chapter 8 oxidation and reduction
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
redox reaction
Oxidation-reduction reaction, in which one molecule accepts electrons (it becomes reduced) from another molecule (which becomes oxidized). Also called electron transfer.
oxidation and reduction always occur
together
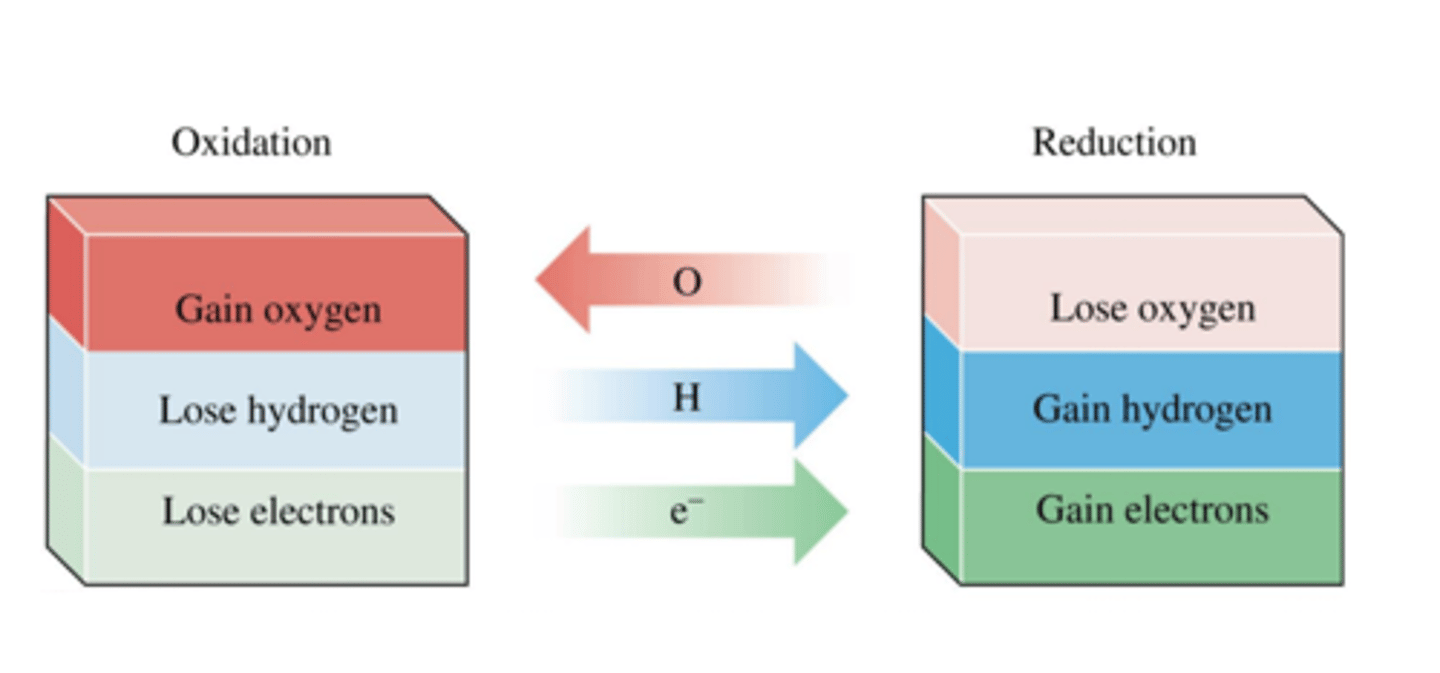
oxidation oxygen atoms
Oxidation electrons
oxidation in terms of hydrogen
oxidation oxidation number
gain of oxygen atoms
loss of electrons
loss of hydrogen
increase oxidation number
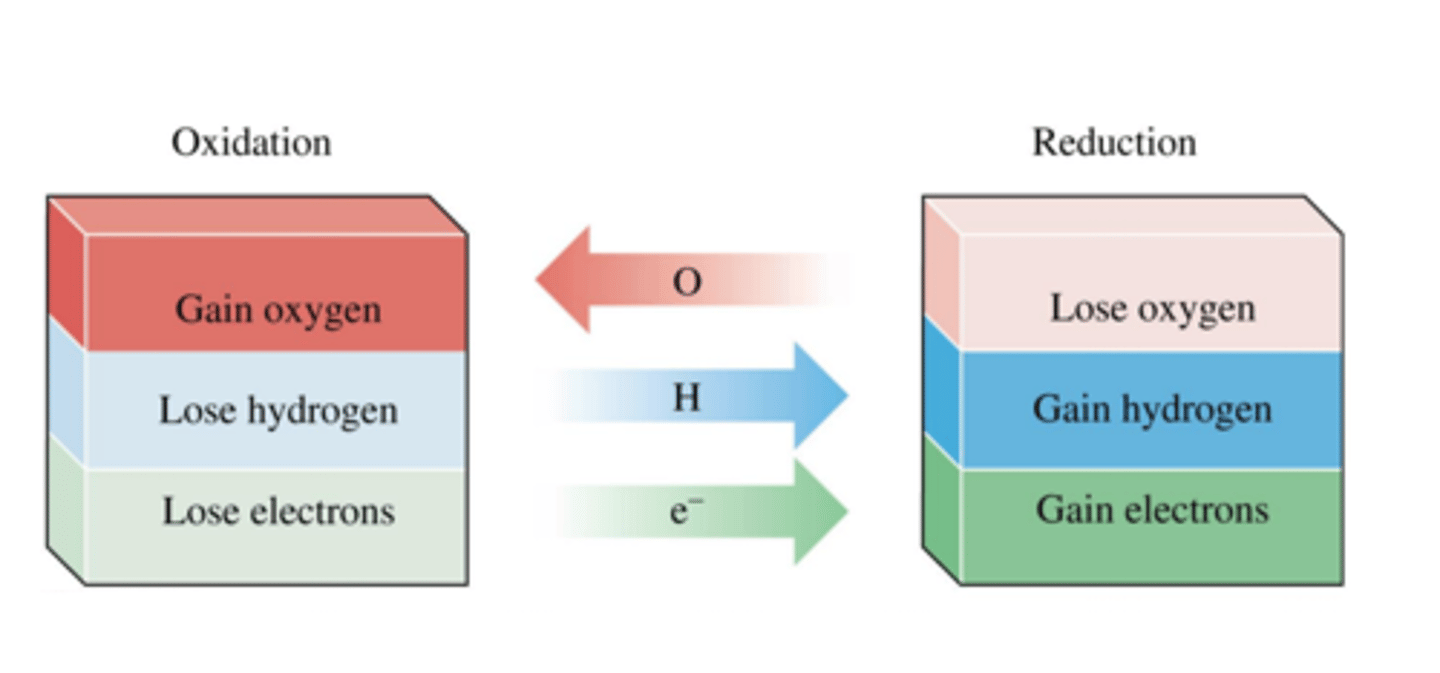
reduction oxygen atoms
reduction electrons
reduction in terms of hydrogen
reduction oxidation number
loss of oxygen atoms
gain of electrons
gain of hydrogen
decrease oxidation number
OIL RIG
oxidation is loss,
reduction is gain
whenever oxidation occurs what also happens
reduction must also occur in exactly the same amount
in monoatomic ions the oxidation number is
the number of unit charges on the atom
an atom in elemental form has an oxidation number of
0
oxidation and reduction reactions can produce electricity true or false
true
an atom in a monoatomic ion has an oxidation number equal to
equal to the charge of the ion
hydrogen has oxidation number of
oxygen?
+1
-2
the sum of the oxidation numbers of the atoms in a poly atomic ion equals
the charge of the ion
patina
surface discoloration caused by age and oxidation
the oxidation number for an element is equal to
8 -its group number
oxidizing agent
The electron acceptor in a redox reaction.
