MIDTERM 3
1/215
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
216 Terms
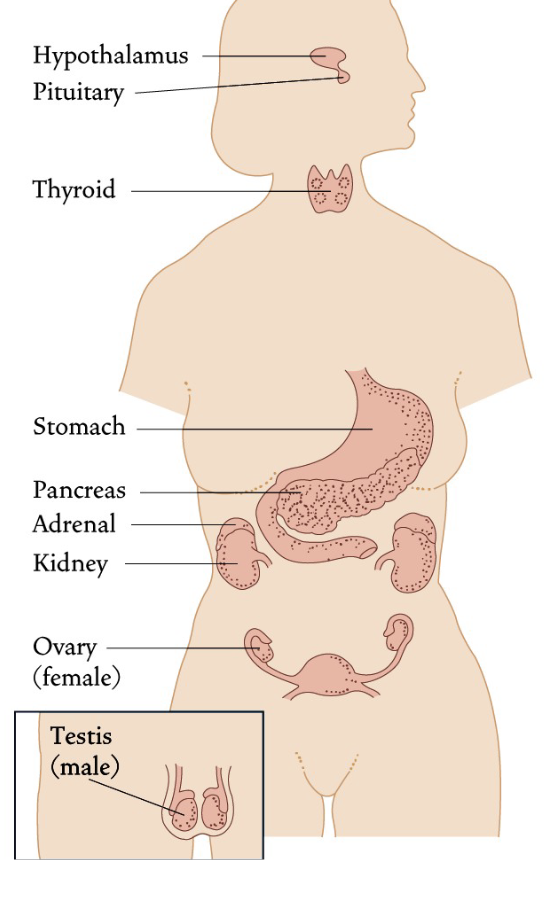
What is the endocrine system?
The endocrine system links the brain to organs that control body metabolism, growth and development, and reproduction
Growth and development: Estradiol and testosterone active during utero
Its mode of communication is via hormones
Hormones are chemical messengers that are released into the bloodstream or tissue fluid affecting target cells some distance away
Ex: Hormones produced in brain → gonads
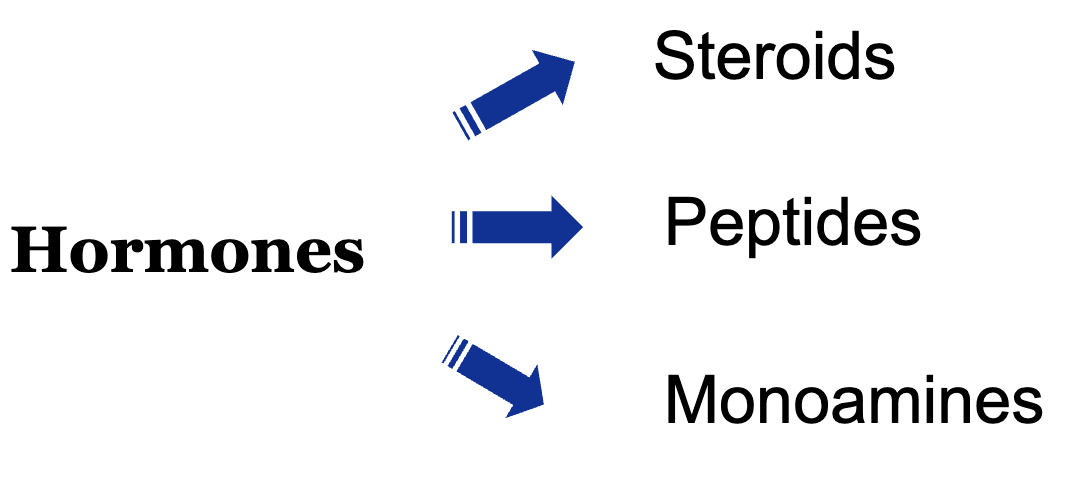
What are the different types of hormones?
Steroids
Peptides
Monoamines (smaller-sized peptides; not released from glands)
What are steroids?
Involved in Stress and Reproduction
Come from two main sources in the body
Testes/Ovaries
Adrenal Cortex
Come into the body as Cholesterol (27-C)
Fat Soluble (can’t easily move around in blood/plasma → cholesterol is cleaved to make it easier)
Small molecules: easily pass the blood-brain barrier, pass from mother to fetus, found in excretions (saliva, sweat, blood, urine, fecal samples)
Blood-brain barrier: Neuron membrane has lots of fat, so fat-soluble steroids can bypass the barrier and enter the neuron
Endocrine toxicants: Chemicals that mimic hormones (hygiene products); can gain access to the same cells and receptors as steroids
Receptors: found within the cells (cytoplasm and nucleus)
What are peptides (and monoamines)?
Major Sources
Hypothalamus
Pituitary
Tend to be larger molecules (compared to steroids)
More fragile: break up within seconds (important for biological action; steroids take longer to break down → longer impact on the cell)
Water soluble: action is much shorter
Receptors: found in the plasma membrane
What are the three groups of sex steroids?
Sex steroid hormones are lipid molecules derived from cholesterol and fall into three categories:
Androgens
Estrogens
Progestins
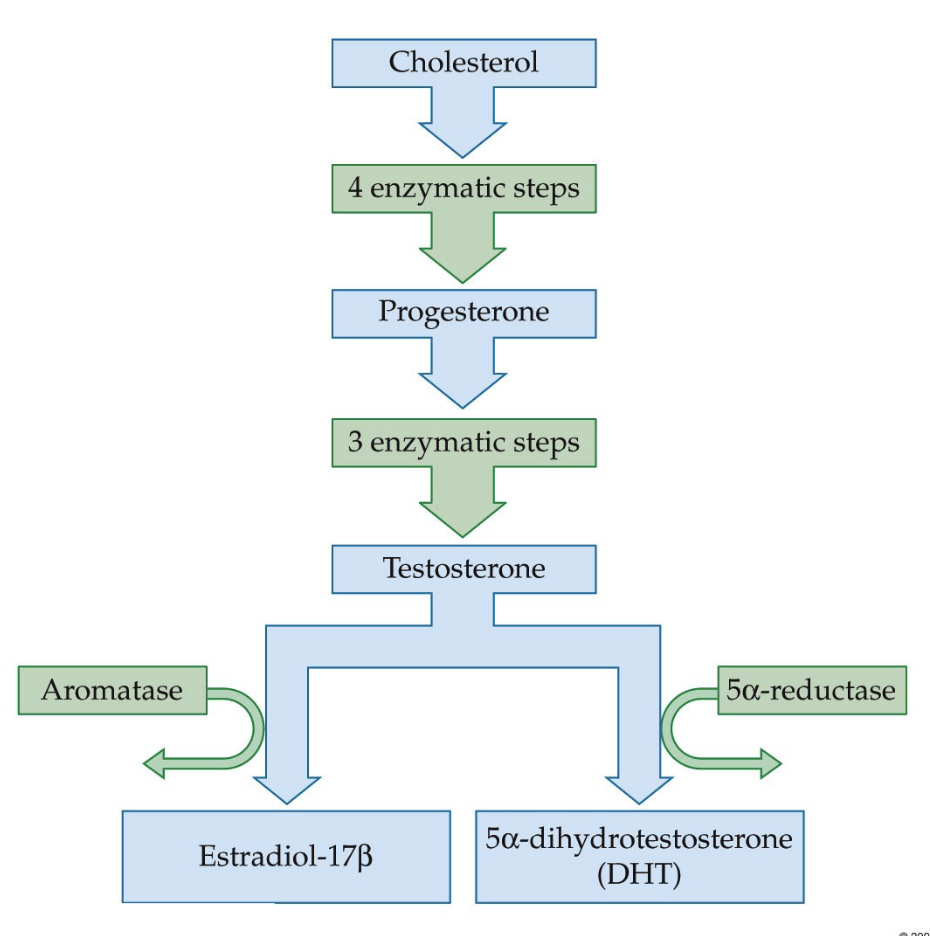
What is the steroid hormone synthesis pathway starting from cholesterol, and how do enzymes determine whether testosterone becomes estradiol or dihydrotestosterone (DHT)?
Hormones become smaller through enzymatic steps
Cholesterol (27C) → precursor molecule for steroid hormones
Progesterone (a progestin) → formed after enzymatic conversion; has fewer carbons than cholesterol
Further enzymatic steps → Testosterone (androgen)
A key branching point occurs at testosterone, where the type of enzyme present determines the final hormone produced:
Aromatase present → Testosterone → 17β-Estradiol
5α-reductase present → Testosterone → 5α-dihydrotestosterone (DHT)
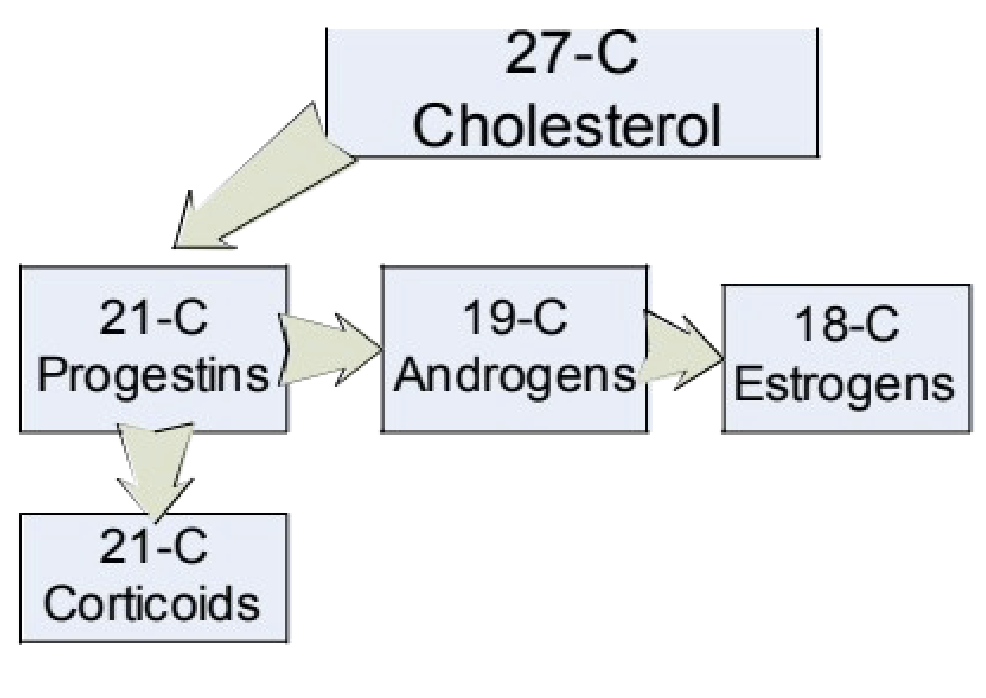
How do the number of carbons change across the steroid hormone pathway, and what hormone classes correspond to each carbon structure?
Steroid hormones derived from cholesterol (27C)
Carbon number decreases during synthesis
27C → Cholesterol
21C → Progestins (progesterone)
19C → Androgens (testosterone)
18C → Estrogens (estradiol)
21C can also form corticoids depending on attached groups
Example: Cortisol = 21-carbon corticoid
Hormone class determined by carbon backbone + attached chemical groups
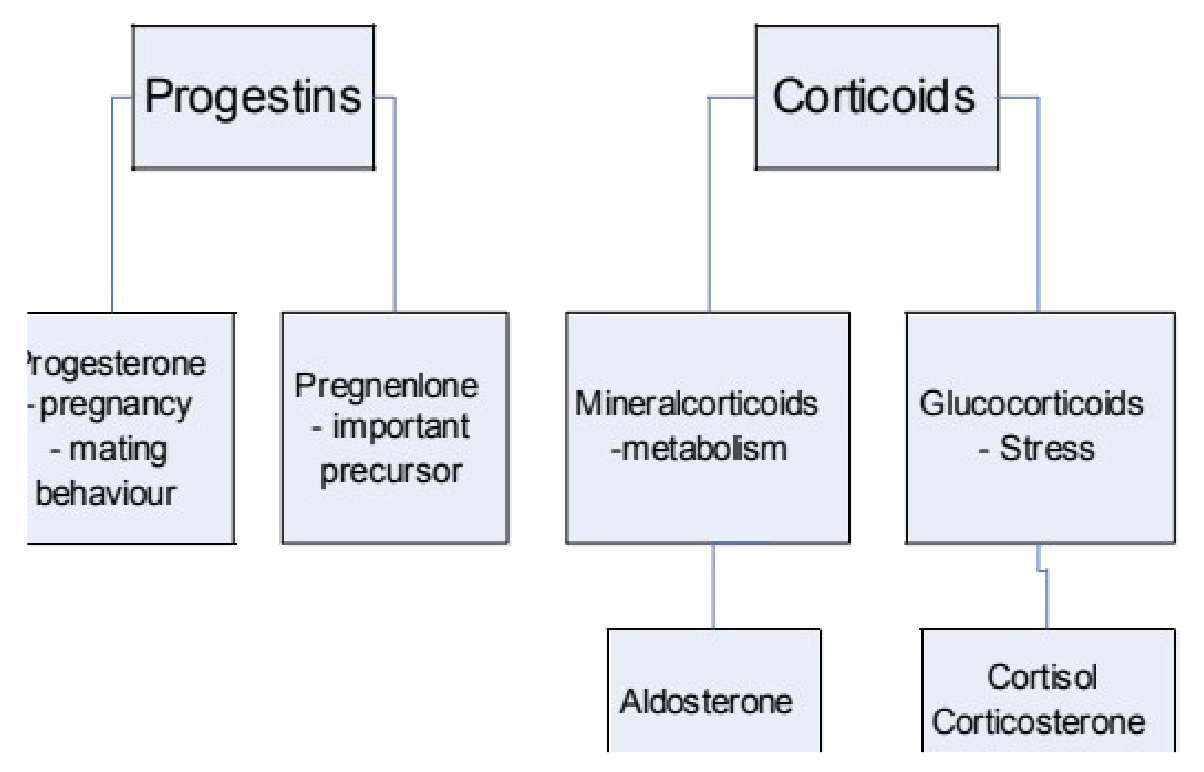
What roles do 21-carbon steroids play, and what are the key hormones and functions within the progestin and corticoid categories?
Progestins:
Progesterone: Pregnancy and mating behaviour (many behavioural findings from laboratory animal studies)
Pregnenolone: Important precursor to progesterone and a precursor for conversion from androgens to estrogens.
Corticoids:
Glucocorticoids: Stress response
Cortisol = main glucocorticoid in humans
Corticosterone = equivalent glucocorticoid measured in laboratory animals (e.g., rats, mice)
Mineralocorticoids: Metabolism (regulate electrolyte, salt, and water balance)
Aldosterone
What are 19-C Steroids?
Androgens → Two major examples: Testosterone + 5a-dihydro-testosterone (DHT)
DHT plays a critical function in the sexual development of XY individuals, beginning early in prenatal life.
The role of DHT differs as the person progresses through the different stages of development.
It has various impacts on their physiology during childhood, puberty, and even throughout adult life.
**In utero, androgens have an impact on male reproductive system for development AND the brain, specifically the hypothalamus (might contribute to sexual orientation).

What are 18-C Steroids?
Estrogens → 17B-estradiol, estrone, estriol
Estrone is the weakest type of estrogen, it’s typically higher after menopause (because estrogen and estradiol are lower). Like all estrogen, estrone supports female sexual development and function.
Estriol is one of three estrogen hormones. Estriol levels rise throughout pregnancy, helping to keep the uterus and unborn baby healthy. The levels are at their highest right before childbirth. They help prepare the body for labor and delivery.
Everyone makes estriol. But in people who aren’t pregnant, the levels are almost undetectable.
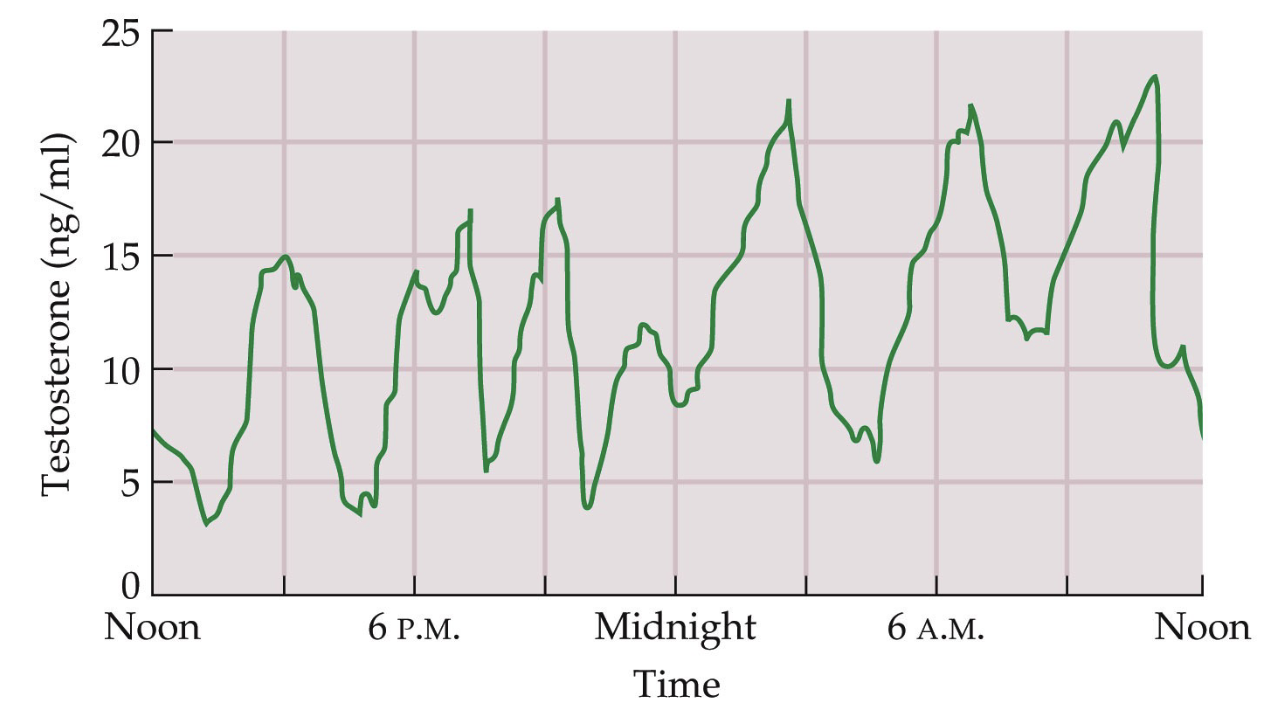
Why is it difficult to define a single “normal” level for reproductive hormones like testosterone, and how are they typically measured in research?
Reproductive hormones do not have a single fixed “normal” level
Levels fluctuate widely over time
Variation occurs:
Across the day (circadian rhythm)
Across menstrual cycles
During pregnancy
Example: testosterone rises and falls throughout the day
Researchers measure patterns (peaks & drops) rather than single values
Hormone sampling often done multiple times per day
e.g., 8 AM, 12 PM, 4 PM, 7 PM
Can be measured in saliva samples
Healthy system = cycling levels
Extremely high or low levels may indicate pathology
Example pathology: PCOS → elevated androgens
What is the pulsatile nature of hormone release, and what supports the follicles?
Hormones are released in bursts rather than continuously. Cycling produces periods of higher and lower hormone levels.
In the ovary, follicles develop to support egg maturation.
A follicle is a structure (container-like) that holds an immature egg (oocyte).
Surrounding the oocyte are granulosa cells, which support egg development, and convert androgens → estrogens
Outside the granulosa cells are thecal cells, which produce androgens from cholesterol.
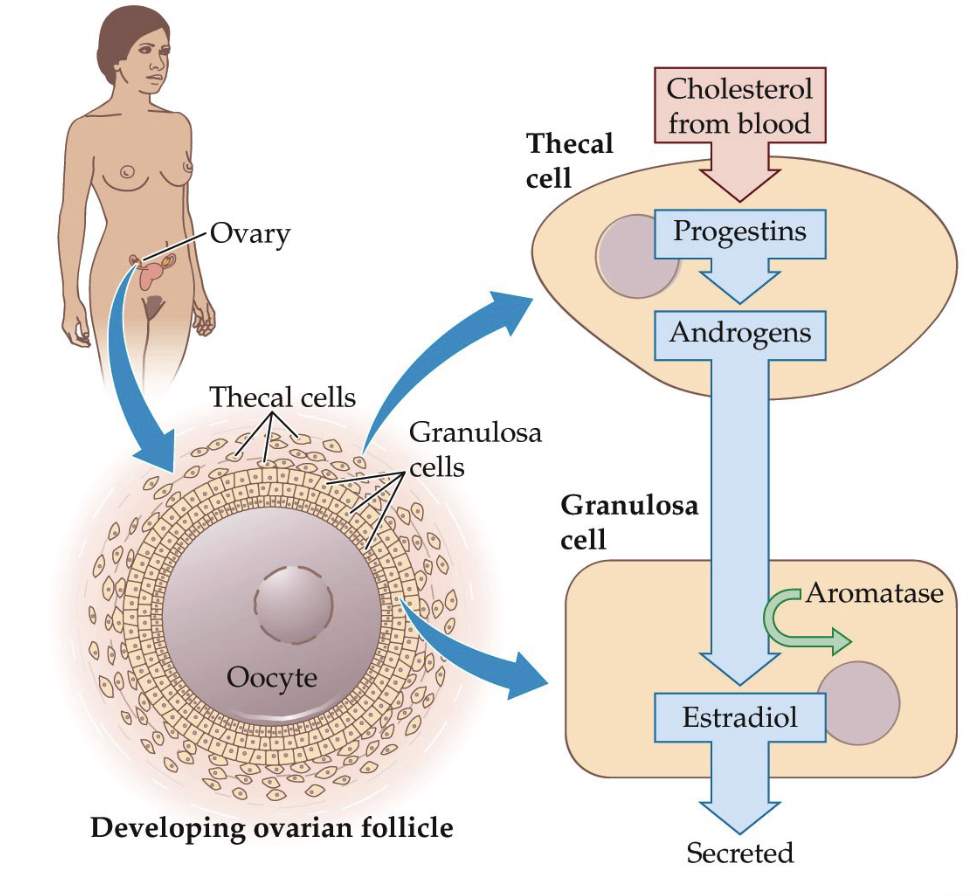
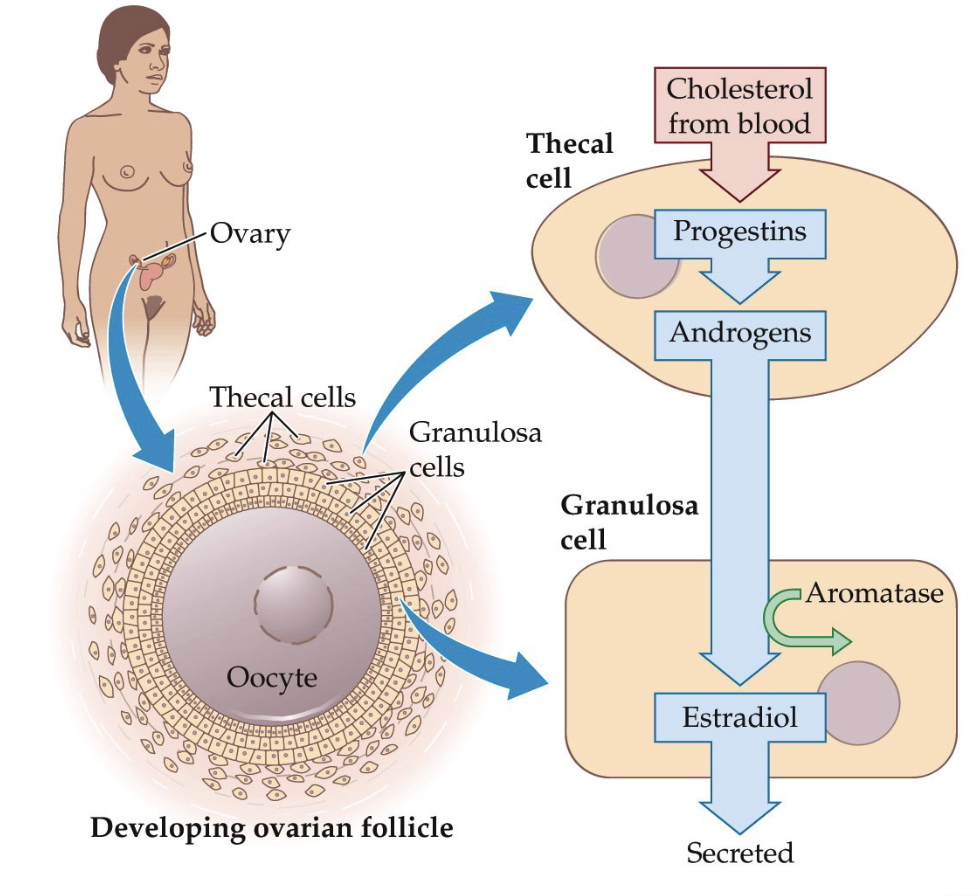
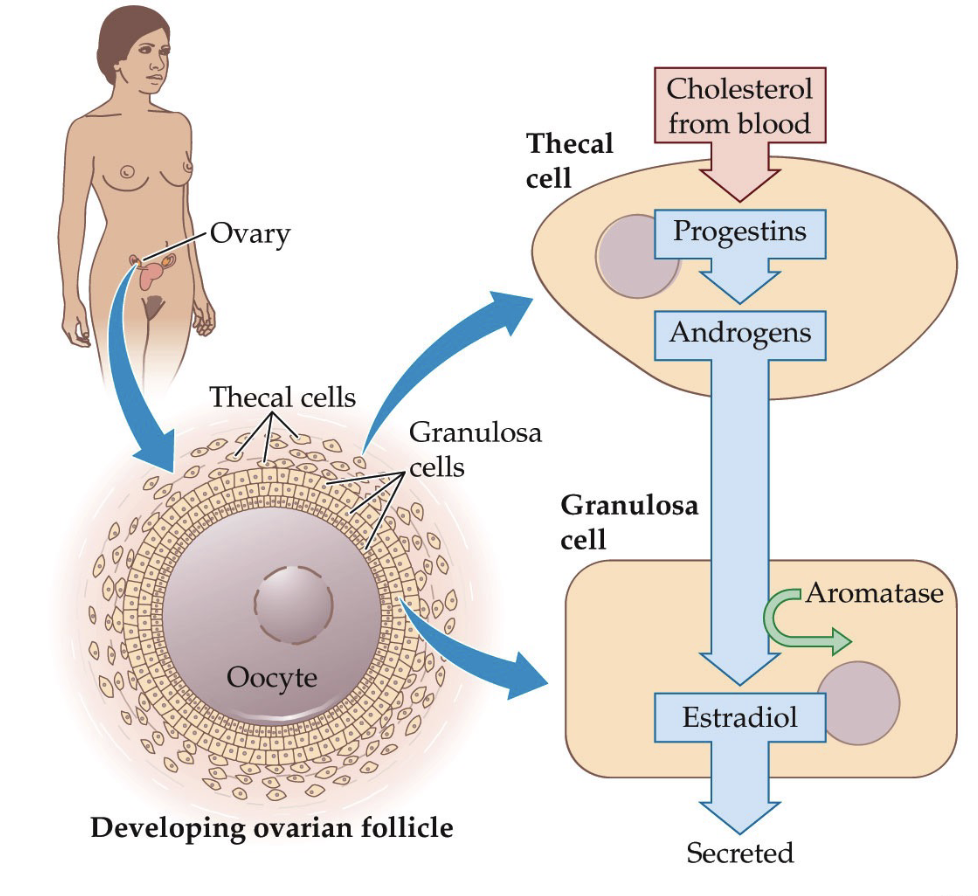
How do ovarian follicles develop and produce estradiol, including the roles of thecal cells and granulosa cells?
