.
Negative cooperativity (Hb): what does it mean?
When a ligand binds to one binding site/subunit on hemoglobin, it reduces the affinity of the remaining sites—so subsequent ligand binding becomes harder.
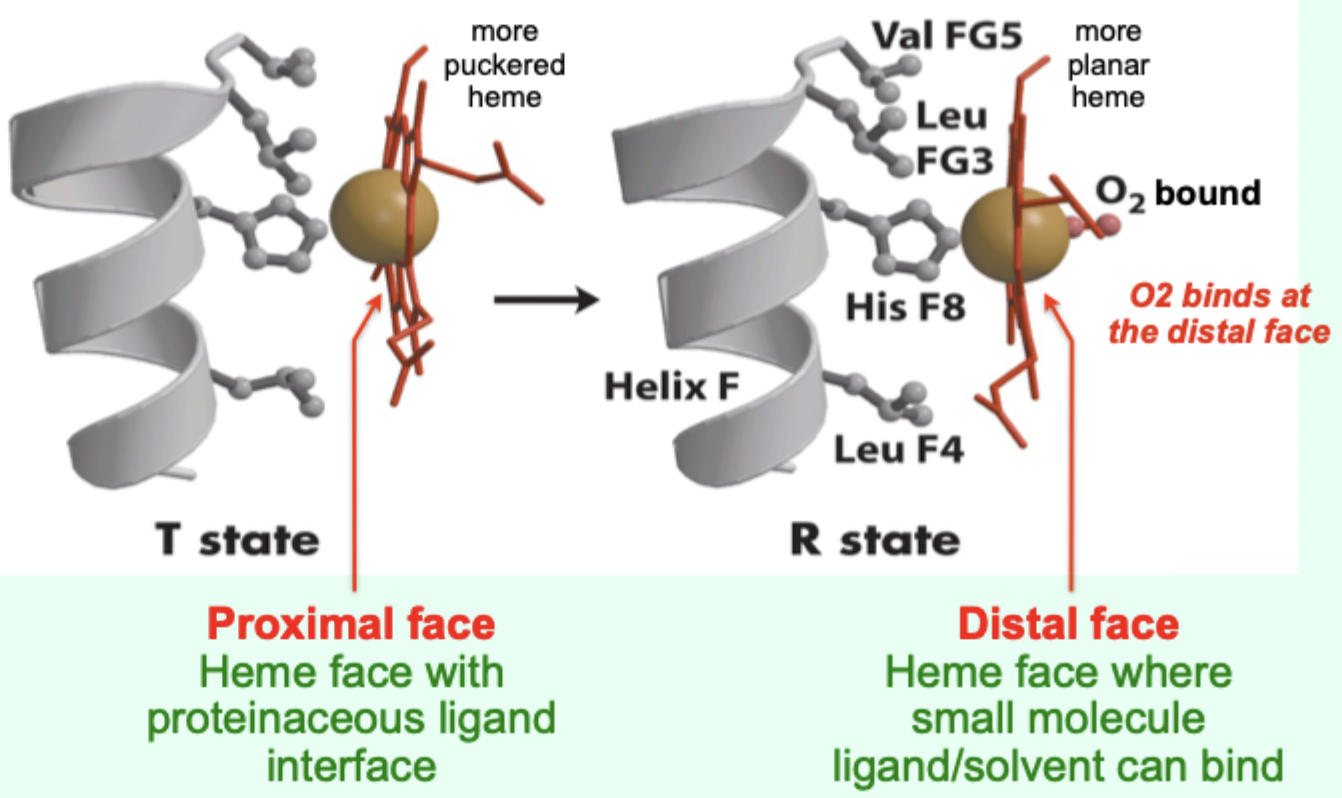
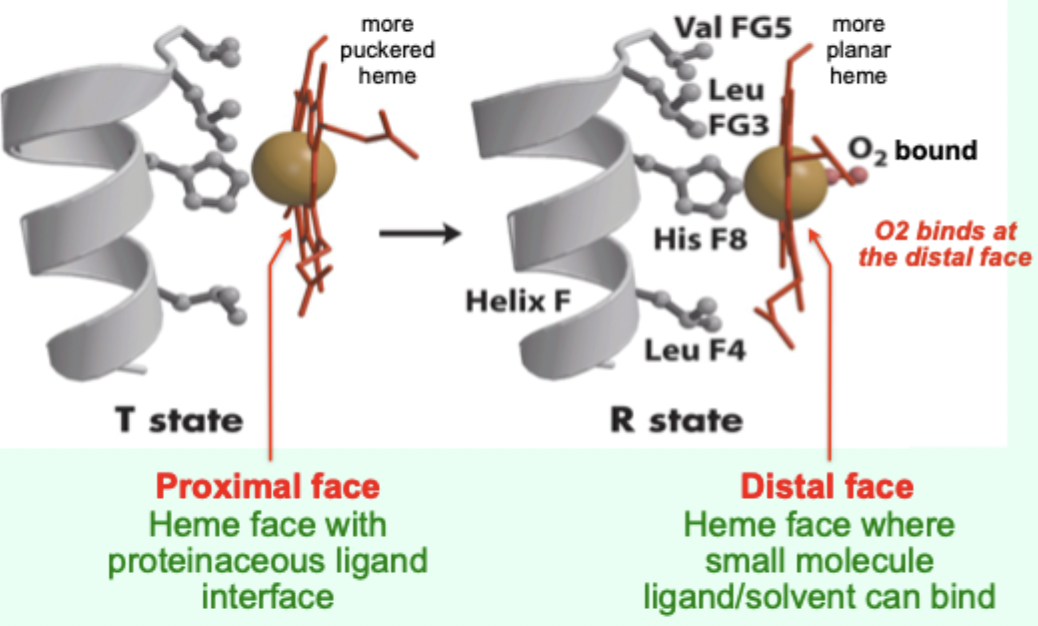
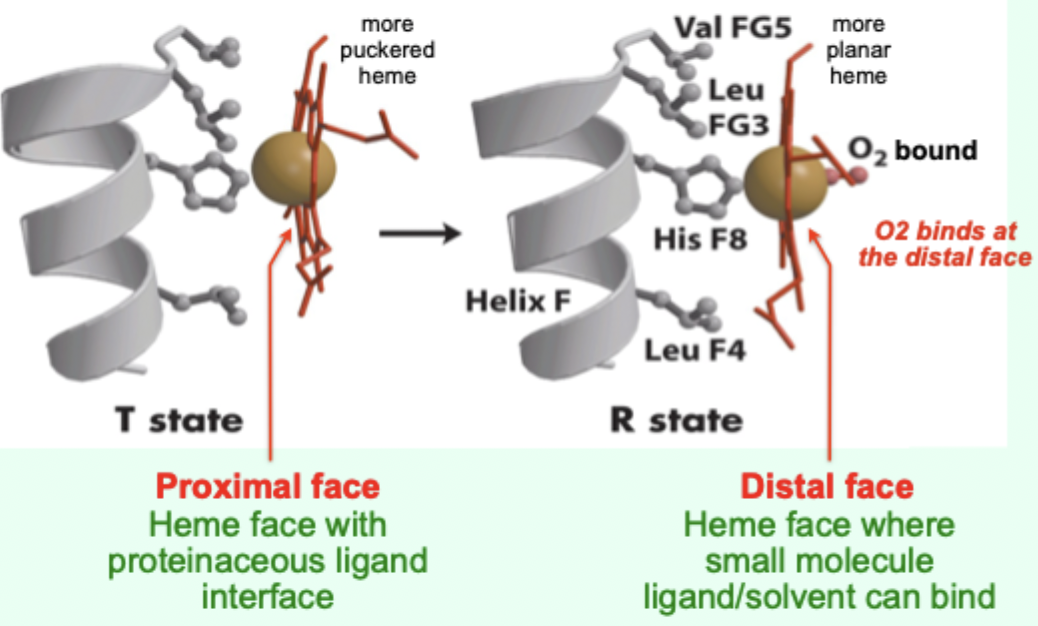
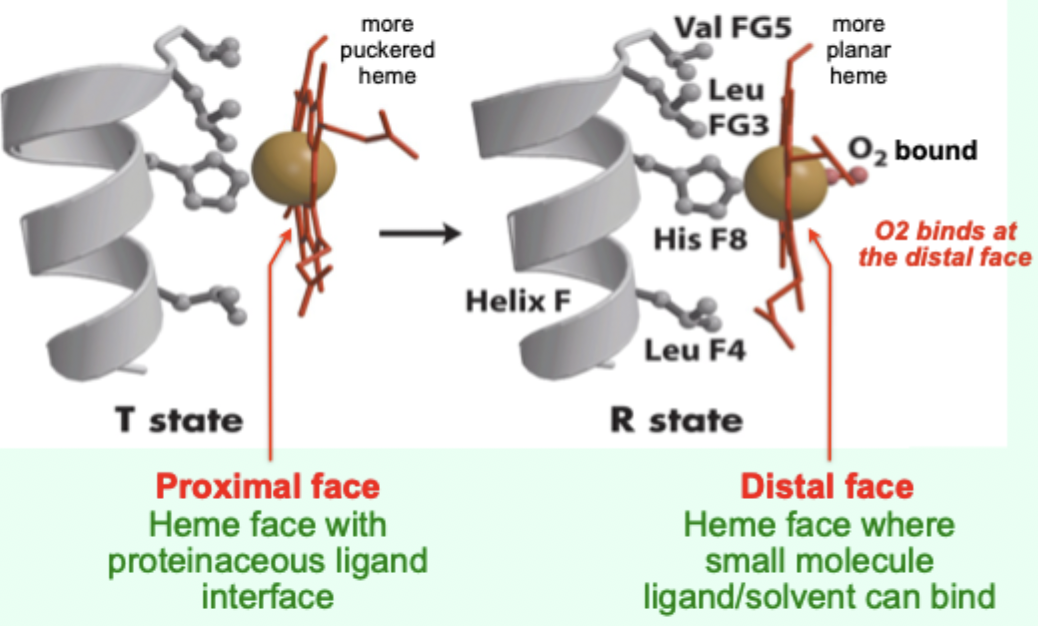
In the T state, what is the heme/porphyrin geometry?
Porphyrin ring is slightly puckered.