reducing agent
electron donor
electrochemical cell
gives an electric current with a steady voltage as a result of an electron transfer reaction
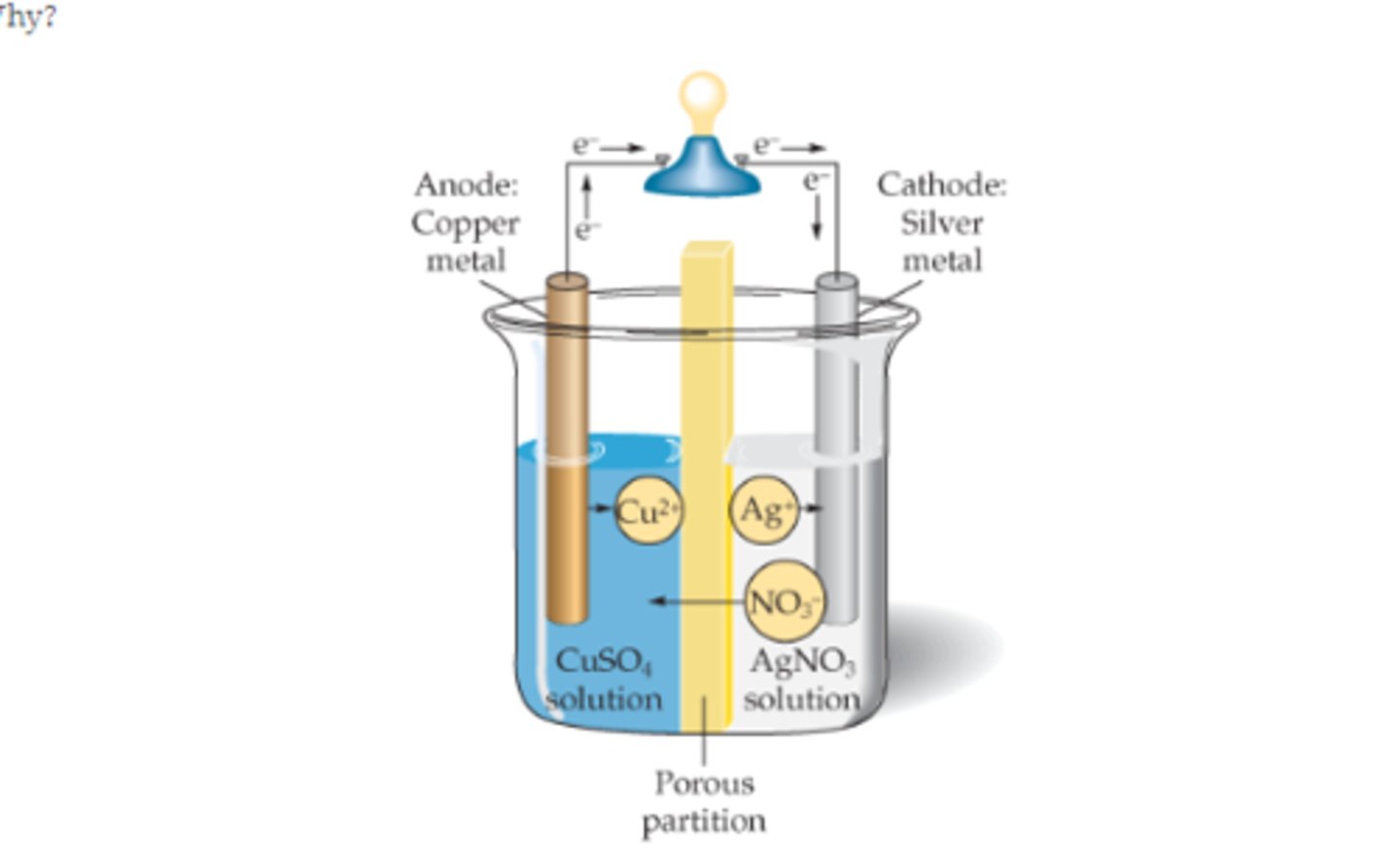
the porous barrier in an electrochemical cell is usally
a salt bridge
electrodes
the two pieces of metal where elctrons are transferred
Anode
Positive electrode, where oxidation occurs.
Cathode
Negative electrode , where reduction occurs.
two half reactions
electrochemical reaction are often split into two
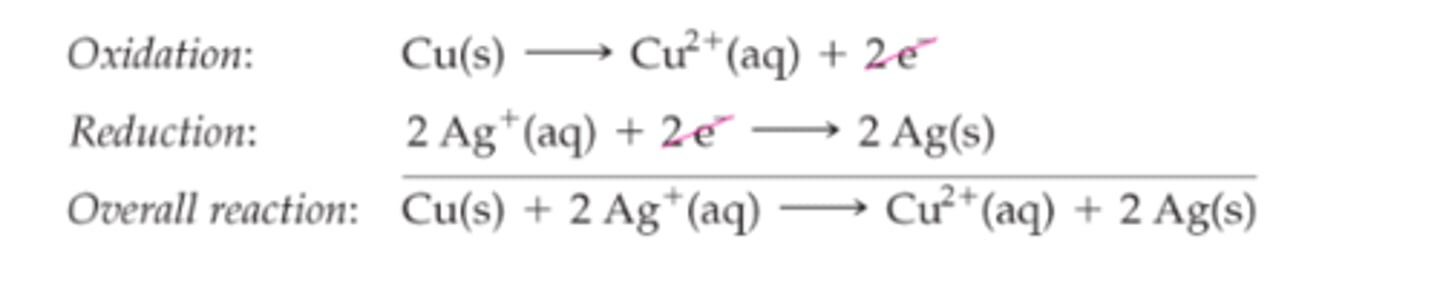
Do metals gain or lose electrons?