Steroid hormone production within the follicle:
Cholesterol from the bloodstream enters thecal cells.
Thecal cells convert cholesterol → progestins → androgens (e.g., testosterone) through enzymatic steps.
Testosterone diffuses into nearby granulosa cells.
Aromatase in granulosa cells converts testosterone → estradiol.
Estradiol is released, supporting follicle growth and egg maturation.
Explain egg development and ovulation.
All immature eggs (oocytes) are present at birth.
At puberty, each cycle recruits several immature follicles.
Usually only 1–2 follicles mature and ovulate each cycle.
The follicle ruptures to release the mature egg.
If fertilization does not occur, the egg breaks down and the cycle repeats monthly.
How does this connect to PCOS?
PCOS involves many follicles and elevated androgens (e.g., testosterone).
Diagnosis involves hormone levels, ultrasound imaging, and symptoms such as excess facial/body hair (hirsutism).
What hormones do the granulosa cells produce?
The granulosa cells also appear to be the source of two peptide hormones.
Inhibin and activin are important in suppressing and enhancing, respectively, hormone secretion from the hypothalamus and pituitary gland.
What are the two parts of the pituitary gland?
Pituitary gland = two parts
Anterior pituitary (front)
Posterior pituitary (back)
Both release peptide hormones
Function differently in regulation and signaling
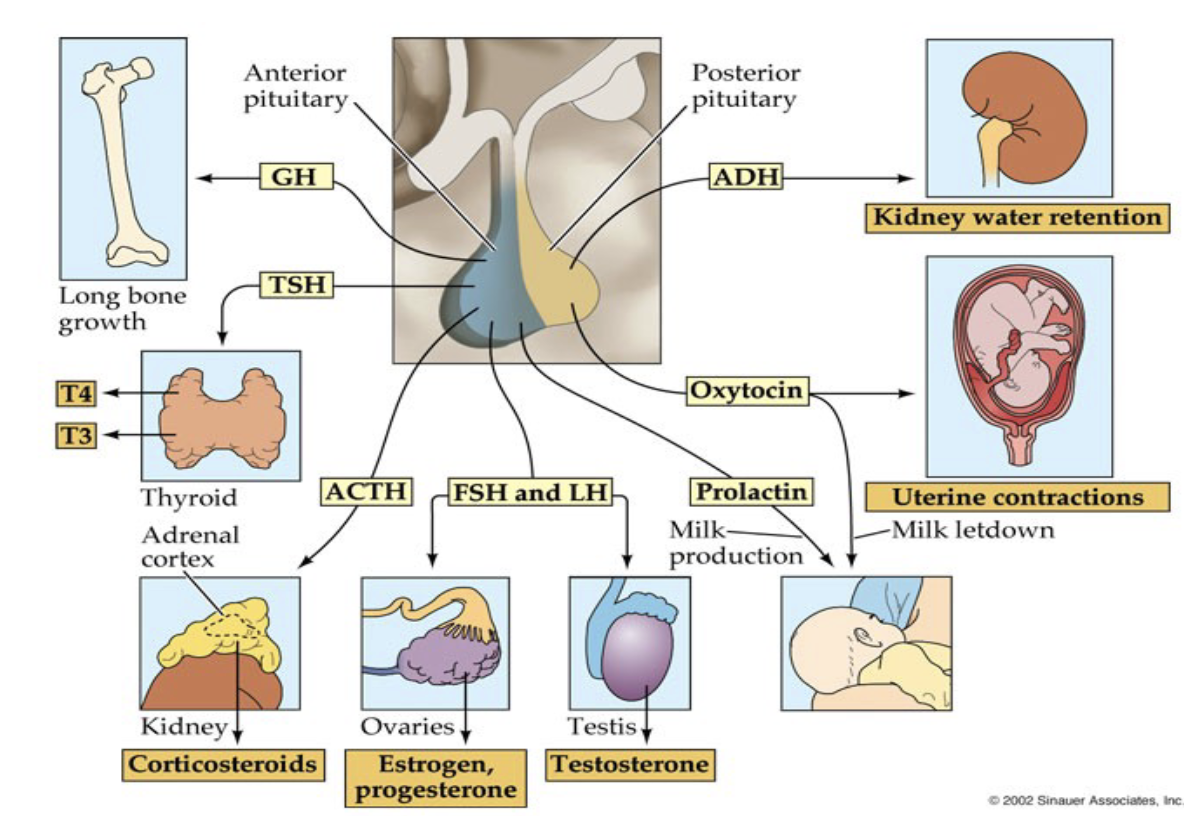
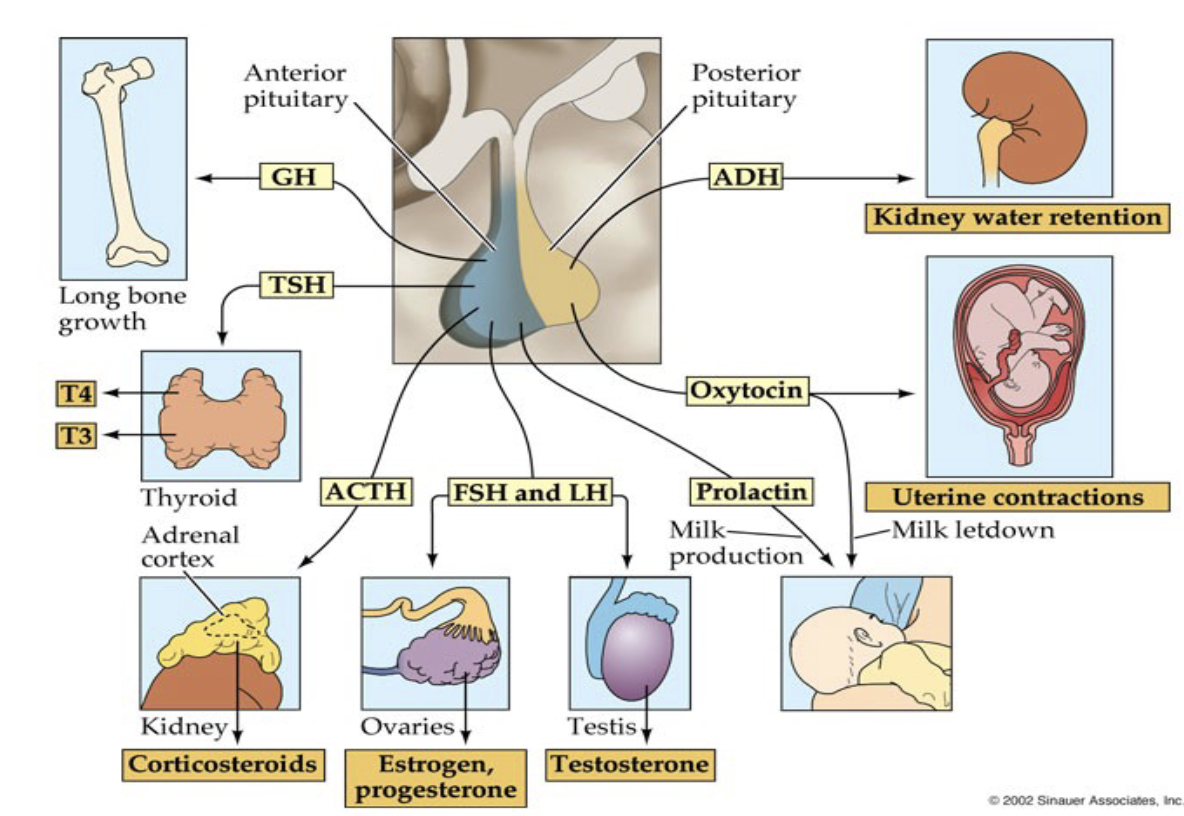
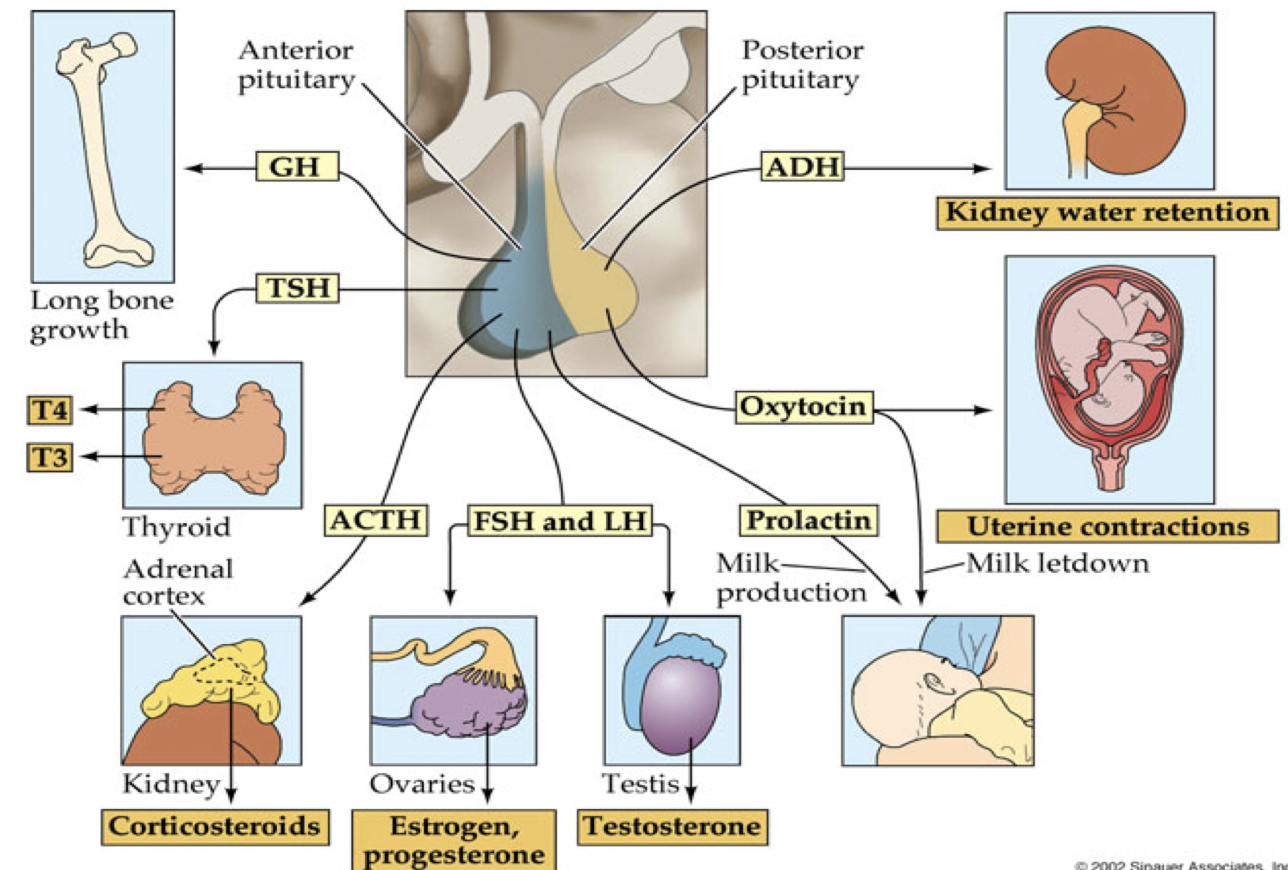
Which peptide hormones does the Anterior Pituitary release?
GH (Growth Hormone)
Important for body growth and development
Supports healthy development of the reproductive system
TSH (Thyroid Stimulating Hormone)
Stimulates thyroid gland
Regulates metabolism and body-wide physiological function
ACTH (Adrenocorticotropic Hormone)
Stimulates adrenal cortex
Causes release of corticosteroids
FSH (Follicle Stimulating Hormone) & LH (Luteinizing Hormone)
Key reproductive hormone
Primary drivers of these systems: Steroid hormones (Estrogen, progesterone, testosterone)
Prolactin
Important for mammary gland function
Stimulates milk production (lactation)
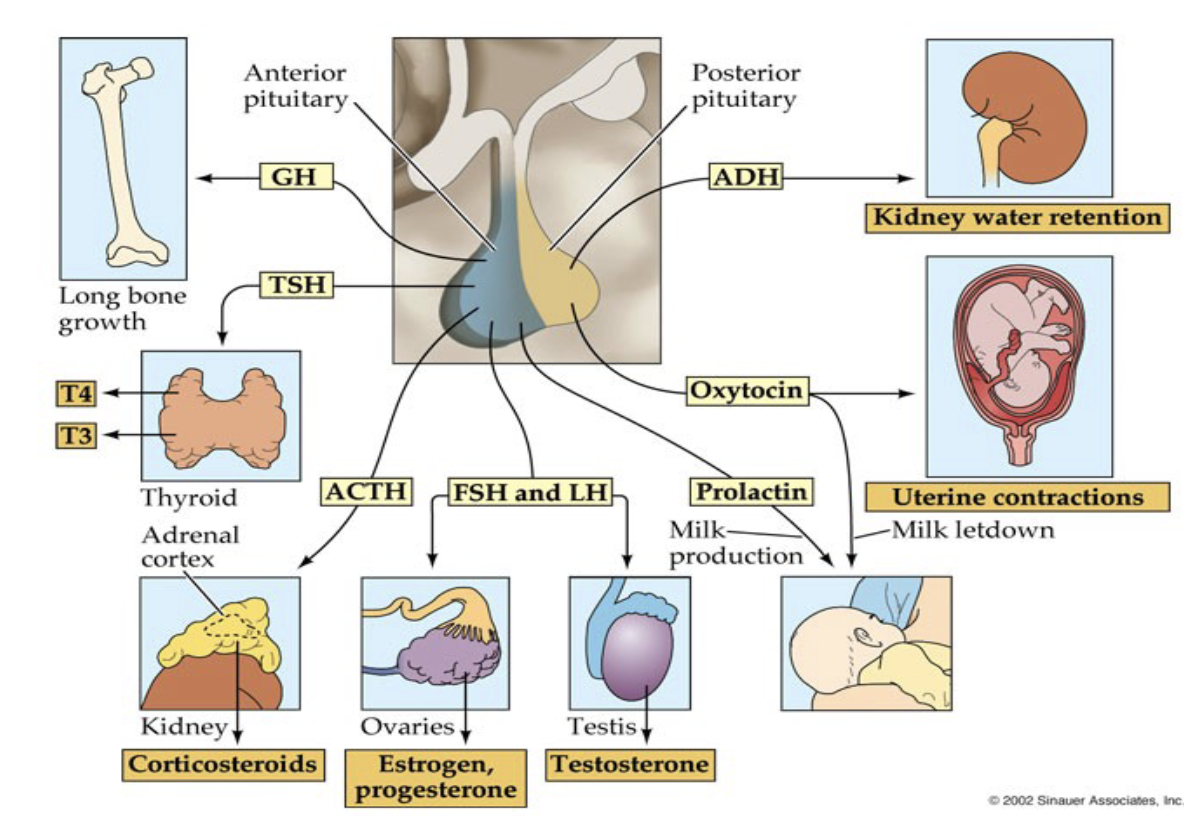
Which peptide hormones does the Posterior Pituitary release?
Oxytocin
Causes uterine contractions during childbirth
Also released during orgasm
May help facilitate sperm movement toward uterus and fallopian tubes
ADH (Antidiuretic Hormone)
Regulates water retention
Maintains fluid balance and metabolism
How does the hypothalamus connect to the anterior vs. posterior pituitary?
Pituitary–Hypothalamus Connection
Hypothalamus located above pituitary gland
Pituitary has two lobes (parts):
Anterior pituitary
Posterior pituitary
Each connects to the hypothalamus in different ways
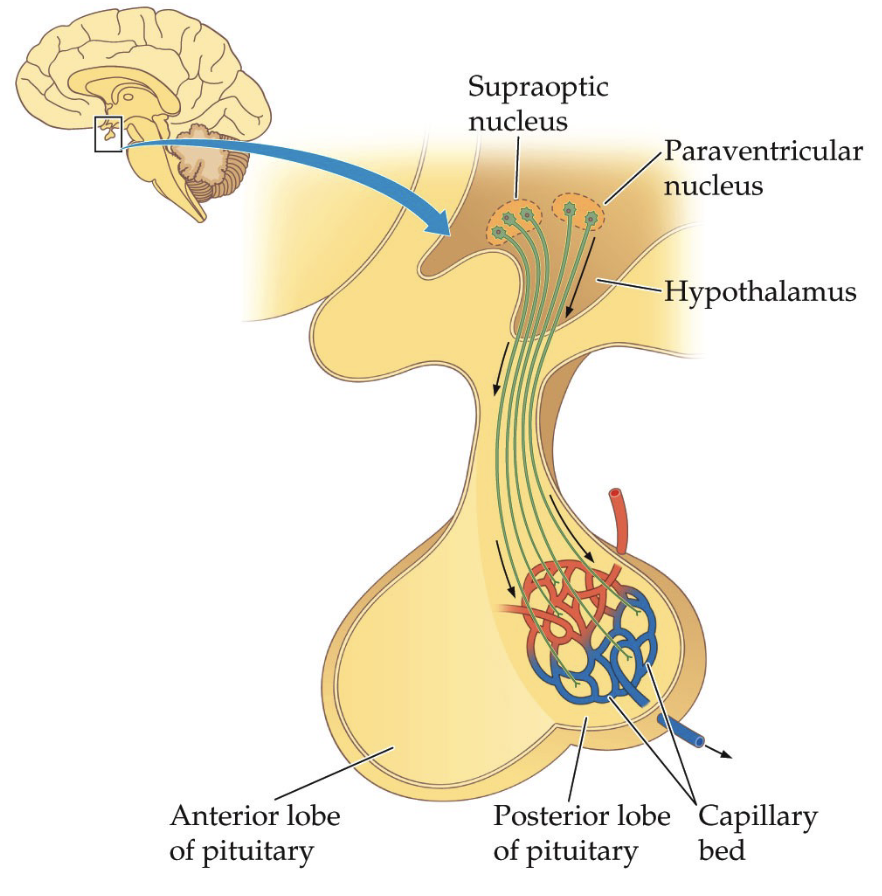
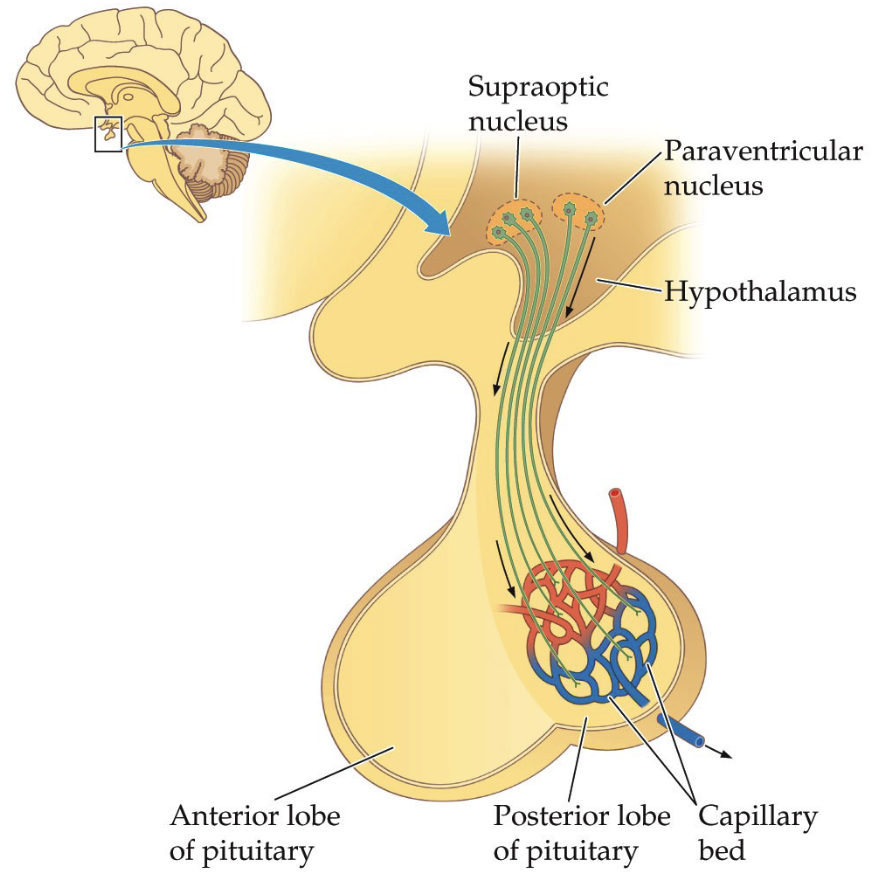
Explain the hypothalamus to the posterior pituitary connection.
Direct Neural Connection
Hypothalamus contains nuclei (clusters of neurons engaged in the same function) involved in hormone production:
Supraoptic nucleus
Paraventricular nucleus
Neurons extend axons directly into posterior pituitary
Hormones are produced in hypothalamic neurons
Hormones are transported down axons and released from posterior pituitary
Posterior pituitary mainly acts as a release site (storage + release)
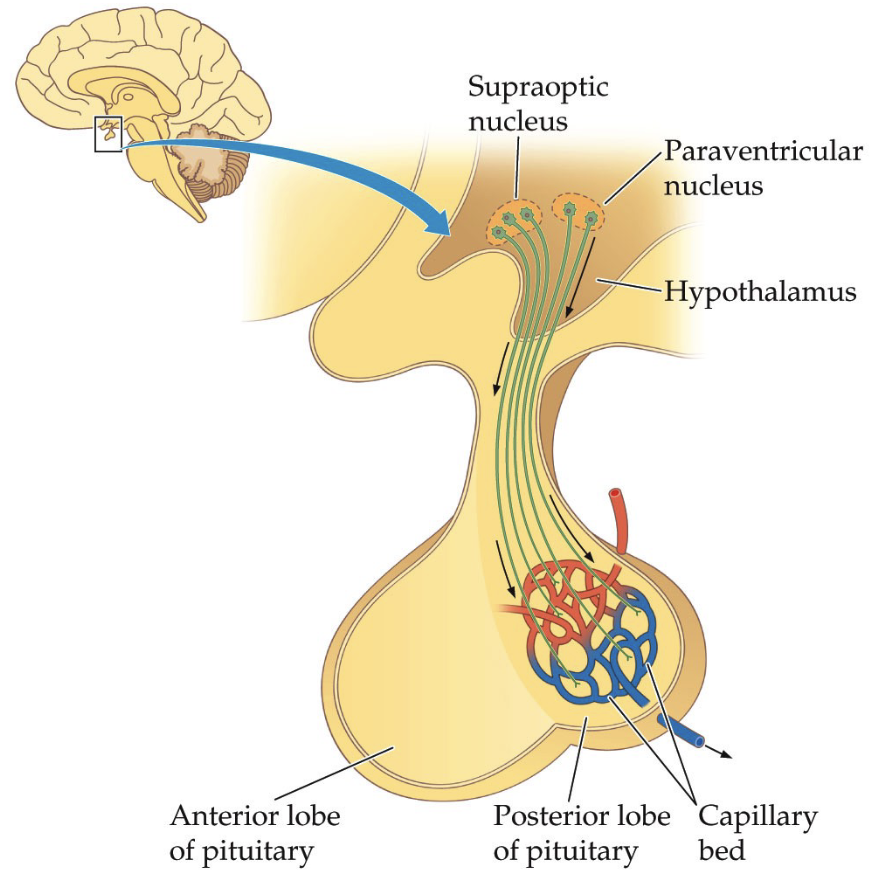
Explain the hypothalamus to the anterior pituitary connection.
