Equilibrium
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Equilibrium Basic Meaning
Balance between things
Types of Equilibrium
Thermal Equilibrium — Heat flow from hot to cool, until temperature is equal
Chemical Equilibrium — Balance between reactants and products in a chemical reaction
Mechanical Equilibrium — An object which is in a state of no movement (no movement caused by itself)
Physical Equilibrium — Balance between states of matter
Thermal Equilibrium
Heat flow from hot to cool, until temperature is equal
Mechanical Equilibrium
An object which is in a state of no movement (no movement caused by itself)
Physical Equilibrium
Balance between states of matter
Two Reaction Types
Reversible — products can reform reactants
Irreversible — Product’s cannot reform reactants
Reversible
A reaction where the products can reform into the reactants
Double sided
Irreversible
A reaction where the Product’s cannot reform reactants
Single sided
Types of Systems
Open — Particles can move & Gain energy with/from surroundings
Closed — Particles confined to container, no outside movement of particles, but energy transfer still possible
Open System
Particles can move & Gain energy with/from surroundings
Closed System
The Particles are confined to the container, cannot leave container
Energy transfer still possible
Closed System allows for reaching of Equilibrium
Prevents leaving of matter, keeps set concentration
Chemical Equilibrium
Balance between reactants and products in a chemical reaction
2 Requirements:
Rates of forward & reverse reactions are equal
Concentrations of reactants & products remain constant
It is Dynamic (Always occuring)
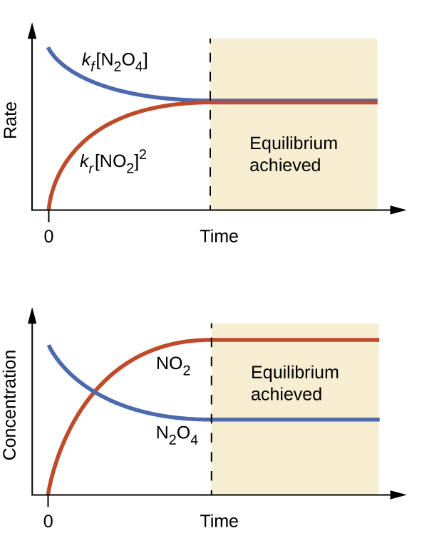
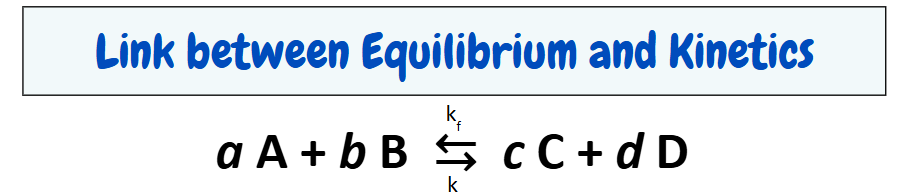
The Law of Mass Action
once at equilibrium, the rate of the reaction in either direction is dependent only on the “active masses” of the reactants and products.
Rate (Forward) = Rate (Reverse)
Leads to Equilibrium Constant
Equilibrium Constant (KC)
Only compounds in the gaseous (g) and aqueous (aq) states are allowed.
All Values must be equilibrium values.
The value of K depends on the particular reaction AND on the temperature.
Concentration or Partial pressures
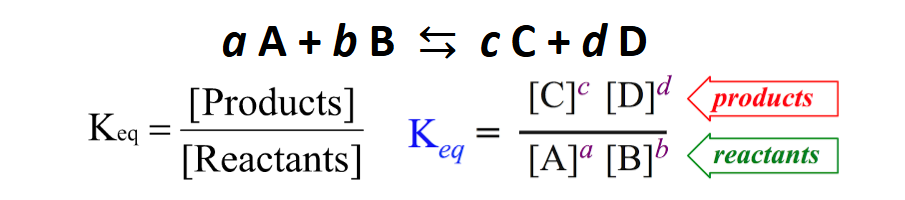
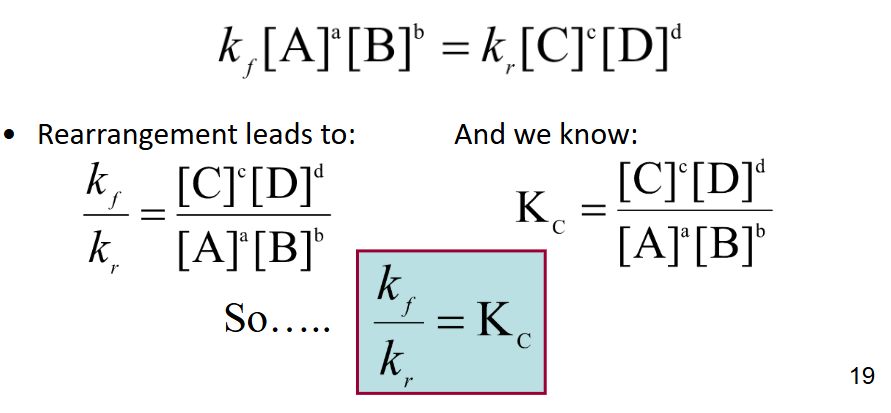
If KC>1
more products than reactants at EQ.