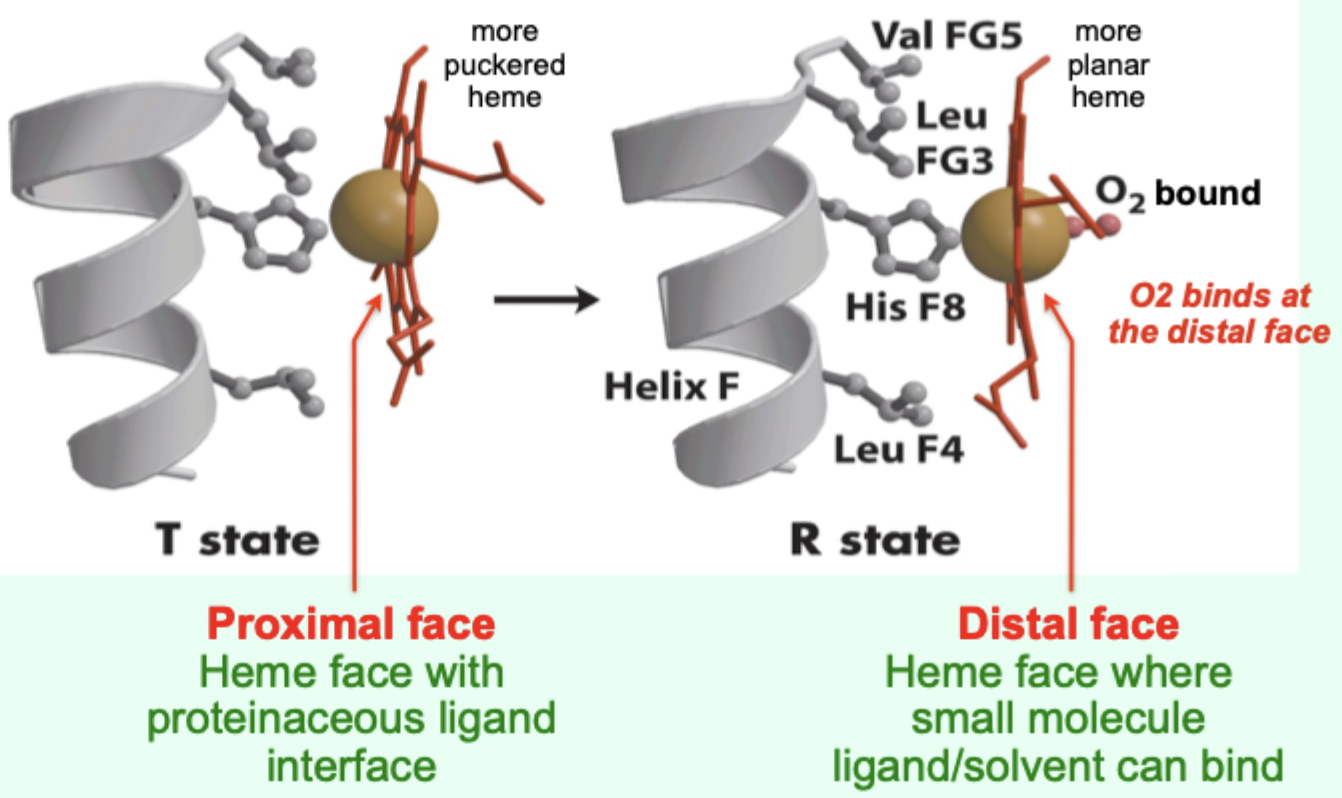
In the T state, where is the iron positioned relative to proximal His?
Iron is pulled toward the proximal His ligand.
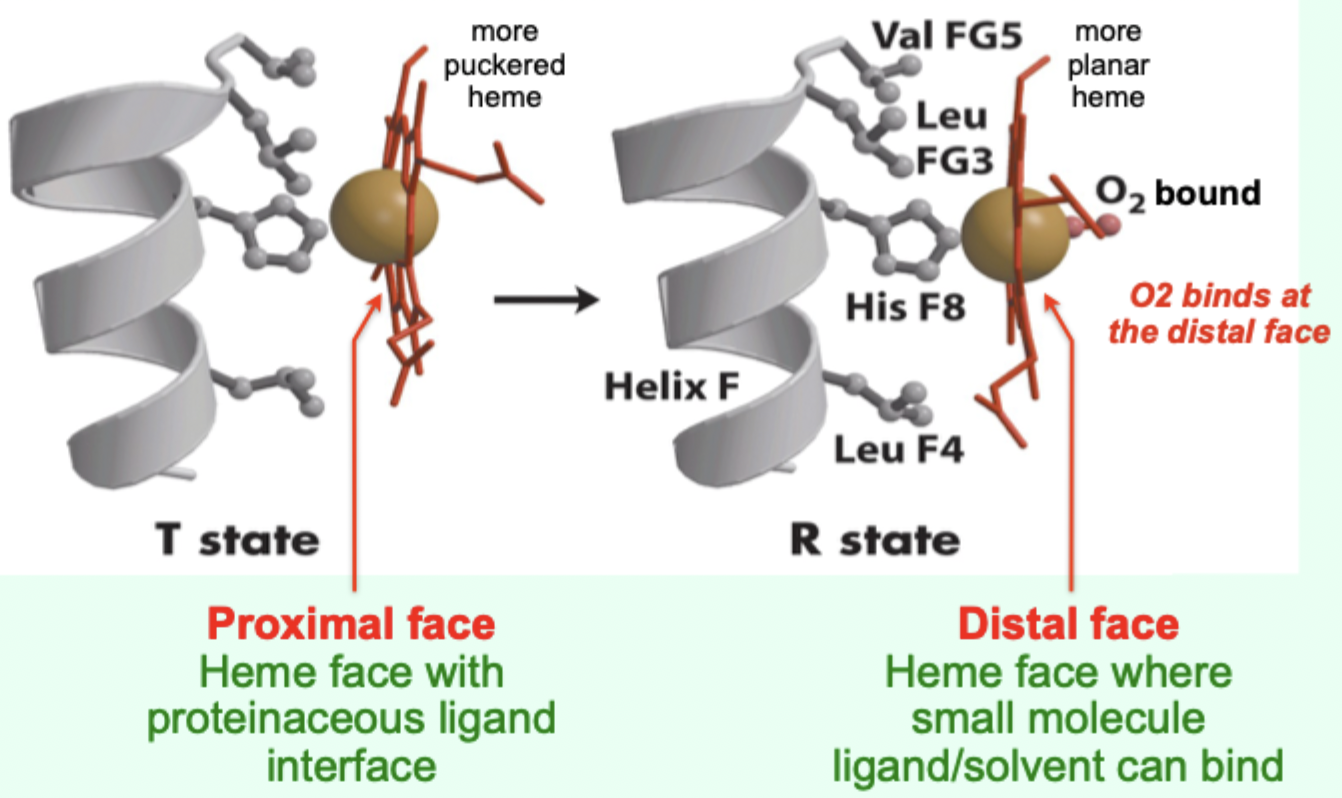
Upon O2 binding, what happens to the porphyrin ring?
It becomes more planar.
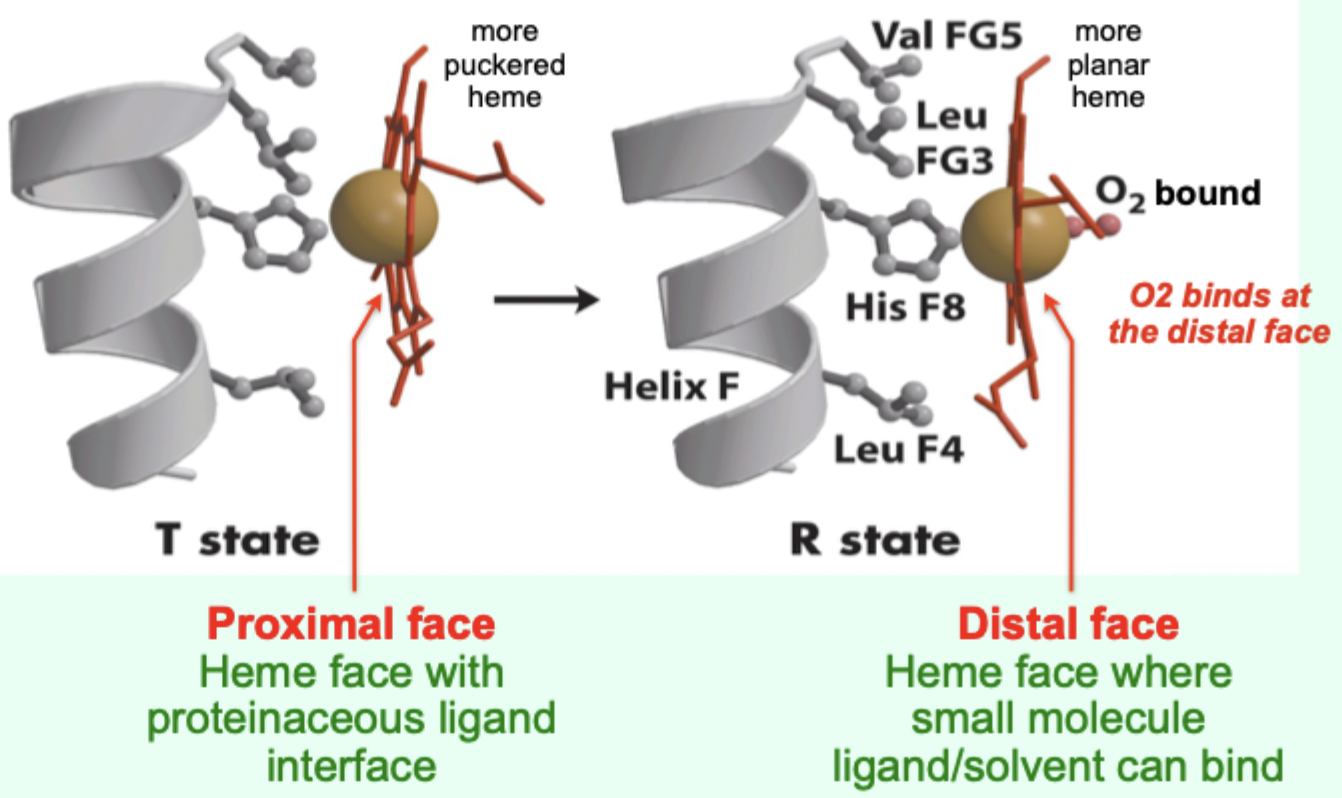
Upon O2 binding, what happens to the iron and proximal His?
Iron and proximal His are pulled into the plane of the porphyrin.