to lose
Which quantities are conserved in all redox reactions?
mass onlyboth
charge and mass
charge only
charge and mass
how does an electric eel creat electricity
uses thousands of stacked cells and a difference between potassium and sodium
photochromic glass
glass that changes its optical properties in response to light intensity...
result of oxidation redcution reactions
use silver chloride and copper the silver is susceptible to oxidation and reduction by light
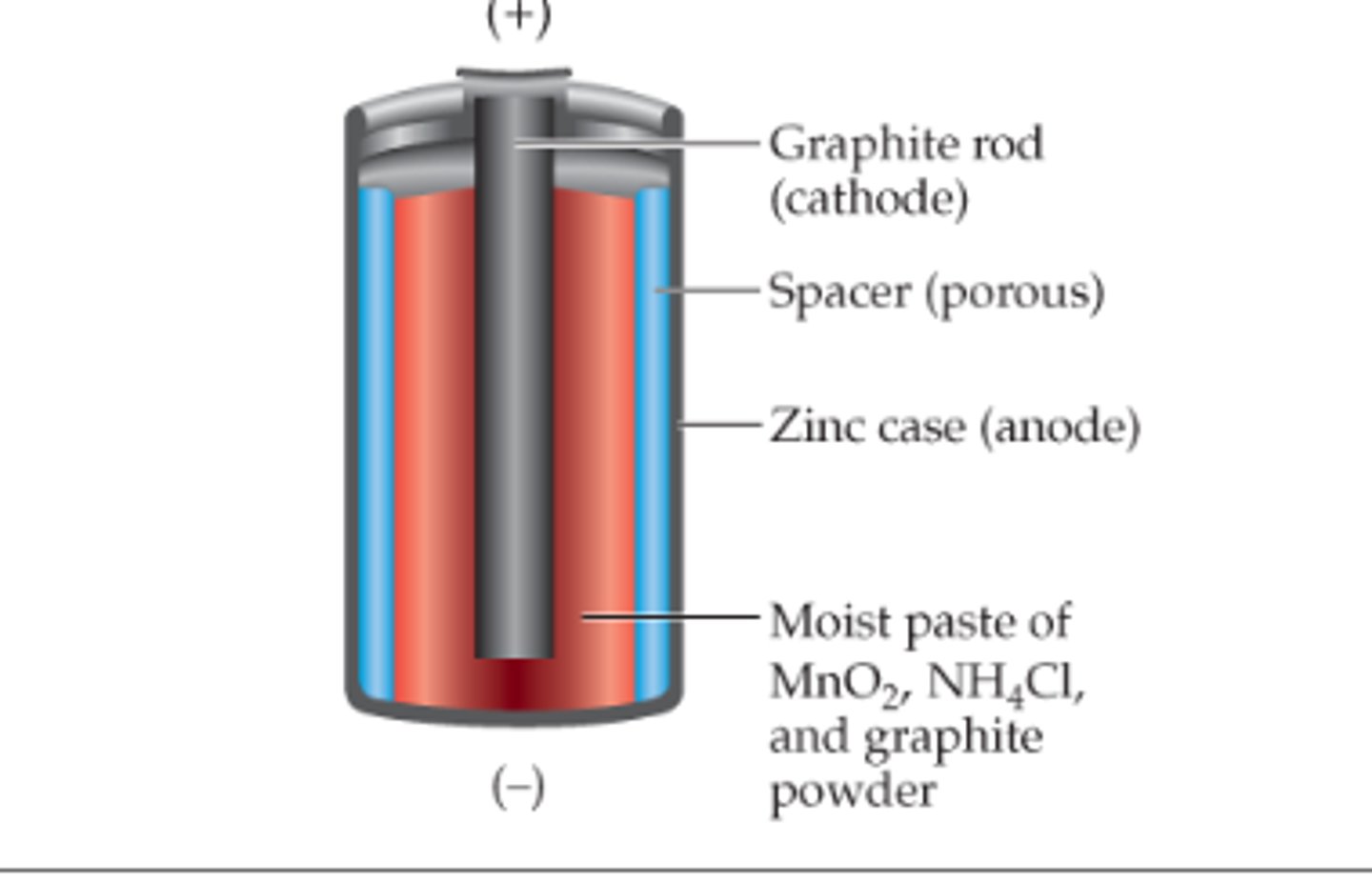
dry cells
an electrochemical cell in which the electrolyte is a paste (graphite powerder)
Battery
dry cell
most important feature of lead storage battery
it can be recharged
In the following reaction that takes place in batteries, which element is being reduced?
