Energy in Reactions
1/14
Earn XP
Description and Tags
Intro thermodynamics and reaction rate chemistry
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|

15 Terms
exothermic
releases energy
Endothermic
Absorbs energy
collision theory
For a reaction to occur, the particles must collide, they must collide with the appropriate orientation, and they must collide with sufficient energy.
kinetic energy
energy of motion (speed of particles)
temperature
measurement of the speed of the particles
activation energy
minimum energy that colliding particles must have in order to react
exothermic energy diagram
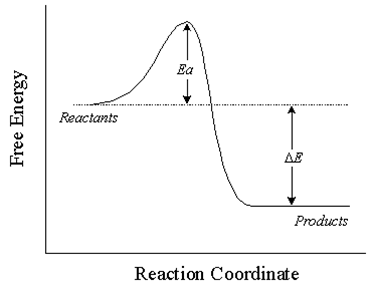
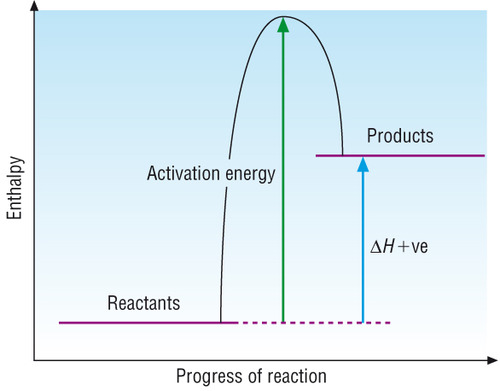
endothermic energy diagram
catalyst
substance that speeds up the rate of a chemical reaction
surface area
The measurement of the outer surface of an object.
reaction rate
the change in reactants and products over time
potential energy
stored energy
orientation
which way a reactant particle is facing
temperature increases (feels hot)
exothermic reaction
temperature decreases (feels cold)
endothermic reaction