Hormonal Signaling via Blood
No direct neuronal axons from hypothalamus
Hypothalamus releases regulatory hormones into blood vessels
These travel through a vascular/capillary system (portal system) to anterior pituitary
Anterior pituitary then produces and releases its own hormones
Key idea:
Two-hormone system
Hormone 1: Hypothalamus → signals anterior pituitary
Hormone 2: Anterior pituitary → releases its own hormones
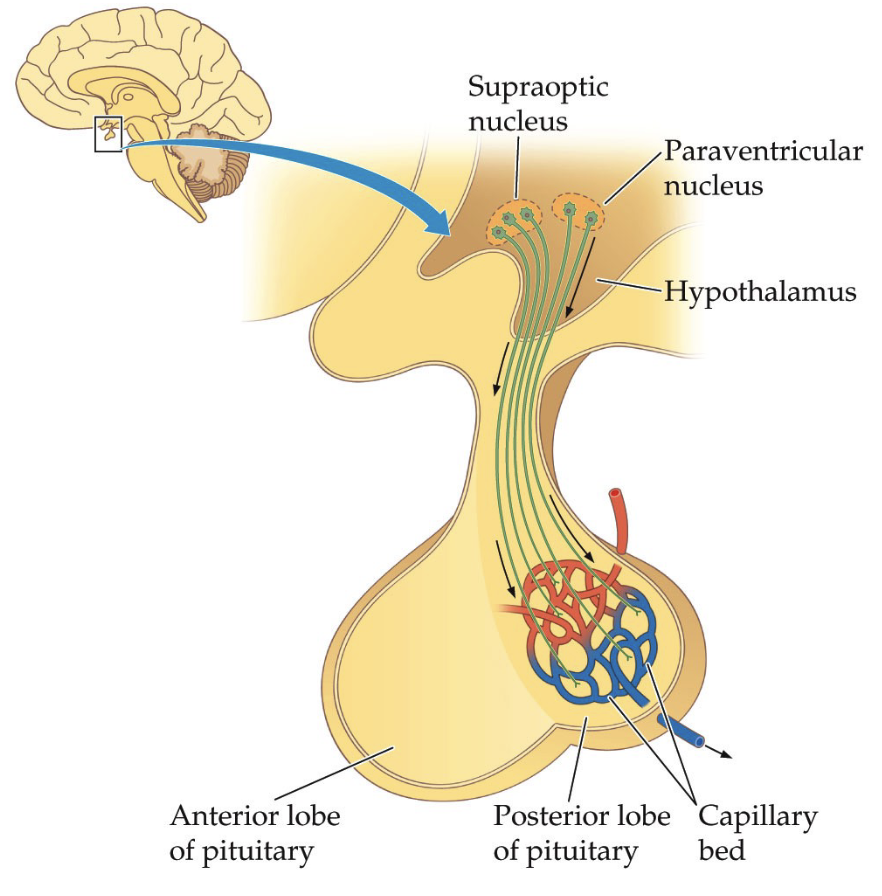
What are the structural differences between the posterior and anterior pituitary?
Posterior pituitary: neural connection (axon projections)
Anterior pituitary: strong vascular (blood flow) connection
Capillary beds support hormone transport and neuron health
What are examples of monoamines?
Monoamines can function as neurotransmitters and hormones.
Epinephrine (aka Adrenaline)
Norepinephrine
Serotonin
Dopamine
Histamine
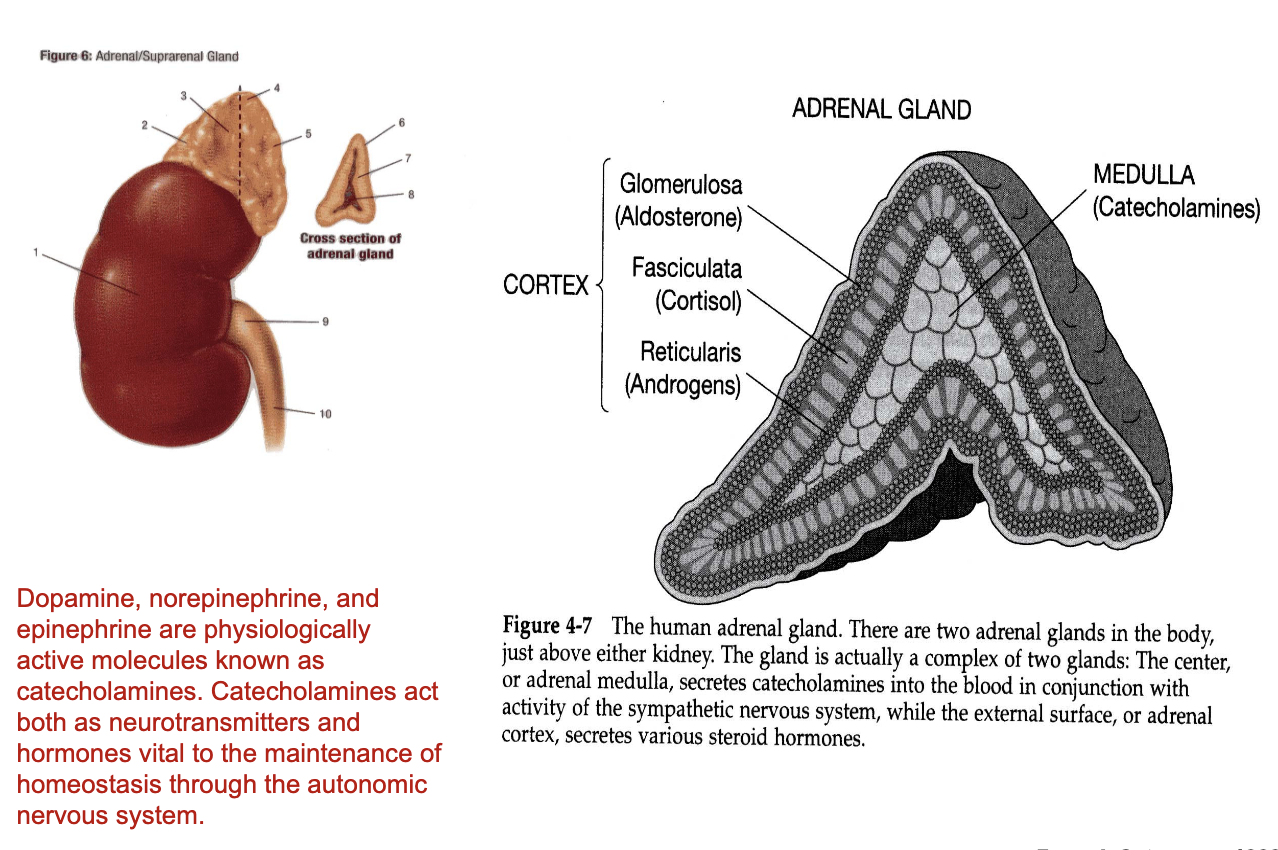
What are the adrenal glands?
Dopamine, norepinephrine, and epinephrine are physiologically active molecules known as catecholamines. Catecholamines act both as neurotransmitters and hormones vital to the maintenance of homeostasis through the autonomic nervous system.
There are two adrenal glands in the body, just above either kidney. The gland is actually a complex of two glands: The center, or adrenal medulla, secretes catecholamines into the blood in conjunction with activity of the sympathetic nervous system, while the external surface, or adrenal cortex, secretes various steroid hormones.
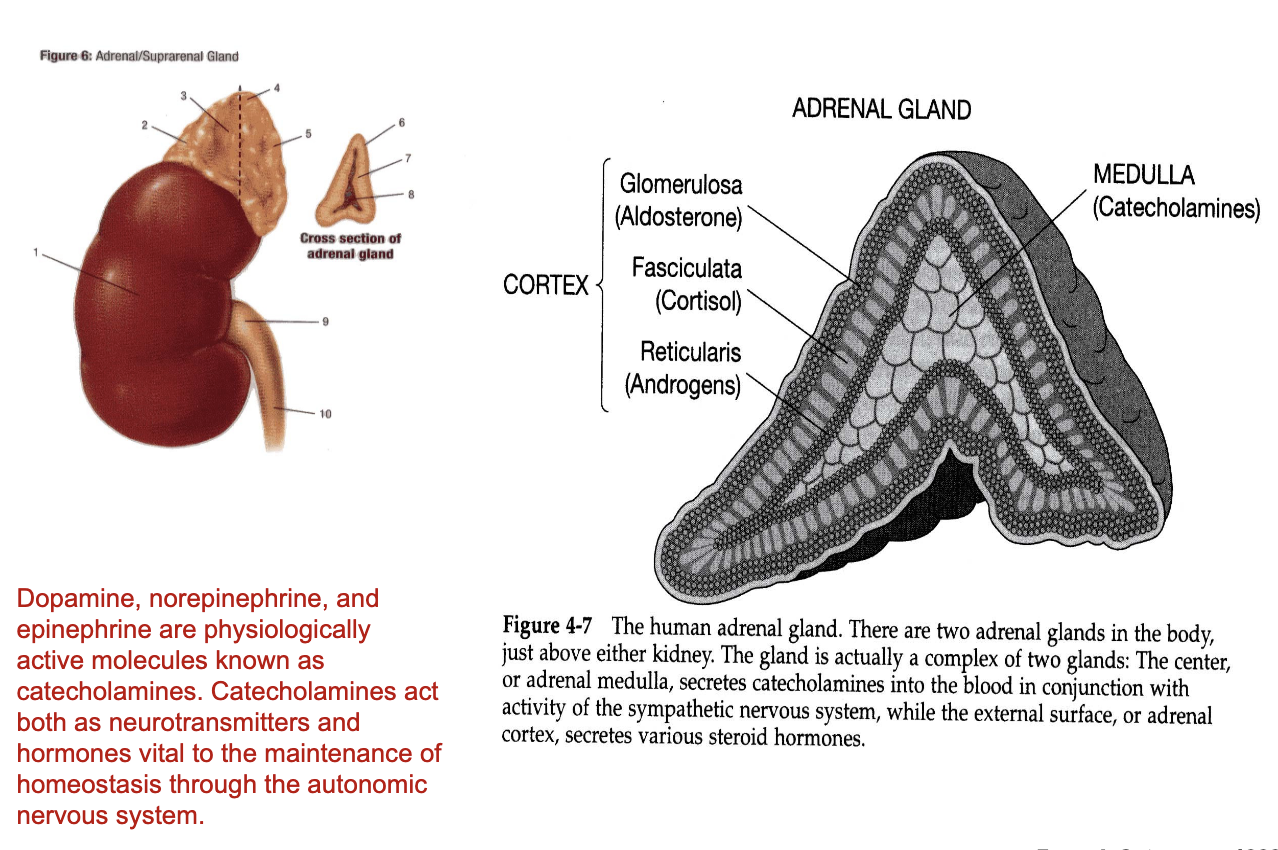
What are the main structures of the adrenal gland?
Adrenal glands
Located on top of the kidneys
Two main regions:
Adrenal cortex (outer region)
Adrenal medulla (inner region)
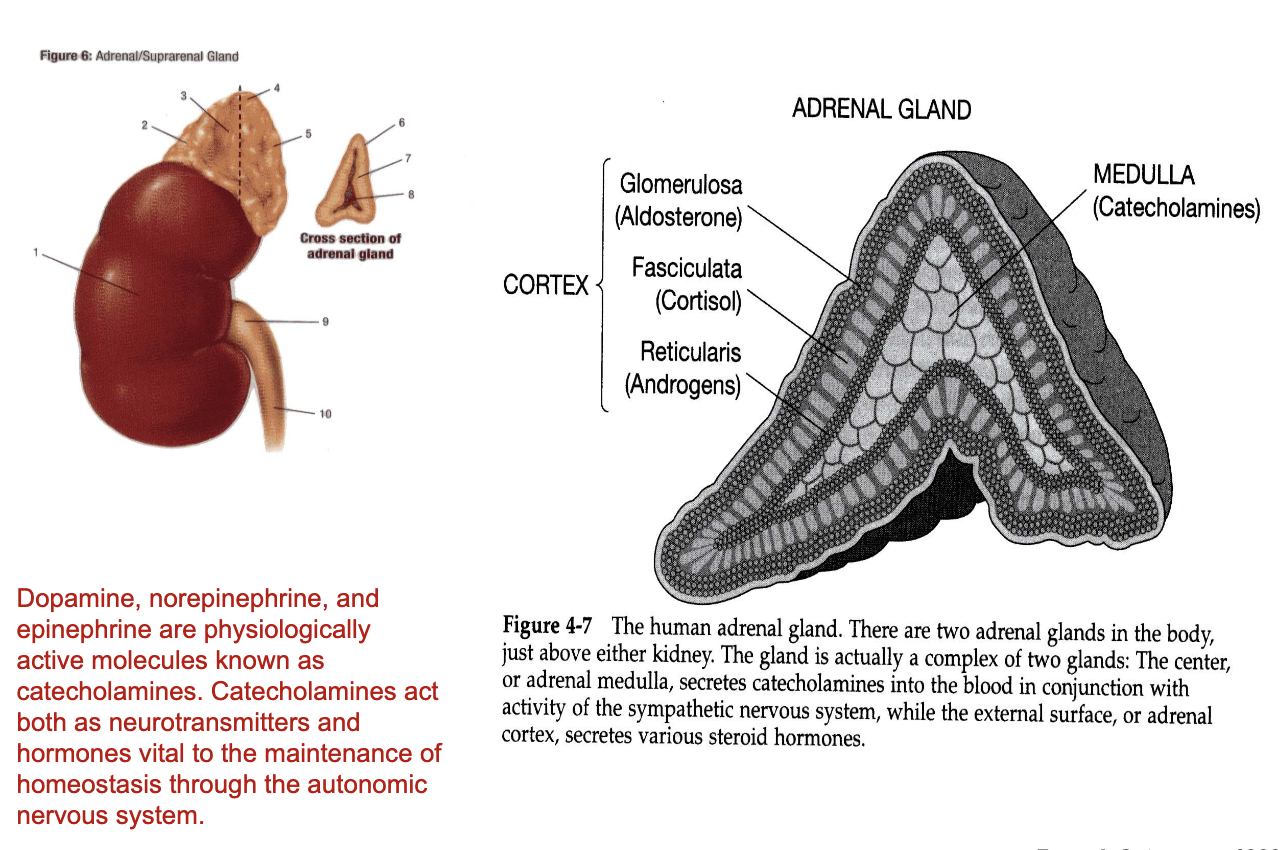
What hormones are produced by the adrenal cortex?
Adrenal Cortex (3 layers – outer → inner)
Each layer produces different steroid hormones:
Glomerulosa
Produces aldosterone
Regulates salt, water, and electrolyte balance
Fasciculata
Produces cortisol (glucocorticoid)
Important for stress response and metabolism
Reticularis
Produces androgens
Contributes to sex hormone production
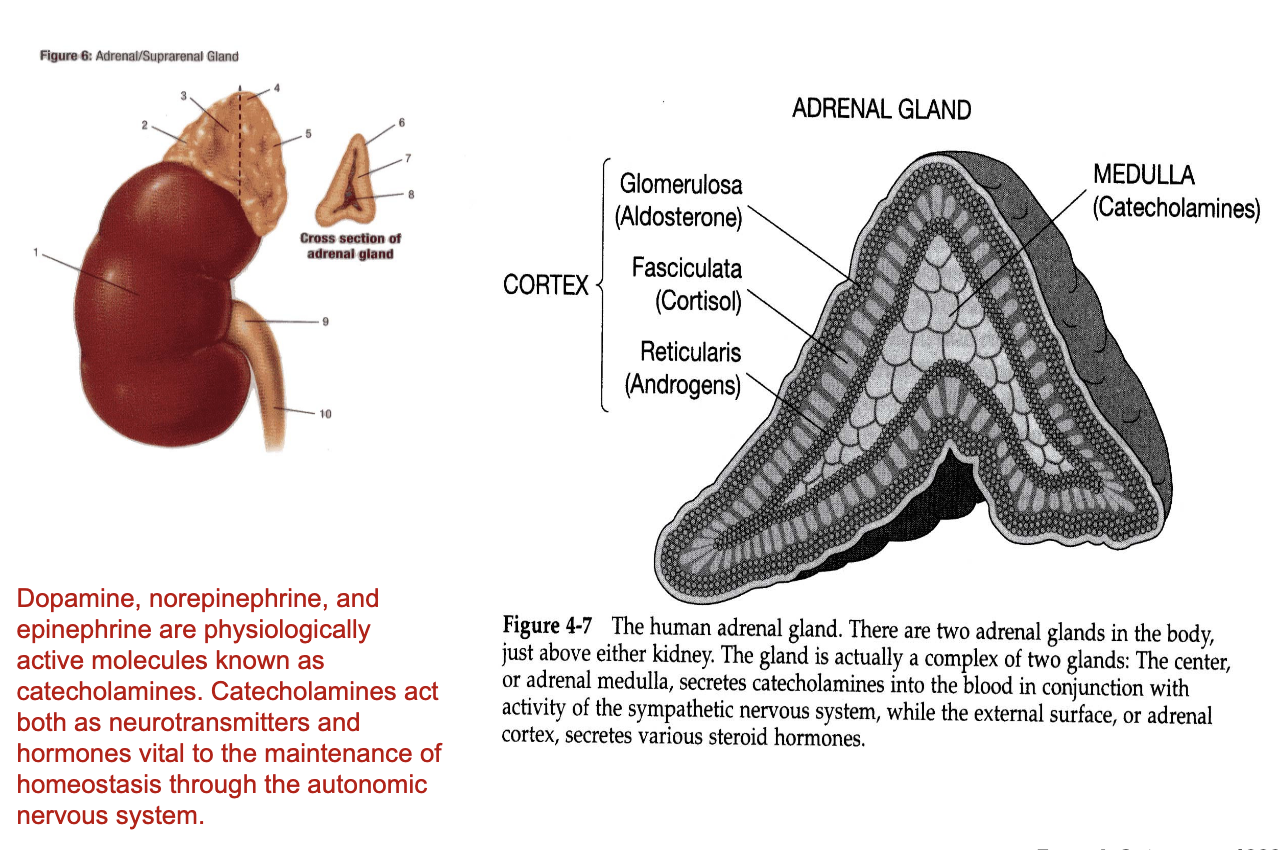
What hormones are produced by the adrenal medulla?
Adrenal Medulla (inner region)
Produces catecholamines:
Dopamine
Norepinephrine (noradrenaline)
Epinephrine (adrenaline)
Catecholamines can function as:
Neurotransmitters when released locally in the brain/synapse
Hormones when released from adrenal gland and travel through bloodstream to distant organs (e.g., heart)
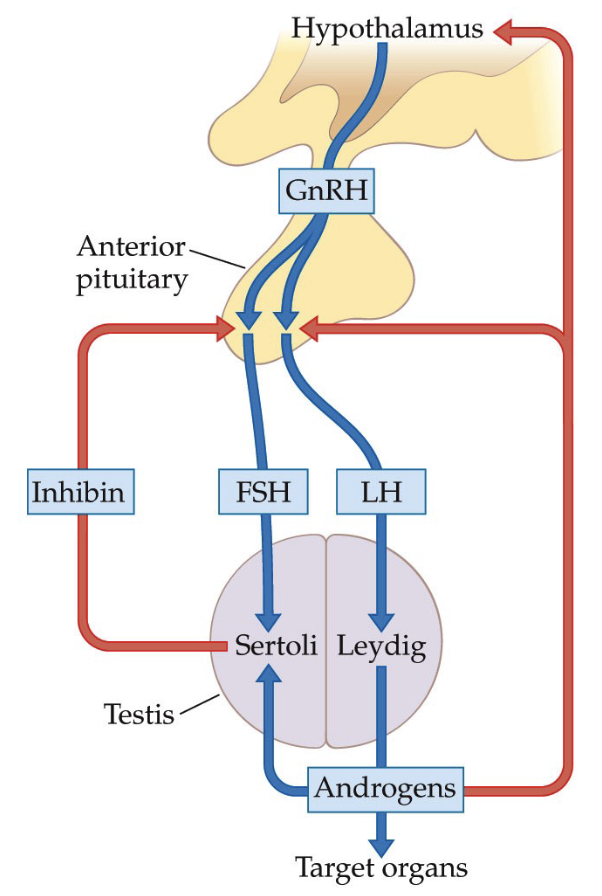
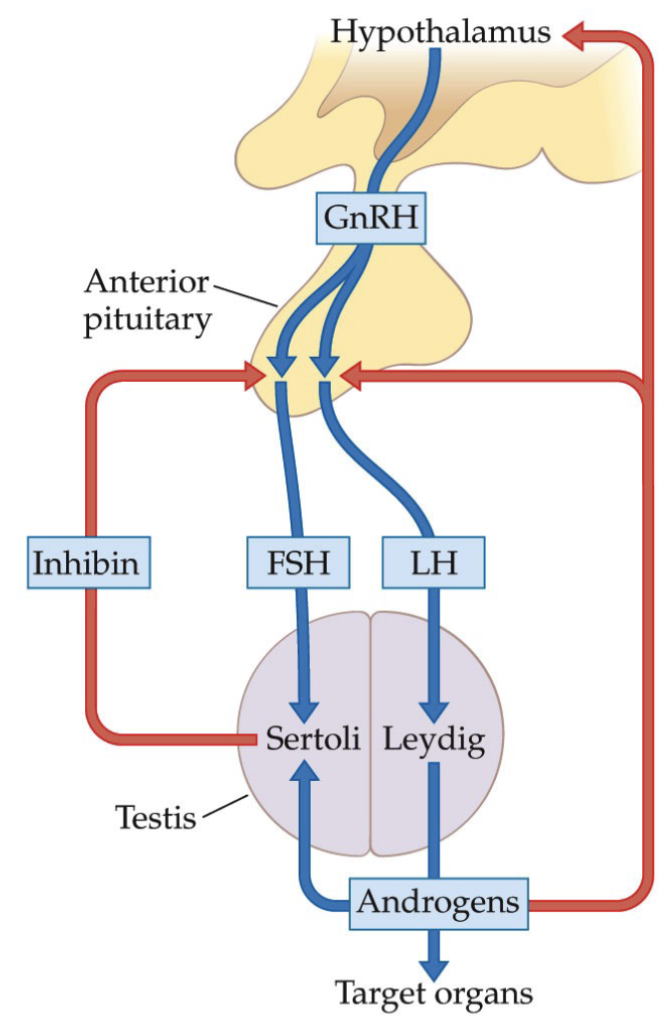
What is the Hypothalamic-Pituitary-Testicular Control System?
GnRH stimulates the release of gonadotropins, such as LH, which then stimulates the Leydig cells to secrete testosterone.
FSH stimulates the Sertoli cells to produce sperm.
Testosterone production and spermatogenesis are regulated via negative feedback.
**This is simplified; in reality, FSH and LH have impact on multiple cells.
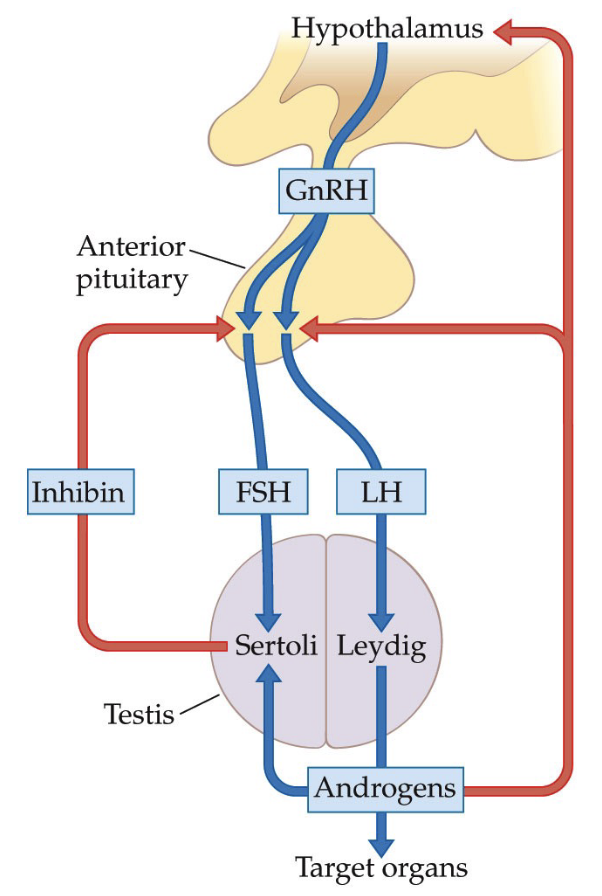
What are the local and systemic effects of testosterone in the male reproductive system, and how does negative feedback regulate its levels?