Manganese
nickel-cadmium battery
A type of rechargeable battery using nickel oxide hydroxide and metallic cadmium as electrodes.
litium ion battery
anode?
cathode?
electrolyte?
graphite
lithium manganese dioxide
mixture of lithium salts
protection of aluminum
Aluminum is more reactive than iron. However, corrosion of aluminum is not a serious problem. Aluminum oxide is very tough and prevents further corrosion.
The black tarnish on silver is
ag2s
Silver tarnishing
oxide on the silver surface
In a reaction, the substance undergoing reduction serves as the A) electron donor. B) proton donor. C) oxidizing agent. D) reducing agent.
oxidizing agent
why does a battery go dead
when one or more of the reactants zinc and MnO2 are used up
Why are TNT explosions so powerful?
TNT decomposes rapidly into several gaseous products.
whya re some batteries rechargeable
they use a reversible redox reaction the starting materials are reformed
How does silver tarnish?
How can the tarnish be removed without the loss of silver?
It is oxidized to silver sulfide.
Use aluminum to reduce the silver sulfide to metallic silver.
rhubarb
used in flow batteries
Which is true about the 12-volt (V) lead storage batteries used in automobiles?
They contain six 2-V cells.
They are filled with hydrochloric acid.
They cannot be recharged.
They contain lead and copper electrodes.
They contain six 2-V cells.
fuel cell
An electrical-chemical device that converts fuel, such as hydrogen, into an electrical current.
the oxidizing agent and reducing agen are supplied continuously to the cell
What is the main active ingredient in common household bleach, also known as chlorine bleach?
NaOCl
Electrolysis
the process in which an electric current is used to produce a chemical reaction, such as the decomposition of water
used to make metals from oxides
The processes of corrosion and explosion undergo oxidation-reduction reactions. Why do explosions tend to be so much more dangerous?
Explosions are often rapid, with huge increases in volume.
In an electrochemical cell, electrons flow from the
anode to cathode
how is a zinc block used to prevent ship rusting
used as a sacrificial anode and gets oxidized instead of iron
what 3 things must be present for corrosion of a metal to happen
water
oxygen
electrolyte
a chemical explosion is what type of reaction
rapid redox reaction
ANFO
ammonium nitrate soaked in fuel oil
exposive mixture
oxygen reacts with nonmetals or metals
both
anaerobic exercise
oxygen is insufficient and leads to weakness
An oxidizing agent that was used in older versions of the Breathalyzer test is

Ozone
powerful oxidizing agent
Which substance is a common reducing agent used in the production of metals from ores?