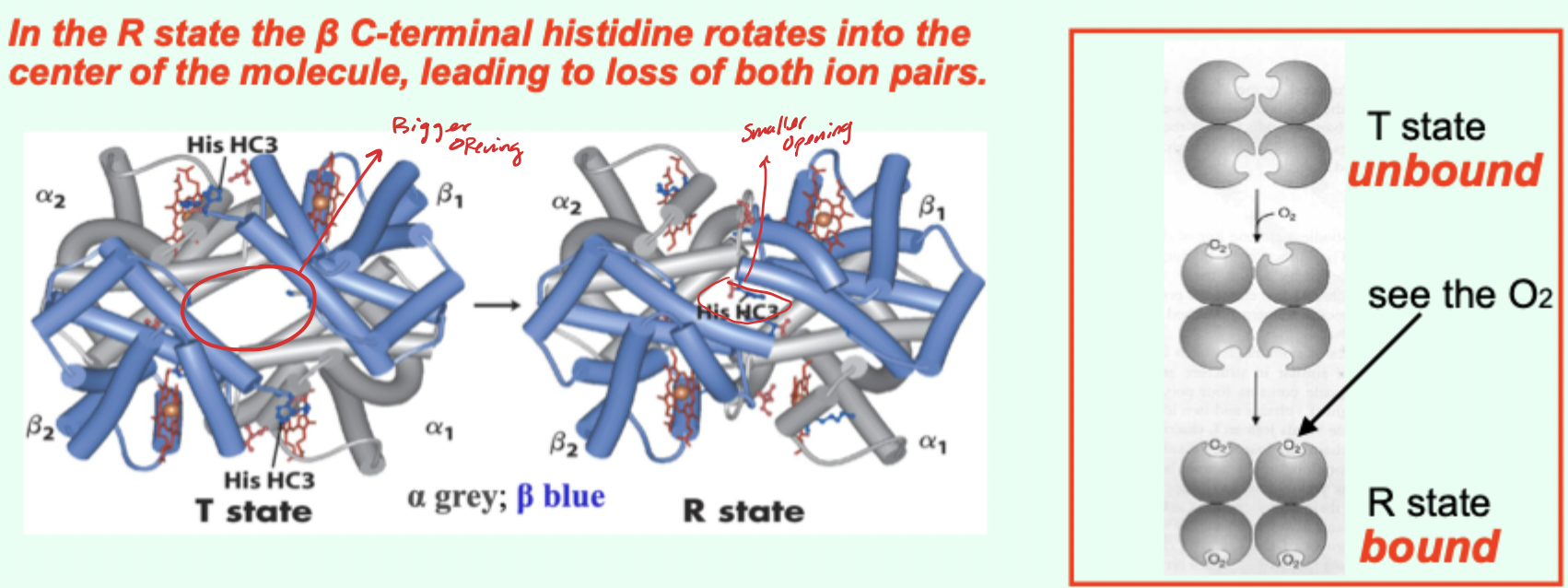
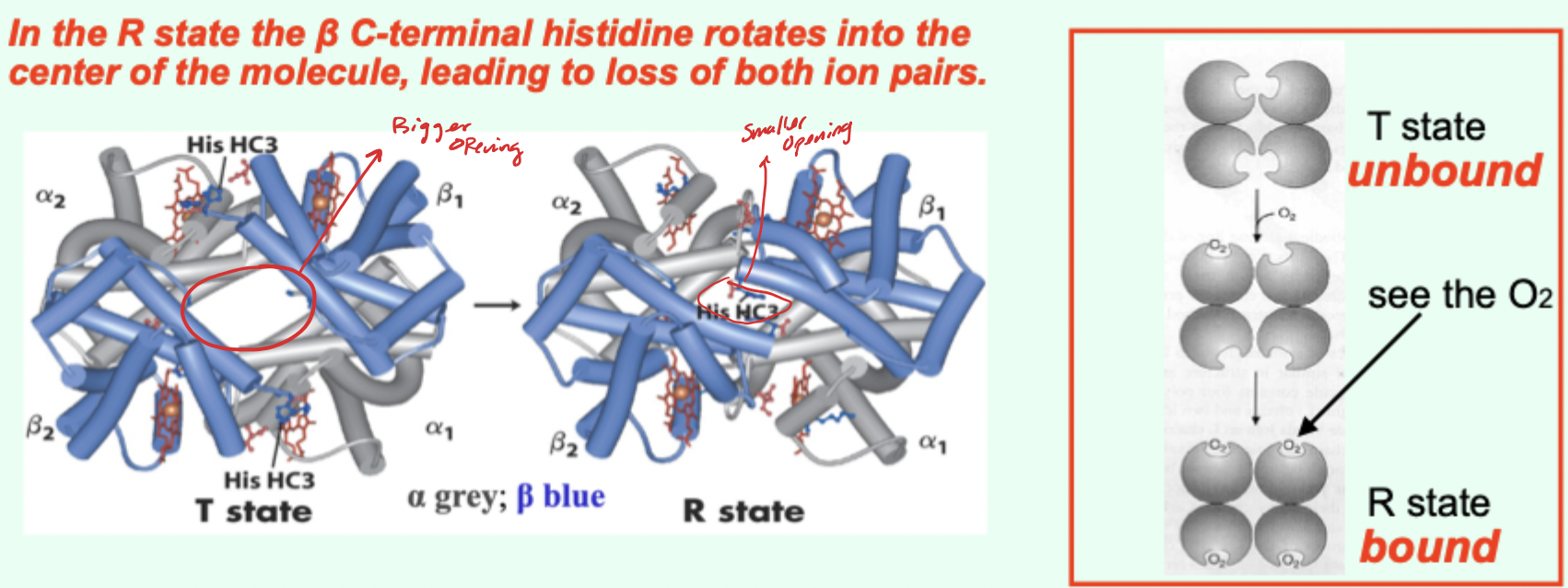
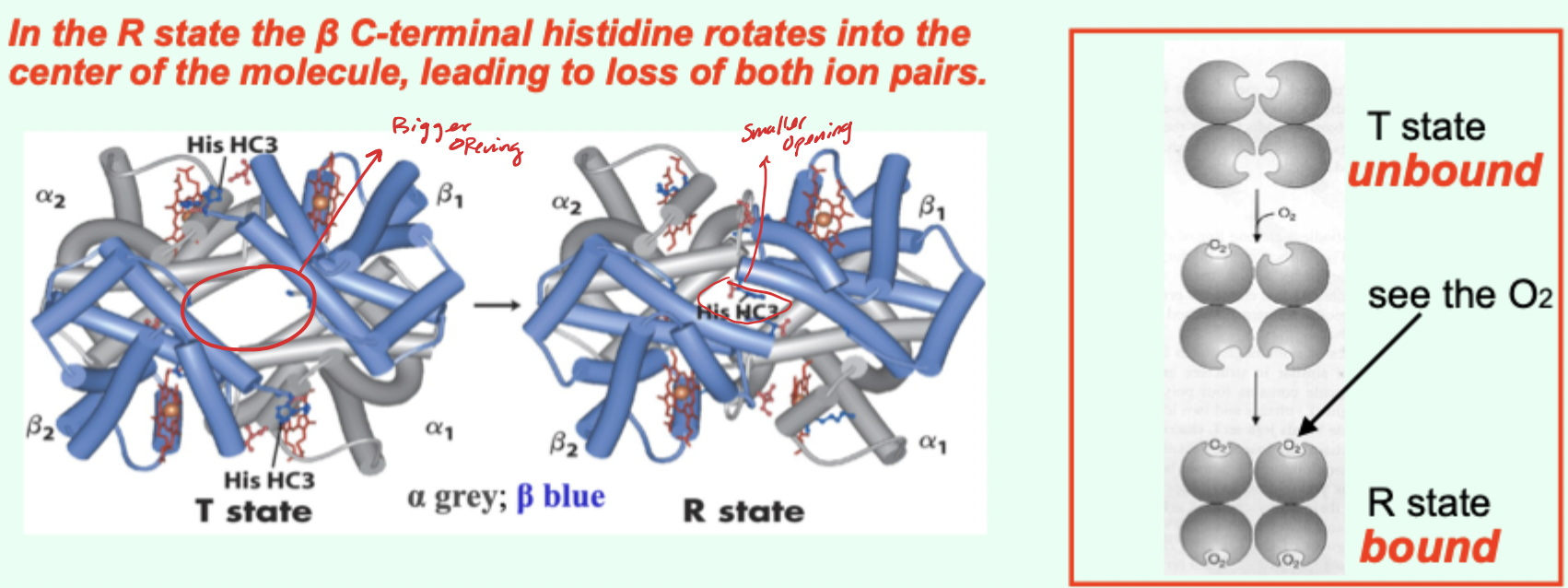
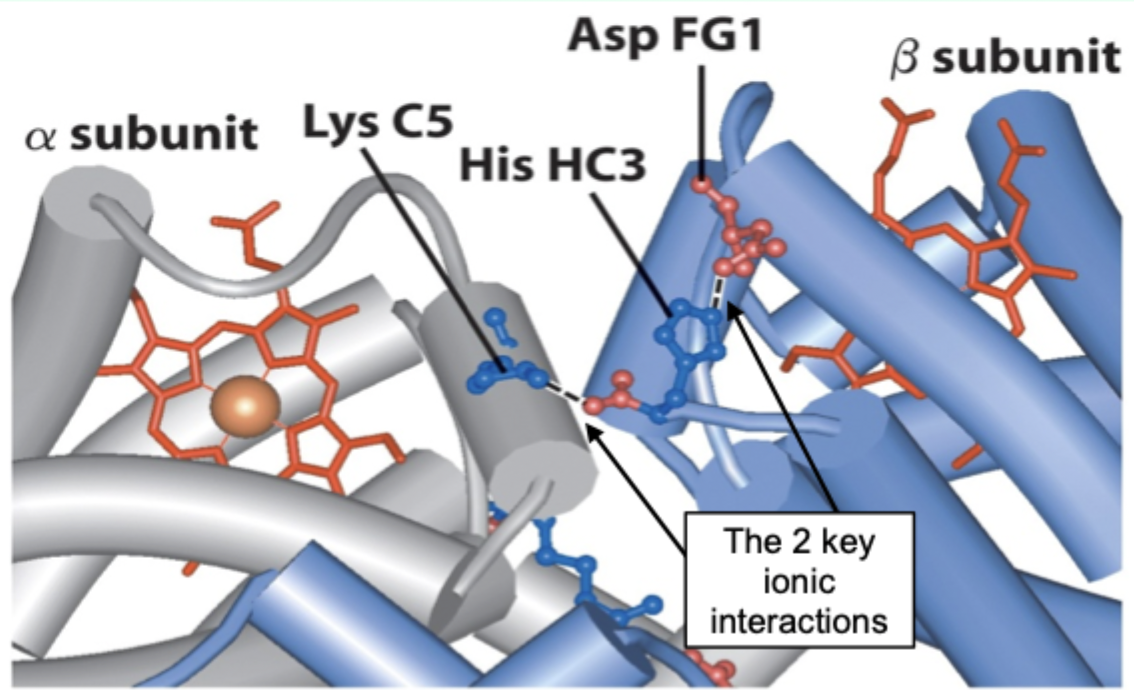
Going from T→R involves the loss of how many key ionic interactions?
2 key ionic interactions.
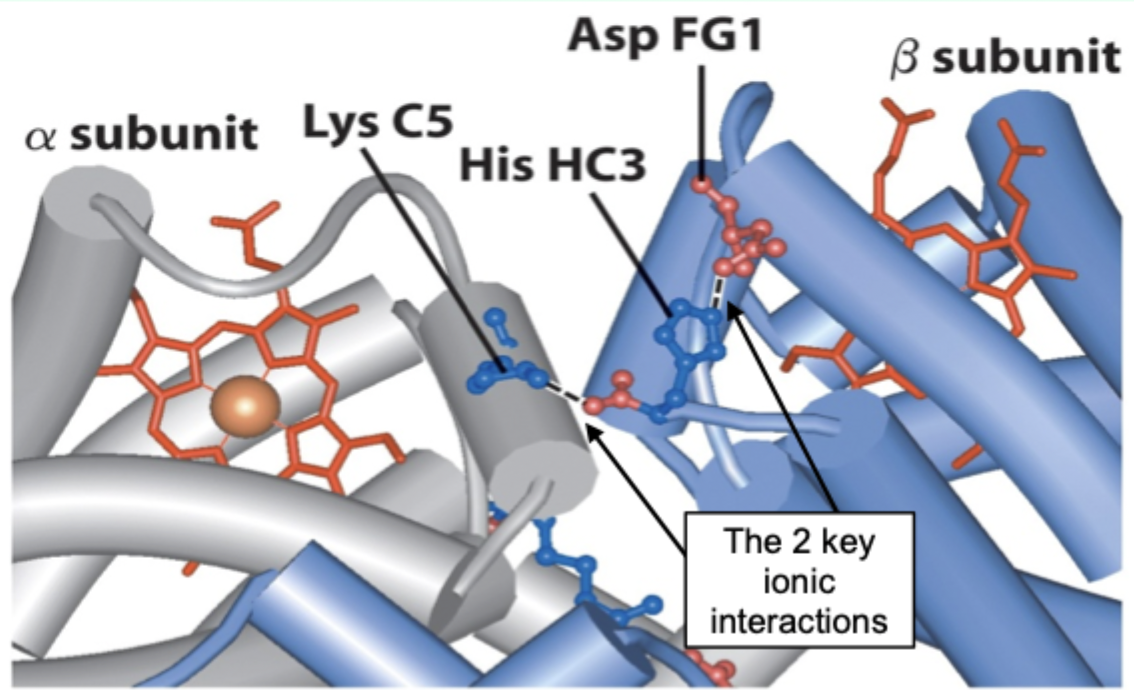
In the R state, what happens to the β C-terminal histidine?
It rotates into the center of the molecule, leading to loss of both ion pairs.
T state is generally associated with what O2-binding status?
Unbound (deoxy) favored.
R state is generally associated with what O2-binding status?
Bound (oxy) favored.