Source of Testosterone
Produced by Leydig (Lydig) cells in testes
Main androgen hormone
Local Effects (within testes)
Testosterone acts on Sertoli cells
Stimulates spermatogenesis (sperm production)
If sperm production becomes excessive:
Inhibin released from Sertoli cells
Inhibin inhibits anterior pituitary
↓ FSH and LH (gonadotropins)
Systemic Effects
Maintains health of male reproductive organs
Supports blood flow to reproductive tissues
Increased circulating RBCs
Delivery of oxygen and nutrients via plasma
Promotes cell growth and tissue maintenance
Stimulates muscle growth and development
Negative Feedback Regulation
Testosterone levels monitored by:
Hypothalamus
Anterior pituitary
High testosterone → negative feedback
↓ GnRH from hypothalamus
↓ FSH and LH from anterior pituitary
Maintains optimal androgen levels
Exogenous Testosterone (e.g., anabolic steroids)
Artificial testosterone ↑ circulating androgens
Body interprets this as excess hormone
Feedback loop suppresses natural hormone production
Can shut down reproductive function
Athletes may cycle steroid use to limit suppression
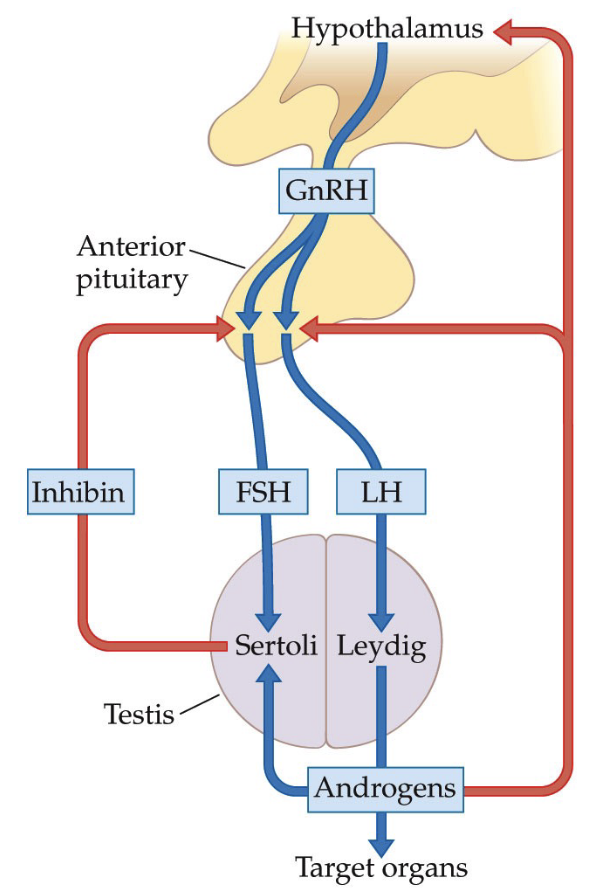
How does the hypothalamus–anterior pituitary–gonad pathway (HPG axis) regulate reproductive hormones?
Step 1: Hypothalamus
Releases GnRH (Gonadotropin-Releasing Hormone)
GnRH = signal sent to anterior pituitary
“Gonadotropin-releasing” → instructs pituitary to release gonadotropins
Step 2: Anterior Pituitary
Responds to GnRH
Releases two peptide hormones (gonadotropins):
FSH (Follicle Stimulating Hormone)
LH (Luteinizing Hormone)
Step 3: Target = Gonads
Gonads = testes (males) and ovaries (females)
FSH and LH regulate reproductive function in both sexes
Male system example
Target cells in testes:
Sertoli cells
Leydig cells
Leads to production of:
Androgens (especially testosterone)
Inhibin (peptide hormone)
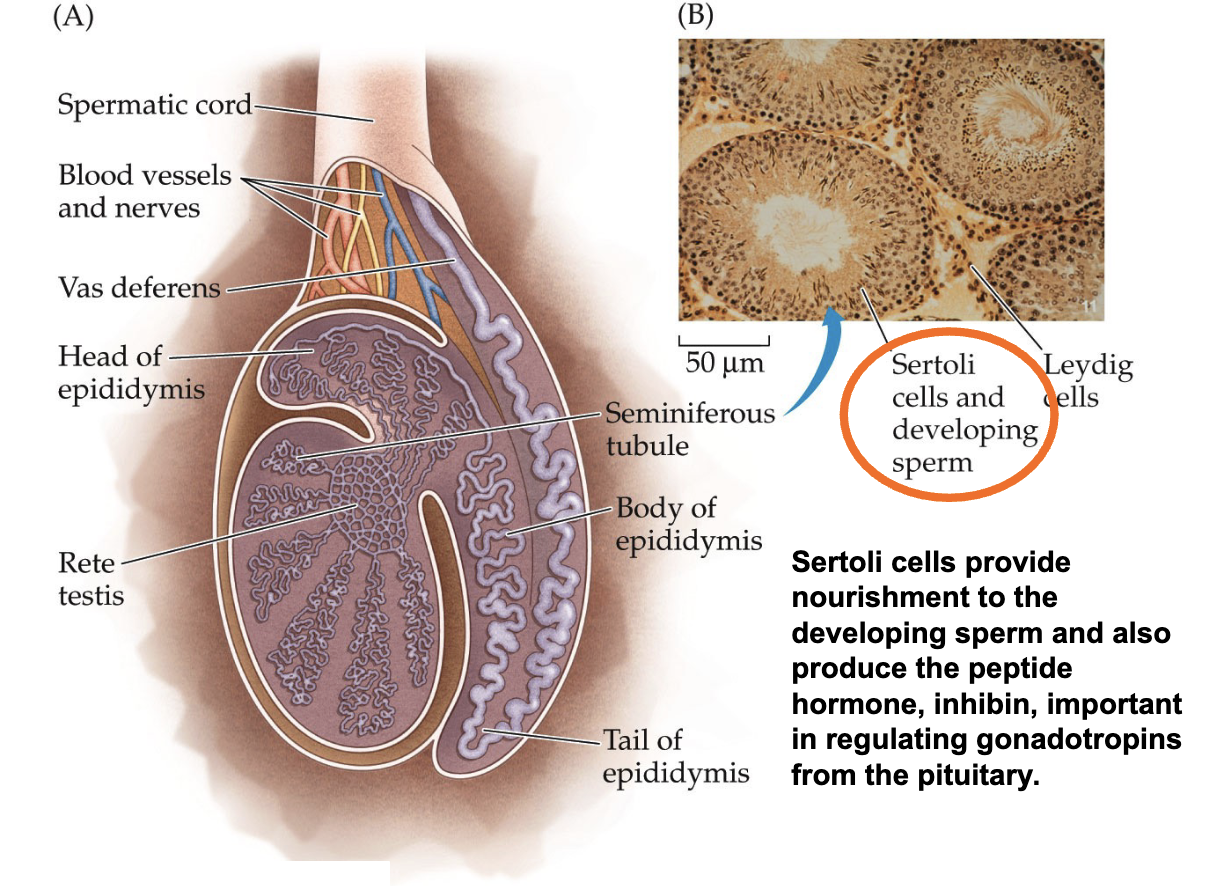
What is the internal structure of the testis (Sertoli cells)?
Sertoli cells provide nourishment to the developing sperm and also produce the peptide hormone, inhibin, important in regulating gonadotropins from the pituitary.
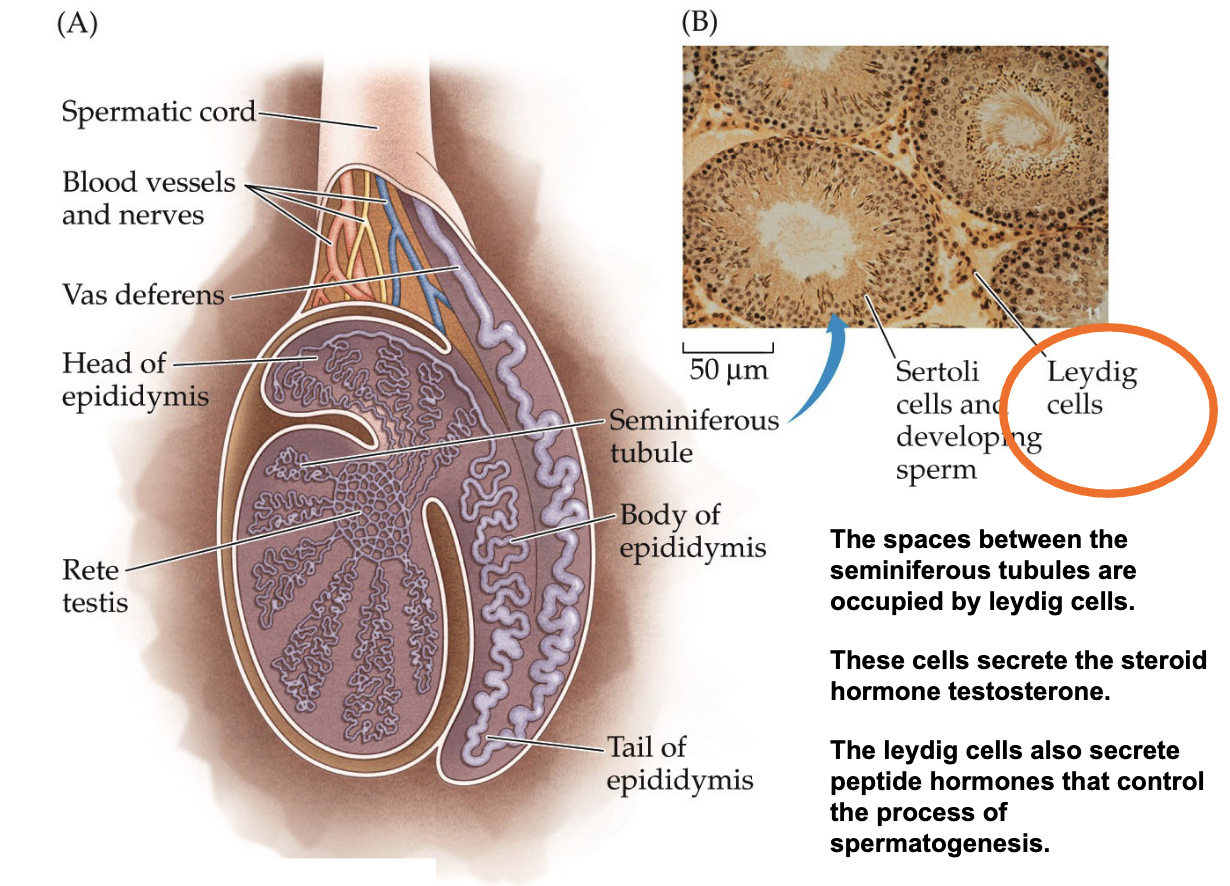
What is the internal structure of the testis (Leydig cells)?
The spaces between the seminiferous tubules are occupied by leydig cells.
These cells secrete the steroid hormone testosterone.
The leydig cells also secrete peptide hormones that control the process of spermatogenesis.
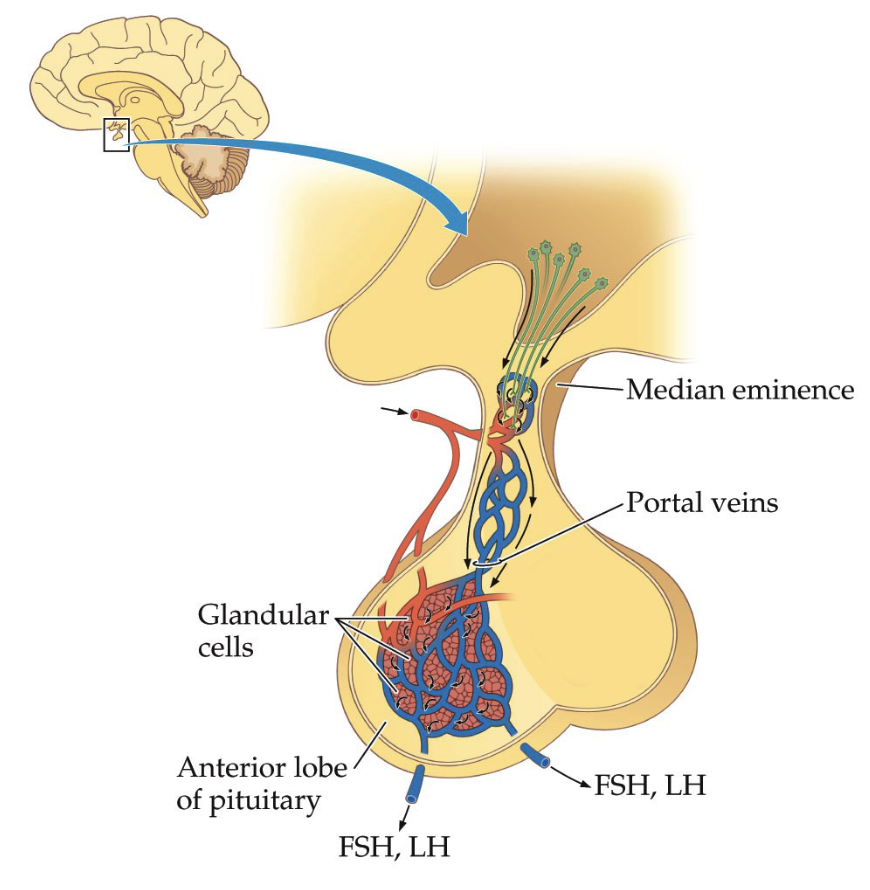
How does the hypothalamus communicate with the anterior pituitary and why is the hypothalamic–pituitary portal system important?
Anterior pituitary is highly vascularized (many capillaries, veins, blood vessels)
Contains glandular cells that produce hormones
Connected to hypothalamus via hypothalamic–pituitary portal system
Portal system
Specialized blood vessel network
Transports hypothalamic releasing hormones directly to anterior pituitary
Median eminence
Region where hypothalamic hormones enter portal blood supply
Hormone pathway
Hypothalamus releases releasing hormones (e.g., GnRH)
Hormones travel through portal blood vessels
Anterior pituitary releases its own hormones (e.g., FSH, LH)
Hormones act on distant organs (gonads)
Regulation
Hypothalamus responds to circulating hormones (e.g., testosterone)
Produces cyclical hormone regulation via feedback loops
Development
System forms in utero, mostly inactive in childhood, activates at puberty.
What are peptide hormones?
Some protein hormones not secreted by the brain include human chorionic gonadotropin (hCG), inhibin, and anti-Müllerian hormone (AMH)
hCG, secreted by the conceptus and placenta, is a useful indicator of pregnancy status
AMH is produced by the granulosa cells in ovarian follicles. The production of AMH is reflective of ovarian reserve
Inhibin is secreted by the Sertoli cells and by the granulosa cells
It inhibits the synthesis and release of the FSH in the pituitary gland and reduces the hypothalamic LH - releasing hormone content
It also inhibits the multiplication of spermatogonia (undifferentiated germ cell) in the gonads and the production of progesterone by the ovaries
What does the term conceptus mean and what are the main developmental stages after fertilization?
Conceptus: umbrella term for all stages of a developing fertilized egg.
Blastocyst: early stage after fertilization when the zygote undergoes multiple cell divisions.
Embryo: stage after implantation in the uterine wall when development continues.
Fetus: later stage of pregnancy when the organism grows, matures, and takes a human-like form.
Pregnancy tests detect hCG (human chorionic gonadotropin), a peptide/protein hormone produced by the conceptus.
What is Hypothalamic-Pituitary-Testicular Control System?
The hypothalamus secretes GnRH, which stimulates the secretion of LH and FSH by the anterior lobe of the pituitary gland.
LH in turn stimulates the synthesis and secretion of androgens (primarily testosterone) by the Leydig cells of the testes.
FSH stimulates the Sertoli cells of the testes to increase the production of sperm (spermatogenesis).
Sertoli cells promote high levels of sperm production only when both FSH and androgens are present.
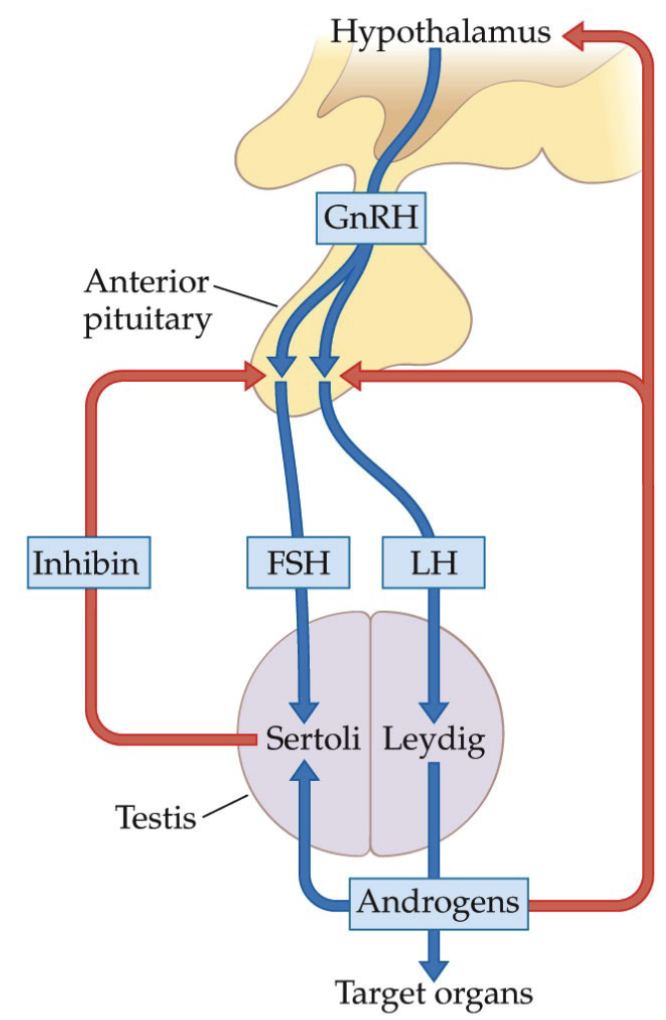
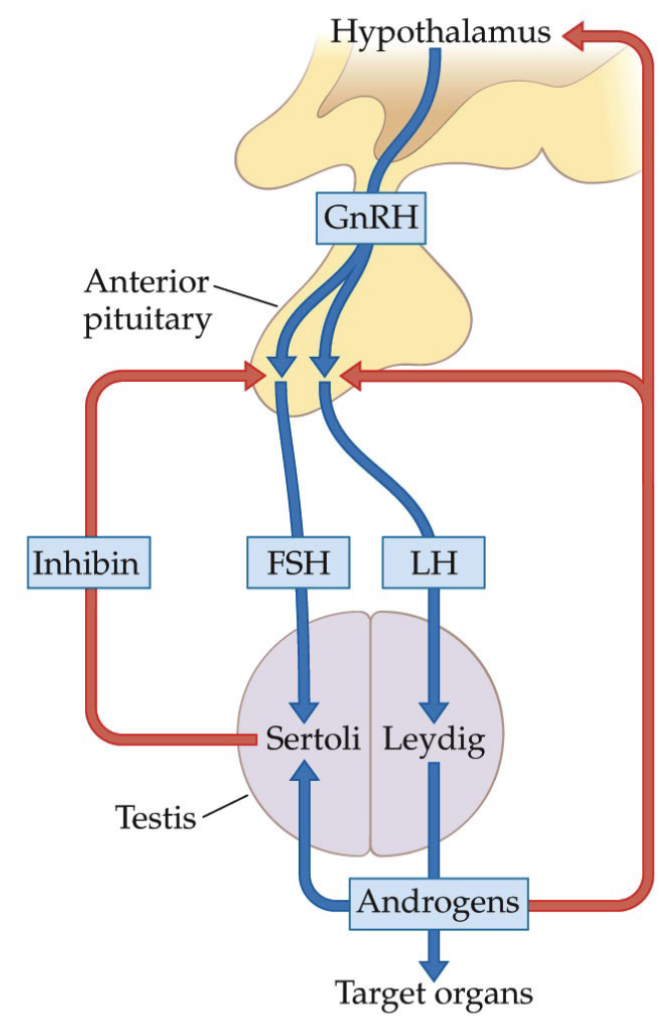
What is the negative feedback of the Hypothalamic-Pituitary-Testicular Control System?
Negative feedback occurs via two routes.
The first is a direct effect of testosterone on the LH-secreting cells in the anterior lobe of the pituitary, depressing their response to the GnRH pulses coming from the hypothalamus.
The second is an effect of testosterone on the hypothalamus, decreasing the frequency of GnRH pulses.
What about the regulation of spermatogenesis?
Because testosterone controls the secretion of GnRH and because GnRH stimulates both LH and FSH secretion by the pituitary gland, increasing testosterone levels tend to have a negative feedback effect on FSH as well as LH.
Sertoli cells also send a signal back to the pituitary gland that is more directly related to spermatogenesis, in the form of inhibin.
What are the functions of testosterone?
Testosterone has multiple functions in XY individuals
Most internal and external genital structures require testosterone for normal functioning
People abusing exogenous testosterone: Musculature will increase everywhere else, but the genital structures will decrease in size
Testosterone influences sexual behaviour
Testosterone promotes tissue growth
What is the role of testosterone in erections?
Testosterone also influences the erectile capacity of the penis
In healthy people, penile erections occur during REM sleep
These nocturnal erections cease some weeks after castration (removal of testes) and return with testosterone treatment
Non-testicular sources—most likely the adrenal glands— for those who can still experience an erection after castration have higher circulating levels of testosterone than those who cannot
How does testosterone promote tissue growth?
Testosterone promotes tissue growth
Anabolic effects are easiest to see in the musculature
Androgens are the main reason why XY individuals tend to be more muscular than XX after puberty
But androgens also promote the formation of red blood cells
This increases the oxygen-carrying capacity of the blood, and they increase the mass of the liver and kidneys as well
The anabolic effects of androgens is the reason they are abused by athletes (XX and XY), but such use carries significant health risks
Do the testes secrete estrogen?
The testes also secrete some estrogens
Estradiol facilitates maturation and concentration of sperm in the epididymis
Estradiol terminates growth at the end of puberty
Estradiol maintains normal bone density
What is the menstrual cycle?
Menstruation is the vaginal discharge of endometrial tissue & blood experienced at roughly monthly intervals during fertile years, except when a person is pregnant or intensively nursing an infant
Brought about by a complex internal mechanism that involves the ovaries, the brain, and the uterus

What are the 2 cycles of the menstrual cycle?
Ovarian Cycle: Hormonal interactions between the ovaries, the hypothalamus, and the pituitary gland
Menstrual Cycle: Changes in the endometrium of the uterus, which is responsible for menstrual periods
What are the key phases of a Menstrual Cycle?
Follicular or preovulatory phase is marked by the maturation of follicles in the ovaries
Luteal or postovulatory phase because it is marked by the presence of a corpus luteum (CL)
A CL is a hormone-secreting structure formed from a follicle ruptured at ovulation (produces progesterone)
Proliferative phase and secretory phase: processes that take place in the uterus
Proliferative phase (Follicular): Increasing the number of cells in the endometrium to prepare for pregnancy
Secretory (Luteal): No pregnancy → gets ready for menstrual phase
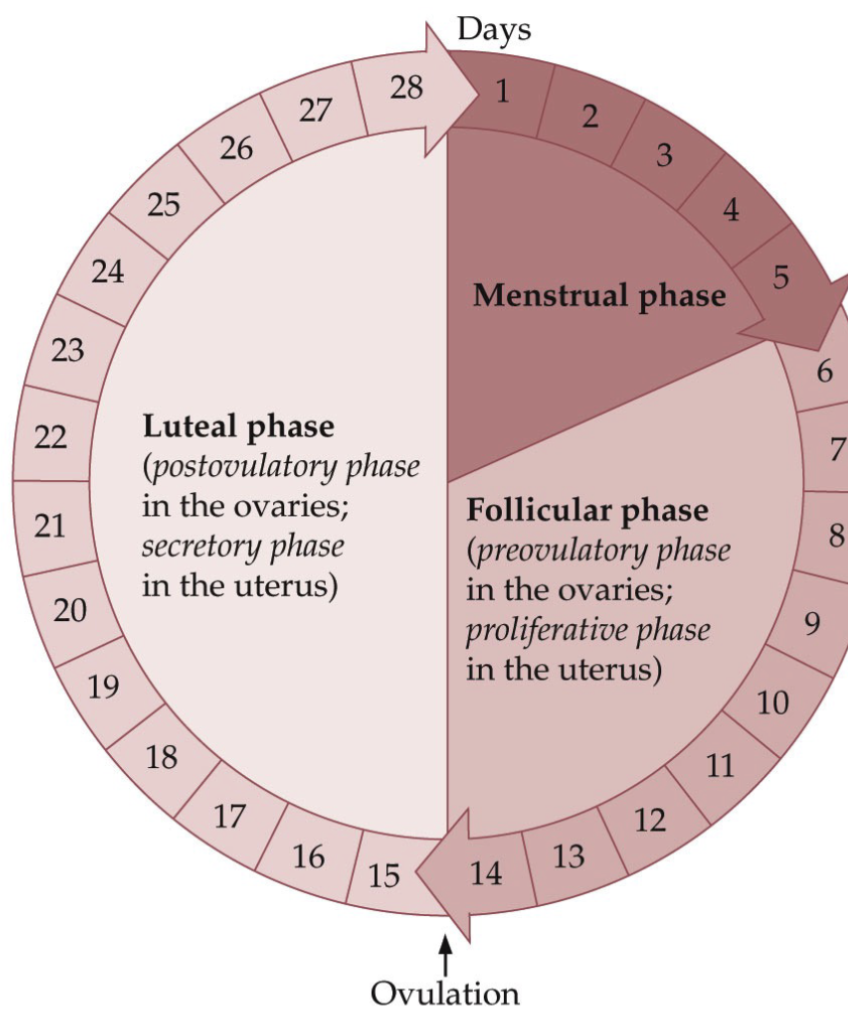
What is the typical 28-day Menstrual Cycle?