Formation of Products is favored at Equilibrium
If KC<1
more reactants than products at EQ.
Formation of Products is not favored
Uses of Chemical Equilibrium Equation
Calculate the Value of K
If you have the equilibrium concentration or pressure values for the reaction components, you can calculate the K value
Determine if the Reaction is at Equilibrium
If the ratio of products to reactants equals the Keq value for a given reaction, then you know the system is at equilibrium
Find Unknown Equilibrium Concentrations
If some concentrations of the reaction components are known at equilibrium, the the K value can be used to calculate an unknown.
Reaction Quotient (Q)
Ratio of Products to Reactants
NOT ALWAYS AT EQULIBRIUM
Same equation
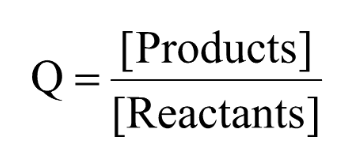
When Q<K
Less Products than at equilibrium
Will favor forward reaction
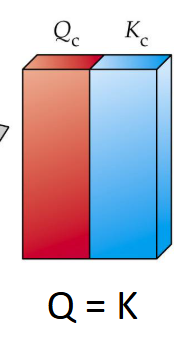
When Qc = Kc
Then System is at equilibrium
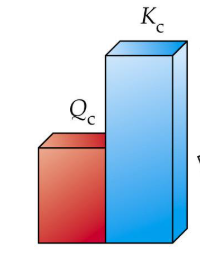
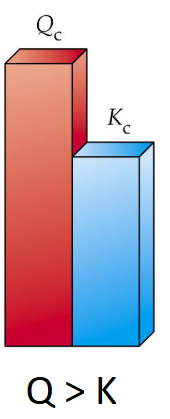
When Qc > Kc
System has more products than equilibrium
Will favor reverse reaction
Rule of Multipole Equilibria
“If a reaction can be expressed as the sum of two or more reactions, the K for the overall reaction is the product of the K values for the individual reactions”
Simply: You can manipulate elementary steps to get overall reactions. What ever you manipulate must be done to the KC values, as well. Then you multiple the Kc values for steps, to get Kc for final
What happens to Kc when equation is flipped?
Kc becomes reciprocal (Kc-1)
What happens to Kc when equation multiplied?
K goes to power of coefficient (nKc)
5% rule
For RICE tables
used when solving for x becomes polynomial
Can make assumption that X is so small it does not need to be calculated
Remove it when added to another value NOT ALONE
After solving, check X is less than 5% of Initial value (All moles)
If not valid, solve polynomial
Always add both initial concentrations (if 2)
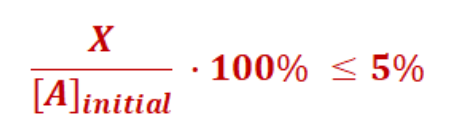
Le Chateleir’s Principle
when a reversible reaction at equilibrium is ‘stressed’ by a change in reaction conditions, the reaction direction must adjust to relieve the stress and bring the system back to equilibrium.
Simply: When something done to one side, the reaction must rebalance itself to re-reach new equilibrium
Depends on equation for Q
How will equation change, directly afterwards
3 methods of Stress:
Changes in Concentrations
Changes in Temp
Changes in Volume
Le Chateleir’s: Adding substance
Will go in opposite direction of substance
+ reactant → increase product
+ product → increase reactant
Le Chateleir’s: Removing substance
Will go in direction of substance
- reactant → decrease product
- product → decrease reactant
Le Chateleir’s: Effect of Temperature
Temp = Energy
Energy treated like addition/subtraction of substance
ONLY METHOD TOP CHANGE KC VALUE OF REACTION
How to change Kc value of an expression
CAN ONLY BE DONE BY CHANGE IN TEMPERATURE
Le Chateleir’s: Effects of Volume
Only affects gasses
Based on Number of Moles on each side of reaction
Decrease in Volume → Reaction favors direction with fewer moles
Increase in Volume → Reaction favors direction with more moles
Equal moles → No change in equilibrium, stays same
Effect of Catalyst on system at Equilbrium
Has no Effect
Solubility Product Constant(Ksp)
Measures how much a solid dissolves into ions at equilibrium
Same equation as Kc
Only for insoluble salts
When Writing chemical Equation, Precipitate always Reactant
Lower Ksp means less soluble
Vice Versa
Ion Solubility Product Qsp
Equivalent of the reaction Quotient (Qc)
If Qsp < Ksp
Unsaturated
Will not form precipitate
If Qsp=Ksp
Saturated
Right amount of Ions present
if Qsp >Ksp
Supersaturated
Precipitate Forms
Common Ion Effect
Increasing the concentration of one of the ions by adding a second solution puts stress on the equilibrium.
Selective Precipitation
chemical technique that separates ions in a solution by adding a reagent that causes one or more of the ions to precipitate. The remaining ions stay in solution.
Use specific other chemicals to form a precipitate → can analyze precipitate to see unknown elements
Vapor Pressure at Equilibrium
Only affected by temperature