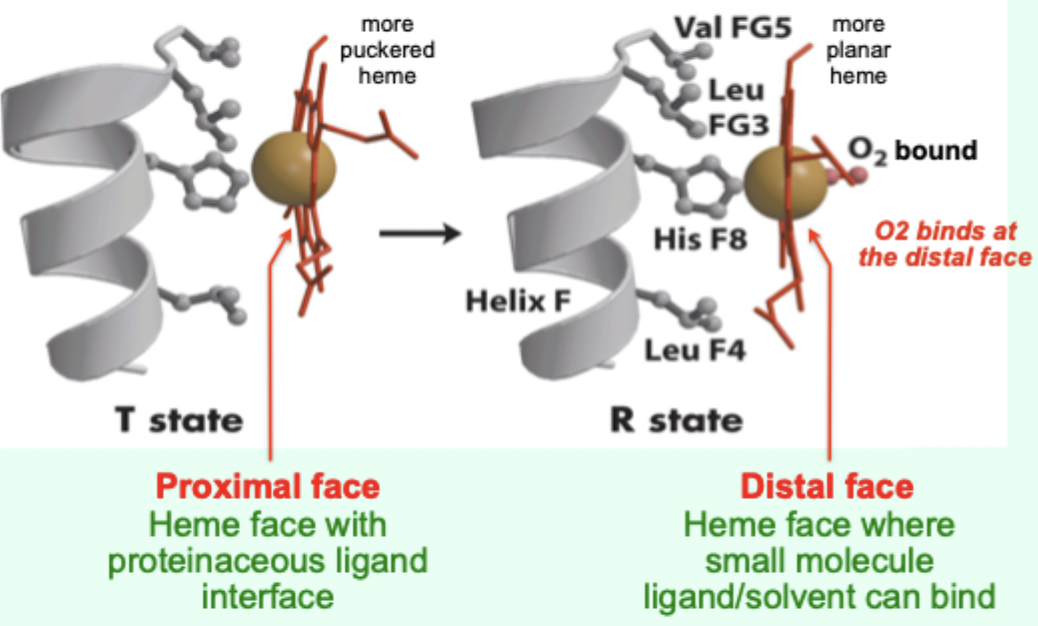
Which heme face is the “proximal face”?
The face with the proteinaceous ligand interface (proximal His side).
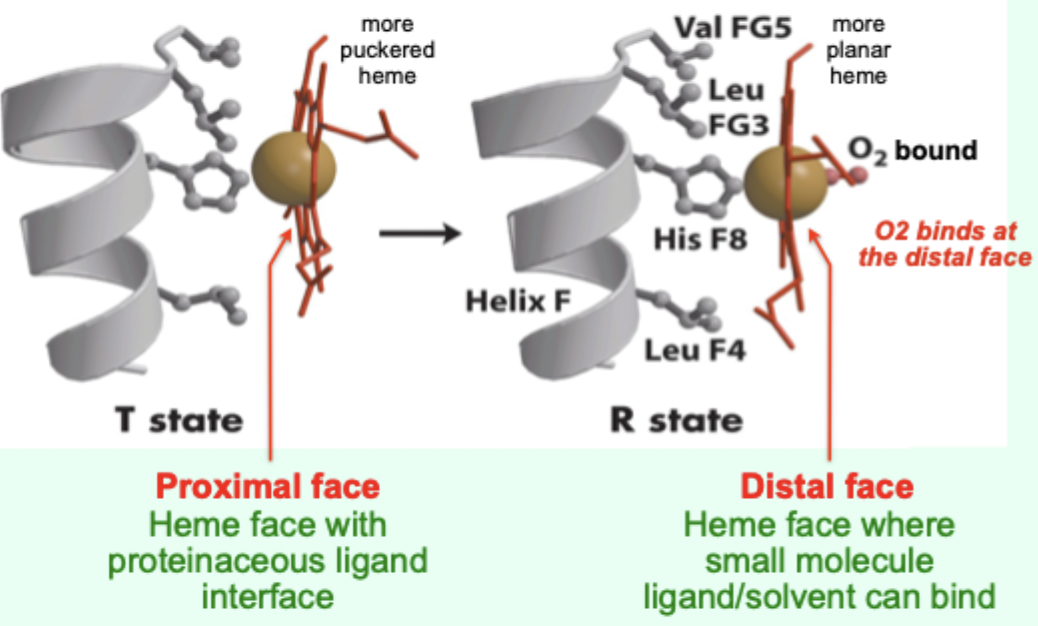
Which heme face is the “distal face”?
The face where small molecule ligand/solvent can bind (O2 binds here).
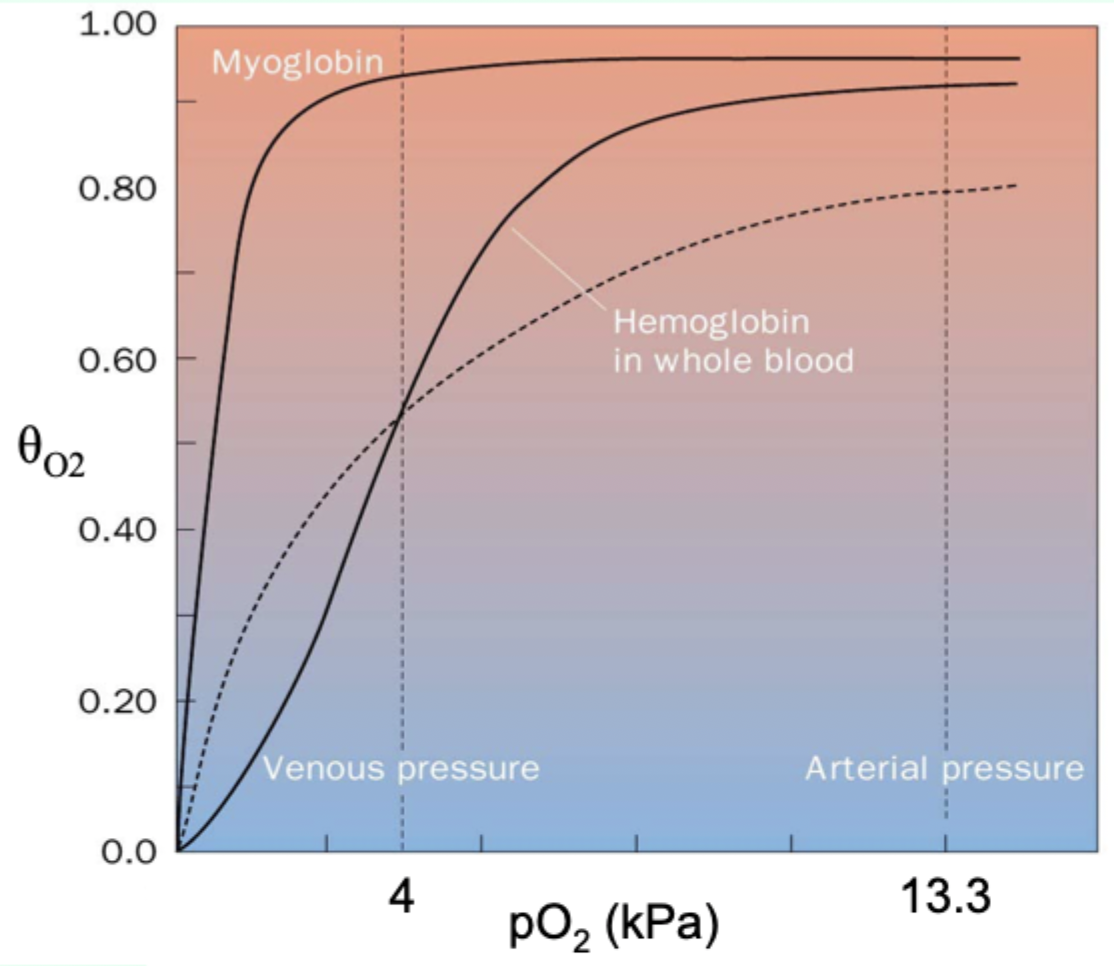
What must Hb do at lungs vs tissues?
Bind O2 efficiently at lungs but release it at tissues.
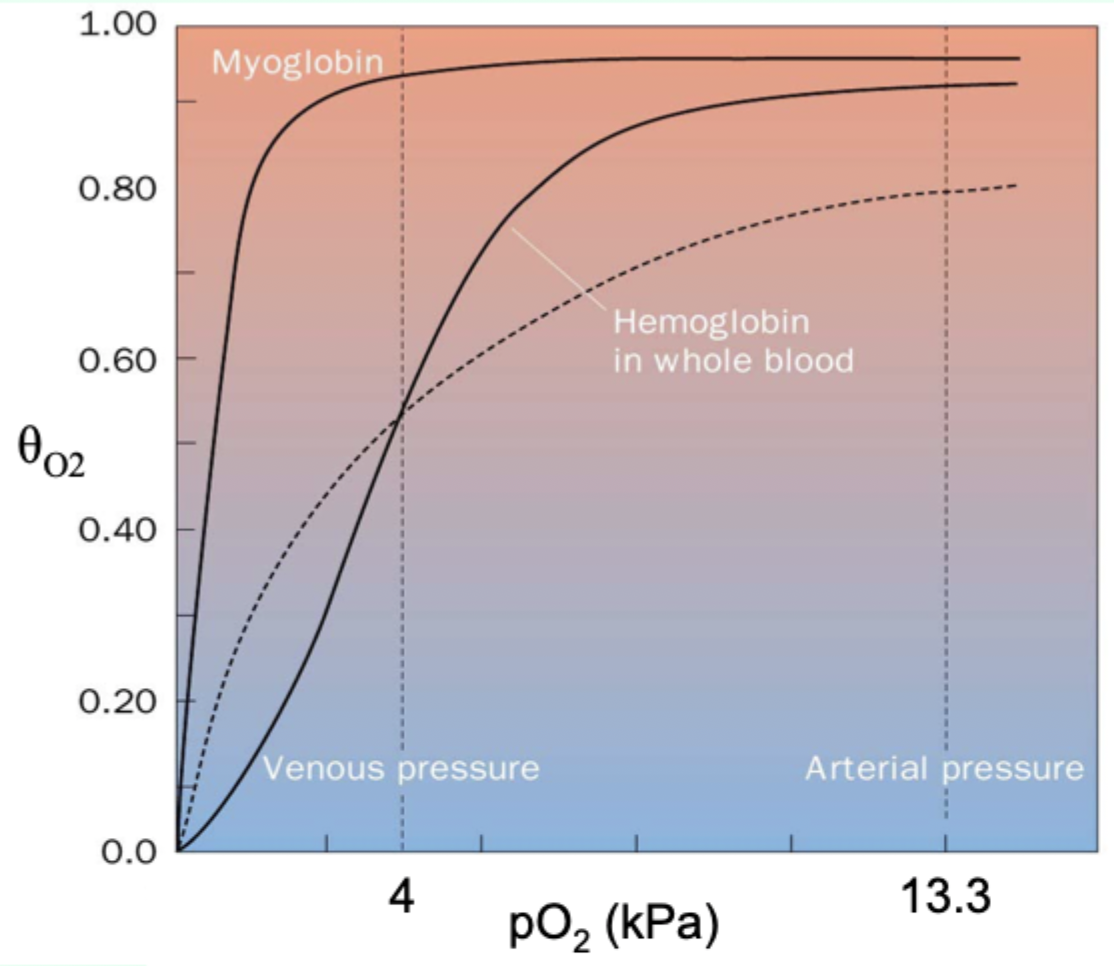
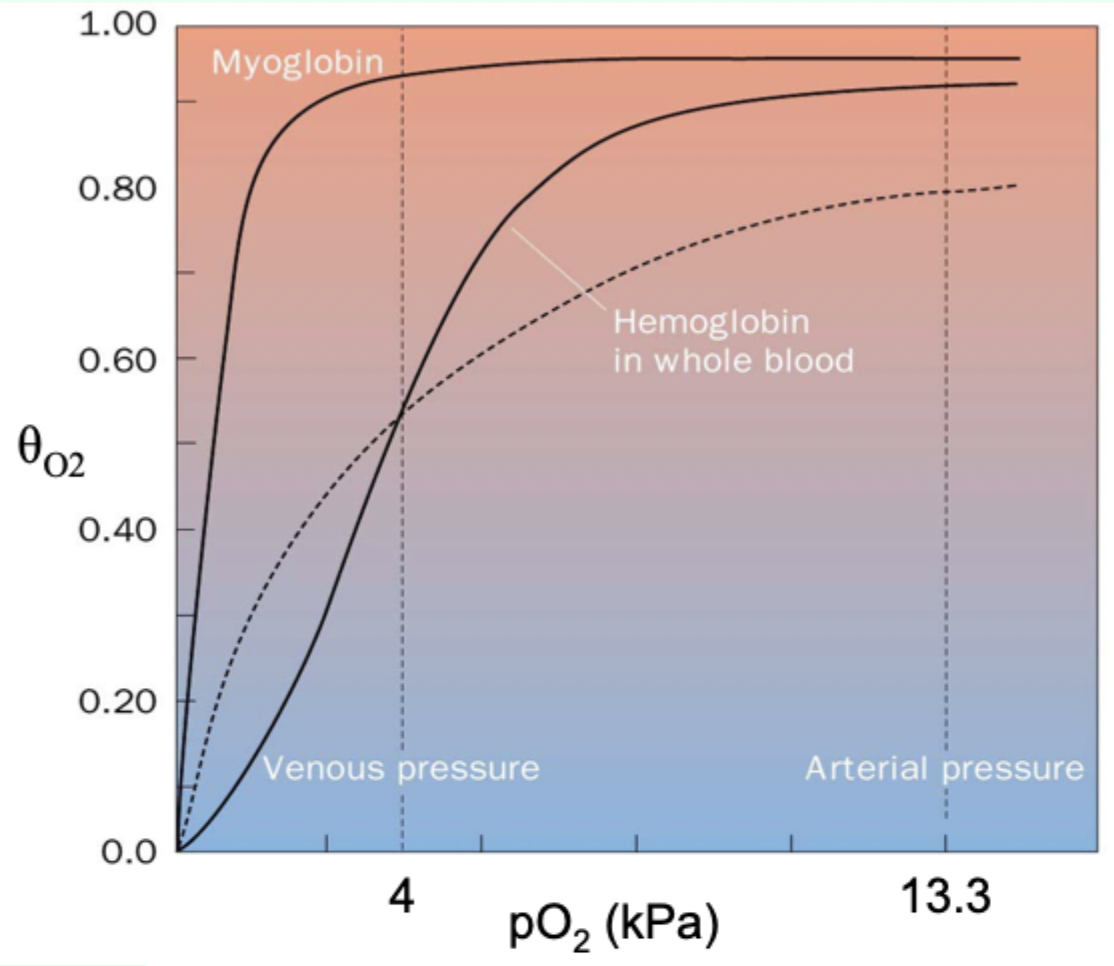
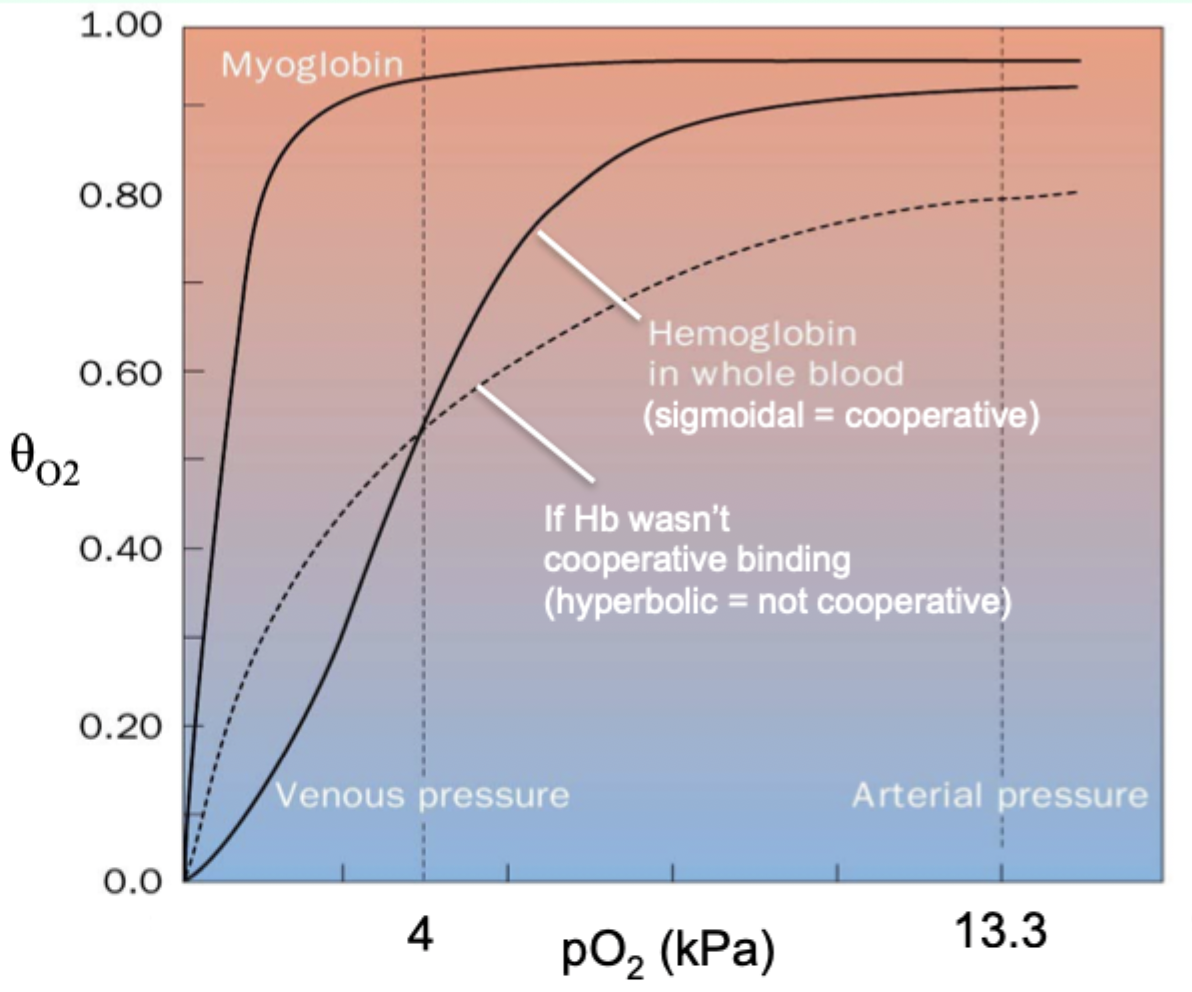
Why is myoglobin ill-suited for oxygen delivery like Hb?
Its hyperbolic curve makes it hold O2 too tightly at lower pO2.
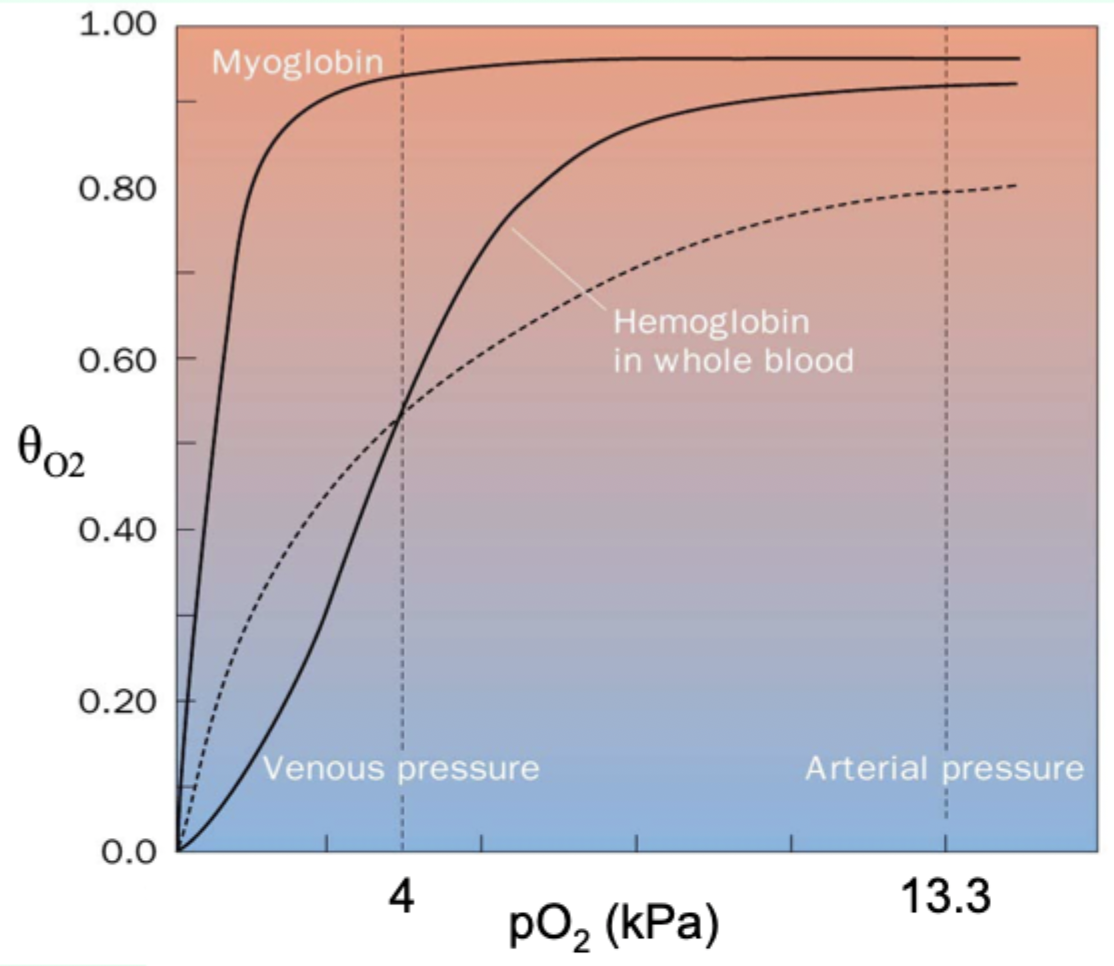
What property of Hb enables efficient loading/unloading across lung→tissue pO2?
Cooperativity via multi-subunit architecture enabling T (low affinity) ↔ R (high affinity) transition.
A “more puckered heme” protein–heme interface corresponds to which Hb state?
T state
Define “allosteric protein”.
Proteins if they have multiple binding sites (homotrophic or heterotrophic) where ligand binding at the first (allosteric) site causes a conformational change that alters binding affinity at a second site.
What is “homotrophic” allostery?
Multiple binding sites for the same ligand.
What is “heterotrophic” allostery?
Multiple binding sites where the regulatory ligand is different from the primary ligand.
A different molecule than the substrate binds to an allosteric site and modulates protein activity.
What is an “allosteric site”?
The first site where ligand binding triggers a conformational change that alters affinity at another site.
Define negative cooperativity.
Binding affinity at the second site is decreased by ligand binding at the first site.
What curve shape do you get when binding is cooperative and you plot θ vs pO2?
Sigmoidal curve.