c (coke)
Metallurgy Reducing Agents
Coke (C) is produced by heating coal to drive off volatile compounds. It is used as a reducing agent in smelting of metals such as tin:
Bleach
oxidizing agents
work by oxidizing carbon
in order to free most metals from compounds we must
the compounds must be reduced
Antioxidants
neutralize free radicals
reducing agents
hydrogen is _____- reducing agent for ____
hydrogen is an excellent reducing agent for both metal and non mental reagents
Reduction in Photography
The silver ions that are exposed to light react with the developer (hydroquinone, C6H4 (OH)2 ) to form metallic silver
A green catalyst is one that
contains earth-abundant elementsmakes reagents react more efficientlydecreases the cost of producing a chemical
hydrogenated
Combined with, treated with, or exposed to hydrogen
catalyst
substance that speeds up the rate of a chemical reaction with out itself being sued up
cellular respiration
important oxidation reduction process where we obtain energy from out food
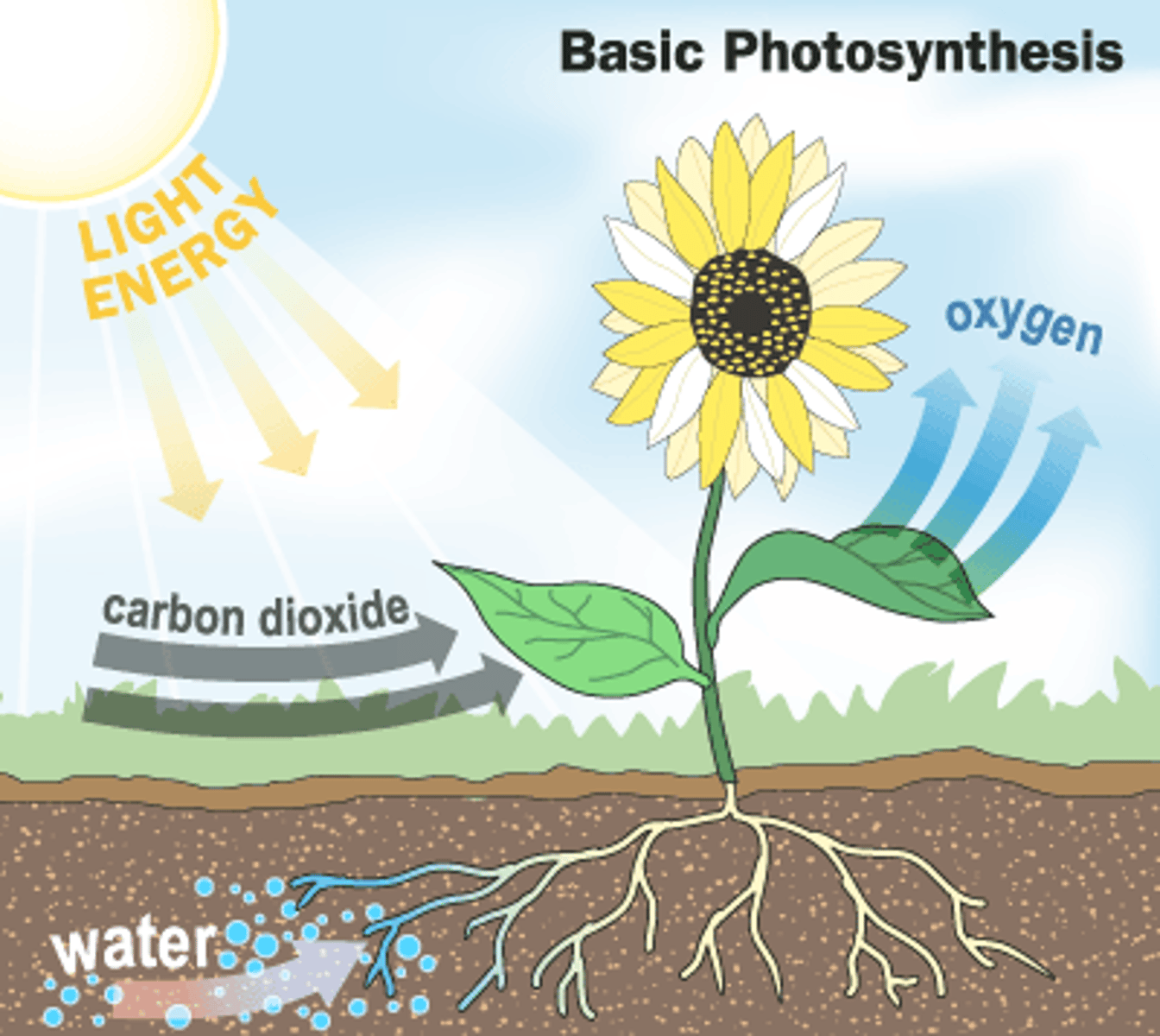
Photosynthesis
plants synthesize carbohydrates
how can we make hydrogen gas
it can be made by reacting zinc with hydrochloric acid
why are catalysis the key to green and sustainable redox chem
it allows us to avoid using toxic or hazardous reagents like chromium
classify the substances as oxidizing or reducing agents
antioxidants
pure metals
disinfectants
bleaches
antispectices
reducing agents
reducing agents
oxidizing agents
oxidizing agents
oxidizing agents
In addition to carbohydrates, which other substance is a product of photosynthesis?
oxygen