The menstrual phase occupies days 1 through 5
The follicular phase occupies days 6 through 14
Luteal phase occupies days 15 through 28
The luteal phase is the most constant—it usually lasts 14 days— give or take 2 days
Most of the variation in total cycle length is accounted for by variation in the other two phases (menstrual and follicular)
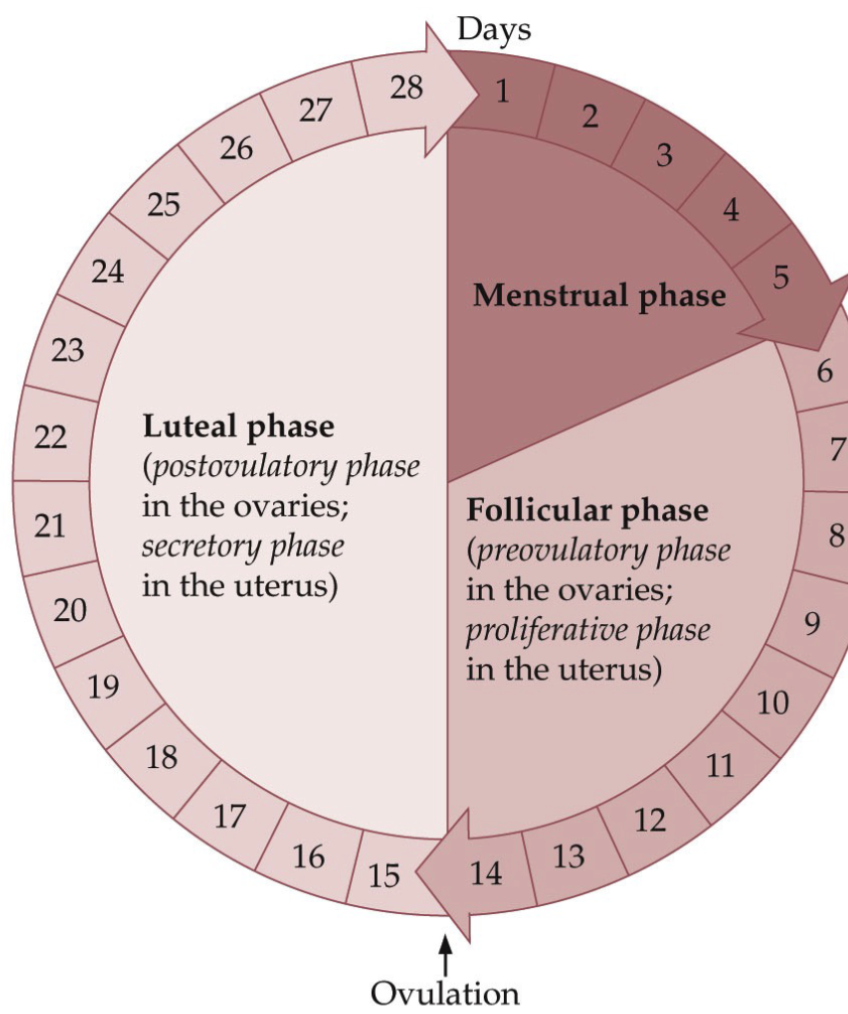
What body parts are involved in the Menstrual Cycle?
The menstrual phase is triggered by a drop in progesterone and estradiol levels →
As estrogen levels fall at the beginning of the menstrual cycle, negative feedback diminishes, and gonadotropins rise →
The follicular phase is marked by the maturation of ovarian follicles
Ovaries of newborns contain about 2 million primary oocytes that are arrested in the first two of their meiotic divisions
What is progesterone’s role in pregnant woman?
Despite pregnancy being a shit experience, progesterone increases and gaslights you into thinking this is amazing because at least you are pregnant
However, after giving birth, dramatic drop of progesterone → hypothesize this might be why some have difficulty connecting with the newborn
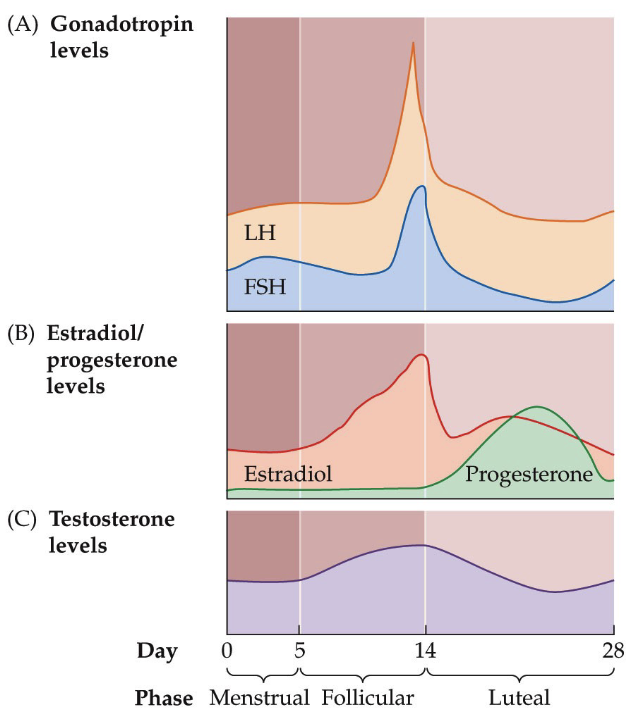
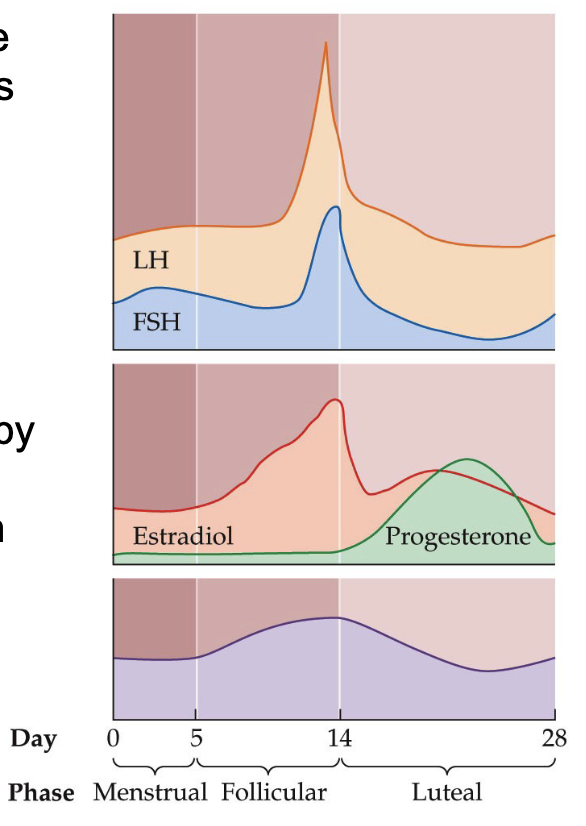
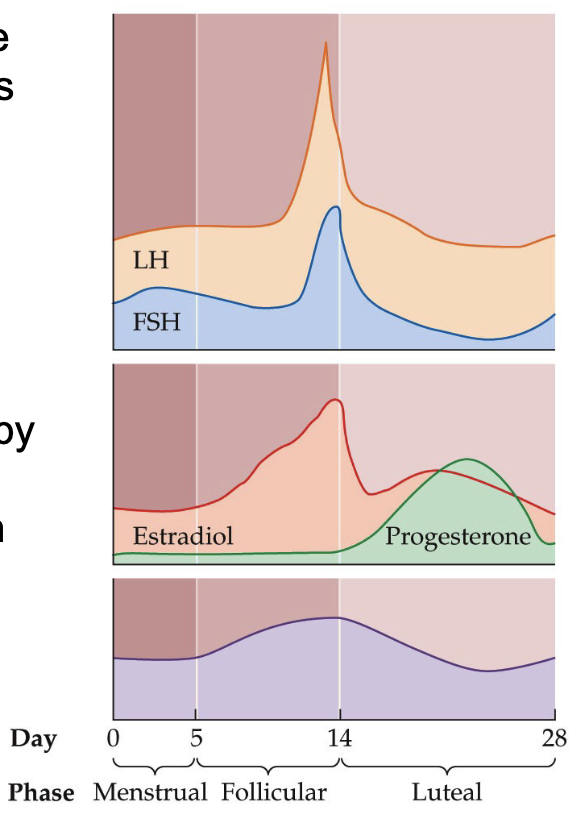
What are the changes in the circulating levels of major hormones in the menstrual cycle?
As estrogen levels fall at the beginning of the menstrual phase the negative feedback effect diminishes
Allows circulating LH and FSH levels to rise
These increased gonadotropin levels—in particular, the increase in FSH—promote the development of follicles in the ovaries
If pregnancy occurs → CL will continue to secrete progesterone to facilitate the implantation process
If pregnancy doesn’t occur → CL dissolves and is recycled; drop in progesterone
**Memorize graphs
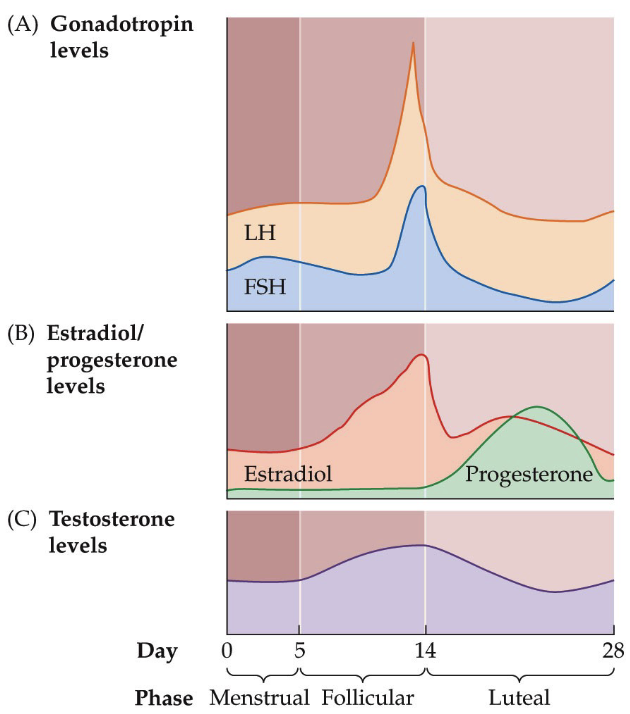
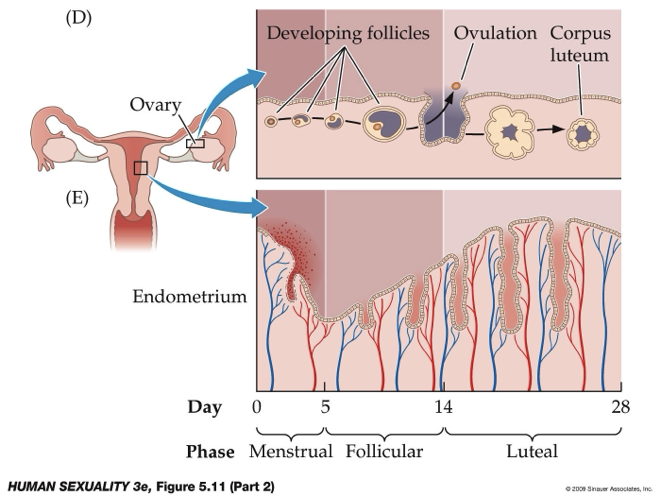
What is the function of the endometrium during the menstrual phase?
During the menstrual phase, much of the inner lining of the uterus—the endometrium—breaks down
There is a transformation where the endometrium goes from a state in which it is capable of sustaining pregnancy to a state in which it facilitates the transport of sperm
The full thickness of the endometrium is not lost during menstruation
The deepest portion remains intact, and this "basal layer" will be the source for regeneration of the endometrium after menstruation ends.
A healthy endometrium is important; lesions → unhealthy tissue releases its own signals telling the body the implantation shouldn’t happen → fertility issues
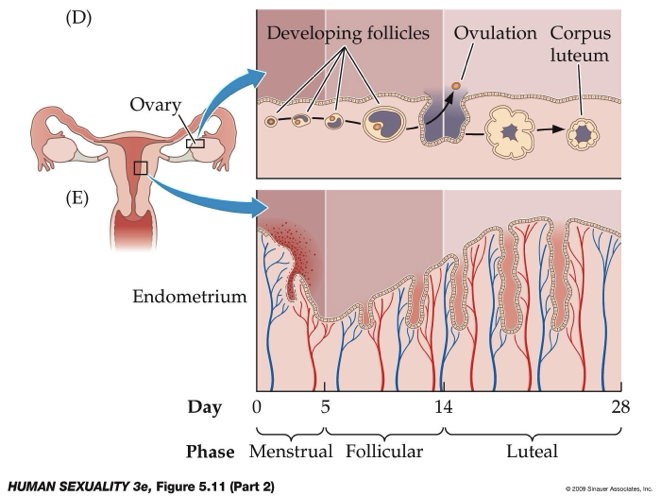
What is the development of ovarian follicles and the endometrium?
Each oocyte is surrounded by a thin layer of granulosa cells, forming a primary follicle
Follicles that become gonadotropin-dependent during the follicular phase of the cycle will develop further
Of the gonadotropin-dependent follicles, one becomes dominant and grows faster than the others.
This follicle is called a preovulatory follicle
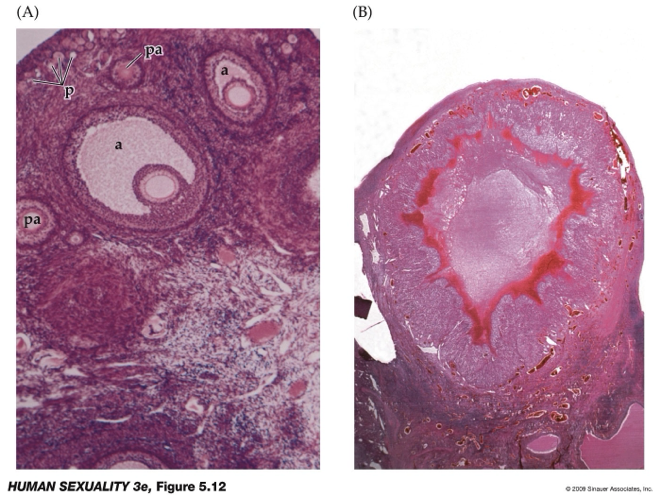
How are ovarian follicles monitored during fertility testing and what is the role of the corpus luteum?
Follicle size indicates stage of development (small = early, larger = more mature).
a = antrum
Monitored using transvaginal ultrasound during fertility testing.
Patients may undergo daily ultrasounds and blood tests for ~7–12 days (psychological effects).
Image B: Corpus luteum; ruptured follicle
After ovulation, the ruptured follicle forms the corpus luteum.
Corpus luteum secretes progesterone, which helps prepare the uterus for implantation.
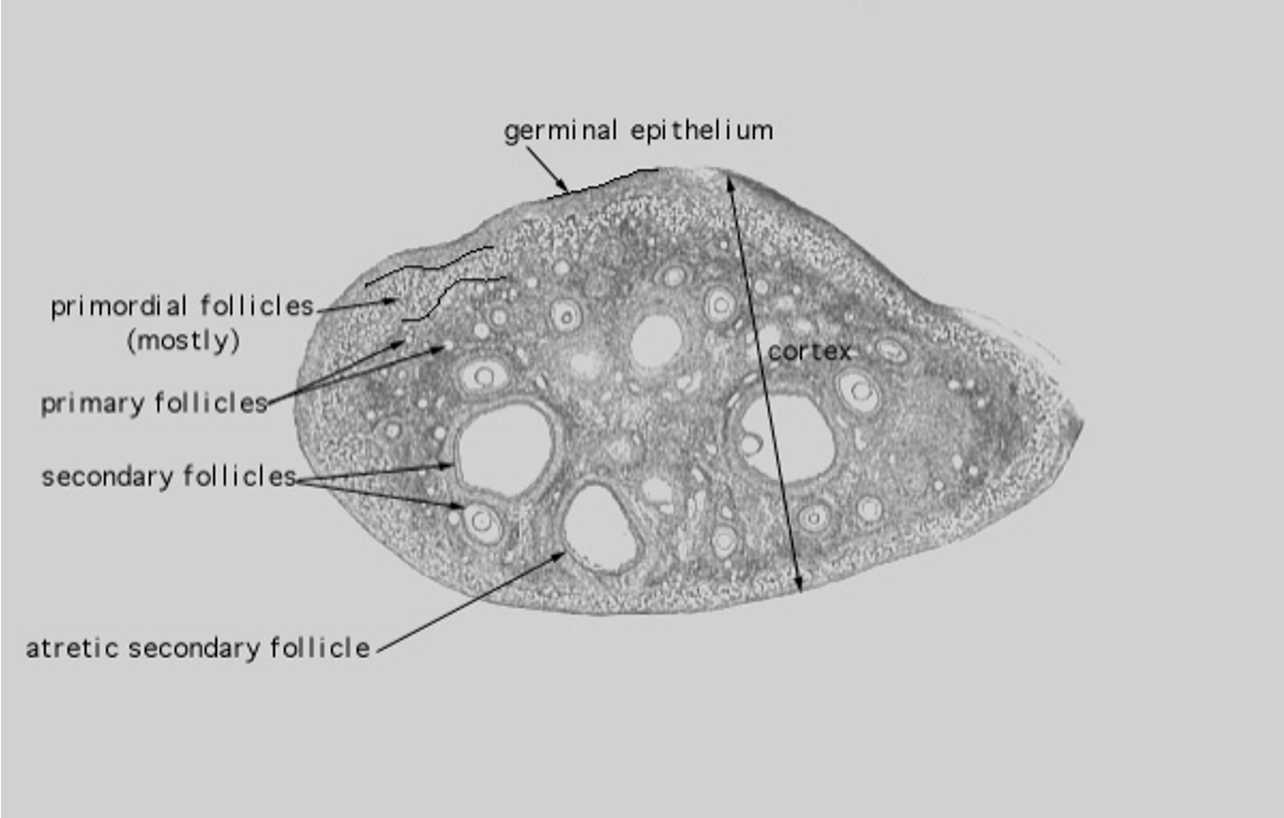
What are the stages of ovarian follicle development and how are they evaluated in fertility testing?
Primordial follicles: present at birth, very small, diminish at menopause.
Primary follicles: recruited follicles that begin to grow.
Secondary → tertiary follicles: continue increasing in size as the egg develops.
Atretic secondary follicles: follicles that stop developing and are reabsorbed.
Transvaginal ultrasound measures follicle size (mm) and ovary structure to monitor development.
Ovarian cysts: fluid-filled sacs; usually common and harmless, but large or blood-filled cysts may require further investigation.
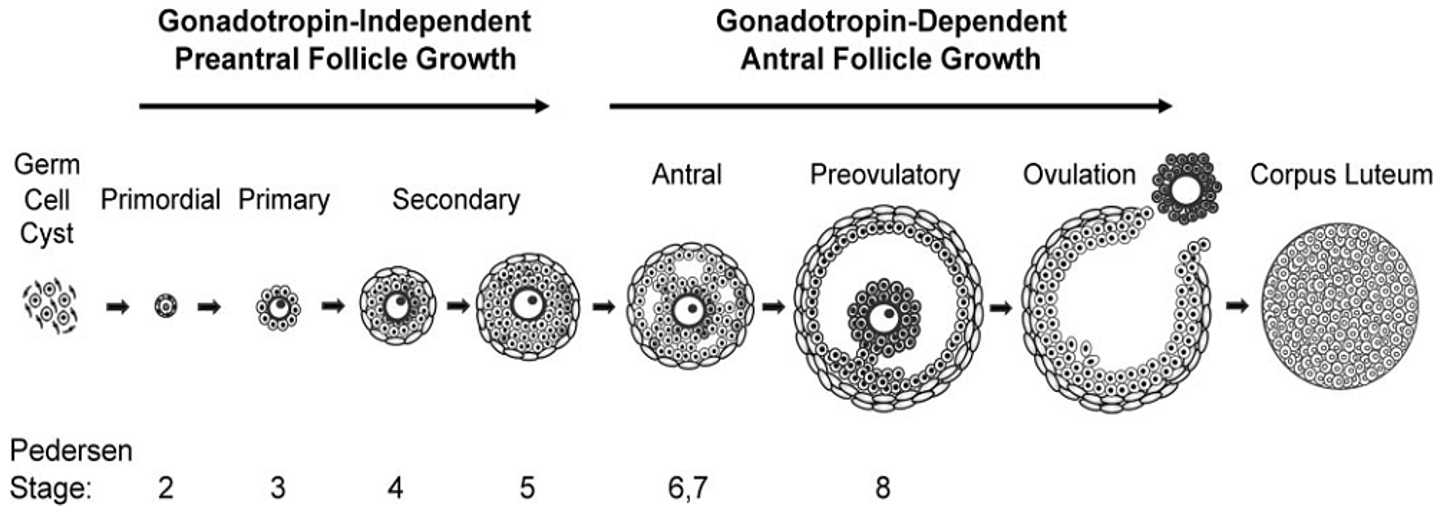
What are the two main phases of follicular development and what is the role of the antrum (Pedersen Stage)?
Phase 1: Gonadotropin-independent (pre-antral) growth — early follicle development controlled mainly by genetic factors.
Phase 2: Gonadotropin-dependent (antral) growth — follicle responds to hormones like FSH/LH.
Antrum: fluid-filled cavity inside the developing follicle containing nutrients and hormones that support follicle growth.
Follicle stage is assessed by size, presence of the antrum, ovulation, and corpus luteum formation.
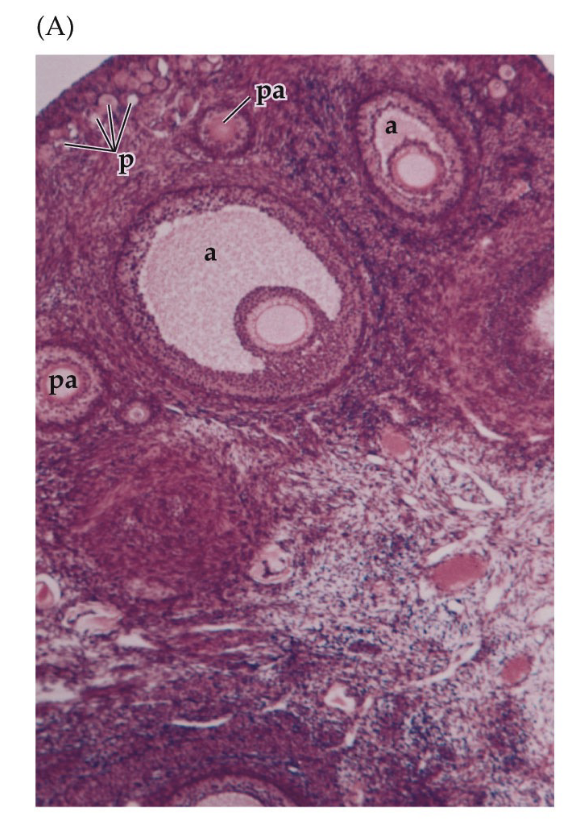
What are the initial stages of follicular development?
Primordial follicles
Leave their long-term "frozen" state and enter a process of renewed maturation
The primary oocyte expands in size
Preantral follicles (pa)
Layer of granulosa cells thickens and attracts an outer layer of thecal cells
Follicle that does not yet have a cavity (antrum)
Antrum (a), forms within the follicle
What happens when the follicle swells?
The entire follicle swells as fluid accumulates
2 mm diameter
The follicular cells begin to produce gonadotropin receptors
Thecal cells begin to produce receptors for LH
Granulosa cells begin to produce receptors for FSH
The follicle is dependent on circulating gonadotropins for survival and further development
What is Gonadotropin-Dependent Follicular Development?
Follicular phase of the menstrual cycle
LH and FSH levels are high
Some follicles will be "rescued" & allowed to develop further; all others will die
Thecal cells synthesize androgens, including testosterone
Granulosa cells pick up some of these androgens and convert them to estrogens
What are the steps of Gonadotropin-Dependent Follicular Development?
The presence of estrogens promotes the proliferation of the granulosa cells, which synthesize more estrogens, and so on.
Estrogen and testosterone levels in the blood rise steadily through the follicular phase of the menstrual cycle.
During this same period, one of the 15 to 20 antral follicles becomes dominant; it grows much faster than the others and is referred to as the preovulatory follicle.
The granulosa cells, in this follicle begin to produce LH receptors, making the follicle sensitive to LH.
Recall that, up to this point, only the thecal cells possessed LH receptors
**Don’t memorize 15-20
What are hormonal loops controlling the menstrual cycle?
When high blood levels of estradiol are maintained for 48 hours, negative feedback on LH switches to positive feedback, and as a result, a surge of LH enters the blood
Feedback loop:
Inhibited at low levels of estradiol
Stimulated at high/sustained levels of estradiol
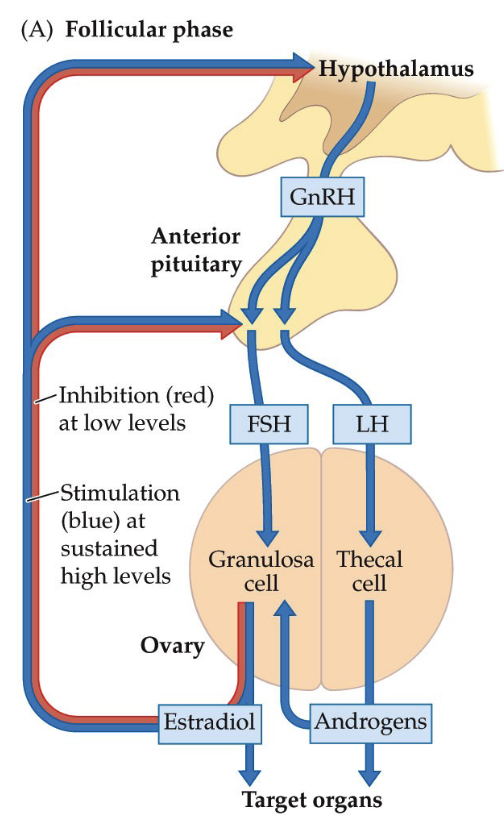
What is the role of thecal cells and granulosa cells in hormone production during follicular development?