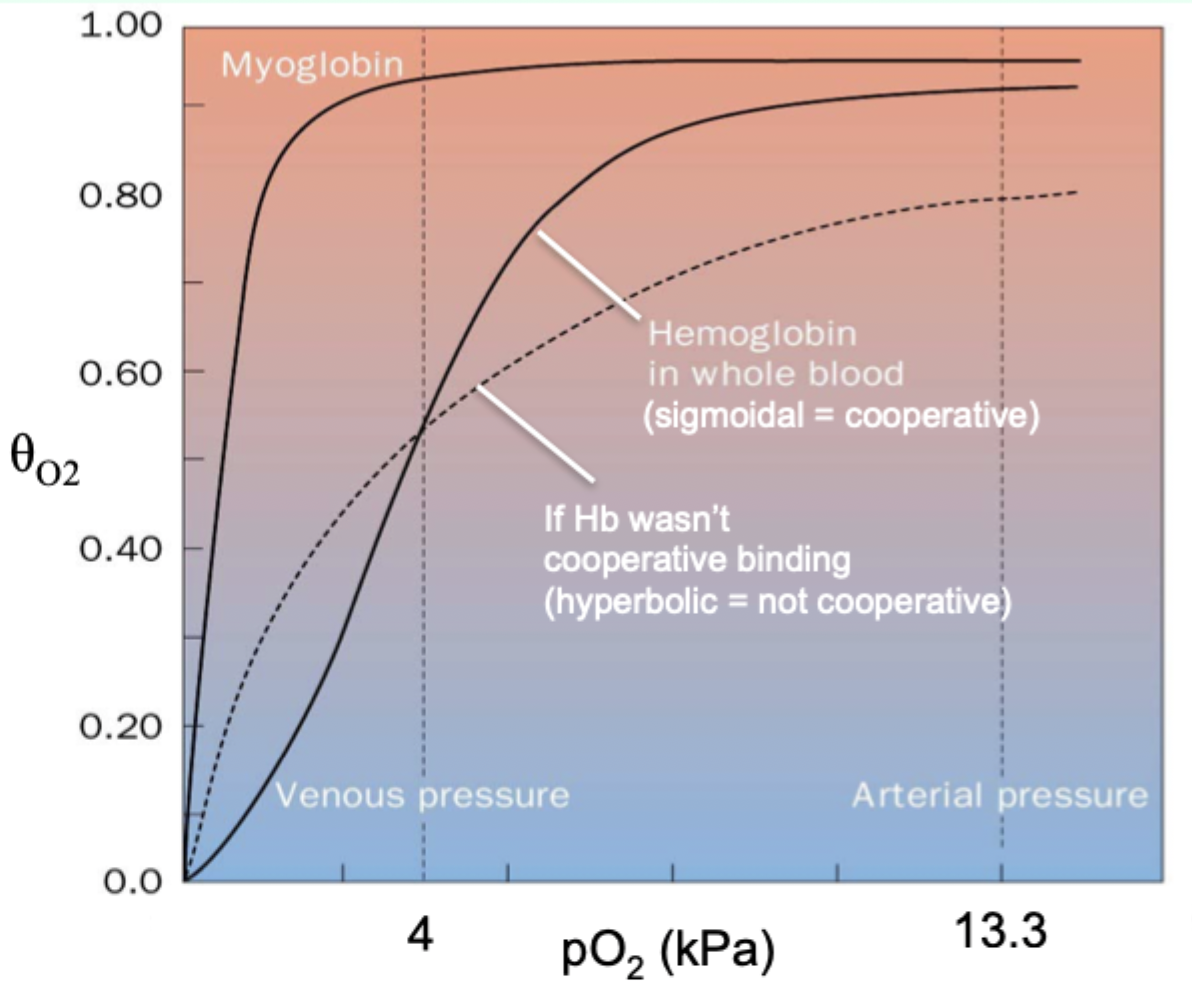
What curve shape do you get when binding is NOT cooperative?
Hyperbolic function.
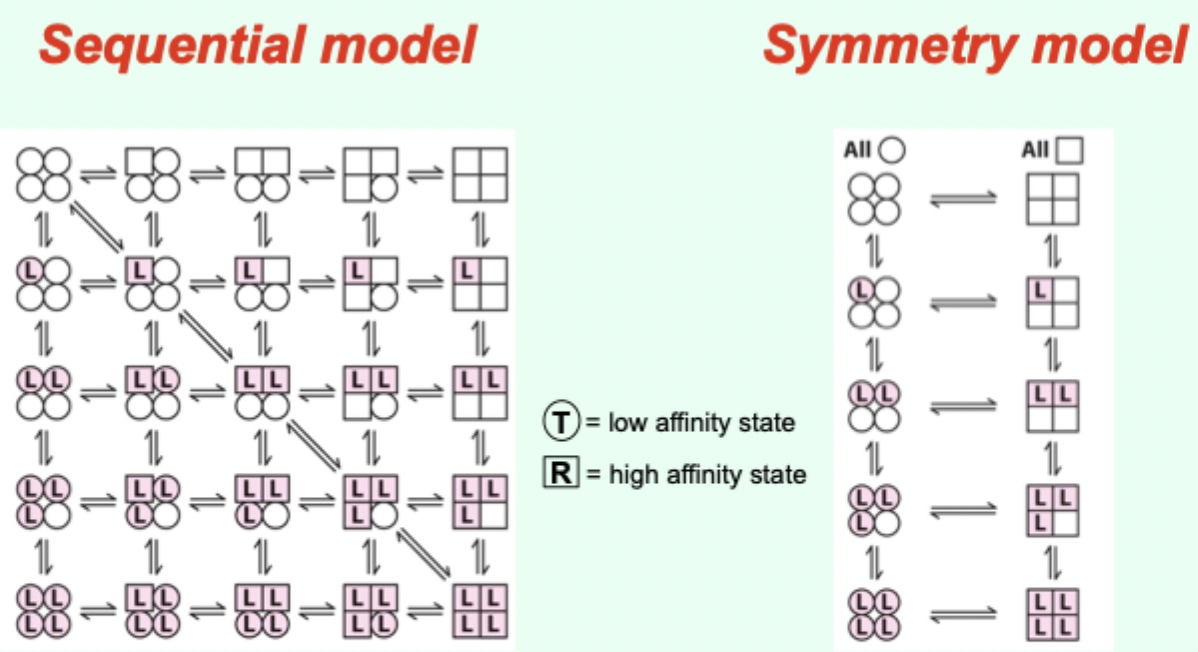
Two proposed models for cooperative binding in Hb (red text)?
Symmetry model
Sequential model.
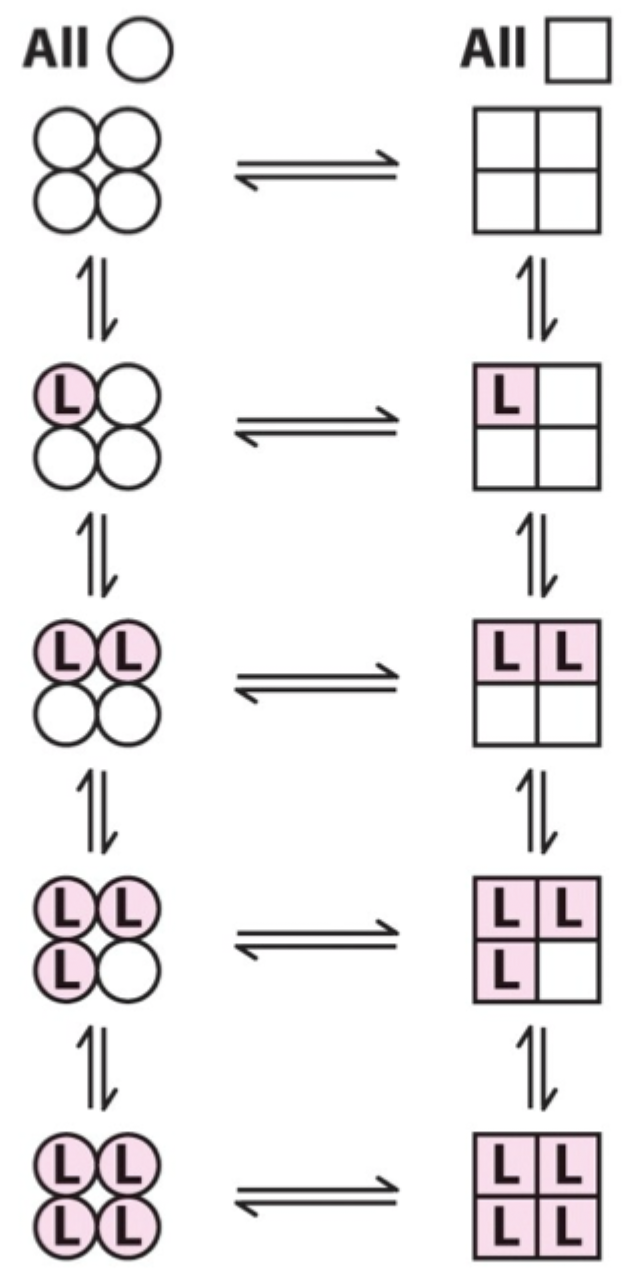
Symmetry model: what happens after one ligand binds?
Binding stabilizes the high-affinity state and alters the T↔R equilibrium, changing overall affinity for the next ligand.
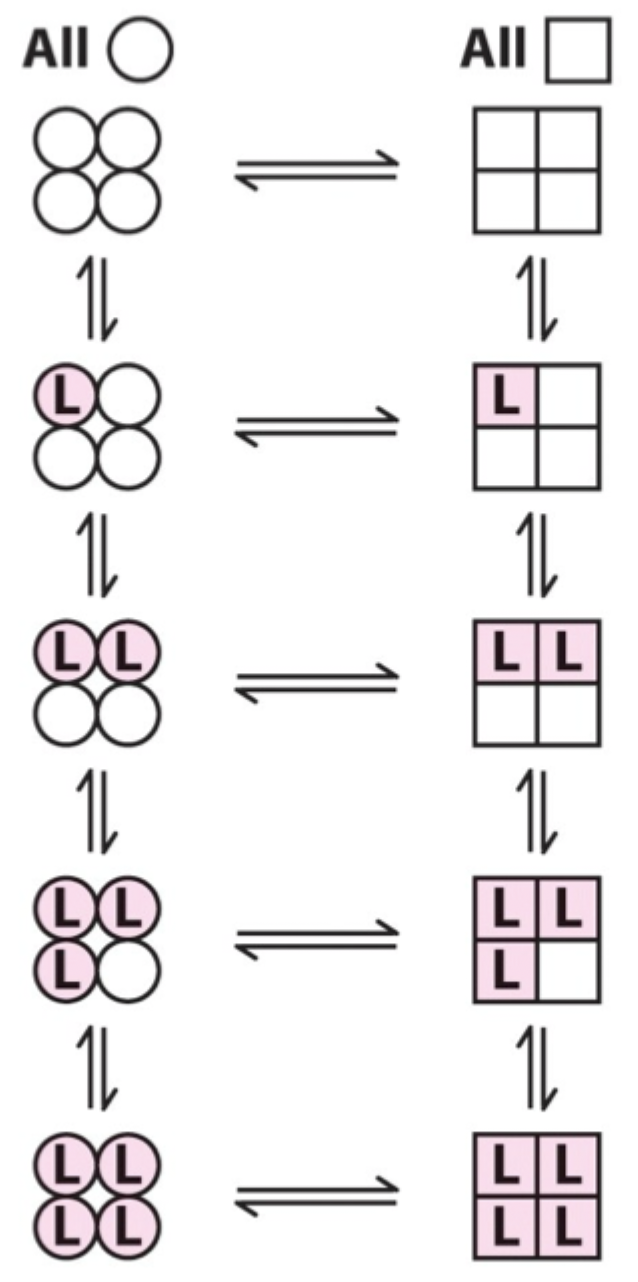
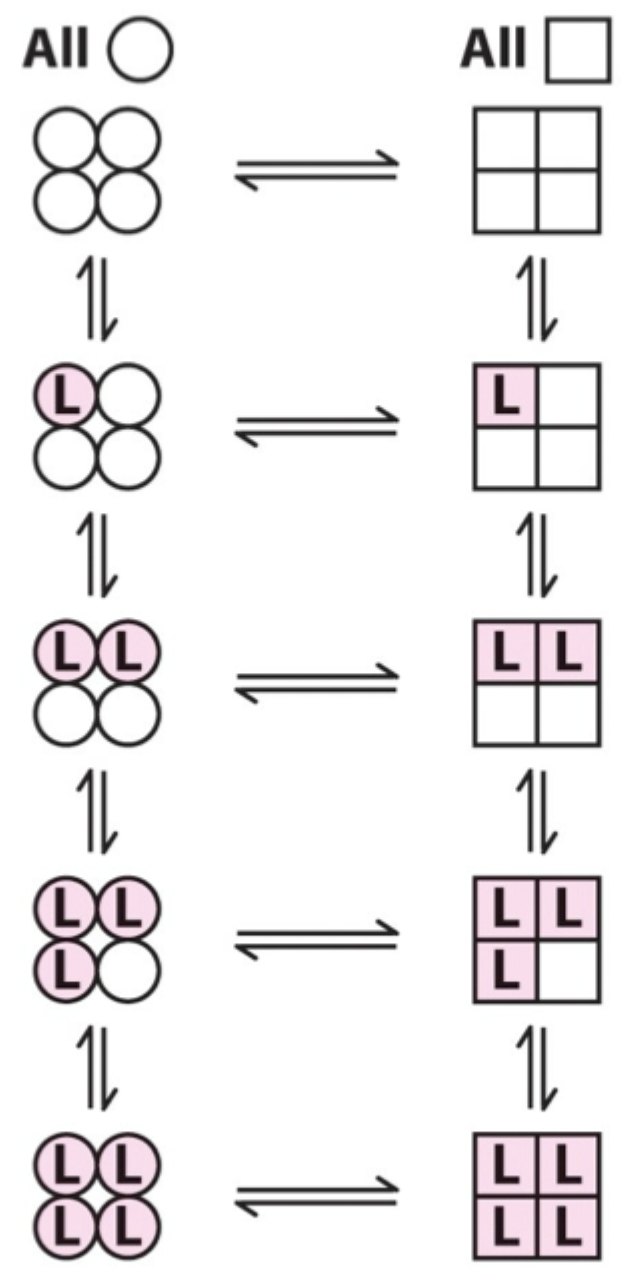
Symmetry model: what does each ligand binding event do to the equilibrium?
Moves the equilibrium in favor of the high-affinity (R) state.
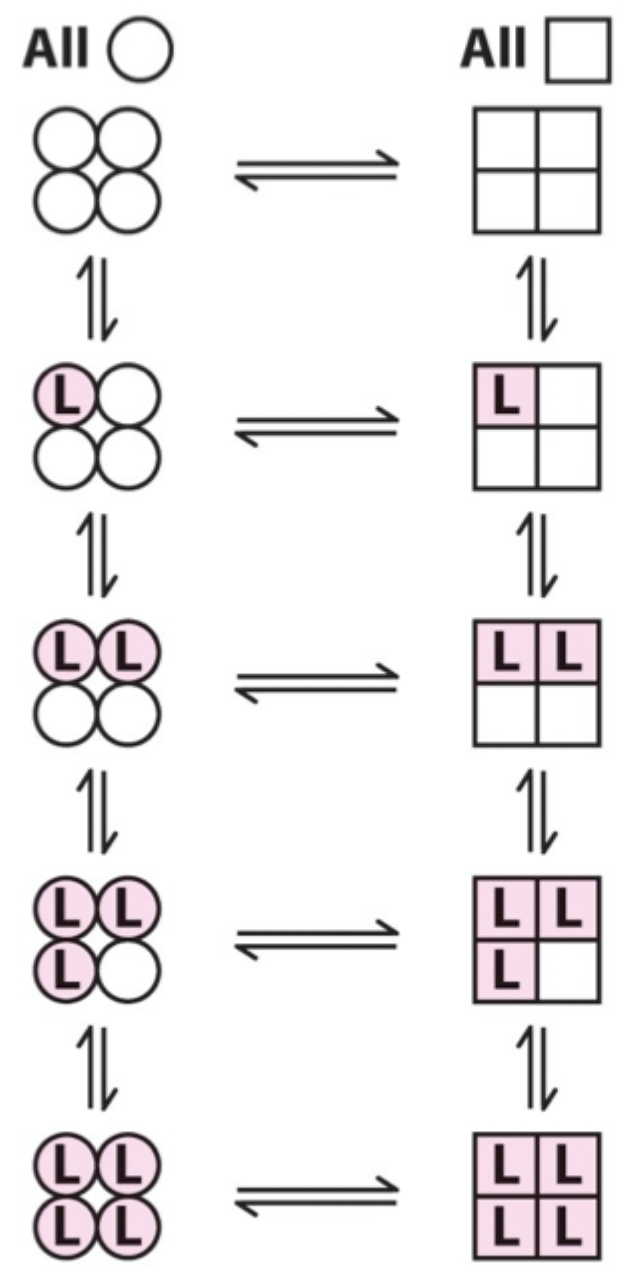
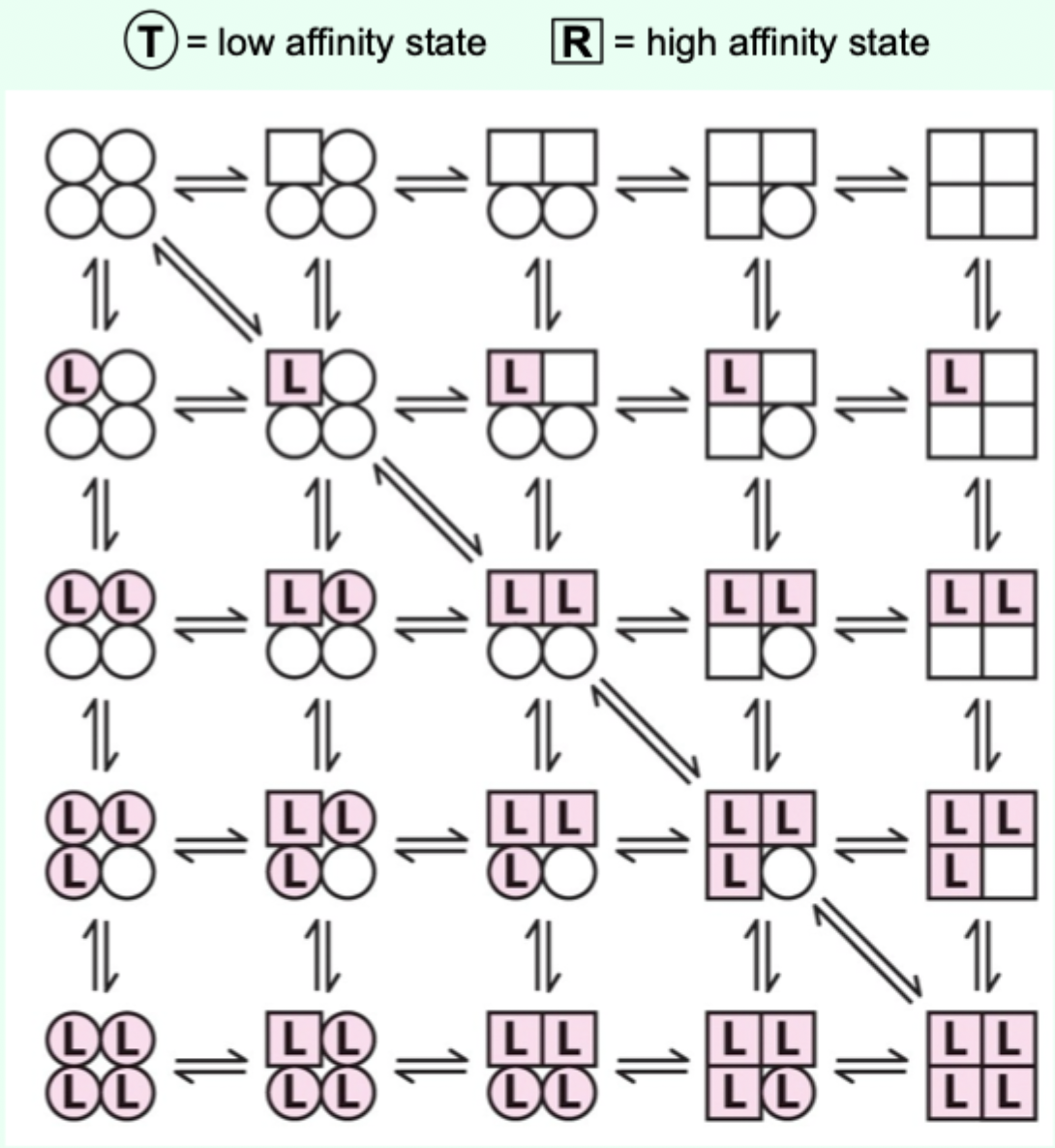
Sequential model: what happens as more ligands bind?
The greater the number of sites in the high-affinity state.
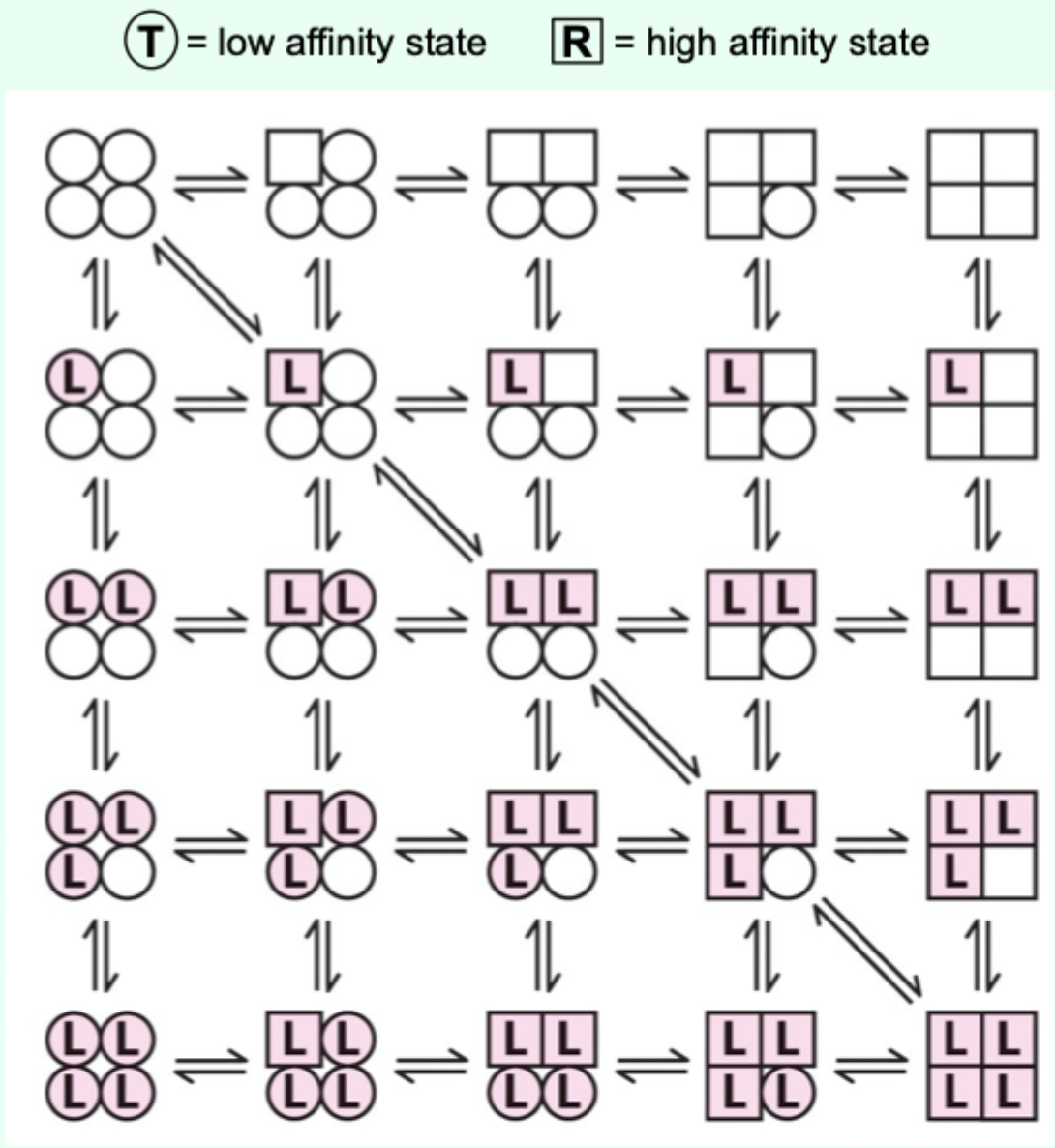
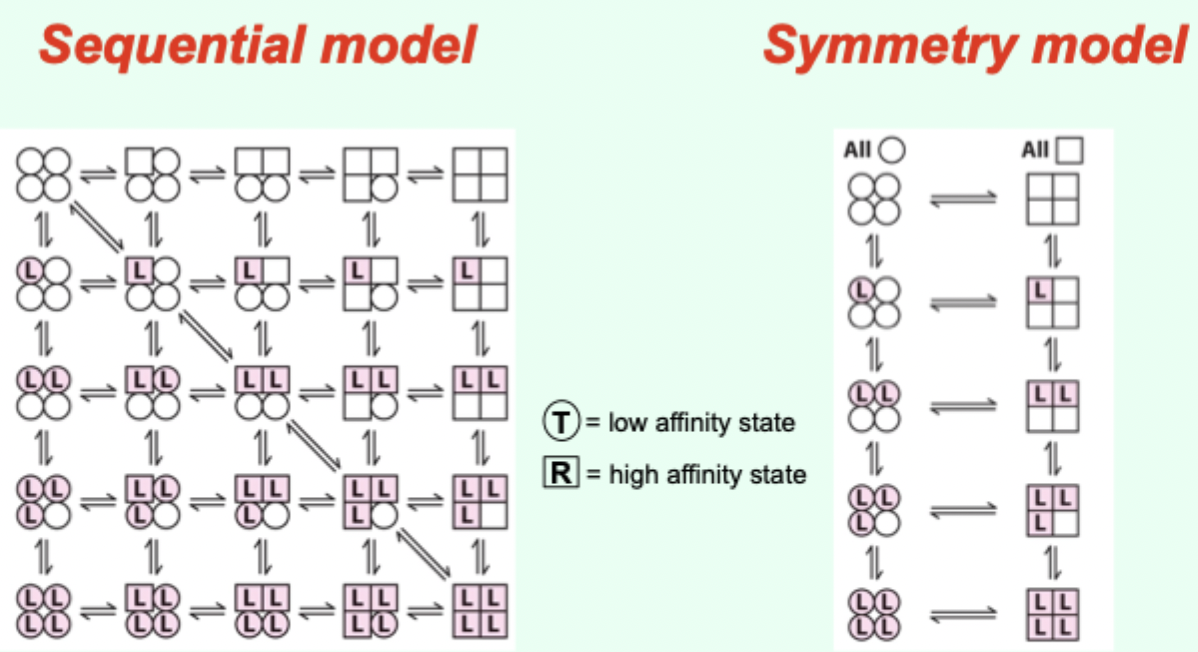
Which model is “right” for Hb?