Thecal cells produce androgens (e.g., testosterone).
Androgens support muscle maintenance and oxygen delivery in reproductive tissues.
Androgens move to granulosa cells, which contain the enzyme aromatase.
Aromatase converts testosterone → estradiol.
Estradiol promotes endometrial cell growth, preparing the uterus for possible implantation.
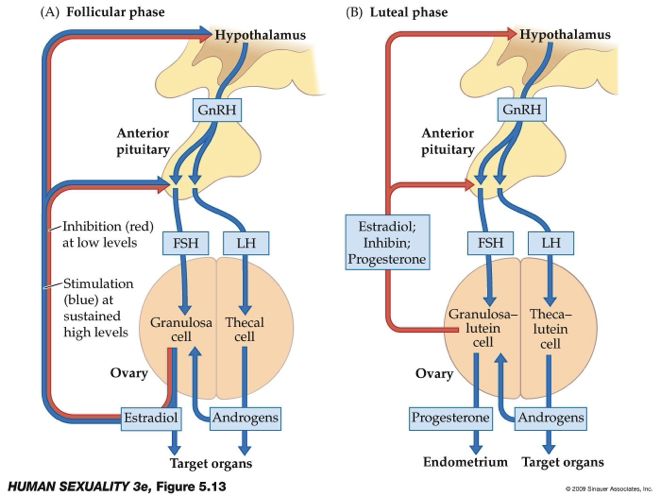
What happens during the luteal phase of the ovarian cycle?
Begins after ovulation when the corpus luteum forms.
Corpus luteum releases progesterone and estradiol to support possible pregnancy.
Granulosa cells → granulosa lutein cells and thecal cells → thecal lutein cells.
High progesterone, estradiol, and androgens create negative feedback on the reproductive hormones.
If no pregnancy occurs, hormone levels fall, leading to shedding of the endometrial lining (menstruation).
How do high estrogen levels trigger an LH surge?
When high blood levels of estradiol are maintained for 48 hours, negative feedback on LH switches to positive feedback, and as a result, a surge of LH enters the blood
This LH surge drive development of the preovulatory follicle
Maturation of the preovulatory follicle requires completion of the first meiotic division
At ovulation, the follicle ruptures
What is the difference between organization and activation in reproductive development?
Organization: occurs in utero; reproductive hormones shape and organize the hypothalamic–pituitary–reproductive system and brain circuits.
Activation: occurs at sexual maturity (puberty) when these systems begin functioning (e.g., ovulation, regular hormone cycles).
Early menstrual cycles may not release eggs despite reaching puberty.
Animal research suggests hormones in utero can influence later sexual behavior, supporting the organization–activation concept.
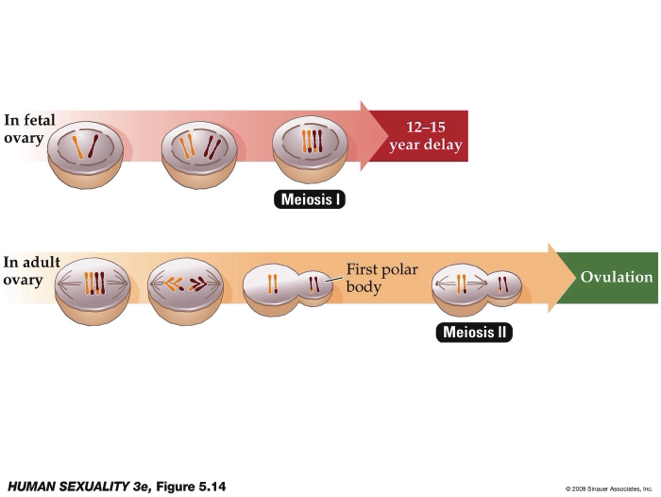
What happens to the follicle under the influence of LH?
Under the influence of LH, the follicles secretion of estrogens drops rapidly
They begins to secrete progesterone, the major sex steroid of the luteal phase
Progesterone is synthesized by the granulosa cells, the same cells that had previously been converting testosterone into estradiol
What happens to the follicle during ovulation?
The follicle ruptures, and the secondary oocyte is extruded
The secondary oocyte is usually called an ovum at this stage
The ovum is swept up by the nearby fimbria of the oviduct and passes into the ampulla, where it lingers (2-3 days) and may be fertilized if any sperm are present
How do the endometrium and cervical mucus change during the menstrual cycle?
Estrogen causes endometrial thickening, growth of epithelial cells, and development of spiral arteries to nourish the lining.
If pregnancy does not occur, the endometrium is shed during menstruation.
Estrogen also increases progesterone receptor production in the uterus.
Cervical mucus changes:
Follicular phase (high estrogen): thin, watery mucus to facilitate sperm transport.
Luteal phase (lower estrogen): thicker mucus after ovulation.
What is the growth of the endometrium?
During the luteal phase, the endometrium of the uterus thickens under the influence of progesterone made from the corpus luteum
Spiral arteries mature and the myometrium thickens
Cervical mucus thickens and becomes cloudy
By then end of the luteal phase, the corpus luteum degenerates and blood levels of progesterone and estrogen drop, thus removing endocrine support for the endometrium
Why is the menstrual phase considered a dramatic event in the cycle, and how does it impact daily life?
Image: Follicular → Luteal phase
Endometrial shedding during menstruation involves thickened endometrium, spiral arteries, and many active cells, causing tissue breakdown.
Can lead to pain and cramping due to muscle contractions.
Despite significant discomfort, society often under-recognizes menstrual pain compared to other ailments like headaches or colds.
Menstrual symptoms can impact daily activities, but awareness and accommodation are often limited.
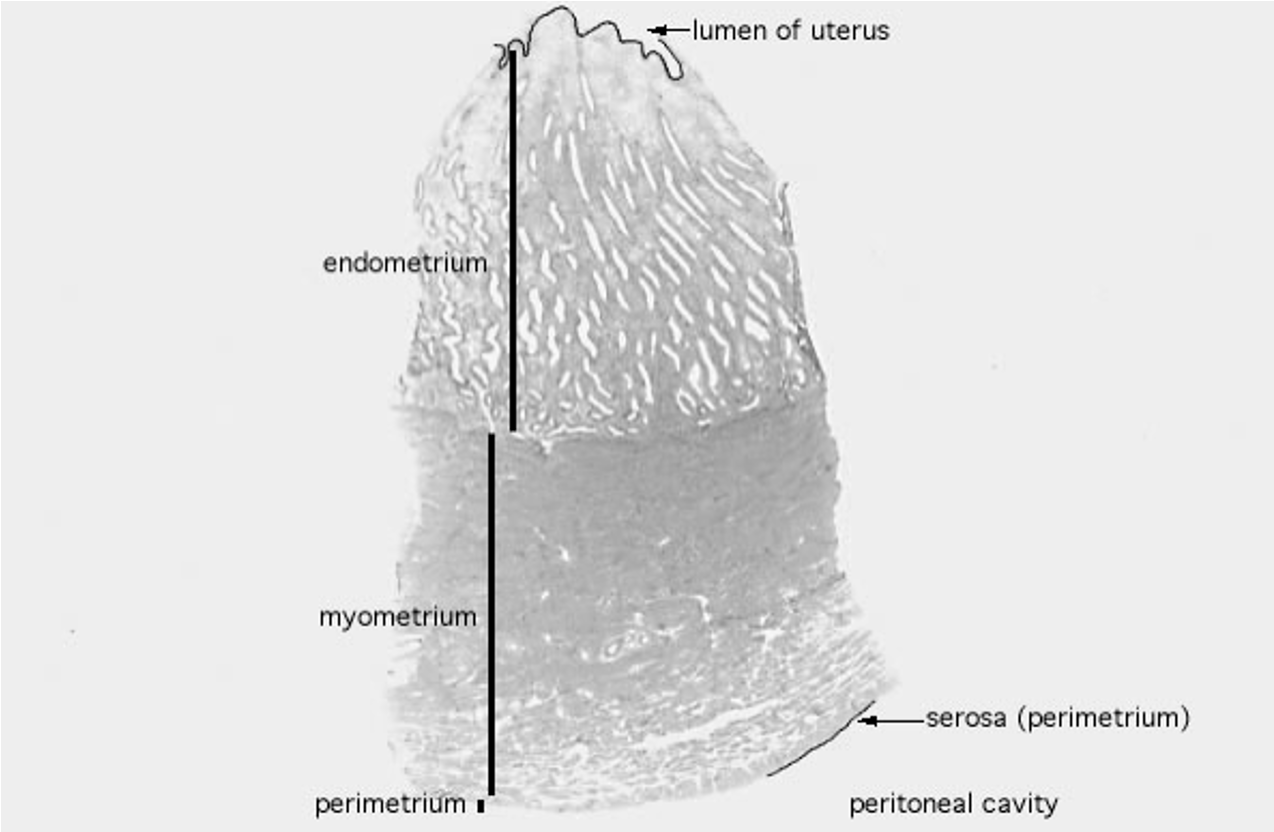
What are the layers of the uterus and what is the lumen?
Endometrium: inner lining that thickens and sheds during the menstrual cycle.
Myometrium: muscular middle layer, supports contractions.
Perimetrium: outer layer, part of the peritoneal/abdominal cavity.
Lumen: hollow central cavity where a fertilized egg implants and develops into an embryo/fetus.
What happens to granulosa and thecal cells in the luteal phase, and why is the estrogen-to-progesterone ratio important?
Granulosa lutein cells: secrete progesterone and inhibin.
Theca-lutein cells: secrete testosterone; estrogen also plays a role.
Corpus luteum focus: hormone production, not ovum release.
Estrogen-to-progesterone ratio: critical for implantation; imbalance can prevent a fertilized egg from successfully docking in the uterine wall.
Docking signals: hormonal balance and environmental factors (e.g., nicotine) influence early pregnancy success.

How do the hypothalamus and pituitary influence the menstrual cycle, and how are they typically assessed?
Hypothalamus & pituitary: control menstrual cycle via gonadotropins (LH & FSH).
Assessment methods:
Measure gonadotropin levels in blood.
Ultrasound of ovaries to check follicle development.
Hormonal profiles for system health.
Neuroimaging is possible but not routinely used.
Impact on cells: gonadotropins act via specific protein receptors that trigger biological activity.
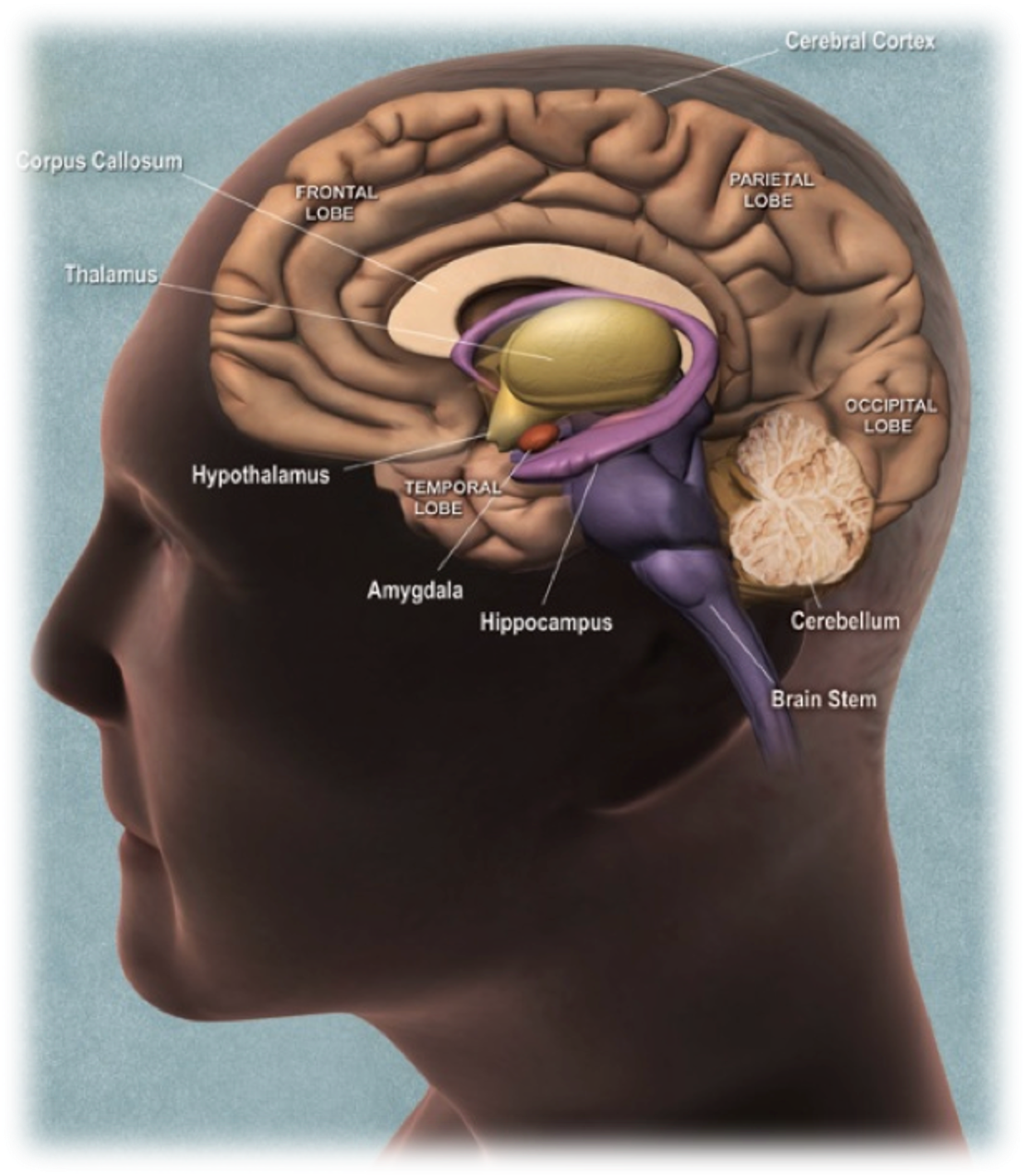
What is the function of the hypothalamus?
The hypothalamus is a small but important part of the brain. It contains several small nuclei with a variety of functions. It plays an important role in the nervous system as well as in the endocrine system. It is linked to another small and vital gland called the pituitary gland.
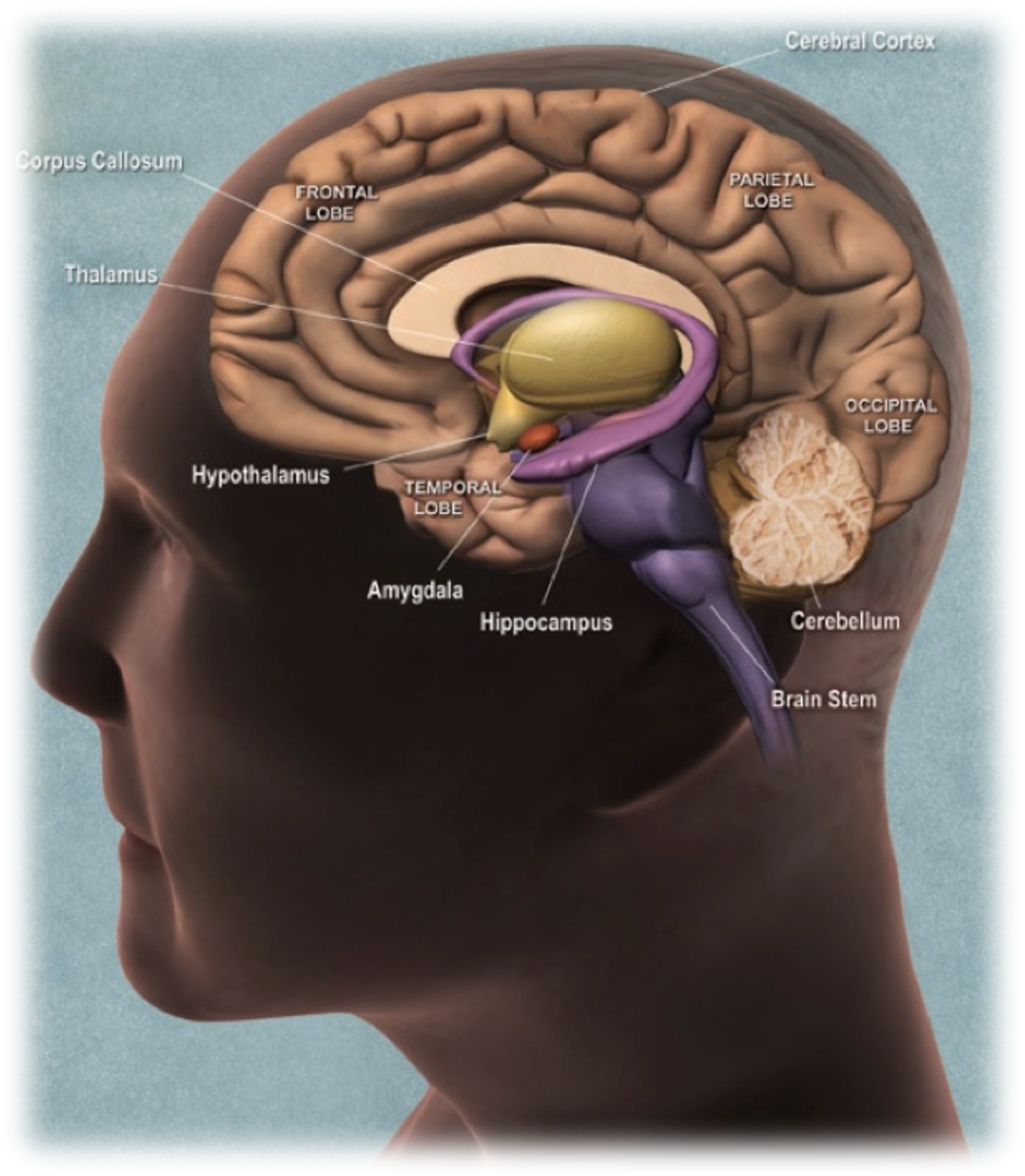
Where are the hypothalamus and pituitary located, and what is their structural organization?
Located in the diencephalon, which includes:
Epithalamus (top)
Thalamus
Subthalamus (under thalamus)
Hypothalamus (important for neuroendocrine control)
Nuclei: clusters of neuron cell bodies in each region perform specific functions.
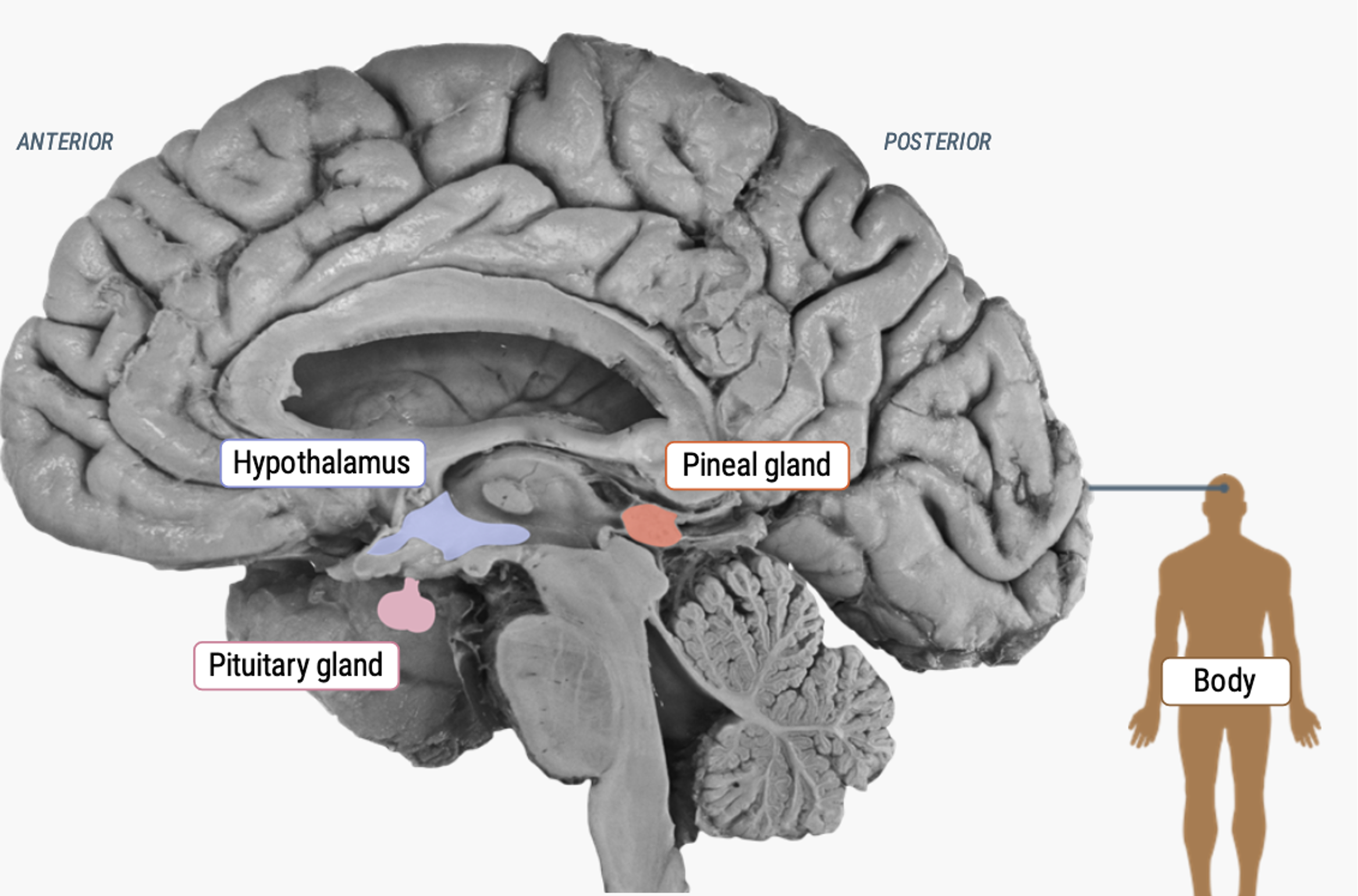
What is the pineal gland, and how does it relate to the hypothalamus and pituitary?
Located near the hypothalamus and pituitary in the diencephalon.
Produces melatonin, regulating sleep-wake cycles.
Activity is mediated by light: low melatonin signals morning.
In non-human animals, important in mating and breeding seasons.
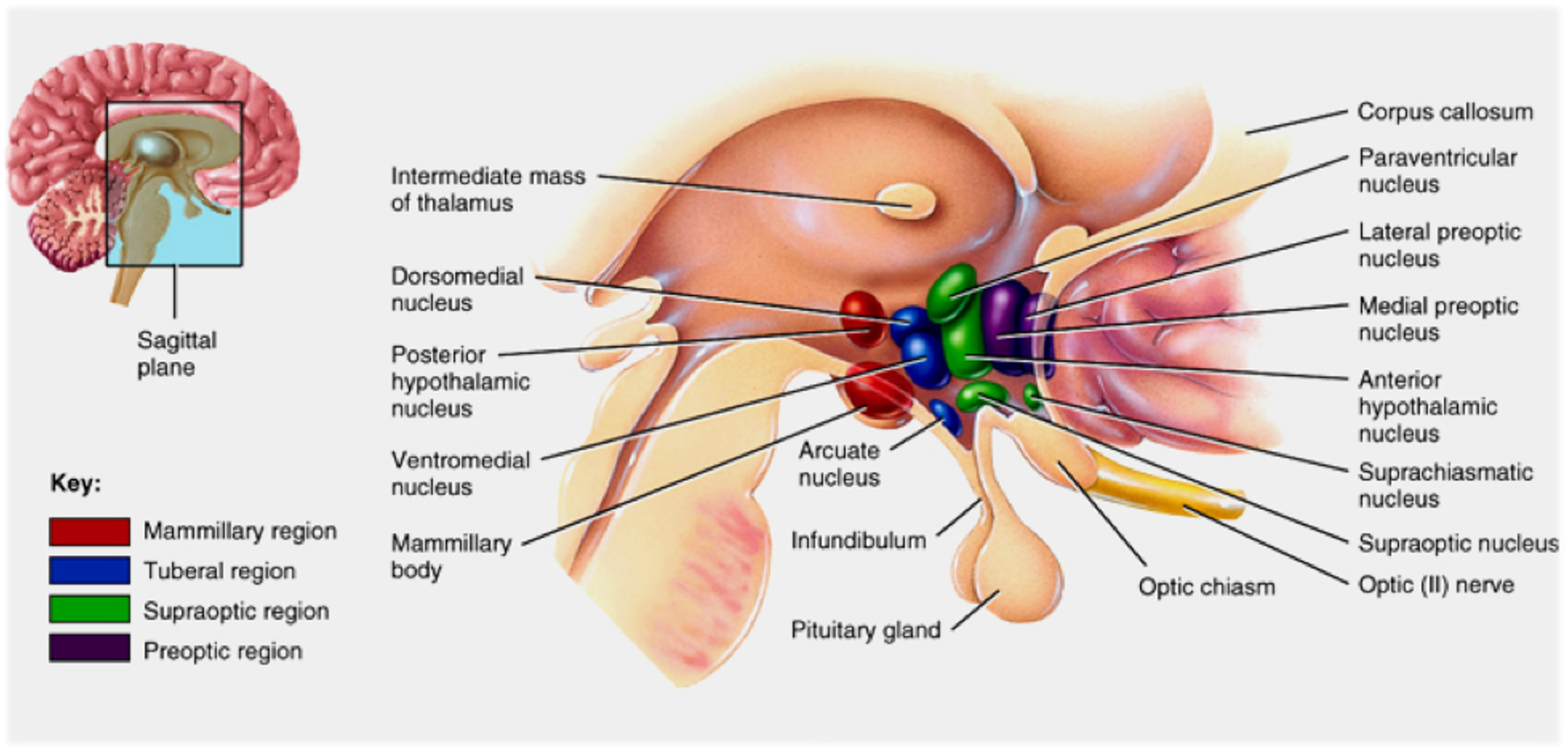
What are parts of the hypothalamus?