Hemoglobin allostery has elements that fit both the symmetry and sequential models.
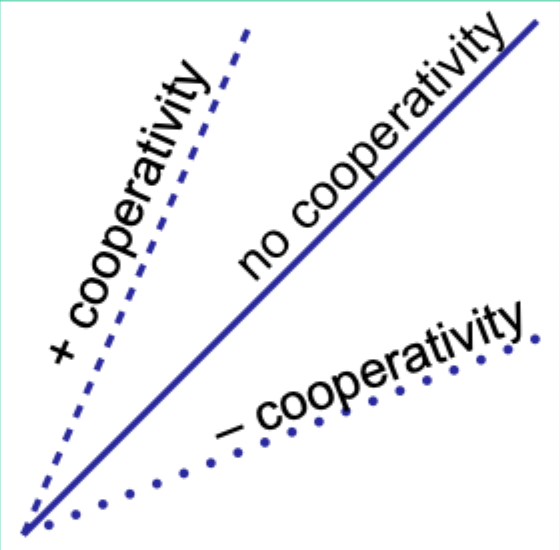
When would the Hill plot be a straight line?
If the binding affinity change is the same as each subsequent ligand binds.
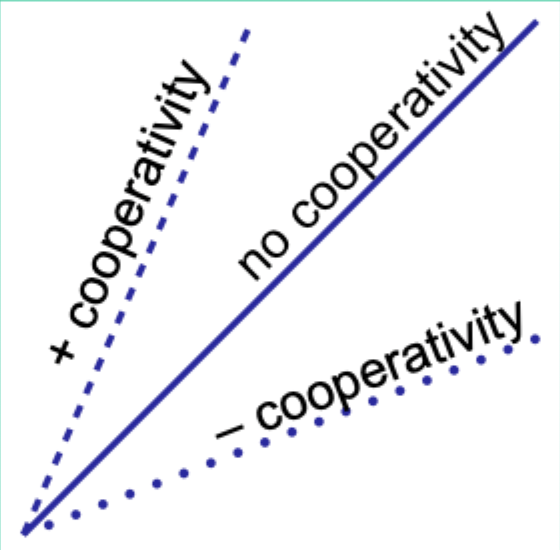
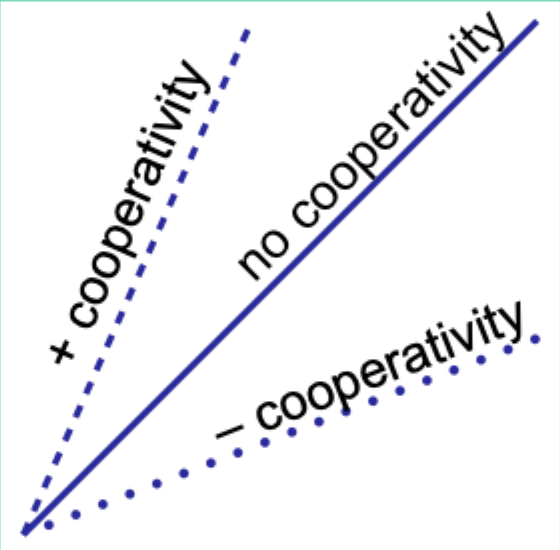
What does the Hill coefficient nH measure?
The degree of cooperativity.
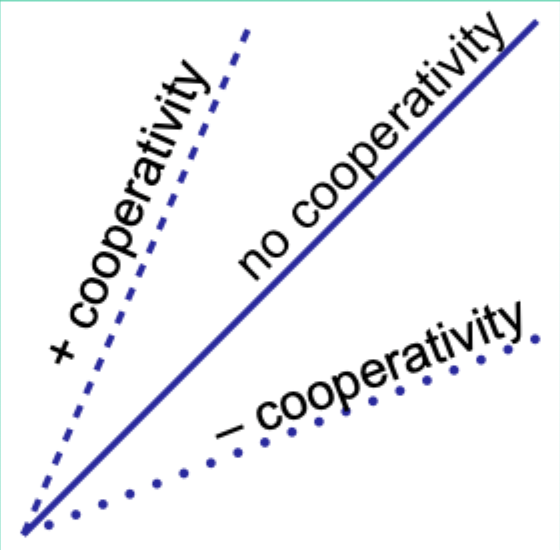
Interpret nH = 1.
No cooperativity.
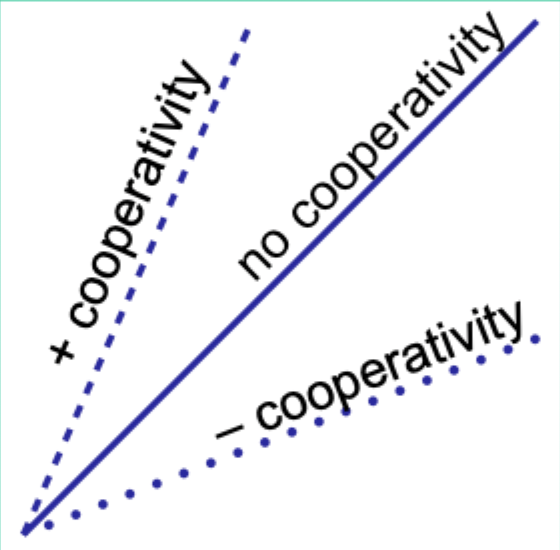
Interpret nH > 1.
Positive cooperativity.
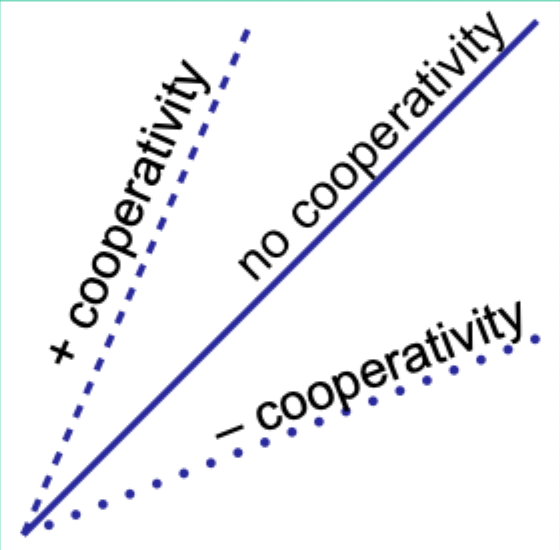
Interpret nH < 1.
Negative cooperativity.
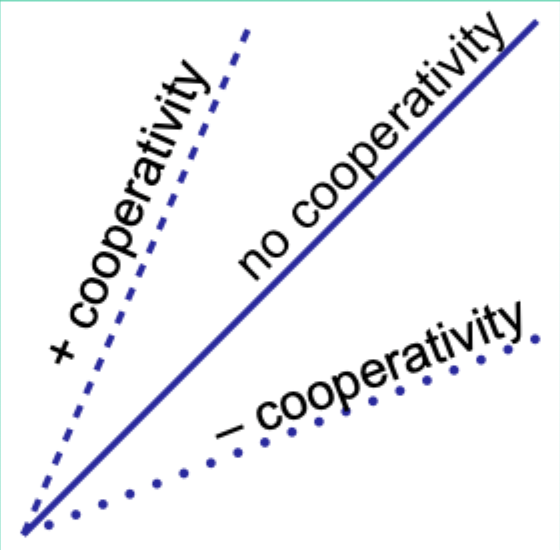
Careful note: is nH equal to n (number of binding sites)?
No. nH ≠ n.
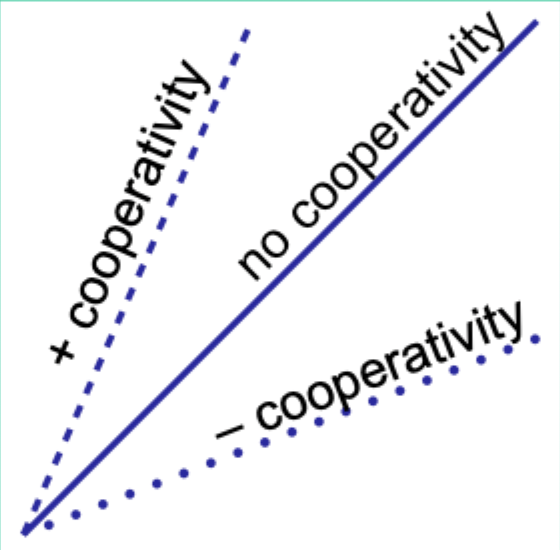
Experimentally, how does nH compare to the actual number of binding sites?
nH is always less than the actual number of binding sites.
Curve-shape link: what does hyperbolic vs sigmoidal indicate?
Hyperbolic = non-cooperative binding (e.g., myoglobin).
Sigmoidal = cooperative binding (e.g., hemoglobin).