Parts:
Mammillary region - resembles mammary glands
Tuberal region - narrow
Supraoptic region - above optic chiasm
Preoptic region - before the optic chiasm
Location:
The Hypothalamus and Pituitary gland are located deep in the brain.
They receive sensory information and have many connections with the Limbic system, which processes emotions.
**Don’t need to label this slide
What are the hypothalamus functions?
[1] release of major hormones
[2] thermoregulation
[3] control of food and water intake
[4] sexual behaviour and reproduction
[5] daily cycles in physiological state
[6] mediation of emotional responses
*Don’t memorize this, will most likely be “All of the above” on MC
What are the major hypothalamic nuclear groups?
“Pharmacological Museum”
The list of putative (possible) neurotransmitters includes the “classical” transmitters ACh, GABA, glutamate, serotonin, and dopamine
And, dozens of peptides that have been identified in recent years
**Memorize the chart
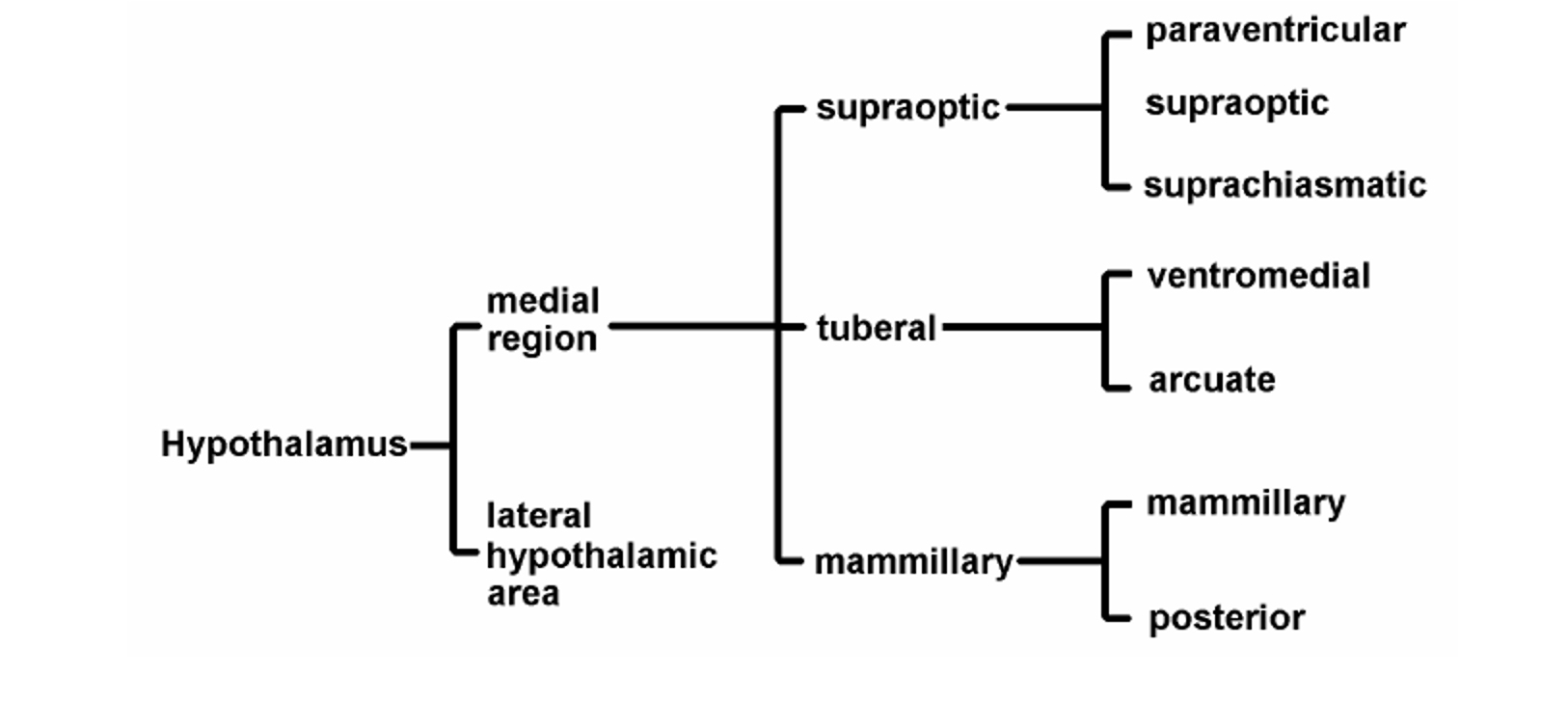
What is the Supraoptic or anterior region?
Location: Near the optic chiasm
Nuclei: Supraoptic & paraventricular
Hormones: E.g., Oxytocin by supraoptic and paraventricular and Corticotropin-releasing hormone (CRH) by paraventricular
What is the Tuberal or middle region?
Location: Near the 3rd ventricle
Nucleus: Arcuate
Hormones: E.g., Gonadotropin-releasing hormone (GnRH)
What are the Mammillary or posterior regions?
Location: Near the mammillary bodies
Nuclei: Do not appear to be closely related to endocrine functions
Why is the anatomy of the Hypothalamus and Pituitary important?
Anterior pituitary: produces its own hormones.
Median eminence: base of hypothalamus where hypothalamic releasing hormones (e.g., Gonadotropin-releasing hormone) enter a blood portal system to regulate the anterior pituitary.
Key parts: pars tuberalis (near hypothalamus), pars distalis (main part, farther from hypothalamus), pars intermedia (boundary between anterior & posterior).
Posterior pituitary: does not make hormones; hormones are made in hypothalamic neurons and released here.
Infundibulum (infundibular stalk): neural connection from hypothalamus to posterior pituitary.
Pars nervosa: bottom portion of the posterior pituitary where hormones are released.
Clinical relevance: Structures near the Optic chiasm and hypothalamus can be affected by tumors, which may disrupt visual processing or hypothalamus–pituitary signaling.
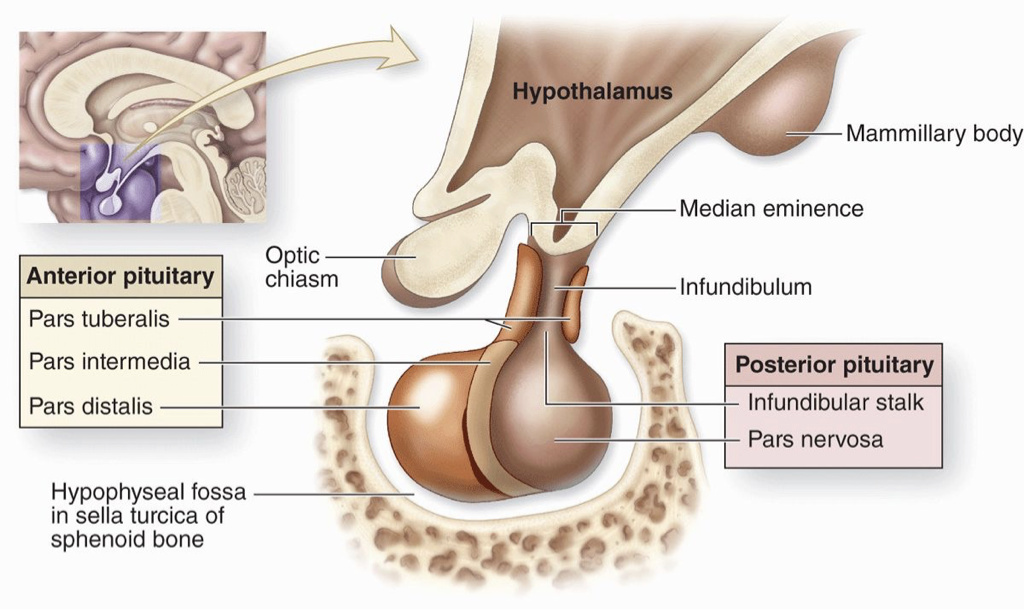
What are important structures of the hypothalamus?
The hypothalamic-pituitary portal system is a specialized network of blood vessels that connects the hypothalamus to the anterior pituitary gland.
The median eminence and the infundibulum are small regions located at the base of the hypothalamus, where it meets the pituitary gland.
These are important structures for the regulation of the endocrine system, as they are the site of the hypothalamic-pituitary portal system.
The neurons of the hypothalamus release a variety of hormones that enter the portal vessels and are transported directly to the anterior pituitary gland, where they [1] stimulate or [2] inhibit the release of pituitary hormones.
![<ul><li><p><strong>The hypothalamic-pituitary portal system </strong>is a specialized network of blood vessels that connects the hypothalamus to the anterior pituitary gland.</p></li><li><p>The <strong>median eminence</strong> and the <strong>infundibulum</strong> are small regions located at the base of the hypothalamus, where it meets the pituitary gland.</p><ul><li><p>These are important structures for the regulation of the endocrine system, as they are the site of the hypothalamic-pituitary portal system.</p></li></ul></li><li><p>The neurons of the hypothalamus release a variety of hormones that enter the portal vessels and are transported directly to the anterior pituitary gland, where they [1] stimulate or [2] inhibit the release of pituitary hormones.</p></li></ul><p></p>](https://assets.knowt.com/user-attachments/1da0b4d8-0866-4310-968d-f7d180262b10.png)
What is the Pars distalis?
Also known as the anterior lobe, the pars distalis is the largest and most prominent part of the anterior pituitary.
It consists of glandular tissue organized into clusters of cells called adenohypophyseal cells or anterior pituitary cells.
Hypophyseal - another name for blood portal system
These cells secrete a variety of hormones that regulate numerous physiological processes, including growth, metabolism, reproduction, and stress response.
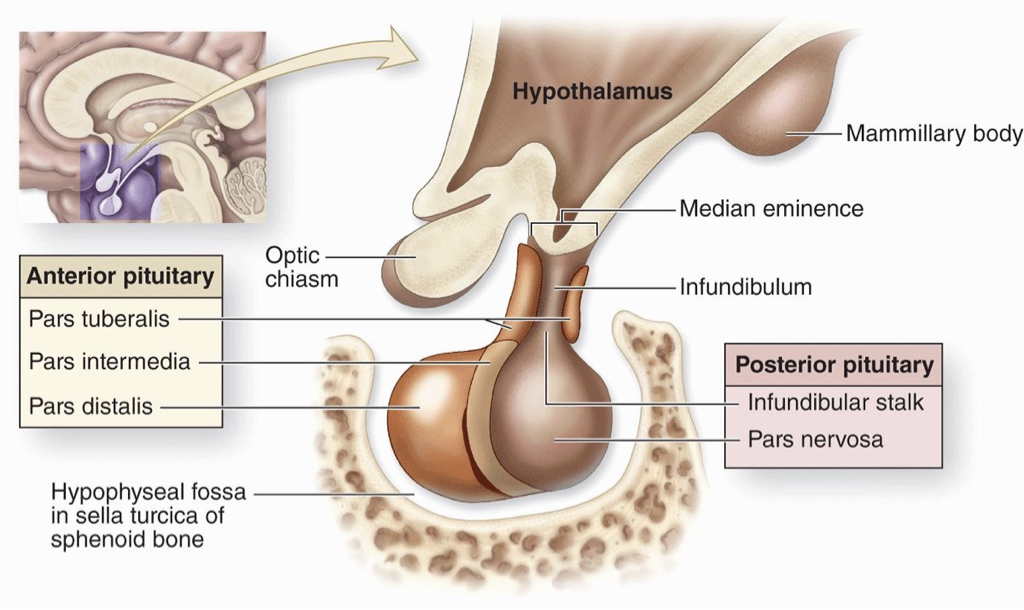
What is the Pars nervosa?
Also known as the neurohypophysis, is the posterior lobe of the pituitary gland.
It is primarily composed of axons and nerve endings of neurons originating from the hypothalamus, specifically the supraoptic and paraventricular nuclei.
These neurons extend their axons through the pituitary stalk (infundibulum) and terminate in the pars nervosa.
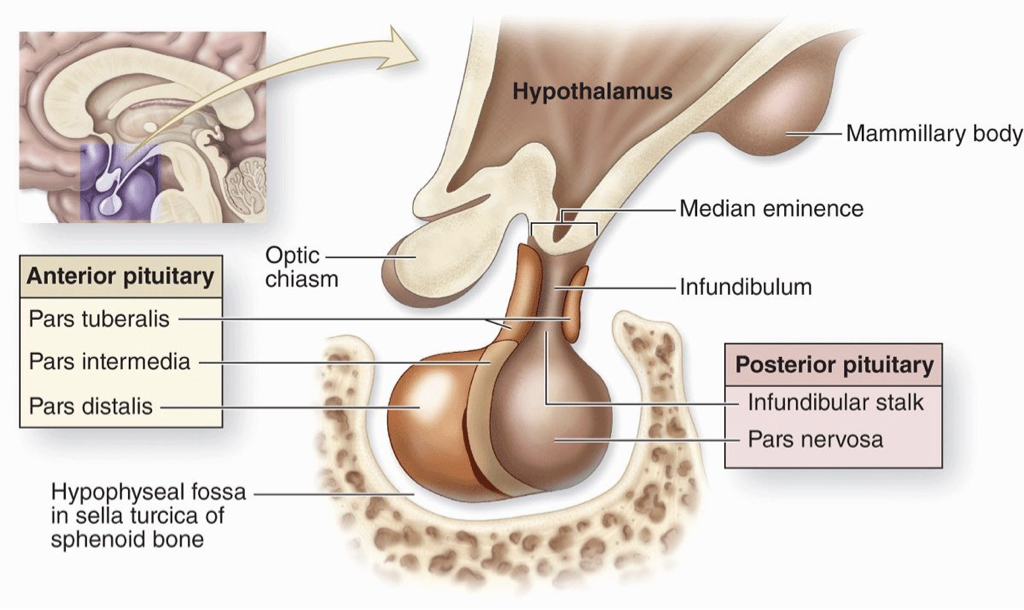
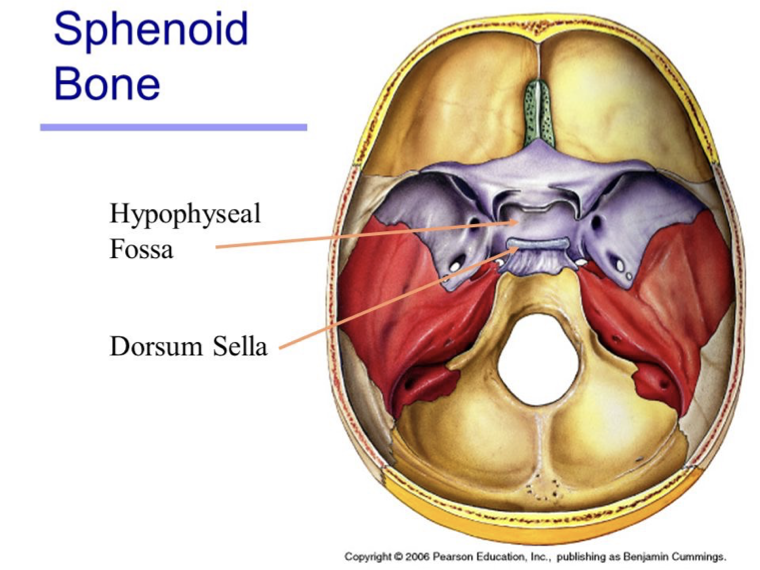
What is the hypophyseal fossa?
The pituitary sits in the hypophyseal fossa, a depression in the Sphenoid bone (within the Sella turcica).
It is surrounded by a sinus (cavity) that contains Cerebrospinal fluid, helping cushion and protect the brain.
This region is also associated with the hypophyseal portal system, which carries hypothalamic hormones to the anterior pituitary.
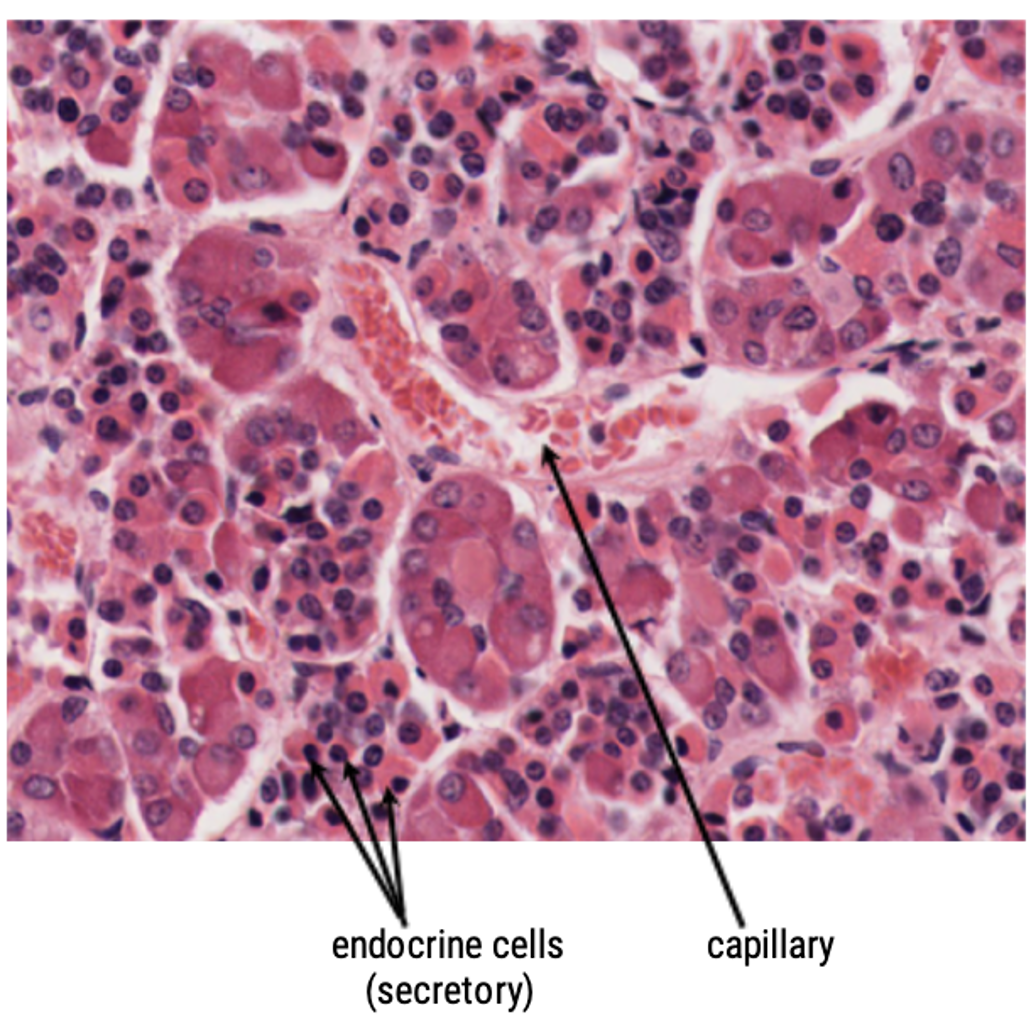
What do the endocrine cells of the anterior pituitary produce and secrete?
6 hormones
Adrenocorticotropic hormone (ACTH)
Thyroid-stimulating hormone (TSH)
Follicle-stimulating hormone (FSH)
Luteinizing hormone (LH)
Growth Hormone (GH)
Prolactin (PRL)
Strong capillary system
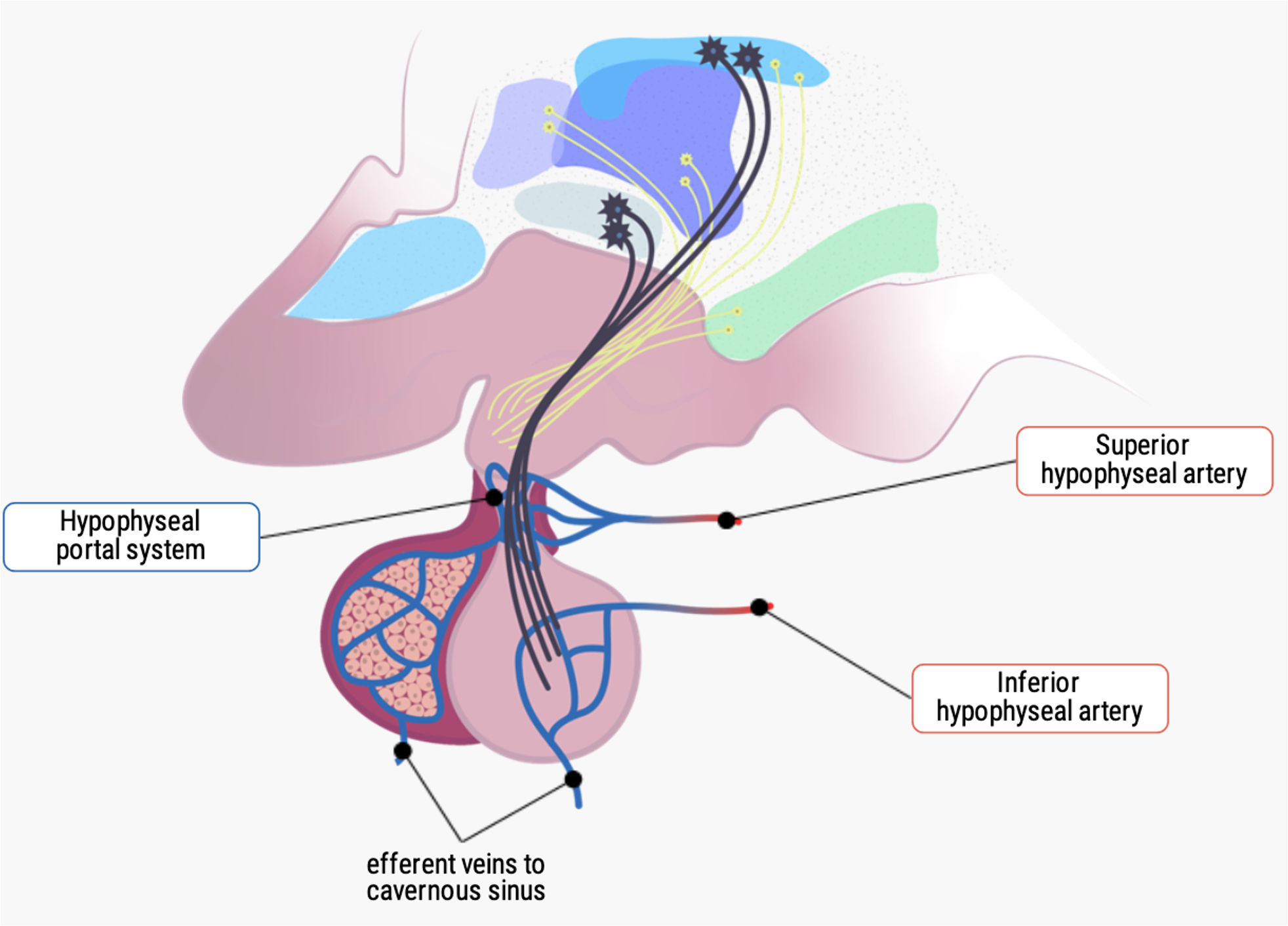
How are these hormones supplied with blood?
Through the extensive arterial and venous system!
Hormone release is regulated by the Hypophyseal portal system, which carries hypothalamic releasing hormones to the anterior pituitary.
The gland has a rich blood supply:
Superior and inferior hypophyseal arteries bring blood in.
Efferent veins carry hormones from the pituitary to the rest of the body.
What hormones does the posterior pituitary secrete?
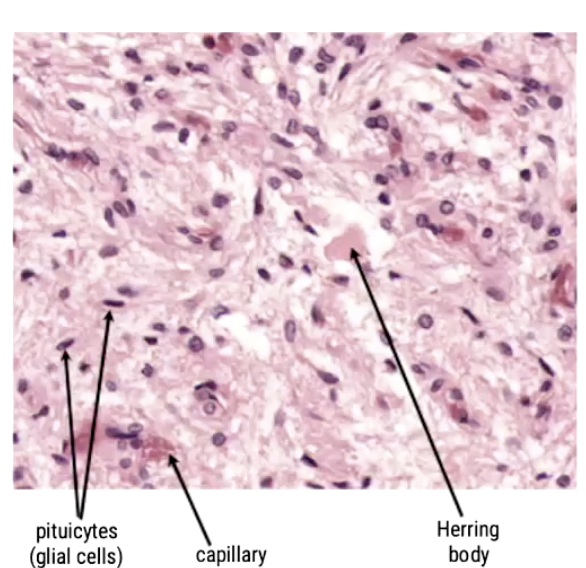
The posterior pituitary does not produce hormones. It secretes 2 hormones that are produced in the hypothalamus and temporarily stored in Herring bodies before release:
Oxytocin (OXT)
Antidiuretic hormone (ADH) - aka vasopressin: prevents the release of fluid from your system
Pituicytes (glial cells): Helper cells to maintain the health, repair and structural integrity of neurons
Capillary
Herring body - neurons which allow release of the hormones
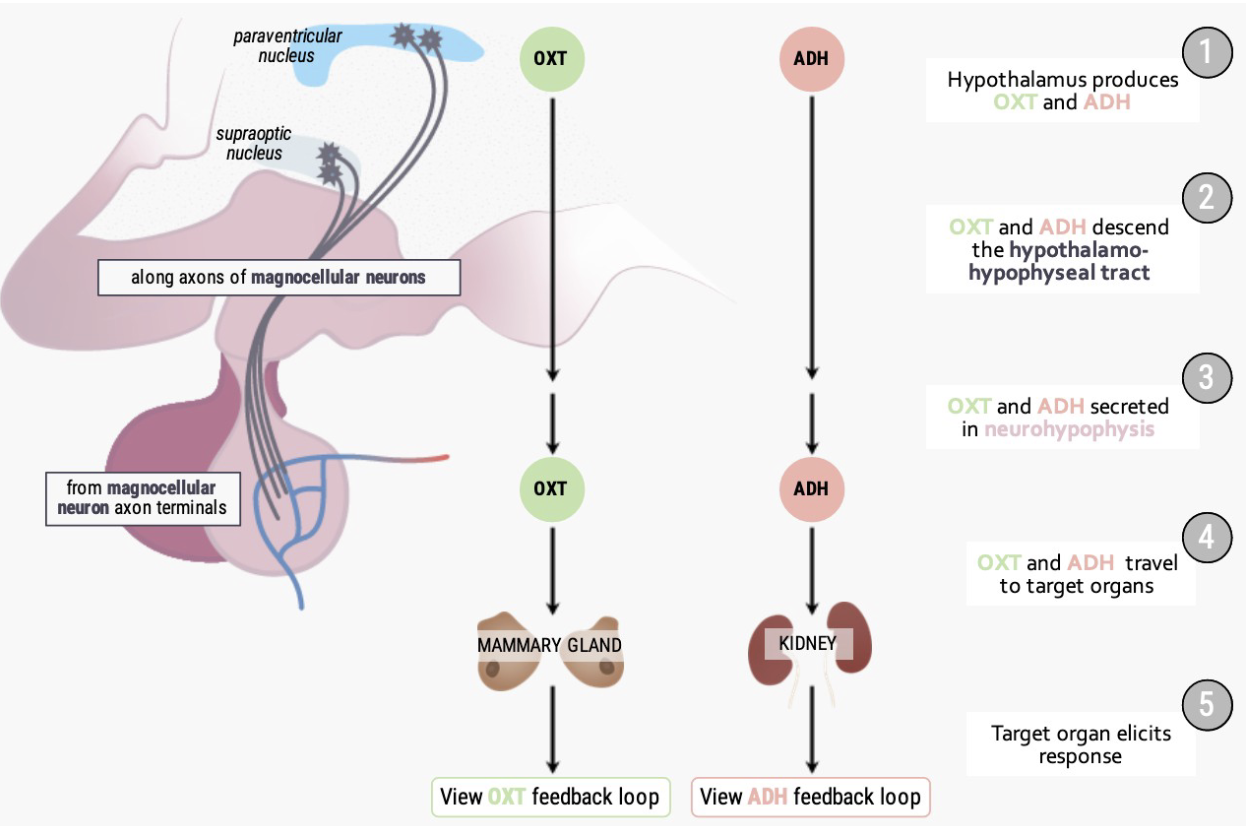
What are the main functions of Oxytocin and Vasopressin, and how are they produced?
Production:
Made by Paraventricular nuclei and Supraoptic nuclei.
Travel along magnocellular neurons to the posterior pituitary for release.
Oxytocin functions:
Milk letdown reflex – triggered by sensory input (sight, sound, smell of infant).
Uterine contractions – during childbirth and orgasm.
Possibly supports fertility by aiding sperm transport during orgasm.
Vasopressin (ADH) functions:
Prevents urine formation, retaining fluid in the body.
Acts mainly at the kidneys to regulate water balance.
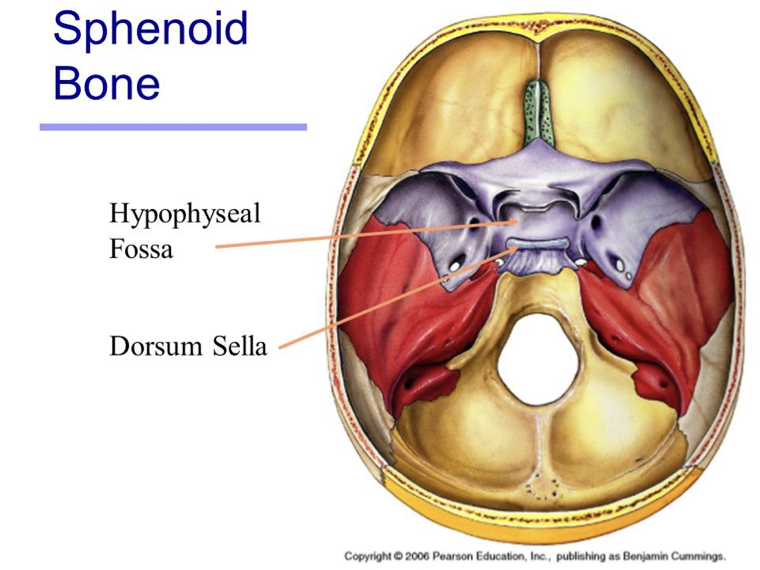
What is the sphenoid bone?
Lies at the base of the skull, in the bone there is a small cup called ‘sella turcica’, the pituitary gland (or hypophysis) lies in the cup
The hypophyseal fossa is a bony depression in the sphenoid bone of the skull that houses the pituitary gland, also known as the hypophysis. It is located at the base of the brain, behind the eyes, and below the optic chiasm.
Dorsum sella: Part of the Sphenoid bone, and Hypophyseal Fossa.
It is located behind the optic chiasm, forming the posterior boundary of the hypophyseal fossa, where the pituitary gland sits.
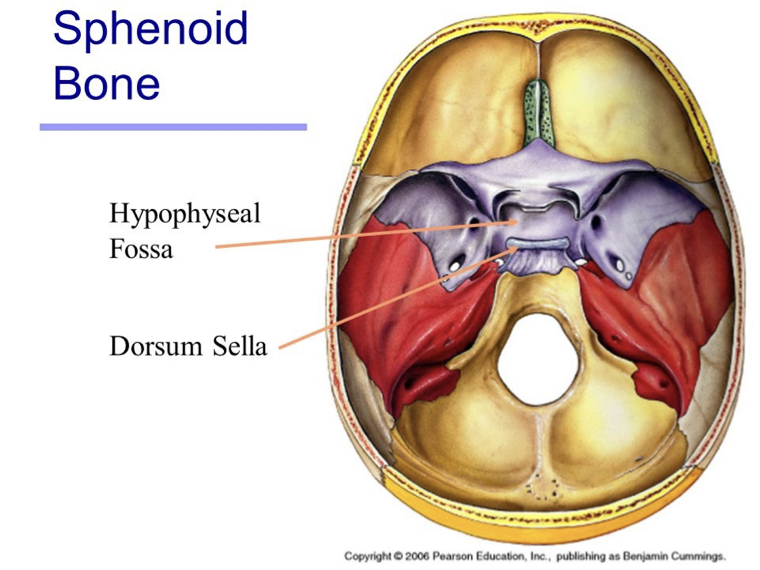
Where does the pituitary gland sit?
The sphenoid bone lies at the base of your skull, and in this bone is a small, cup-shaped depression.
Lying in this depression is a round ball of tissue, about 1.3 cm (0.5 in) in diameter, called the pituitary gland or hypophysis.
What is the hypophyseal fossa?
The hypophyseal fossa is a crucial structure for the diagnosis and treatment of pituitary disorders.
Abnormalities of the pituitary gland, such as tumors or cysts, can be detected through imaging techniques such as magnetic resonance imaging (MRI) or computed tomography (CT) scans of the hypophyseal fossa.
These imaging techniques can also guide the surgical removal of pituitary tumors, which can cause hormonal imbalances and other health problems (common in ages 10-12).
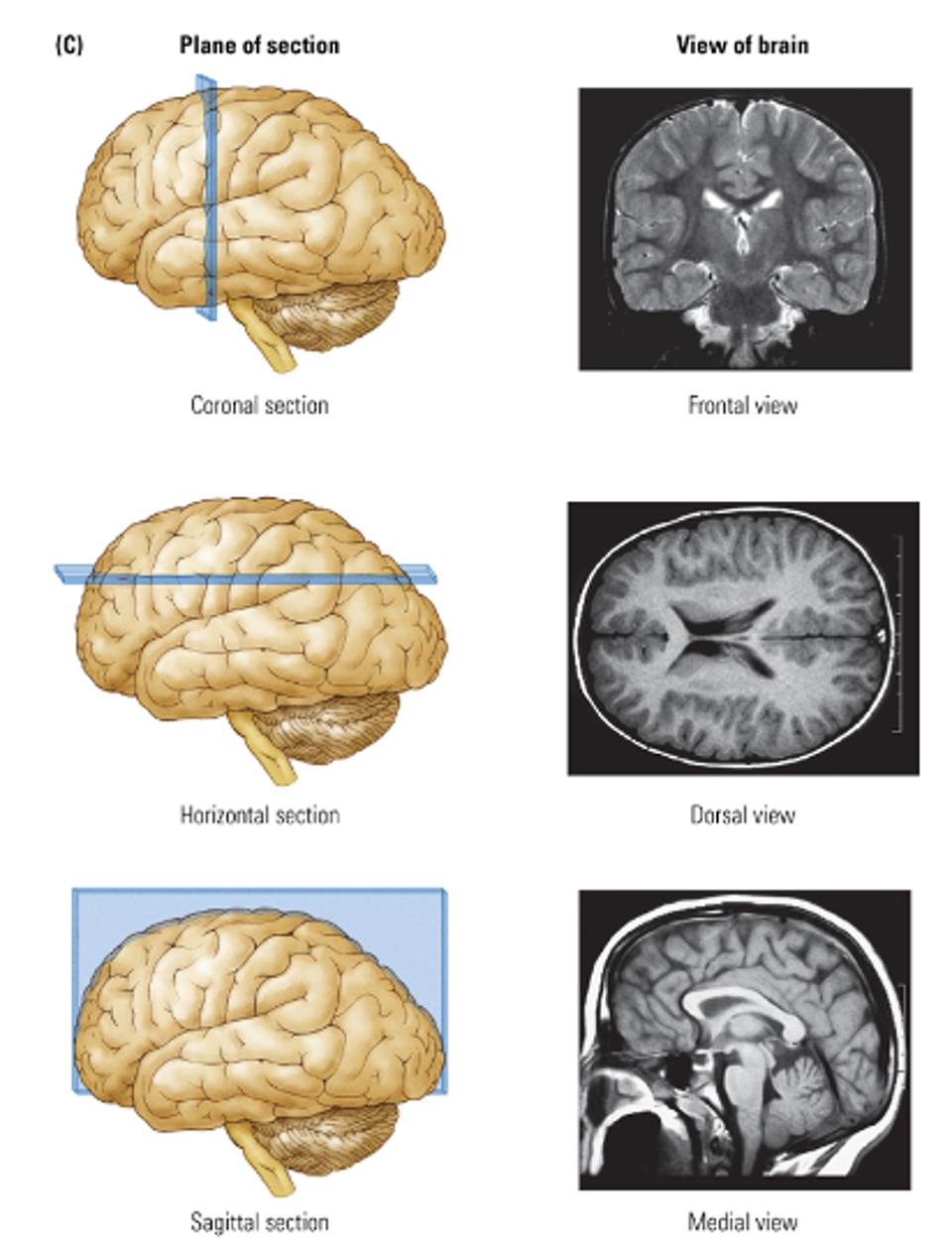
What are the planes of the brain?
Coronal section → Frontal view
Horizontal section → Dorsal view
Sagital section → Medial view (clear access to hypothalamus pituitary)
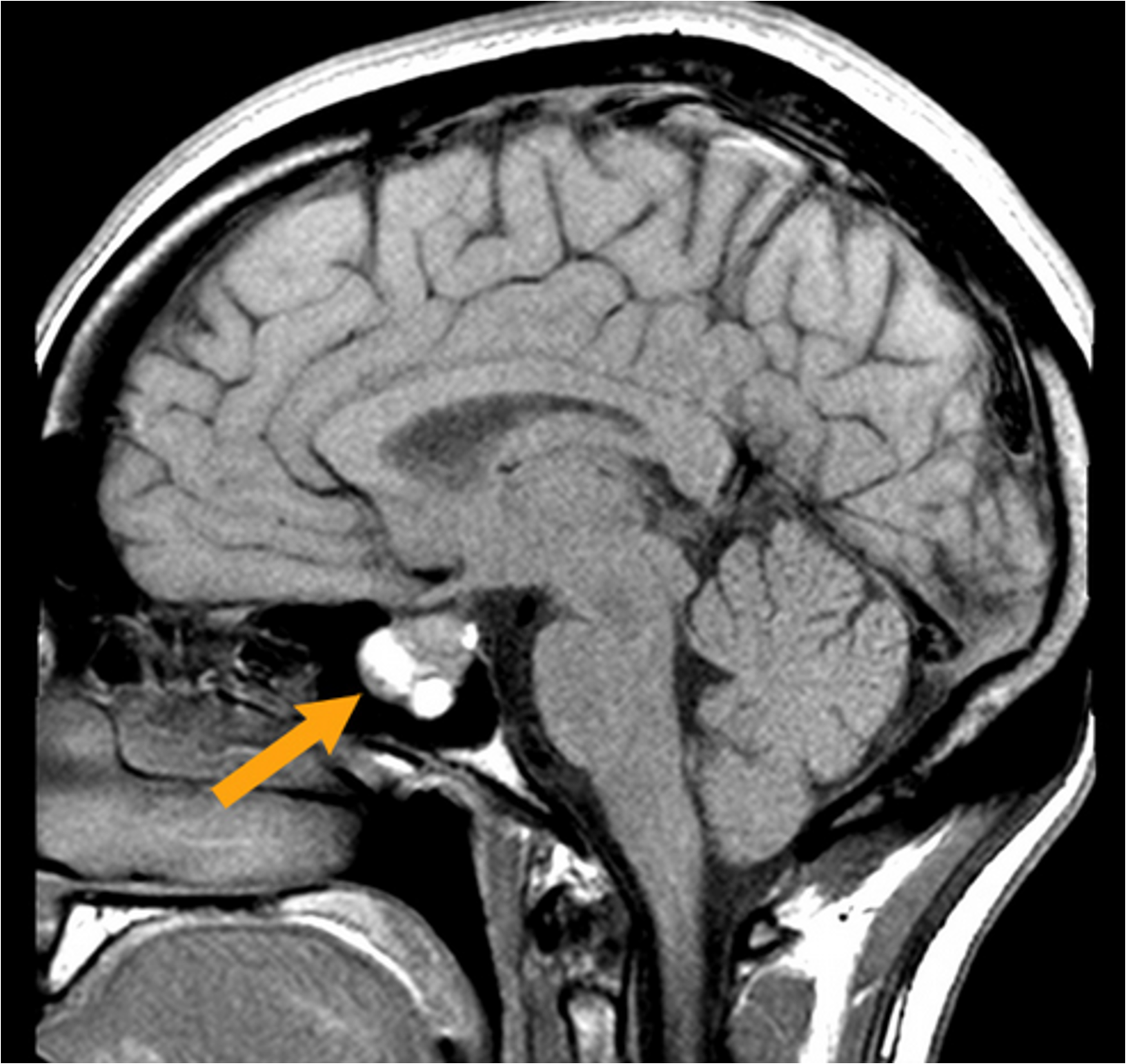
What is a functional pituitary adenoma?
A functional pituitary adenoma is a benign (non-cancerous) tumor of the pituitary gland.
“Functional” means it increases hormone production, unlike non-functional tumors which are just masses.
Can affect endocrine function, causing symptoms related to overproduction of pituitary hormones.
Even though it does not spread, it can impact brain function and hormone balance.
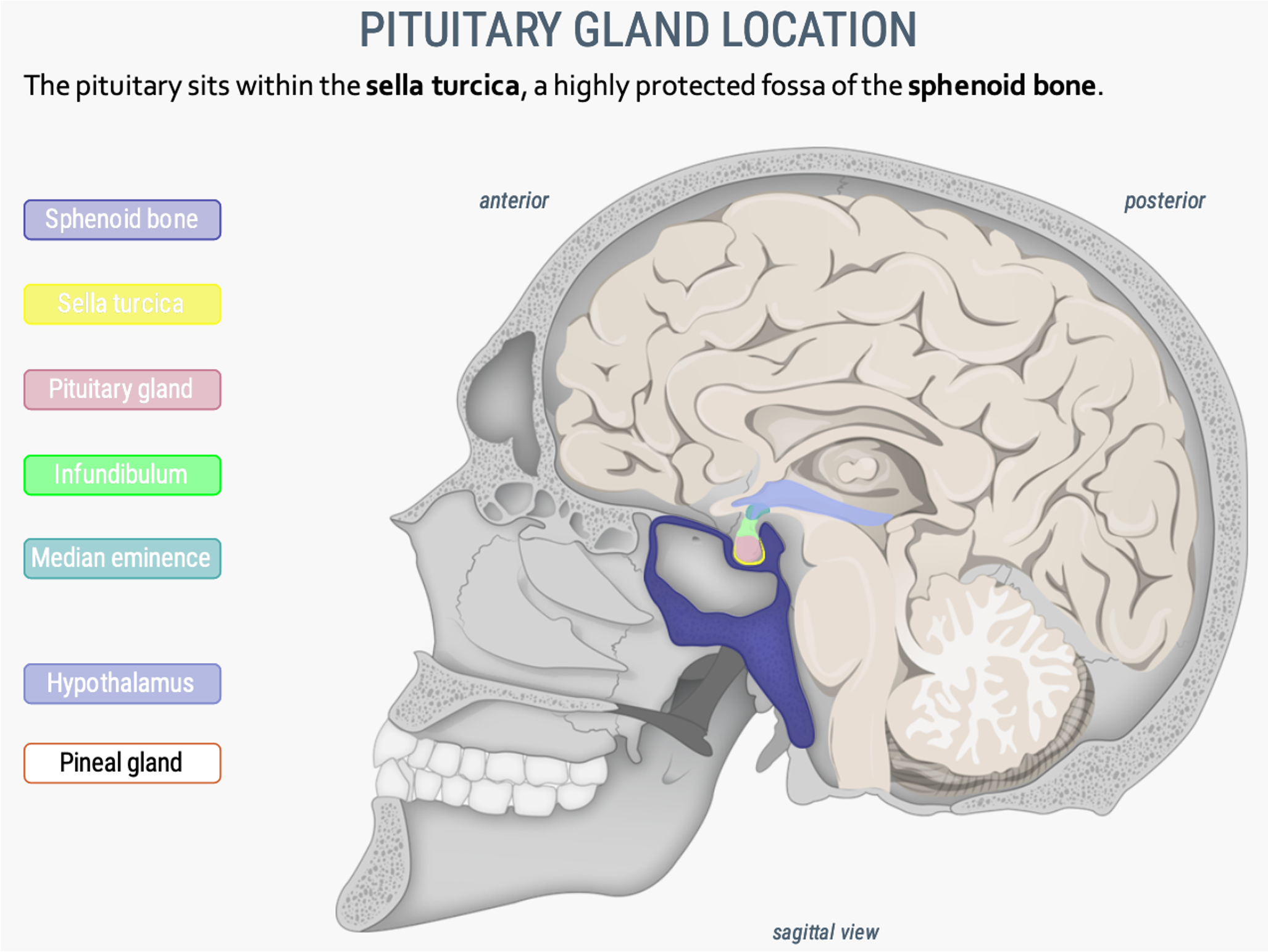
What is the pituitary gland location?
The pituitary sits within the sella turcica (yellow), a highly protected fossa of the sphenoid bone (dark purple).
**Check image
What are protein hormones?
Most vertebrate hormones are proteins.
Protein hormones that are only a few amino acids in length are called peptide hormones.
Whereas the larger ones are called protein hormones or polypeptide hormones.
Protein hormones: insulin, the glucagons, the neurohormones of the hypothalamus, the tropic hormones of the anterior pituitary, inhibin, calcitonin, parathyroid hormones, the gastrointestinal hormones, ghrelin, leptin, adiponectin, and the posterior pituitary hormones.
Protein and peptide hormones are produced in endocrine cells and are released into the circulatory system by means of exocytosis.
What are general features of protein hormones?
Tend to be larger molecules (than steroid hormones)
More fragile: break up within seconds
Water Soluble: action is much shorter
Receptors: found in the plasma membrane
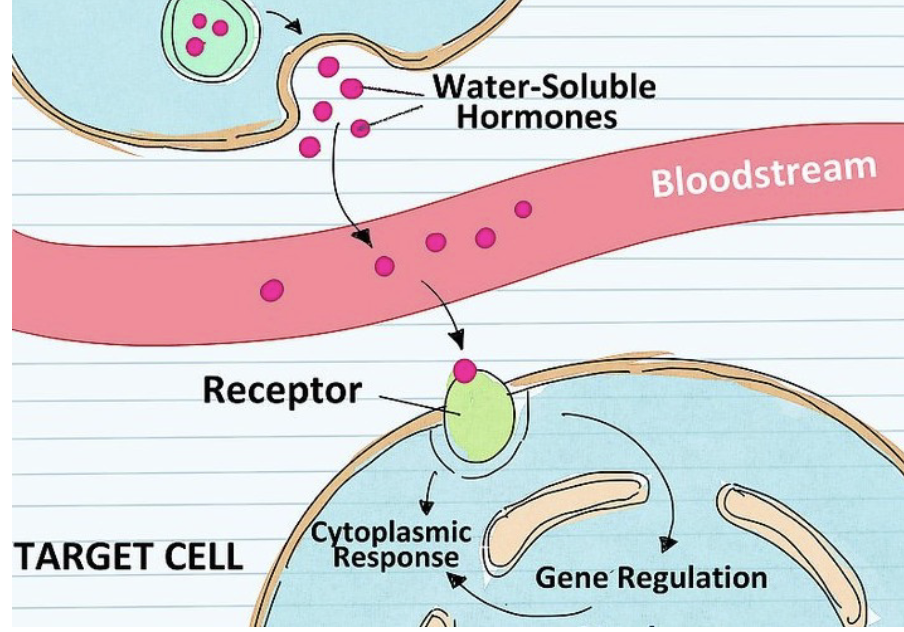
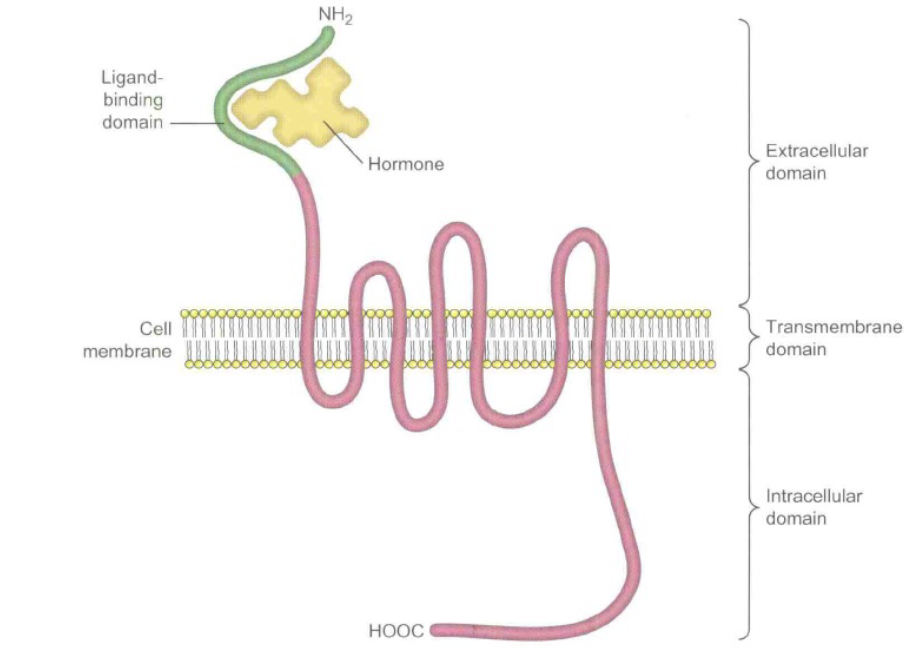
Where are these receptors located?
In the plasma membrane of all these structures
Where are the anterior pituitary hormones (luteinizing hormone and follicle-stimulating hormone) secreted?
Are secreted by the basophils.
They consist of 200-220 amino acids.
Approximately 10-20% (MEMORIZE) of the molecular structure of each of these hormones is carbohydrate and they are known as glycoproteins.
Each glycoprotein is composed of 2 subunits: alpha and beta.
These subunits have no biological activity separately; both subunits are necessary to produce a biological response
The alpha subunits of LH and FSH are identical.
The beta subunit imparts the specific biological function of the molecule and also determine the species specificity.
LH and FSH are also known as gonadotropins because in response to GnRH they stimulate steroidogenesis in the gonads as well as the development and maturation of gametes.